Abstract
Objective
Antitumor effects of the medicinal herb Prinsepia utilis Royle (P. utilis) have been reported. This study aimed to identify the potential functional components and molecular mechanisms of P. utilis in the treatment of melanoma through network pharmacology and experimental verification.
Methods
Bioinformatics database and pharmacology analysis platform of the Chinese medicine system were used to obtain the active components of P. utilis, and the potential targets were predicted by the Swiss Target Prediction database. Disease targets associated with melanoma were retrieved from the Genecard database. Network topology analysis and functional enrichment analysis were conducted to screen out the core targets and related signal pathways. The molecular docking method was employed to evaluate the core target binding to bioactive ingredients of P. utilis, and a molecular biology experimental approach was performed to predict and verify the potential molecular mechanism of P. utilis in treating melanoma.
Results
KEGG pathway analysis revealed the enrichment of multiple cancer-related pathways and signaling pathways. Oleanolic acid (OA), Ursolic acid (UA), Arbutin are the active ingredients of P. utilis. The crude extract of P. utilis was able to inhibit melanin in A375 cells. The active compounds OA and UA in P. utilis can significantly inhibit the growth and migration ability of human melanoma cells A375 while inducing apoptosis and inhibiting tyrosinase activity and melanin synthesis. In contrast, arbutin does not show obvious performance in these biological activities.
Conclusion
These findings suggest that OA and UA are promising as more effective anti-melanoma active ingredients in P. utilis. In conclusion, these studies suggest that OA and UA can protect against melanoma by regulating tyrosine to inhibit melanogenesis.
Introduction
Melanoma is the deadliest form of skin cancer, accounting for over 287,723 cases and 60,712 deaths in 2020 worldwide.1–3 Among all subtypes of skin cancers, melanoma is the most rare subtype globally. Ultraviolet rays are one of the environmental factors that induce melanoma.4,5 The incidence of melanoma is increasing faster than that of any other solid tumor. 6 Surgery alone can effectively treat melanoma in its early stages, with a high survival rate. However, survival rates sharply decline after metastasis. 7 Surgical resection, photodynamic therapy, immunotherapy, chemotherapy, and targeted therapy are some of the current therapeutic modalities. Depending on the patient's condition, the tumor's stage, and its location, the therapeutic approach may involve a combination of therapies or solitary drugs. 8 Despite the adoption of multimodal therapy regimens, they persist and are linked to a bad prognosis. Medicinal plants are crucial for people living in mid-altitude and highland regions, particularly in terms of subsistence. 9 Melanoma is linked with exposure to UVA and UVB radiation from the sun or sunbeds, mid-altitudes, and the highlands have higher UV radiation intensity. Therefore, there is an urgent need to identify anti-melanoma lead compounds from natural products.
Prinsepia utilis Royle (P. utilis) is a medicinal and edible herb commonly distributed in the Himalayan mountains at 1000–3000 m above sea level.10,11 P. utilis has been utilized in Chinese and Indian folk medicines for a long time to treat various diseases including dermatitis skin diseases, 12 diarrhea, stomach ache, 13 inflammation, leprosy, 14 rheumatism, pains resulting from over-fatigue, fractures,15,16 arthritis, bone disorders, joint pain, 17 atherosclerosis and is also potentially effective for high blood pressure. 18 Ethnopharmacological studies of P. utilis revealed the use of its leaves, seeds, roots, and fruits exhibited antioxidant, anti-inflammatory, and analgesic activities.19,20 Further, different parts of the plant used to cure various ailments, such as roots and seeds of P. utilis are used to cure diarrhea, stomachache, 13 and arthritis. 17 The leaves of P. utilis are used to cure osteoporosis 17 and prostatic hyperplasia. 21 P. utilis has a wide range of biological activities and is also a plant of interest in the melanin drug discovery neighborhood.
To date, hemiterpenoids, monoterpenoids, triterpenoids, phenolic acids, anthocyanins, flavonoids, fatty acids, γ-hydroxy nitrile glucosides, lignans, sterols have been isolated from different parts of P. utilis. Several studies have shown triterpenes inhibit proliferation and induce apoptosis of tumors, and medicinal plants play a significant role in the subsistence economy of the people, especially those living in the mid-altitudes and the highlands. 9 However, the potential mechanism by which P. utilis improves the prognosis of melanoma remains unknown. This study investigates and demonstrates the drug-target relationships using the network pharmacology method. Furthermore, in vitro experiments were conducted to validate the in vitro antimelanoma effects of Oleanolic acid, Ursolic acid, and Arbutin, which were the active ingredients of P. utilis.
Materials and Methods
Data Preparation
Chemical components in stems and leaves P. utilis were obtained based on literature and database searches (Supporting Table S1). The screened compounds were analyzed for canonical SMILES information retrieved from PubChem (https://pubchem.ncbi.nlm.nih.gov/), which was then utilized for target prediction through the SwissTarget Prediction web server (http://www.swisstargetprediction.ch/).22,23 Melanoma-related targets were identified using the Genecards database (http://www.genecards.org/). We then intersected these melanoma-related targets with those associated with the components of P. utilis and visualized the overlap using a Venn diagram (http://bioinformatics.psb.ugent.be/webtools/Venn/).
Protein-Protein Interaction and Functional Enrichment Analysis
To delve deeper into the shared targets of active compounds in P. utilis and melanoma, we extracted their interactions from the human genome database STRING (Version 11.0, https://string-db.org/). 24 We then constructed a protein-protein interaction regulation network using Cytoscape software (Version 3.7.2). The Network Analyzer tool in Cytoscape was used to analyze the topological importance of nodes in the network, as indicated by their degree, betweenness centrality, and closeness centrality. 25 Gene ontology (GO) analysis and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analysis were performed using the Metascape database to identify the biological functions and clarify the crucial pathways involving the candidate targets. The cluster profile package in R was used to carry out gene enrichment analysis, KEGG pathway, and GO keywords visualization, which are related to biological processes (BP), cellular components (CC), and molecular functions (MF).
Network Construction and Analysis
Compound-target and pathway interaction networks were constructed using Cytoscape software (Version 3.7.0). The NetworkAnalyzer tool was employed to calculate key topological parameters including “degree”, “betweenness centrality”, and “closeness centrality” for network analysis. 26
Molecular Docking Simulation
Molecular docking simulation was performed following a protocol described in a previous study. The chemical structures of the bioactive ingredients were screened with oral bioavailability (OB) > 30% 27 or druglikeness (DL) > 0.18. 28 The potential targets were chosen in the protein-protein interaction (PPI) and component-targets-pathways networks based on being ranked in the top 20 degrees. Bioinformatics analysis and molecular docking were conducted using AutodockTools (Version 1.5.6). A heat map showing the affinity between potential targets and predicted compounds was created using R.
Preparation of P. utilis. Extract
The dried stems and leaves of P. utilis were ground into a fine powder and sieved with a 40-mesh sieve. The ground fine powder (P. utilis) was extracted with 95% ethanol under reflux for 2 h. The extracted extract was filtered and concentrated under reduced pressure at 65 °C using a rotary evaporator. The extract was freeze-dried and stored in a refrigerator at 4 °C.
Identification of OA,UA and Artubin in the Extracts by LC-MS/MS
OA, UA, and arbutin in P. utilis were analyzed using liquid chromatography-tandem mass spectrometry (LC-MS/MS, Supporting fig S2). Agilent Technologies 6420 Triple Quadrupole LC/MS and ZORBAX SB-C18 column (880975-902, Agilent) were used to detect and analyze the crude extracts of stems and leaves of P. utilis, including OA, UA, and arbutin. The crude extracts were qualitatively analyzed with the corresponding standards as controls. The mass spectrometry conditions were as follows: injection volume was 0.5 ul, flow rate was 1 ml/min, methanol was used as the mobile phase, and elution conditions were 0–4 min with 100% methanol.
Cell Culture and Viability Assay
Human melanoma A375 cells (Cat no. SCSP-533, shanghai, China) were purchased from the National Infrastructure of Cell Line Resource. A375 cells were cultured at 37 °C in a humidified environment containing 5% CO2 in DMEM medium (C3113-0500, Viva Cell Biosciences, China), supplemented with 10% fetal calf serum (FBS, C04001-500, VivaCell Biosciences, China) and glutamine (E607004-0500, sangon biotech, China). All consumables are purchased in NEST Biotechnology (Wuxi, China). The cytotoxicity of P. utilis was evaluated using sulphorhodamine B (SRB, MB1808, MeilunBio, China) assay. A375 cells (4 × 103/well) were cultured in 96-well plates. After that, the cells were then treated with different concentrations of the bioactive ingredients of Oleanic acid (OA, A010031, Energy Chemical, China), Ursolic Acid (UA, MB6981, MeilunBio, China), and Arbutin (P2039551, adamasbeta, China) and incubated for 48 h. The viability of the cells was assessed using the percentage of the control values.
Flow Cytometric Detection of Cell Apoptosis
Cell apoptosis was quantified using Annexin V: FITC apoptosis detection kit I (40302ES20, Yeasen Biotechnology, China) and assayed according to manufacturer protocols. Annexin V was viewed as a reliable marker for identifying early-stage apoptotic cells, whereas PI served as a marker for detecting late-stage apoptotic cells. The cells were placed in 500 μl binding buffer and then stained with 5 μl Annexin V-FITC followed by 10 μl PI Staining Solution. Then, cells were analyzed by flow cytometry using FACS Aria III (Beckman Coulter, Brea, CA). Both HMVECs stained Annexin+/PI– and Annexin+/ Pl + in the flow cytometric analyses were apoptotic cells.
Scratch Wound Assay
A375 cells (3 × 105 cells/well) were seeded in 6-well culture plates and incubated overnight. When the cells have grown to fill or nearly fill the bottom of the dish, the monolayers of cells were scraped with a cross in the middle of each well with a 10μL sterile tip. Next, remove the exfoliated cells after the scratch. OA, UA, and Arbutin were used to incubate A375 cells for 24 h. The scratch in each field was imaged under an inverted microscope at 0 and 24 h, respectively. The area of the scratch was measured on the images.
Tyrosinase Activity and the Cell Melanin Content Analyses
Tyrosinase activity was measured by the oxidation rate of levodopa. Briefly, after A375 cells were grown for 24 h, different concentrations of OA, UA, and Arbutin were incubated for 48 h. Then, A375 cells were reacted with 100 μL 1% triton (CT321318200, Coolaber, China) for 30 min at −80°C. After melting at room temperature, the cells were reacted with 10 μL 0.2% L-DOPA (M0113521, MeilunBio, China) at 37 °C for 3 h. The absorbance was measured at 490 nm using a microplate reader. The melanocyte's melanin synthesis was measured by NaOH (MB3333, Energy Chemical, China) lysis method. The cells were incubated with 200 μL different concentrations of OA, UA, and Arbutin for 72 h in 6 well plates at 37 °C. Then, the Cells were pre-treated with the 200 uL 1 M NaOH reacted for 30 min at 80°C, and 800 μL pure water corresponds to 2 fold of NaOH in A375 cells. The absorbance was measured at 475 nm using a microplate reader. Tyrosinase activity and the cell melanin content of the cells were assessed using a percentage of the control values.
Statistical Analysis
GraphPad Prism 8 (San Diego, CA, USA) was used for analyzing the data (Mean ± SD). One-way analysis was used to analyze Multiple groups, and P < 0.05 was regarded as statistically significant.
Results
Potential Targets of P. utilis in Melanoma Treatment
A total of 71 compounds were reported in stems and leaves of P. utilis (Supporting Table S1). As shown in Figure 1A, 559 targets of P. utilis were predicted from the Swiss target prediction database, 1260 melanoma-related targets were summarized from the Genecards database, and 154 common targets were found by making Venn diagrams. The protein-protein interaction (PPI) network involving targets linked to melanoma and P. utilis was created and displayed in Figure 1B. Nodes and edges within the network signify protein associations. Following the removal of disconnected nodes, the PPI network consists of 153 nodes and 2267 edges. The average node degree is 29.634, and the local clustering coefficient is 0.592. As the degree value increased, the color of a node in the topological property network changed from blue to orange and the size of the node increased from small to big. Furthermore, based on the degree centrality, the 4 highest degree value targets were AKT1, VEGFA, TNF, and IL-6.
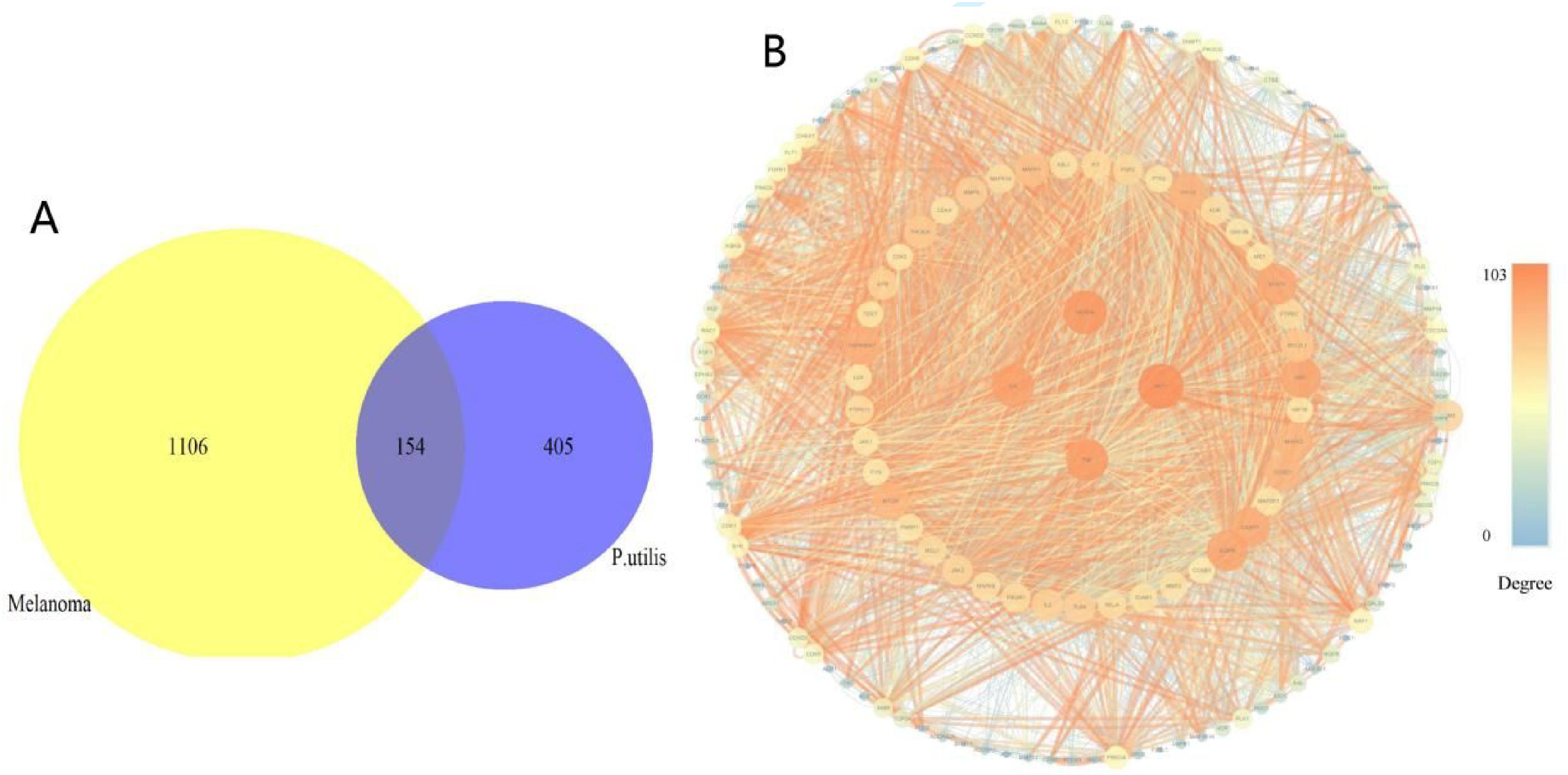
Venn diagram and PPI network graph. (A) Venn diagram showing overlap of candidate targets in melanoma and P. utilis. The yellow refers to the disease melanoma targets. The blue represents targets of P. utilis. The purple represents 154 putative targets of P. utilis for the treatment of melanoma. (B) Protein-protein interaction (PPI) network. Nodes in the PPI network are colored according to degree, the colored edges represent protein-protein associations. As the combined score increased, the color of associations shifted from blue to orange and the size of the associations grew larger.
Compound-Target-Pathway Network Analysis
The enrichment analysis of the intersection 154 targets was conducted using the Metascape database. A total of 128 KEGG pathways were enriched. The top 20 enriched KEGG pathways included the PI3K-Akt signaling pathway, Ras signaling pathway, Rap1 signaling pathway, and MAPK signaling pathway (Figure 2A). The GO enrichment analysis of the 154 targets, involved biological processes related to protein phosphorylation and regulation, specifically focusing on the transmembrane receptor protein tyrosine kinase signaling pathway (Figure 2B). A compound-target-pathway network was performed based on the compounds, corresponding targets, and the top 20 enriched KEGG pathways. Figure 3 displays that there are 169 nodes in the network, which include 49 compounds, 100 targets, and 20 pathways. The bioactive compounds, targets, and pathways are represented by the blue, red, and yellow nodes, respectively, while the interactions among them are depicted by the edges.
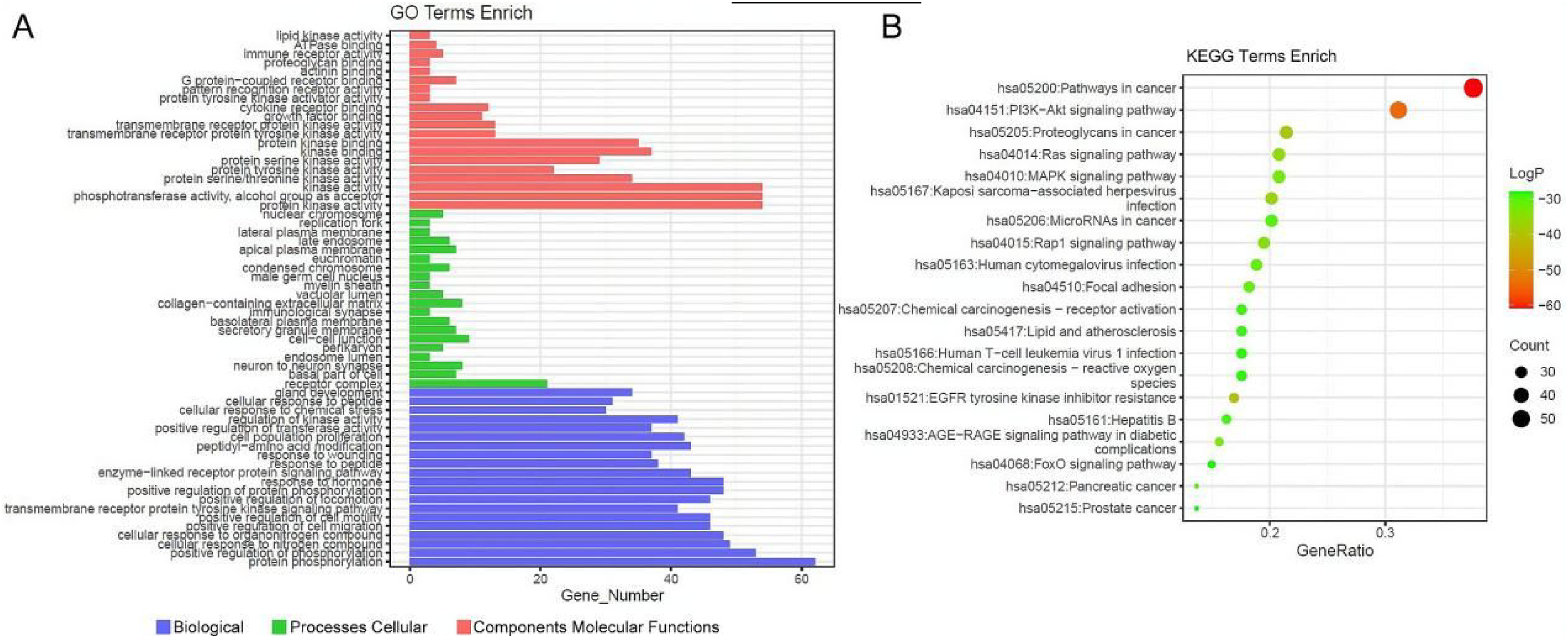
Go and KEGG functional enrichment analysis. (A) The top 20 Gene Ontology terms are enriched in each category. (B) A bubble plot displays the top 20 enriched targets and Genomes in the Kyoto Encyclopedia.

The network consists of potential active compounds of P. utilis, their shared targets with melanoma, and associated signaling pathways.
The Evaluation of Molecular Docking
A simulation of molecular docking was conducted to evaluate how well the bioactive ingredients of P. utilis could bind to the predicted hub proteins. Most of the bioactive ingredients in P. utilis showed consistent binding abilities with the predicted proteins, including MAPK3, MAPK1, PIK3CA, AKT1, EGFR, VEGFA, STAT3, CCND1, and TNF (Figure 4A). The results indicated that Oleanolic acid (OA) had a stable binding ability with VEGFA (−12.08, Figure 4B). Additionally, Ursolic acid (UA) also had a stable binding ability with VEGFA (−11.96, Figure 4C).The above results indicate that the inhibitory effect of P. utilis on melanoma growth is theoretically supported.

The binding energy of the potential active components of P. utilis extract and the hub proteins and molecular docking. (A) The docking score of bioactive ingredients binding with hub targets. (B) Molecular docking simulation for Oleanolic acid-VEGFA (OA-VEGFA). (C) Molecular docking simulation for Ursolic acid-VEGFA (UA-VEGFA).
Active Compound from P. utilis Inhibits Cell Proliferation in Human Melanoma Cell Lines
P. utilis also has antitumor activity 11 and skin protective effects. 29 After completing the flow of network pharmacology, cellular experiments were performed to validate it, and the flow chart is shown in Supporting fig S1. Based on the network pharmacology, we first evaluated the effect of P. utilis extract on melanin production (Figure 5A). Then, we determined that P. utilis extract contained Oleanolic acid (OA) and Ursolic acid (UA) by ESI (Supporting fig S2). To investigate the inhibition of active compounds from P. utilis on melanoma cell growths, A375 cells were treated with different concentrations of Oleanolic acid (OA), Ursolic acid (UA), and Arbutin for 48 h (Figure 5B). Figure 5C shows the cytotoxicity of dacarbazine to A375 cells. As shown in Figure 5D and E, the cell viability decreased with increasing concentrations of OA and UA. Thus, The 50% growth-inhibitory concentrations (IC50) after treatment with OA for 48 h was 33.13 ± 0.35 μg /ml, and IC50 for UA was 5 ± 0.03 μg/ml. Compared with OA and UA, Arbutin did not show significant cytotoxicity (Figure 5F). These results suggested that Oleanolic acid and Ursolic acid inhibited the growth of human melanoma cells in vitro.

P. utilis active ingredients inhibit cell proliferation in human A375 cells. (A) P. utilis inhibits melanin. (B) Chemical structures of the active ingredients of P. utilis, Oleanolic acid (OA), Ursolic acid (UA), and Arbutin. Cell viability was assessed by SRB assay. (C) The cytotoxicity of dacarbazine to A375 cells. A375 cells were treated with 2.28, 4.57, 5.71, 11.42, 22.84, 45.67, 91.34 μg/ml Oleanolic acid (D) and Ursolic acid (E) for 48 h. (F) Inhibitory effect of Arbutin on the growth of A375. Data are mean ± SD, *Compared with the NC group, ***p ˂ 0.001.
Active Compound Extract P. utilis Induces Apoptosis in A375 Cells
The A375 apoptosis rate was detected by flow cytometric analysis. The evaluation of the results indicated the difference in cell apoptosis rate between the NC group and OA, UA, and the Arbutin administration group. From Figure 6 and B, it can be found that similar to the results of the SRB experiment, OA and UA can significantly induce A375 cell apoptosis (p ˂ 0.001), while Arbutin didn't have this ability. The in vitro anticancer activity of OA and UA can be determined by combining flow cytometry apoptosis and SRB cell viability experiments.
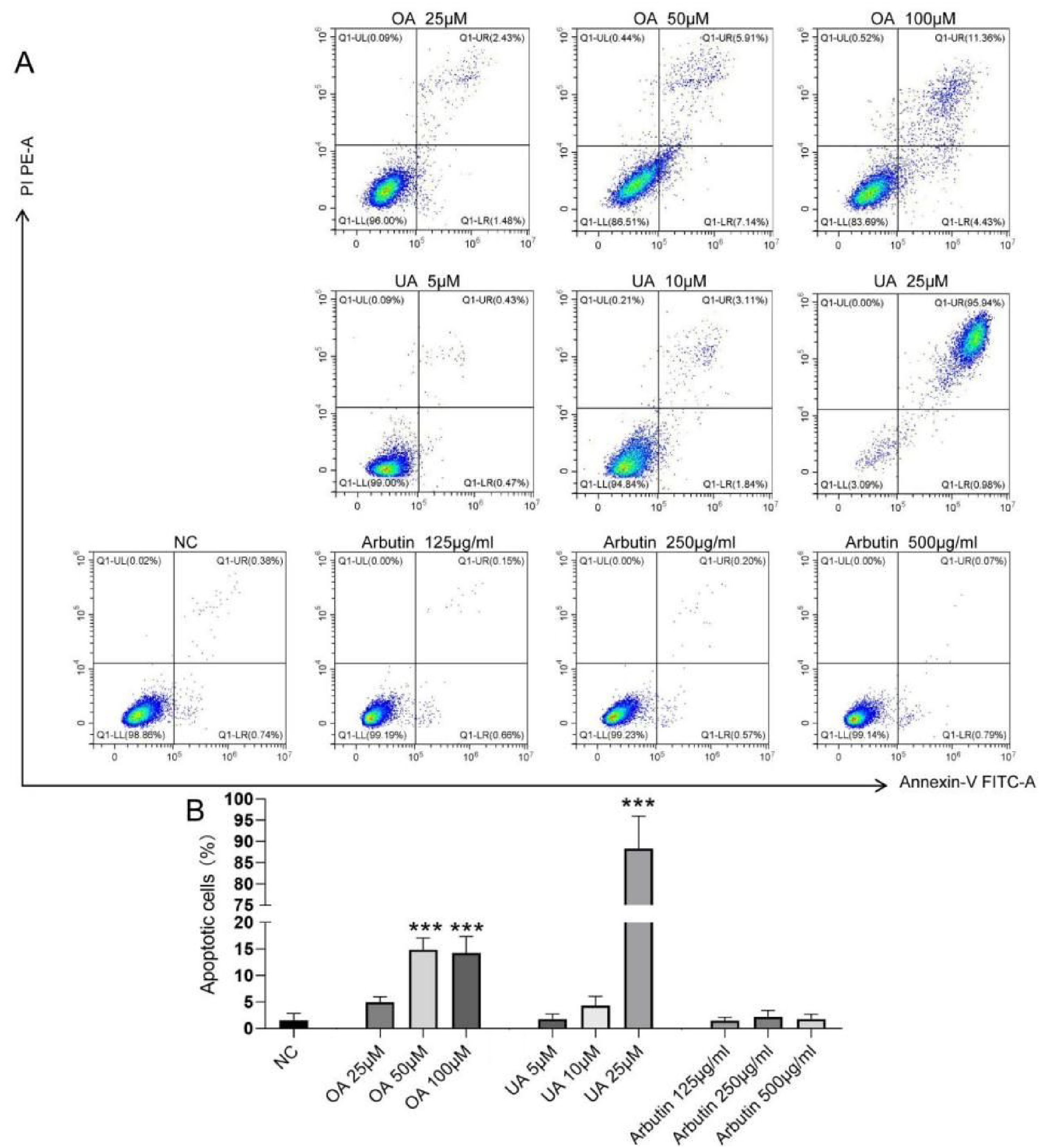
Active compounds from P. utilis induce cell apoptosis in A375 cells. (A) Representative results of apoptosis detection by flow cytometry. (B) Statistical results of cell apoptosis. The cell ratio is indicated by the results following staining with propidium iodide and deoxyuridine triphosphate-fluorescein. Data are mean ± SD, *Compared with the NC group, ***p ˂ 0.001.
Active Compounds from P. utilis Inhibits Cell Migration
As shown in Figure 7A to C, compared with the NC group, 25 μM and 50 μM OA can significantly inhibit the migration of A375 cells (p ˂ 0.001), 5 μM and 10 μM UA can significantly inhibit the migration of A375 cells (p ˂ 0.001), while Arbutin does not affect the migration of A375 cells. The above results show that OA and UA inhibit the migration and repair ability of A375 cells.

Wound healing test assay. Effects of OA (A), UA (B), and Arbutin (C) on the migration ability of A375 cells after 24 h of treatment. Data are mean ± SD, *Compared with the NC group, ***p ˂ 0.001.
Active Compounds from P. utilis Inhibits Tyrosinase and the Cell Melanin Content
Based on the earlier results, We further examined the growth-inhibitory effect of OA, UA, and Arbutin. As shown in Figure 8A, we confirmed that tyrosinase was downregulated in A375 cells following treatment with OA and UA, while Arbutin had no obvious effect. Further investigation of the content of melanin synthesis, Figure 8B shows that OA, UA, and Arbutin have an inhibitory effect on the inhibition of cellular melanin. In conclusion, OA and UA are the better anti-melanoma active compounds in P. utilis, which can inhibit the synthesis of tyrosinase and melanin.
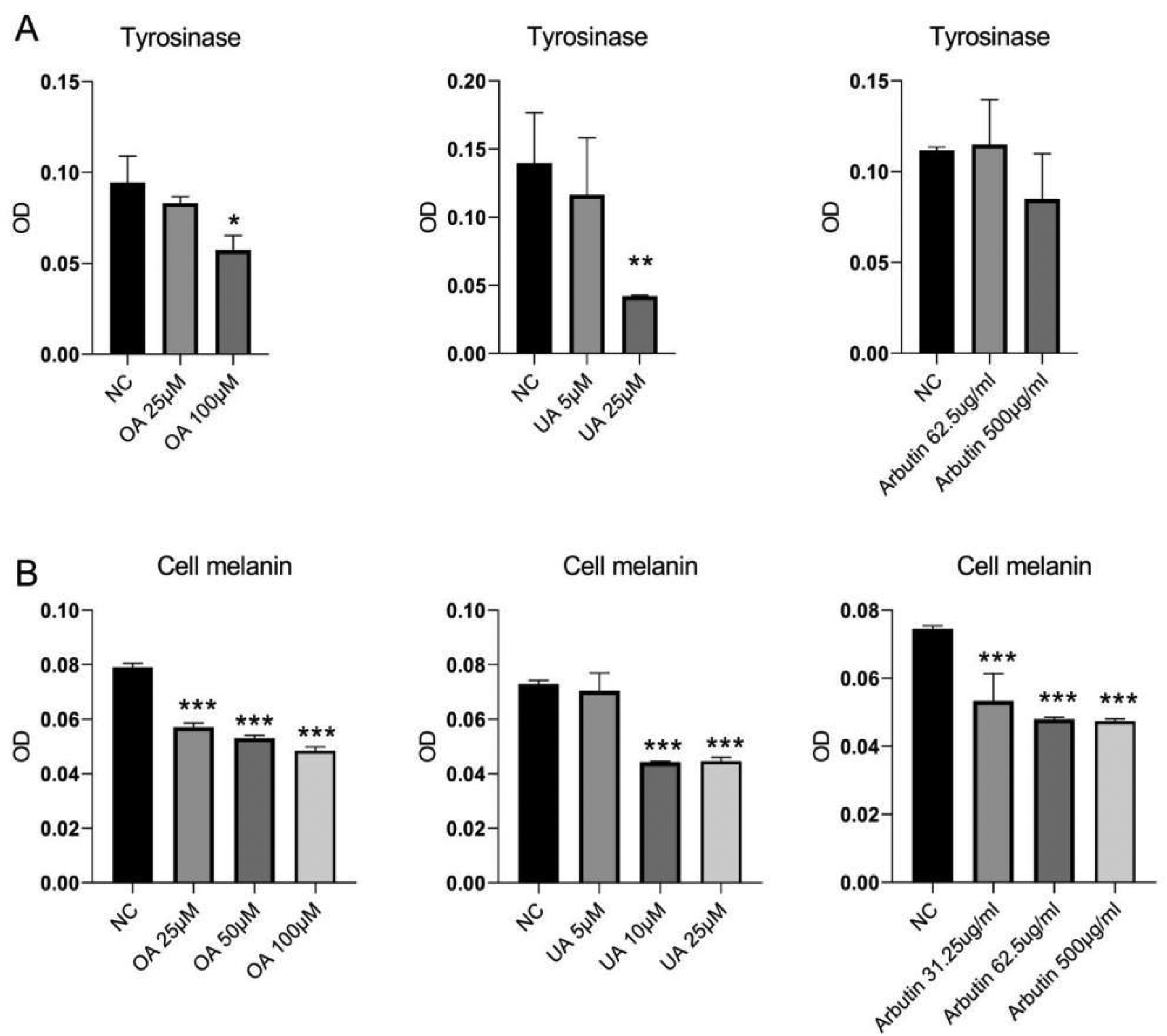
The tyrosinase activity and the cell melanin content. (A) Effects of OA, UA, and Arbutin on tyrosinase in A375. (B) Effects of OA, UA, and Arbutin on cell melanin in A375. Data are mean ± SD, *Compared with the NC group, *p ˂ 0.05, **p ˂ 0.01, ***p ˂ 0.001.
Discussions
Significant advancements in experimental and clinical treatment of melanoma have greatly assisted in discovering the underlying mechanisms of the disease. In the early 2000s, although localized lesions could be surgically removed with a cure rate close to 100%, 30 with the development of targeted therapy, followed by the introduction of immunotherapies,31–34 it is still a challenge to provide better recommendations for treatments in patients with melanoma. Traditional Chinese Medicine can serve as a practical substitute for conventional cancer treatment, providing fewer side effects. Herbal medicines derived from plant extracts are being increasingly used to treat a wide variety of clinical diseases, with relatively little knowledge of their modes of action. Many studies have demonstrated that P. utilis exerts anticarcinogenic and antiproliferative effects, and can reduce deleterious effects of reactive oxygen species.
Network pharmacology is a useful method for discovering various elements and studying the mechanisms of Chinese herbal medicine. Through network pharmacological analysis, 154 potential targets related to melanoma were discovered. The compound found in P. utilis plays a crucial role in treating melanoma by controlling multiple signaling pathways. As one component of P. utilis, Oleanolic acid and Ursolic acid exhibit antitumor and antimetastatic effects on infectious via enhancing cell apoptosis and decreasing cell growth, and migration. Arbutin showed only inhibition of melanin. These observations are consistent with those of several previous studies in which Oleanolic acid and Ursolic acid-induced apoptosis in mouse tumor cell lines. Ursolic acid induces apoptosis by activating p53. 35 EGFR is a tyrosine kinase involved in regulating cell homeostasis. The results of the network pharmacological analysis also demonstrated that extracts of P. utilis regulate the EGFR signaling pathway to decrease the tyrosinase activity and cell melanin content of melanoma.
An extensive phytochemical investigation has been conducted on the seeds and leaves of P. utilis. However, a few phytochemical studies have been done on the stem, and the phytochemical studies on the root have not been reported yet despite its traditional use in skin treatment. For the first time in this study, a network pharmacology analysis was used to identify potential targets of P. utilis for treating melanoma. The mechanisms for the effects of the compound extract from P. utilis against melanoma were outlined based on the network pharmacological analysis and experiments conducted. Firstly, compounds from P. utilis might inhibit cell growth, and migration and induce cell apoptosis by regulating the expression of Ras, Rap1, and MAPK signaling pathways. Among Rap1 signaling pathway is an important participant in the process of tumor cell migration, further analysis of scratch wound assay has confirmed the mechanism against cell migration of melanoma via in vitro experiment. Secondly, another signaling pathway, which was identified through network pharmacological analysis, reducing the content of tyrosinase activity and the cell melanin content, was selected to further analyze and confirm the mechanism against melanoma via in vitro experiment. KEGG enrichment analysis revealed the enrichments of multiple cancer-related pathways. Enrichment of the Ras pathway genes is of paramount importance in the pathogenesis of sporadic melanomas. 36 Besides, the MAPK pathway and PI3 K pathway in skin cutaneous melanoma suggest the RAS pathway has a broad influence, regulating various cellular processes such as cell proliferation, growth, survival, apoptosis, angiogenesis, and cell migration. Dysregulation of this pathway can result in malignant transformation. 37 In brief, network pharmacology identified the active components and potential pathways of P. utilis, focusing on three compounds (OA, UA, and Arbutin), and tyrosinase which was the rate-limiting enzyme in melanin synthesis.
Different standards make it difficult to compare and integrate TCMS and related databases, thereby resulting in many false positive and false negative interactions. Therefore, the results of the network pharmacological analysis were further verified by experimental evaluation. Efforts have been made to verify the effectiveness of commercially available OA, UA, and Arbutin mechanisms, in human skin cutaneous melanoma by vitro experiment. Dacarbazine is a common melanoma treatment drug in clinical practice, 38 while OA and UA have anti-melanoma activity in vitro, and UA also shows competitiveness. However, OA, UA, and Arbutin all showed inhibition of melanin production, which may be the reason for the synergistic effects of the compounds in P. utilis. Arbutin's melanin-inhibiting potential and low cytotoxicity also suggest its potential in the field of whitening skin care. 39 Rajan Logesh 40 reported that exposure to ultraviolet light stimulates the biosynthesis of melanin, which can also stimulate the local production of hormone factors, thereby stimulating the development of melanoma by changing the chemical properties of eumelanin and pheomelanin. Moreover, MITF (Microphthalmia-associated transcription factor) is a basic helix-loop-helix leucine zipper transcription factor that can bind to the M-box motif in the promoter region (a highly conserved sequence shared by TYR, TRP-1, and TRP-2 in the promoter region, namely 5′-AGTCATGTGCT-3′) to regulate the expression of TYR, TRP-1, and TRP-2, thereby regulating the production of melanin. 41 In addition, the PI3 K/Akt signaling pathway has been reported to synergize with MITF to stimulate the tyrosinase promoter, enhance its binding to the tyrosinase promoter, and thus regulate melanin production. 42 Meanwhile, the MAPK signaling pathway plays a role in activating melanocyte receptors. When the ligand binds to the extracellular domain of the receptor, it triggers a complex mechanism (Ras-Raf-MEK-ERK), ultimately causing an increase in MITF expression. 43 In this study, the network pharmacology of P. utilis demonstrated its regulation of PI3 K/Akt and MAPK signals and also demonstrated anti-melanoma and melanin inhibition in experiments. Combined with previous studies, these evidences are evidence of the anti-melanoma mechanism of P. utilis. Hence, one limitation of network pharmacology analysis is that the potential active/functional components may not completely control the predicted pathways. Nevertheless, further advanced research activities, with great priority on an animal model and an understanding of the mechanism of action of anti-melanoma activity, are mandatory to validate the traditional utilization of this medicinally significant plant and continue the long research-based expedition of medicinal plant-derived anti-melanoma medicine discovery for ensuring human safety and protecting their health.
Conclusion
In summary, our findings indicate that compounds from P. utilis exert their antitumor/antimetastatic effects on melanoma by tyrosinase activity and cell growth, migration, and cell apoptosis via a mechanism that might involve multi-repression or activation of the PI3K-Akt, Rap1, MAPK signaling pathway. It is concluded that Ursolic acid from P. utilis is implicated as a promising drug for the treatment of melanoma.
Footnotes
Acknowledgments
We express our gratitude to Qiutuan Ma, Yunge Jiang, Yunmei Sun, and Yang Tao for their technical support.
Authors’ Contributions
Xingyan Yang and Qi Lai designed the study, performed the network pharmacological analysis and wrote the manuscript. Junjie Ma performed the in vivo experiment. Yang Tao provided technical assistance. Jihong Wang assisted some network pharmacological analysis. Guangzhi Zeng participated in its design, revised the manuscript. All authors read and approved the final manuscript.
Availability of Data and Materials
The data that support the findings of this study are present in the paper. Any information for this study is available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by grants from the Bureau of Science and Technology of Yunnan Province (202101AU070052).
Yunnan Key Laboratory of Chiral Functional Substance Research and Application: 202402AN360010.
Statement of Human and Animal Rights
There are no human and Animal subjects in this article.
Statement of Informed Consent
Not applicable.
