Abstract
Chemical investigation of the leaves and stems of Rubus hirsutus Thunb has resulted in the isolation and characterization of a rare new tricyclic thienocyclopentapyran (
Sulfur contributes significantly to the chemical diversity of natural compounds thanks to its particular features which allow fundamental biological reactions.
1
Plant-derived sulfur-containing secondary metabolites constitute a small group of low-molecular weight natural products, which play a vital role in plant-pest interactions in numerous plant families and represent major defense molecules in the Asteraceae, Alliaceae, and Brassicaceae families.
2
Both primary and secondary S-containing compounds have great effects on plant health and regulatory mechanisms when under stress.
3
In addition to antibacterial activity, some sulfur-containing natural products have shown promising antifungal activities against a wide range of plant pathogenic fungi. For instance, the phytoalexins sinalbins A and B were proved to have antifungal activity against the plant pathogenic fungus Leptosphaeria maculans.
4
Sulfur-containing monoterpenoids have been reported as potential antithrombotic drugs.
5
Sulfur-containing natural products have emerged as a new class of compounds that have the potential to exert strong anti-inflammatory effects and may, therefore, stand as a potential “goldmine” for the discovery of future anti-inflammatory drugs.
6
In this study, we investigated the chemical composition of the leaves and stems of Rubus hirsutus Thunb, leading to the isolation and characterization of a rare new tricyclic thienocyclopentapyran (
The MeOH extracts of the leaves and stems of R. hirsutus were subjected to preparative HPLC (C18), affording a new sulfur-containing compound

The structures of compounds 1‐
Results and Discussion
Compound
the NMR Data (600 MHz, MeOH-D 4) of Compound 1.

The 10 signals for the tricyclic thienocyclopentapyran domain comprised 2 aromatic methines at δC 115.2 (C-4), and 120.8 (C-3), an oxygenated methine at δC 82.8 (C-8), six quaternary aromatic carbons at δC 151.5 (C-5), δC 121.5 (C-4a), δC 136.7 (C-8a), δC 127.8 (C-8b), δC 144.9 (C-3a), and δC 150.6 (C-2), corresponding to 3 sets of double bonds, and a lactone carbonyl carbon at δC 171.8 (C-6). Among the six quaternary aromatic carbons, an enol carbon at δC 151.5 (C-5) was observed. In the 1H, 1H COSY spectrum of
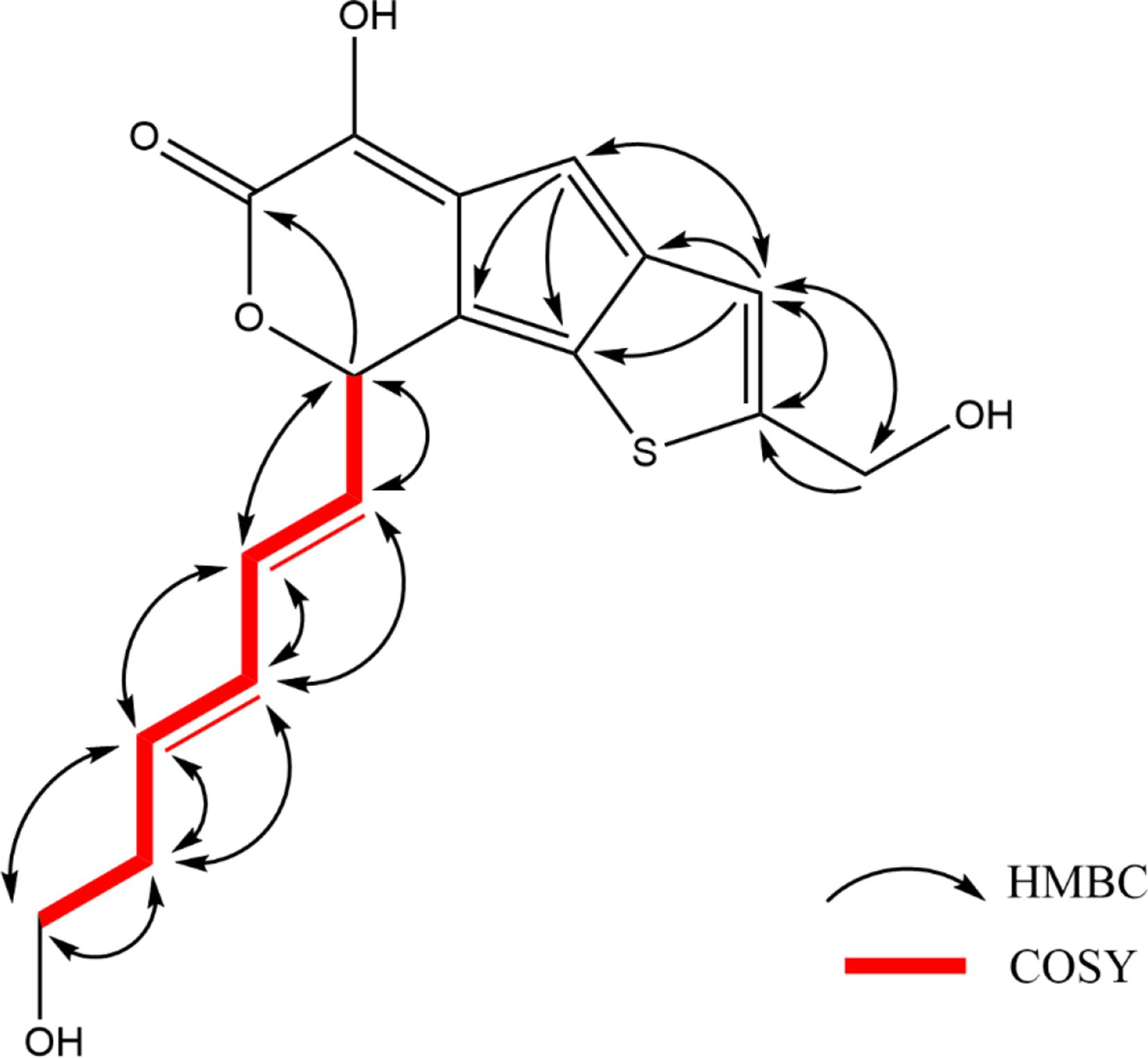
Key 1H, 1H COSY and HMBC correlations of compound 1.
HMBC analyses showed the formation of the tricyclic thienocyclopentapyran skeleton with a characteristic (E)-hexa-3,5-dien-1-ol pedant (Figure 2). HMBC cross peaks between olefinic H-9,10 and the oxygenated methine C-8 confirmed that the side chain was located at C-8 of ring A. In the 1H NMR spectrum of

Experimental and calculated ECD spectra of (8R)−
The isolated new and known compounds were tested for their bacteriostatic as well as bactericidal abilities against 3 pathogenic bacteria namely methicillin resistant Staphylococcus aureus MRSA, Pseudomonas aeruginosa, and Klebsiella pneumoniae. Hirsuthienopyran (
Experimental
General
The melting point (uncorrected): Reichert apparatus. Optical rotations: Perkin-Klmer-341 polarimeter. IR spectra: NicoletAvatar-360FT-IR spectrometer. 1H NMR (600 MHz) and 13C NMR (150 MHz) spectra: Agilent 600 NMR spectrometer with TMS as internal standard (at 25 °C). Mass spectra: AB Sciex 5500 Q-TRAP and LC/MS Q-Orbitrap (Q Exactive Focus, ThermoFisher Scientific). TLC: Merck precoated plates (silica gel 60 F254) of 0.25 mm thickness. Chromatography: Waters 600 Preparative HPLC, with a Shim-pack PREP-ODS (250 × 20 mm) column; Sephadex LH-20 (Amersham).
Plant Material
The leaves and stems of Rubus hirsutus were collected in Zhejiang Province, China, in September 2018 and identified by Prof. Changxi Zhang (Jinhua Medical College, Jinhua, China.). A voucher specimen (No. zju 7541) is kept in the College of Agriculture and Biotechnology, Zhejiang University, Hangzhou, China.
Extraction and Isolation
The shade-dried leaves and stems (5 kg) were extracted with methanol (20 L), and 134 g extract was obtained, which was partitioned with light petroleum (1200 ml), EtOAc (1500 ml) and n-BuOH (1000 ml), successively. The light petroleum extract (27 g) was subjected to column chromatography (4 × 40 cm, 700 g, 200‐300 mesh) over silica gel, eluting with light petroleum/EtOAc (10:0-0:10, gradients, 3000 ml) to afford 4 fractions. Fraction 3 was purified preparative HPLC (flow rate 8 mL/min, UV detector 250 nm), using CH3OH-H2O (75:25) as eluent, to afford compound
Computation Section
The geometry was optimized starting from initial conformations, with DFT calculations at the B3LYP/6‐31 + G(d) level using the Gaussian 09 program. Frequency analysis was made at the same level of theory to verify that these optimized structures are real minima on the potential energy surface. Time-dependent DFT calculations were performed on the lowest-energy conformations for each configuration using 30 excited states and in methanol solution. ECD spectra were generated using the program SpecDis by applying a Gaussian band shape with 0.2 eV width, from dipole-length rotational strengths. 10 -12
Antibiotic Activity Screening of Stress Metabolites
The conventional broth dilution assay was used to evaluate the antibacterial activities. Three clinical pathogens namely MRSA, P. aeruginosa [CMCC(B)10104], and K. pneumoniae [CMCC(B)46117] were cultured and left overnight to grow. Each pathogenic culture was then diluted in 0.9% saline to an inoculum density of 5 × 105 cfu by comparison with a McFarland standard. Tetracycline was dissolved in Mueller–Hinton broth with a starting concentration of 512 µg/mL and was used as positive control, whereas methanol was used as negative control. A total of 125 µL of MHB was distributed into the 96-well plates. A total of 125 µL of the different samples was dispensed into well 1 and serially diluted across the plate. Ultimately, addition of 125 µL of the bacterial inoculum was made and the plates were incubated at 37 °C for 18 hours. The bacteriostatic abilities of the compounds were noted as MICs, which were made in triplicate. Five mg per mL of a methanolic solution of 3-[4,5-dimethylthiazol-2-yl]−2,5-diphenyltetrazolium bromide (MTT; Lancaster) was used to detect bacterial growth by a change in color from yellow to blue. 13
Hirsuthienopyran (1)
Brown powder
Rf : 0.40 (CHCl3-MeOH, 2:1).
UV/Vis λmax (MeOH) nm (log ε): 210 (4.24), 265 (2.47), 324 (3.14).
ECD (c 50 ppm, MeOH) λmax (Δε) 224 (-1.56), 215 (+0.01), 203 (−3.57) nm.
ESI MS m/z 331.0639 [M - H]- (calcd. 331.0640).
Product yield: 0.02%.
Footnotes
Acknowledgments
We thank Dr Yaqin Liu from the Department of Chemistry for the NMR measurement and Prof. Hujun Xie from Zhejiang Gongshang University for his help on ECD calculation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science Foundation of Lishui Science and Technology Bureau (2017ZDYF17) and Jinhua Science and Technology Project (2020-2-019).
