Abstract
The effects of groundnut (Apios americana Medik) extract on osteoblast differentiation were examined using MC3T3-E1 cells. MC3T3-E1 cells were treated with the crude extract along with other differentiating reagents. The alkaline phosphatase (ALP) activity of cells cultured in a differentiation medium supplemented with 0.01% crude groundnut extract was 1.5‐1.6 times higher than that of cells cultured in a differentiation medium without the extract. Crude groundnut extract was further separated into aqueous and methanol fractions. The methanol fraction enhanced ALP activity, osteocalcin, integrin-binding sialoprotein, and type I collagen expression, and calcium mineralization. Conversely, the aqueous fraction did not show such effects. Groundnut extract may enhance osteoblast differentiation, and this effect is likely conferred by water insoluble substance(s).
Groundnut (Apios americana Medik, family Fabaceae), a plant of North American origin, forms a tuber, which is consumed as food. There are some reports on the physiological activities of groundnut. For instance, groundnut extract could decrease lung inflammation caused by influenza virus H1N1 infection and local exposure to lipopolysaccharides (LPSs). 1 Groundnut flower polysaccharide showed an immunoenhancement effect in mice, 2 and groundnut tuber polysaccharides activated autophagy and suppressed the release of inflammatory cytokines by LPS-induced RAW264.7 macrophages. 3 Lupinalbin A isolated from groundnut inhibited LPS-induced interferon (IFN)-β production in RAW264.7 macrophages. 4 These biological activities of groundnut are mainly an inflammatory suppression effect. These immune modulation effects of groundnut are mainly caused by polysaccharide.
On the other hand, another important biological activity of groundnut is caused by flavonoid. Flavonoids from groundnut showed anti-inflammatory and antioxidant activities, 5,6 and isoflavonoids from groundnut inhibited xanthine oxidase activity. 7 In particular, isoflavones are important physiologically active substances in groundnut. Since groundnut is a legume, its tubers show a high isoflavone content. A key groundnut tuber isoflavone is genistein-7-O-gentiobioside (G7G), a glycoside of genistein. 8 Groundnut also contains genistin, another glycoside of genistein. 9 Groundnut isoflavones have been reported to enhance cellular antioxidant activity by enhancing HO-1 expression. 10 Groundnut isoflavones exhibit a partial agonistic activity against estrogen, antioxidant properties, and an α-glucosidase inhibitory potential. 11 Together, these reports indicate that groundnut contains various physiologically active compounds, with isoflavones being the distinct functional molecules.
Furthermore, legume isoflavones serve as phytoestrogens to enhance osteoblastogenesis. Daidzein enhanced osteoblast differentiation of and calcium mineralization in MC3T3-E1 cells. 12 Similarly, genistein could enhance osteoblastogenesis. 13 -15 For instance, genistein stimulated osteoblastic differentiation in bone-marrow culture. 16,17 Gegnistin also stimulated osteogenic differentiation of bone marrow stromal cells. 18 Although groundnut does not contain daidzein, G7G is unique to this legume. However, the effect of G7G in enhancing osteoblastogenesis remains unknown.
Additionally, groundnut tubers show high calcium content. 19 If groundnut isoflavones indeed show promoting effects on osteoblastogenesis, groundnut can potentially enhance bone formation and prevent osteoporosis. Therefore, in this study, we examined the potential effects of groundnut on osteoblastogenesis using ME3T3-E1 cells.
Results and Discussion
Effect of Crude Extract on the Differentiation of MC3T3-E1 Cells
Crude extract of groundnut tubers was added to the differentiation medium at final concentrations of 1.0%, 0.1%, and 0.01%, and MC3T3-E1 cells were cultured in this medium. After incubation for 7 days, intracellular ALP activity was measured as an osteoblast differentiation marker (Figure 1). After 7 days of incubation, the ALP activity of MC3T3-E1 cells cultured in the differentiation medium supplemented with 0.1% and 0.01% crude extract was 1.5‐1.6 times higher than that of the control cells. On the other hand, the ALP activity of MC3T3-E1 cells cultured in the differentiation medium supplemented with 1% crude extract was significantly lower than that of the control cells. Additionally, the cells were incubated in differentiation medium including crude extract for 21 days and osteocalcin level in culture supernatant was evaluated as another osteoblast differentiation marker. When the cells were cultured with crude extract at concentrations of 0.01% and 0.1% for 21 days, osteocalcin level in culture medium was approximately 2.5‐3.0 times higher than that of control cells (Figure 2). Next, differentiation-inducing activity of the crude extract was evaluated. Addition of crude tuber extract to the maintenance medium showed no enhancement of ALP activity (Figure 3). Tuber extracts of groundnut enhanced osteoblast differentiation of MC3T3-E1 cells in the presence of ascorbic acid, hydrocortisone, and β-glycerophosphate. On the other hand, tuber extracts alone could not induce the differentiation of MC3T3-E1 cells to osteoblasts. Thus, the physiological activity of tuber extracts is enhancing differentiation rather than inducing it.

Evaluation of enhancement effects of crude groundnut extract on osteoblast differentiation of MC3T3-E1 cells. MC3T3-E1 cells were treated with a differentiation medium supplemented with 1%, 0.1%, and 0.01% crude extract of groundnut tuber for 7 days. Subsequently, ALP activity and cell viability were measured. The values of ALP activity and cell viability were normalized to the value of cells which had been cultured in a differentiation medium without crude extract. Negative control (NC) cells were cultured in a maintenance medium without crude extract. The values of ALP activity were corrected for the influence of cell number by values of WST-1 assay. **P < 0.01 (vs NC cells, ANOVA, Dunnett’s test), ## P < 0.01, # P < 0.05 (vs 0% cells, ANOVA, Dunnett’s test).
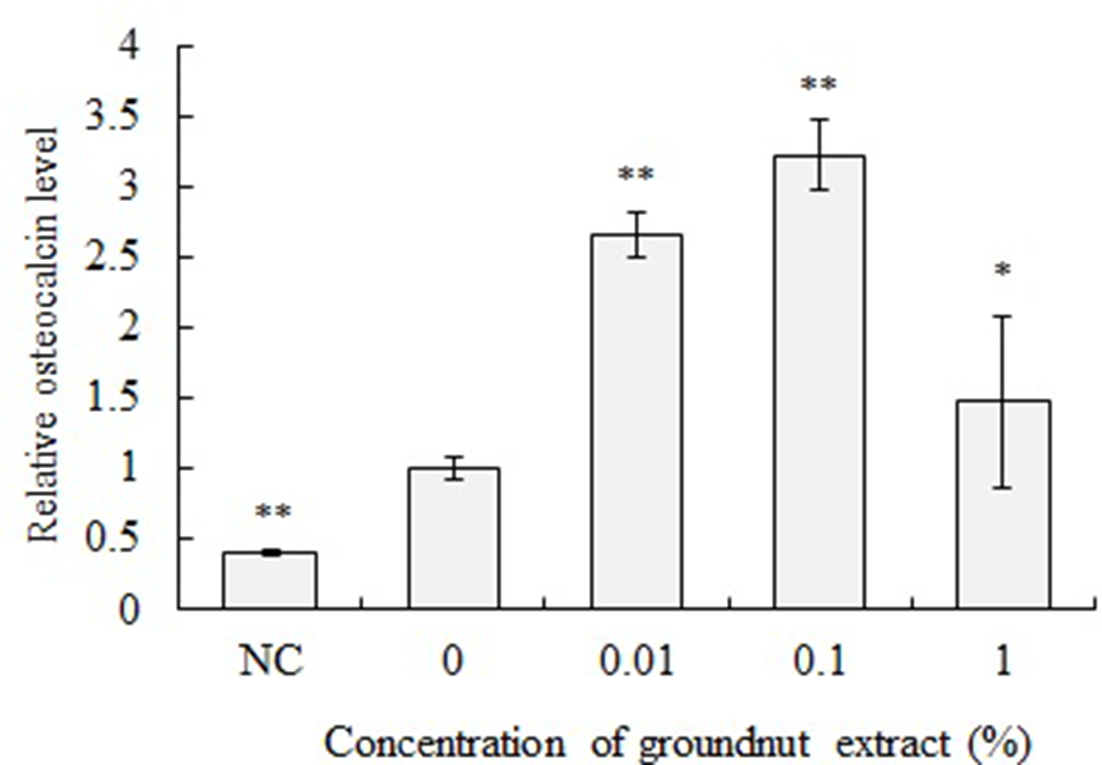
Effect of groundnut crude extract on osteocalcin level secreted by MC3T3-E1. MC3T3-E1 cells were treated with differentiation medium including 1%, 0.1% and 0.01% of crude extract of groundnut for 21 days. Osteocalcin levels in the culture supernatant were determined by ELISA. The osteocalcin level value was normalized to the value of cells which had been cultured in a differentiation medium without crude extract. Negative control (NC) was cultured with maintenance medium. **P < 0.01, *P < 0.05 (vs 0% cells, ANOVA and Dunnett’s test).

Evaluation of induction effects of crude groundnut extract on osteoblast differentiation of MC3T3-E1 cells. MC3T3-E1 cells were cultured in a maintenance medium supplemented with 1%, 0.1%, and 0.01% crude groundnut tuber extract for 7 days. Subsequently, ALP activity and cell viability were measured. Positive control (PC) cells were cultured in a differentiation medium without crude extract. The values of ALP activity and cell viability was normalized to the value of PC. The values of ALP activity were corrected for the influence of cell number by values of WST-1 assay. **P < 0.01 (vs 0% cells, ANOVA and Dunnett’s test).
Effects of crude groundnut extract on the viability of MC3T3-E1 cells were measured as mitochondrial activity using the WST-1 assay (Figures 1 and 3). When the cells were cultured in the differentiation medium supplemented with 1.0% crude extract for 7 days, viability was significantly decreased. Viabilities of cells cultured in the differentiation medium supplemented with 1% crude extract for 7 days were 78% of control cells. Addition of 0.1% and 0.01% crude extract did not reduce cell viability.
Effects of the Aqueous and Methanol Fractions of Tuber Extract on the Differentiation and Calcium Mineralization of MC3T3-E1 Cells
Next, groundnut tuber extract was separated into aqueous and methanol fractions, and the effects were examined of each fraction on osteoblast differentiation of MC3T3-E1 cells. The aqueous and methanol fractions were added to the differentiation medium at a 400-fold dilution, which was equivalent to the dilution ratio of 0.01% crude extract, and MC3T3-E1 cells were cultured in this medium. After incubation for 7 days, the ALP activity of cells was measured (Figure 4). The methanol fraction showed an ALP activity enhancement effect similar to that of the crude extract. However, the aqueous extract did not enhance ALP activity. Thus, the causal substance(s) of osteoblast differentiation enhancement activity in the tuber is water insoluble.

Effect of aqueous and methanol fractions of groundnut extract on osteoblast differentiation of MC3T3-E1 cells. MC3T3-E1 cells were cultured in a differentiation medium supplemented with 0.01% crude tuber extract and either its aqueous or methanol fraction (diluted 400 times; equivalent to 0.01% crude extract) for 7 days. Subsequently, ALP activity was measured. Control cells were cultured in a differentiation medium without tuber extract. Negative control (NC) cells were cultured in a maintenance medium. The values of ALP activity were corrected for the influence of cell number by values of WST-1 assay. **P < 0.01, *P < 0.05 (vs control cells, ANOVA and Dunnett’s test).
The methanol fraction of the tuber extract also enhanced expression of osteoblast differentiation markers in MC3T3-E1 cells. Expression levels of Bglap, Bsp, and Col1a1 at 7 days after incubation were determined (Figure 5). The expression levels of Bglap in cells treated with the crude extract and methanol fraction were respectively 5.3 and 9.2 times higher than those in untreated cells. Similarly, the expression levels of Bsp in cells treated with the crude extract and methanol fraction were approximately 2.3 times higher than those in untreated cells. The aqueous fraction did not enhance the expression of either of these genes. The expression of Col1a1 was also enhanced by treatment with the crude extract and methanol fraction.

Effects of aqueous and methanol fractions of groundnut extract on gene expression in MC3T3-E1 cells. MC3T3-E1 cells were cultured in a differentiation medium supplemented with 0.01% crude tuber extract and either its aqueous or methanol fraction (diluted 400 times; equivalent to 0.01% crude extract) for 7 days. Subsequently, the expression levels of Bglap, Bsp, and Col1a1 were evaluated by real-time PCR. Control cells were cultured in a differentiation medium without tuber extract. Negative control (NC) cells were cultured in a maintenance medium. **P < 0.01, *P < 0.05 (vs control cells, ANOVA and Dunnett’s test).
Calcium mineralization in the MC3T3-E1 cells treated with the groundnut extract was evaluated (Figure 6). Calcium mineralization in the MC3T3-E1 cells treated with the crude extract and methanol fraction was higher than that observed in the untreated cells. Meanwhile, the aqueous extract could not enhance calcium mineralization.

Effect of aqueous and methanol fractions of groundnut extract on calcium mineralization. MC3T3-E1 cells were cultured in a differentiation medium supplemented with 0.01% crude tuber extract and either its aqueous or methanol fraction (400 times; equivalent to 0.01% crude extract) for 21 days. Control cells were cultured in a differentiation medium without tuber extract. Negative control (NC) cells were cultured in a maintenance medium. Cells were stained with alizarin red S. **P < 0.01 (vs control cells, ANOVA and Dunnett’s test).
Osteocalcin, which is encoded by the Bglap gene, is secreted by osteoblasts and enhances bone formation and calcification. 20,21 Bone sialoprotein-1 (BSP) expresses in the early stage of calcification and is involved in calcification. 22 Type I collagen is also important for bone formation. Additionally, groundnut tubers have a high content of calcium, and, therefore, may be effective for prevention of brittle-bone disease.
. Although the causal substance is unclear in the present study, one of the possibilities may be isoflavone. Groundnut contains a unique isoflavone, G7G, which is a glycoside of genistein. 8 Genistein has been reported to enhance osteoblast differentiation. 13 The isoflavone content of the crude extract and each fraction is presented in Table 1. The methanol fraction of the groundnut extract included isoflavones, whereas the aqueous fraction included hardly any. Specifically, the G7G content of the methanol fraction was the highest. The differentiation enhancement effects of the crude extract and methanol fraction were similar. Concentrations of G7G, genistin and genistein in 0.01% crude groundnut extract were approximately 0.70, 0.14 and 0.01 µg/mL, respectively, and those in the methanol fraction diluted to an equivalent degree were 0.28, 0.05 and 0.01 µg/mL, respectively. Dosage of these isoflavones to the cell was in a similar range, approximately; cellular responses to the tuber extract were also similar.
Concentration of Isoflavones in Groundnut Extracts.

Effects of G7G, Genistin and Genistein on the Differentiation and Calcium Mineralization of MC3T3-E1 Cells
Next, the effect of these isoflavones on differentiation of MC3T3-E1 cells was evaluated. G7G, genistin and genistein were added to the differentiation medium at a final concentration of 0.1, 1.0 and 10 µg/mL and MC3T3-E1 cells were cultured in this medium. After incubation for 7 days, intracellular ALP activity was measured (Figure 7). After 7 days of incubation, the ALP activity of MC3T3-E1 cells cultured in the differentiation medium supplemented with 10 µg/mL of genistin and 1 and 10 µg/mL of genistein was significantly higher than that of the control cells. On the other hand, G7G did not affect ALP activity. Genistein showed cytotoxicity at a concentration of 10 µg/mL. When cells were incubated with 0.1 µg/mL of G7G, 0.1 and 1.0 µg/mL of genistin and 1.0 µg/mL of genistein, the osteocalcin level in the culture medium was significantly higher than that of control cells. Additionally, calcium mineralization was evaluated in the MC3T3-E1 cells treated with G7G, genistin and genistein (Figure 8). Calcium mineralization in the MC3T3-E1 cells treated with genistin and genistein was higher than that observed in the untreated cells. Meanwhile, G7G could not enhance calcium mineralization. These results suggest that genistein is most effective for differentiation of MC3T3-E1 cells to osteoblasts. The effect of G7G is smaller than that of genistin. On the other hand, there is the possibility that other undetected molecules in the groundnut extract are involved in the effect.
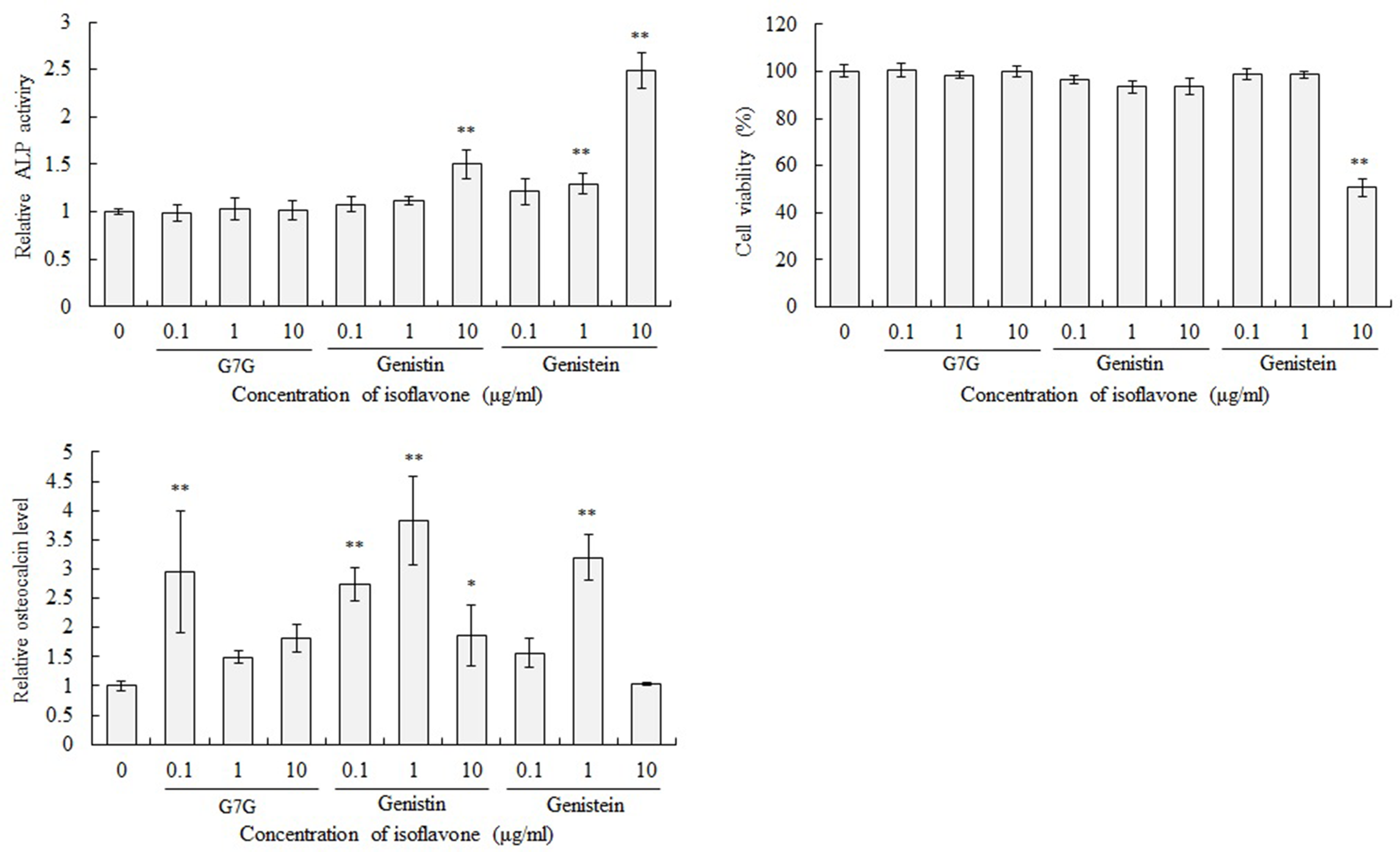
Evaluation of enhancement effects of isoflavones on osteoblast differentiation of MC3T3-E1 cells. MC3T3-E1 cells were treated with a differentiation medium supplemented with 0.1,1.0, and 10 µg/mL of G7G, genistin and genistein for either 7 (ALP activity and cell viability) or 21 (osteocalcin level) days. The values of ALP activity, cell viability and osteocalcin level were normalized to the value of cells which had been cultured in a differentiation medium without isoflavones. The values of ALP activity were corrected for the influence of cell number by values of cell viability. **P < 0.01, *P < 0.05 (vs 0 µg/mL cells, ANOVA, Dunnett’s test).
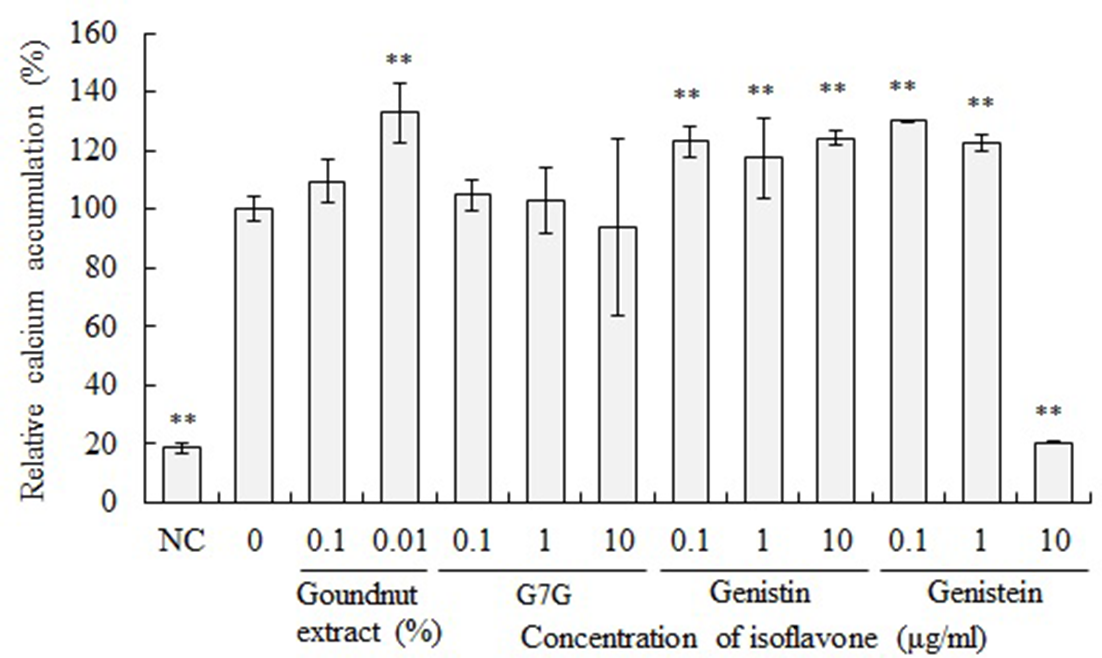
Effect of isoflavones on calcium mineralization. MC3T3-E1 cells were cultured in a differentiation medium supplemented with 0.1,1.0, and 10 µg/mL of G7G, genistin and genistein for 21 days. Control cells were cultured in a differentiation medium without isoflavones. Negative control (NC) cells were cultured in a maintenance medium. Cells were stained with alizarin red S. **P < 0.01 (vs 0% cells, ANOVA and Dunnett’s test).
Experimental
Preparation of Groundnut Extracts
Groundnut plants were separated into tubers and roots and stored at −20℃ until further use (Figure 1). The tubers were used in this study. These were chopped into small pieces with a knife before homogenizing. The pieces (100 g) were added to 3 volumes of methanol and homogenized in a blender. Following vacuum filtration, the residues were treated with 80% methanol for 2 successive extractions. The filtrates were combined and evaporated in vacuo, and the methanol extract was suspended in distilled water (crude extract fraction). Additionally, the crude extract fraction was applied to a Diaion HP-20 (20 × 2.5 cm i.d.) column and successively eluted with water and methanol to obtain 2 (aqueous and methanol) fractions. Both the fractions were evaporated in vacuo and suspended in distilled water. Purified G7G was prepared as in a previous study. 8 Genistein and genistin were purchased from FUJICCO Co., Ltd. (Kobe, Japan).
Cell Culture
Mouse calvarium-derived fibroblast-like MC3T3-E1 cells were purchased from RIKEN BioResource Center (Tsukuba, Ibaraki, Japan). MC3T3-E1 cells were cultured in minimum essential medium alpha (Gibco, Thermo Fisher Scientific Inc., Waltham, MA, USA) supplemented with 10% heat-inactivated fetal bovine serum (FBS; HyClone Laboratories, GE Healthcare, IL, USA), 100 units/mL penicillin, 100 µg/mL streptomycin, and 250 ng/mL amphotericin B (antibiotics mixture; Nacalai Tesque Inc., Kyoto, Japan). The medium, supplemented with 10% FBS and antibiotics, is referred to as the “maintenance medium” in this study. MC3T3-E1 cells were differentiated into osteoblasts by treatment with “differentiation medium” using Osteoblast-Inducer Reagent (for animal cells) (Takara Bio Inc., Kusatsu, Japan). “Differentiation medium” was prepared by adding ascorbic acid, hydrocortisone, and β-glycerophosphate to the maintenance medium, according to the manufacturer’s protocol. The cells were seeded in 96-, 12-, or 6-well plates at a density of 1 × 105 cells/mL in maintenance medium and incubated at 37 °C for 24 hours in an atmosphere containing 5% CO2. Subsequently, the maintenance medium was changed to the differentiation medium with or without groundnut extract, and the differentiation medium was replaced with fresh medium once every 2‐3 days.
Evaluation of Alkaline Phosphatase (ALP) Activity
ALP activity was measured using the TRACP & ALP Assay Kit (Takara Bio Inc.). MC3T3-E1 cells were differentiated with or without groundnut extract in a 96-well plate. After incubation for 7 days, the culture medium was removed and washed once with normal saline (0.9% NaCl). Then, 50 µL of extraction solution (normal saline with 1% of NP-40) was added, and the cells were lysed by pipetting. Next, 10 µL of cell lysate was transferred to a new 96-well plate, and 50 µL of ALP buffer [0.2M Tris–HCl (pH, 9.5) and 1 mM MgCl2] containing p-nitrophenyl phosphate as a substrate (supplied by manufacturer) was added, and the cells were incubated for 15 minutes at 37 °C. Reaction was stopped by adding 50 µL of 0.5M NaOH. Absorbance at 405 nm was measured using an Infinite F200 PRO microplate reader (Tecan Group Ltd., Männedorf, Switzerland).
Measurements of Cell Viability
To measure viability, cells were seeded in a 96-well plate and incubated for 24 hours. The maintenance medium was removed and replaced with differentiation medium with or without groundnut extract, and the cells were incubated for an additional 7 days. Cell viability was measured as mitochondrial activity using the WST-1 assay with a PreMix WST-1 Cell Proliferation Assay System (Takara Bio Inc.). After discarding the culture supernatant, WST-1 medium was added, and the cells were incubated at 37 °C for 1 hour. The optical density of formazan was measured at 450 nm using an Infinite F200 PRO microplate reader (Tecan Group Ltd.).
Analysis of Gene Expression
The cells were seeded in 6-well microplates and incubated for 24 hours at 37 °C. The maintenance medium was removed and replaced with the differentiation medium with or without groundnut extract, and the cells were incubated for an additional 7 days. The expression of target genes was determined using real-time polymerase chain reaction (PCR). Total RNA was extracted from the cells using the RNeasy Mini kit (Qiagen GmbH, Hilden, Germany). cDNA was synthesized using the High-Capacity cDNA Reverse Transcription kit (Applied Biosystems, Carlsbad, CA). Real-time PCR was performed on a 7300 Real-Time PCR System (Applied Biosystems), and the amplicons were detected using a TaqMan® gene expression assay (Applied Biosystems). The mouse β-actin gene was used as an endogenous control. The codes of TaqMan® probes for gene expression assays for mouse β-actin, type I collagen (Col1a1), bone gamma carboxyglutamate protein (Bglap1), and integrin-binding sialoprotein (Bsp) were Mm02619580_g1, Mm00801666_g1, Mm03413826_mH, and Mm00492555_m1, respectively.
Quantification of Osteocalcin in Culture Supernatant
Cells were seeded in a 24-well plate and incubated for 24 hours. Then the culture medium was removed. Subsequently, differentiation medium with/without groundnut extract was applied, and the cells were incubated for an additional 21 days. The culture supernatant was collected. Secretion of osteocalcin in the cell culture medium was determined by enzyme-linked immunosorbent assay (ELISA) using a Mouse Gla-Osteocalcin High Sensitive EIA Kit (Takara Bio Inc.).
Evaluation of Calcium Mineralization
Calcium mineralization in MC3T3-E1 cells was evaluated by alizarin red S staining. Cells were seeded in a 12-well plate and incubated for 24 hours. Then, the maintenance medium was removed and replaced with the differentiation medium with or without groundnut extract, and the cells were incubated for an additional 21 days. After discarding the culture supernatant, cells were washed with PBS twice. Then the cells were fixed with 70% ethanol for 1 hour at 4 °C and subsequently with distilled water, twice. Then, 300 µL of 1% alizarin red S solution (Muto Pure Chemicals Co., Ltd., Tokyo, Japan) was added to each well and incubated for 15 minutes at room temperature. After incubation, the cells were washed with distilled water 5 times. Next, 500 µL of PBS was added, and the cells were incubated for 15 minutes at room temperature. Excess PBS was removed by washing, and the cells were allowed to dry. Alizarin red S was eluted by incubation with 500 µL of 10% 1-ethylpyridinium chloride (Tokyo Chemical Industry Co., Ltd., Tokyo, Japan) for 15 minutes at room temperature. Then, 100 µL aliquots were transferred to a 96-well plate, and absorbance at 562 and 415 nm was measured using an Infinite F200 PRO microplate reader (Tecan Group Ltd.).
Statistical Analysis
The data are expressed as mean ± standard deviation. Statistical analyses were performed via analysis of variance (ANOVA) using Dunnett tests for multiple comparisons. The corresponding calculation method is described in each figure legend.
Conclusion
Groundnut extract showed an osteoblast differentiation enhancement effect in MC3T3-E1 cells. Additionally, groundnut extract also enhanced calcium mineralization. Isoflavones, particularly genistin, are involved in this effect.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Japan Society for the Promotion of Science (JSPS) Grant-in-Aid for Scientific Research (C) JSPS KAKENHI (Grant Number 16K00825).
