Abstract
Ginsenoside Rg5 is a rare secondary ginsenoside with good pharmacological activity. However, preparation of Rg5 is time consuming, gives a low yield and has poor selectivity and efficiency. Therefore, the current study describes an efficient method of preparing ginsenoside Rg5 through conversion of Protopanaxadiol (PPD) type saponins in the presence of a molecular sieve and hydrochloric acid. The PPD type saponins were transformed on the surface of a molecular sieve to obtain ginsenoside Rg5 and most of the generated secondary ginsenoside was dissociates into the reaction solution, avoiding further decomposition. In addition, the optimal experimental conditions were identified to be as follows; 0.5 ml of absolute ethanol, 0.2 mol/L of hydrochloric acid, 12.5 mg of a 4A molecular sieve, a reaction time of 2.5 hours and a reaction temperature of 70 ℃. Under these experimental conditions, yields of upto 63.2% were obtained for ginsenoside Rg5. Moreover, the hydrochloric acid-catalyzed preparation of ginsenoside Rg5 effectively inhibited its decomposition in the presence of a molecular sieve. The method is therefore suitable for laboratory and industrial production with the advantages of large-scale preparation and high-yield.
Keywords

Ginseng is the rhizome of Panax ginseng C. A. Meyer and has a complicated chemical composition. In addition, it has a wide range of biological activity, unique pharmacological properties and thousands of years of medicinal history in north-east Asia. 1,2 Notably, ginsenosides are the most important active ingredients of ginseng and have widely been studied with regard to such aspects as the separation processes, structural transformation and pharmacological activity. In particular, it has been found that rare ginsenosides such as Rg5, Rk1, Rg3, and Rh2, which can be formed by degradation of major ginsenosides, and were shown to have better pharmacological properties than the major ones. 3 -7
Among these unique compounds, ginsenoside Rg5 was one of the main components isolated from red ginseng by a Korean research team in 1996. 8 Rg5 has since been used for the prevention and treatment of various types of cancer, cardiovascular diseases, diabetes, memory impairment, and allergic diseases. 9 -15 Additionally, a previous study reported that ginsenoside Rg5/Rk1 had the strongest ability to rescue memory deficits in mice, induced by ethanol, out of the 3 compounds studied, that is, 20(S)-Rg3, 20(R)-Rg3, and Rg5/Rk1. 16 Moreover, Shin-Jung Kim et al. 17 reported that ginsenoside Rg5 has a greater impact than 20(S)-Rg3 in promoting apoptosis in the MCF-7 (HER2/ER+) and MDA-MB-453 (HER2 +ER) breast cancer cells. All these studies showed that ginsenoside Rg5 has a great potential as a natural remedy. However, it is yet to be fully developed due to its limited supply although the less active ginsenoside Rg3 has already been developed in China as a new anti-cancer drug known as the Shen-Yi Capsule. Therefore, it is necessary to develop a simple and efficient strategy for the synthesis of ginsenoside Rg5 in order to enhance research on the pharmacological activity of ginsenosides and related drugs.
Currently, ginsenoside Rg5 is mainly obtained by isolation from steamed ginseng (red ginseng or black ginseng). However, the processing time is relatively long 18 and the yield is also low. Ginsenoside Rg5 can also be prepared by structural transformation of Protopanaxadiol (PPD) saponins but the yield is similarly relatively low. For instance, Guan et al. 19 in 2015 utilized acid catalysis to synthesize ginsenosides Rk1 and Rg5 using a 50% ethanol solution and saponin from the stem and leaves of ginseng. They obtained a total yield of 19.88%. Additionally, Gui et al. 20 in 2020 explored the preparation of ginsenoside Rg5 from ginsenoside Rb1 with ferric chloride as the catalyst and obtained yields of up to 57.4%. Moreover, Huang et al. 21 in 2016 developed a method for the in situ preparation of ginsenoside Rg3 and Rg5 by extraction of PPD and Protopanaxatriol (PPT), with tartaric acid as the catalyst. The team reported a 94.5% conversion rate. For nearly a decade, our research group has been committed to the extraction and modification of rare ginsenosides, 22 -26 especially Rg5. 24 -26 As a result, our group 24 developed a methodology for the preparation of ginsenoside Rg5 from PPD with lemonade as a catalyst and yields of 30.8%. However, subsequent studies showed that hydrochloric acid might accelerate the hydrolysis of glycosidic bonds, 25,26 leading to more decomposition of Rg3 and Rg5.
Therefore, as an extension of our ongoing efforts to efficiently prepare Rg5, the current study speculated that a new synthetic strategy could be achieved if a suitable adsorbent was designed carefully (Figure 1(A)). The study hypothesized that a highly acidic reaction site would be formed on the surface of the adsorbent after which the PPD type saponins would be converted to Rg5 on this surface. Rg5 would then eventually diffuse into the solution with a low acid concentration, further inhibiting its decomposition. Notably, a molecular sieve is a crystalline aluminosilicate with a large surface area, regular hole structure as well as good selective adsorption and hydrophilic properties which make it an excellent adsorbent, catalyst and catalytic carrier. 27,28 Therefore, the effect of molecular sieve and hydrochloric acid on the preparation of Rg5 was investigated, the preparation method of Rg5 was optimized, and the role of molecular sieve in the preparation of Rg5 was discussed theoretically in present study. The preliminary results of this research are presented herein.

Experimental design for the transformation of PPD type saponins to ginsenoside Rg5.
Materials and Methods
Materials and Instruments
The PPD type saponin was prepared in our laboratory at Jilin Institute of Chemical Technology (93.7% purity, containing 47.65% ginsenoside Rb1, 19.80% ginsenoside Rc, 3.06% Rb2, 4.31% Rb3, and 18.88% Rd). The standards for Rb1, Rb2, Rb3, Rc, Rd, 20(S)-Rg3, 20(R)-Rg3, and Rg5, all at 98% purity, were purchased from the Chengdu Must Biotechnology Co., Ltd. (China), and the analytical grade type A molecular sieves (ground finely before use and activated for 2 hours at 400 °C), CaCl2, hydrochloric acid, anhydrous ethanol, n-butanol, and chemically pure blue silica gel, were purchased from the Tianjin Damao Chemical Reagent Factory (China). In addition, analytical grade MgSO4 and Na2SO4 were obtained from the Liaoning Quanrui Reagent Co., Ltd., China, while the High-performance Liquid Chromatography (HPLC)-grade acetonitrile was purchased from the Oceanpak Company (Germany), and HPLC-grade methanol was obtained from the Tianjin Damao Chemical Reagent Factory (China).
The High-performance liquid chromatography carried out with a P230P high-pressure constant flow pump; UV230 +ultraviolet-visible detector; EC-2000 LU workstation (P230, Dalian Elite). The morphology of molecular sieve was examined by scanning electron microscopy (SU3500, Hitachi). Silica gel 60 F254 thin-layer plate (Merck) was used for thin layer chromatography (TLC). The rotary evaporator (Re-2000A, Shanghai Yarong) and high-speed centrifuge (H2050R, Hunan Xiangyi) were used for concentration and centrifugation.
Preparation of Standard Stock Solutions
The ginseng saponin standards, Rb1 (1.955 mg), Rb2 (1.879 mg), Rb3 (1.534 mg), Rc (2.044 mg), Rd (2.147 mg), S-Rg3 (2.133 mg), R-Rg3 (2.144 mg), and Rg5 (2.612 mg) were accurately weighed, transferred into 10 ml volumetric flasks then dissolved in 5.0 ml of HPLC grade methanol. The solutions were sonicated in a bath sonicator for 10 minutes to ensure complete solubilization. After sonication, the volume was adjusted to 10 ml using HPLC grade methanol. Thereafter, the mixed stock solution was serially diluted (dilution factor = 1, 4, 10, 20, 40) to prepare working solutions of reference compounds. The working solutions were then filtered through a 0.22 µm PTFE syringe filter before subjecting them to HPLC analysis.
Thin-Layer Chromatography (TLC) Analysis
Solutions of the ginsenoside standards and reaction products were chromatographed on silica gel 60 F254 thin-layer plates (Merck) with CHCl3-MeOH-H2O (65:35:10, v/v/v, lower phase) as the development solution. The compounds were then visualized by spraying with 10% H2SO4, followed by heating at 105 ℃.
The HPLC Analytical Method for Rg5
The following conditions were used for chromatography 29 : inverse chromatographic column (250 mm × 4.6 mm, 5 µm), a detection wavelength of 203 nm in the UV detector, injection volume of 20 µL, a column temperature of 35 ℃ and a volumetric flow rate of 1 mL/min. The mobile phase consisted of acetonitrile (A) and water (B). Additionally, the following gradient elution procedure was used: 0∼10 minutes, 22% A; 10∼20 minutes, 22%∼27% A; 20∼25 minutes, 27%∼31% A; 25∼45 minutes, 31%∼38% A; 45∼60 minutes, 38%∼52% A; 60∼65 minutes, 52% A; 65∼75 minutes, 52%∼55% A; 75∼82 minutes, 55%∼60% A; 82∼82.10 minutes, 60% ∼90% A; 82.10∼100 minutes, 90% A; 100∼100.10 minutes, 90%∼22% A and 100.10∼115 minutes, 22%A.
In addition, equations for the calibration curves of each compound were as follows: yRb1 = 4.6529x + 7.8339 (R2 = 0.9999), yRb2 = 4.1232 x-4.8534 (R2 = 0.9997), yRb3 = 4.6275 x-1.88 (R2 = 0.9999), yRc = 4.8784 x-5.6868 (R2 = 0.9999), yRd = 5.5473 x-0.716 (R2 = 0.9998), y S -Rg3 = 7.312x + 5.8851 (R2 = 0.9999), y R -Rg3 = 8.3824 x-7.3741 (R2 = 0.9999), and yRg5 = 16.286 x-8.2291 (R2 = 0.9997). Good linearity was obtained for Rb1, Rb2, Rb3, Rc, Rd, S-Rg3, R-Rg3, and Rg5 within the range of 4.8875‐195.5 µg/mL, 4.6975–187.9 µg/mL, 3.835‐153.4 µg/mL, 5.11–204.4 µg/mL, 5.3675‐214.7 µg/mL, 5.3325–213.3 µg/mL, 5.36‐214.4 µg/mL, 6.53–261.2 µg/mL, respectively. Moreover, the limit of detection (LOD) under the aforementioned analytical conditions were determined at a S/N (signal to noise ratio) of 3. The LODs of Rb1, Rb2, Rb3, Rc, Rd, S-Rg3, R-Rg3, and Rg5 were 0.6, 0.6, 0.5, 0.6, 0.5, 0.4, 0.4, and 0.3 µg/mL, respectively. On the other hand, the limit of quantitation (LOQ) under the aforementioned analytical conditions were determined at a S/N of 10. The LOQs of Rb1, Rb2, Rb3, Rc, Rd, S-Rg3, R-Rg3, and Rg5 were 2, 2, 2, 2, 2, 1.5, 1.5 and 1 µg/mL, respectively.
The Strategy for Preparing Ginsenoside Rg5
Hydrochloric acid and molecular sieves were added to the PPD type saponin solution (0.5 ml, 10 mg/mL) while heating at 300 rpm for a certain time. The reaction mixture was then neutralized with a saturated sodium carbonate solution after which it was extracted using water-saturated n-butanol (3 × 500 µL). Thereafter, combined organic extracts were added into appropriate amounts of water then concentrated in vacuo at 45 ℃. Following this, Rg5 and the main by-products, 20(S)-Rg3 and 20(R)-Rg3, were qualitatively and quantitatively analyzed through TLC and HPLC. The residue was then dissolved in methanol (16 ml) and filtered using a 0.22 µm PTFE syringe filter.
Results and Discussion
Selection of a Solvent and Molecular Sieve for the Preparation of Ginsenoside Rg5
Ginsenoside Rg5 was synthesized under the following conditions: 5 mg of PPD type saponin in 0.5 ml of water, 50% ethanol and anhydrous ethanol, respectively, in the presence of 0.2 mol/L hydrochloric acid, without a molecular sieve. The experimental results are shown in Figure 2, Table 1, Supplemental Figure S1. Results from TLC (Supplemental Figure S1) and HPLC analysis (Figure 2, Table 1) showed that the type of solvent had a significant influence on the structural transformation of the ginsenoside. Additionally, quantitative analysis of the conversion products showed that the yields of 20(S)-Rg3 (retention time-61 min), 20(R)-Rg3 (retention time-62 min), and Rg5 (retention time-73 min) were 29.6%, 31.7%, and 9.6%, respectively, when water was used as the solvent, (Figure 2(A-e), Table 1, entry 1). The results showed that the total yield of ginsenoside Rg3 was 6.40 times more than that of Rg5. However, when 50% ethanol was used, yields of 25.4%, 27.1%, and 26.9% for 20(S)-Rg3, 20(R)-Rg3, and Rg5, respectively, were obtained (Figure 2(A-d), Table 1, entry 2). This indicated that the yield of ginsenoside Rg3 was 1.96 times more than that of Rg5. Moreover there was a significant decrease in the ratio of Rg3 to Rg5 in the ethanol-water solutions, suggesting that an increase in ethanol concentration led to an increase in the quantity of Rg5. When anhydrous ethanol was used as the reaction solvent, the yield of ginsenoside Rg5 increased further and became the main product (Figure 2(A-c), Table 1, entry 3). The results showed that ethanol was beneficial in improving selectivity for Rg5. Notably, the PPD raw materials were almost completely consumed. Furthermore, the total yield of ginsenosides Rg3 and Rg5 was only 41.7%, indicating that the 2 compounds were easily decomposed under catalysis by hydrochloric acid, consistent with existing literature. 21
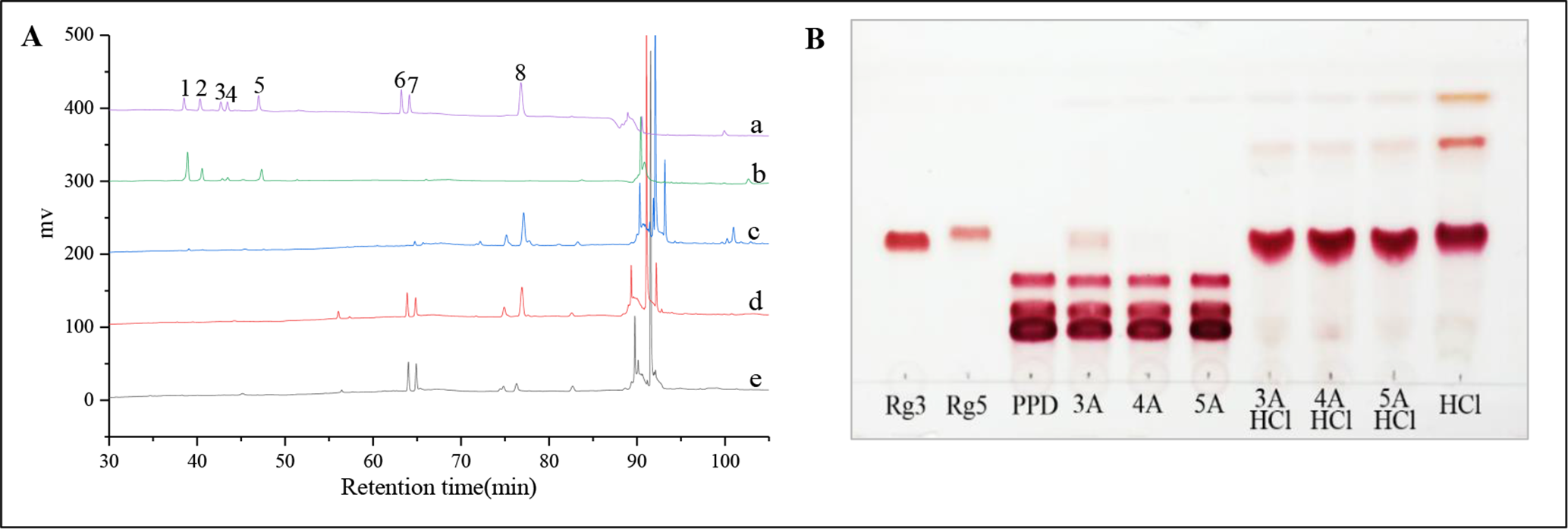
(A) A HPLC chromatogram of the conversion of PPD type saponin in different solvents (a, ginseng saponin standards; b, the raw PPD type saponins; c, conversion in EtOH; d, conversion in a mixed solvent containing ethanol and water (1:1, v/v); e, conversion in H2O. Peaks: 1, Rb1; 2, Rc; 3, Rb2; 4, Rb3; 5, Rd; 6, S-Rg3; 7, R-Rg3; 8, Rk1/Rz1; 9, Rg5.); (B) TLCs of the preparation of ginsenoside Rg5 in the presence of molecular sieves (3A, 4A and 5A were 3A MS, 4A MS, and 5A MS).
The Effects of Solvents and Molecular Sieves on the Yield of Ginsenoside Rg5. a

aReaction conditions: 5 mg of PPD type saponin, 0.5 ml of solvent, 15 mg of molecular sieve, 0.2 mol/L of hydrochloric acid, reaction temperature of 70 ℃, and a reaction time of 2 hours, unless otherwise stated;
b“-” means: no HCl was added; “+” means: HCl was added.
cH2O: EtOH = 1:1, v/v;
Moreover, 3A, 4A, and 5A molecular sieves (15 mg) were added separately into the solution of PPD type saponin in ethanol (0.5 ml, 10 mg/mL). Comparative experiments were then performed in triplicate under the same conditions, by adding a solution of hydrochloric acid (0.2 mol/L).
The results in Figure 2(B) and Table 1 show that the molecular sieves on their own had a very weak catalytic effect on the structural conversion of PPD type saponins (Figure 2(B); Table 1, entries 4‐6). However, when the transformation was catalyzed by hydrochloric acid coupled with the molecular sieves, most of the raw materials were consumed. Compared to the absence of molecular sieves, TLC analysis showed that the color of the decomposition product spots above ginsenoside Rg5 was significantly lighter. In addition, HPLC quantitative analysis showed that combining a molecular sieve with hydrochloric acid (Table 1, entries 7‐9) led to a significant improvement in the yield of ginsenoside Rg5, compared to the 34.2% obtained in the absence of a molecular sieve (Table 1, entry 3). The yield was also higher than that obtained from traditional methods. 18 -21 This indicates that addition of molecular sieves, especially 4A MS, may improve the yield of Rg5 by inhibiting its decomposition (Table 1, entries 8). Notably, PPD type saponins were mainly transformed into ginsenoside Rg5 and 20(S)-Rg3, with high stereoselectivity in a de of 92% for the 3A and 4A molecular sieves and 59% for 5A.
Optimization of Conditions for the Preparation of Ginsenoside Rg5
Optimization of the reaction temperature
Fifteen milligrams of the 4A molecular sieve was added into 0.5 ml of the PPD type saponin solution in ethanol (10 mg/mL) and 0.2 mol/L of hydrochloric acid. The solution was then stirred at 40‐80 ℃ for 2 hours. The results showed that there was an increase in the yield of ginsenoside Rg5 with a rise in the reaction temperature, reaching a maximum at 70 ℃ (Supplemental Table S1). Notably, the yield of ginsenoside Rg5 began to decrease when the reaction temperature increased to 80 °C. This indicates that the reaction efficiency can be accelerated with the increase of reaction temperature, but the decomposition of Rg5 will be increased at the same time.
Optimization of the amount of 4A molecular sieve
10 to 20 mg of the 4A molecular sieve were used for optimization. The sieve was added into 0.5 ml of the PPD type saponin in ethanol solution (10 mg/mL) and 0.2 mol/L of hydrochloric acid. The solution was then stirred at 70 ℃ for 2 hours. The results showed that the amount of molecular sieve had a specific effect on the yield of ginsenoside Rg5 (Supplemental Table S2). When 12.5‐15.0 mg of molecular sieve was used, yields of up to 62% were obtained. However, when the amount was either increased or decreased beyond this range (12.5, 15.0 mg), there was a decrease the yield of ginsenoside Rg5.
Optimization of the reaction time
Reaction times ranging from 1 to 3 hours were tested. Briefly, 12.5 mg of the 4A molecular sieve was added into 0.5 ml of the PPD type saponin solution in ethanol (10 mg/mL) and 0.2 mol/L hydrochloric acid (Supplemental Table S3) then stirred at 70 ℃. The yield of ginsenoside Rg5 increased with time up to 2.5 hours, after which it began to decrease, probably due to the decomposition of ginsenoside Rg5 in the presence of hydrochloric acid. In contrast, the yield of 20(S)-Rg3 increased with a rise in temperature indicating that 20(S)-Rg3 was more stable than ginsenoside Rg5. When the reaction time was 2.5 hours, the yield of ginsenoside Rg5 was 63.2% while that of by 20(S)-Rg3 was 11.2%, in a de of 72.4%.
Moreover, the experiment was repeated 3 times under the optimized conditions in order to analyze the repeatability of the reaction. The yields of Rg5 were 63.2%, 62.8%, and 63.1%, respectively, with an average yield of 63.0%. In addition, the Standard Deviation (SD) was 0.19 while the Relative Standard Deviation (RSD) was 0.30%, indicating that the reaction was repeatable.
Analysis of the roles for 4A molecular sieves in the conversion process
In order to clearly understand the role of 4A molecular sieves in the preparation of Rg5, several comparative experiments were carried out. First, the study selected several common water absorbents (MgSO4, Na2SO4, CaCl2, silica gel) for the control test, to ascertain the effect of water absorption on the preparation of Rg5. TLC analysis showed that the water absorbents did not effectively inhibit decomposition of the secondary ginsenosides, Rg3 and Rg5, in the absence of a molecular sieve (Supplemental Figure S2).
Additionally, HPLC analysis (Supplemental Table S4) showed that the yields of Rg5 and 20(S)-Rg3 both dropped by more than 60%, in the absence of the 4A molecular sieve. This suggested that secondary ginsenosides decomposed more in the presence of water-removing agents such as MgSO4, Na2SO4, CaCl2, and silica gel. This also showed that the 4A molecular sieve did not simply act as a water absorbent during the reaction process.
Second, a comparative experiment (Supplemental Figure S3) was designed to investigate the effect of adsorption to the molecular sieve. After 10 minutes of reaction in the presence of the 4A molecular sieve, the reaction mixture was centrifuged then the supernatant A was neutralized, concentrated and dissolved in HPLC-grade methanol (16 ml) for HPLC analysis. The remaining molecular sieve residue was washed twice with water-saturated n-butanol then the supernatant B was concentrated and dissolved in HPLC-grade methanol (2 ml) for HPLC analysis (Figure 3(A)). The results in Figure 3(A-a) show that only the secondary ginsenosides such as S-Rg3 and Rg5 appeared in the supernatant. In contrast, both secondary ginsenosides (S-Rg3 and Rg5) and PPD type saponins such as Rb1, Rc, Rb2, Rb3, and Rd were observed in the dissociated products of molecular sieves (Figure 3(A-b)). This indicated that molecular sieves can adsorb ginsenosides. In addition, PPD type saponins only appeared in the eluate of molecular sieves, indicating that they had a greater adsorption capacity for PPD type saponins than secondary ginsenosides such as Rg3 and Rg5.
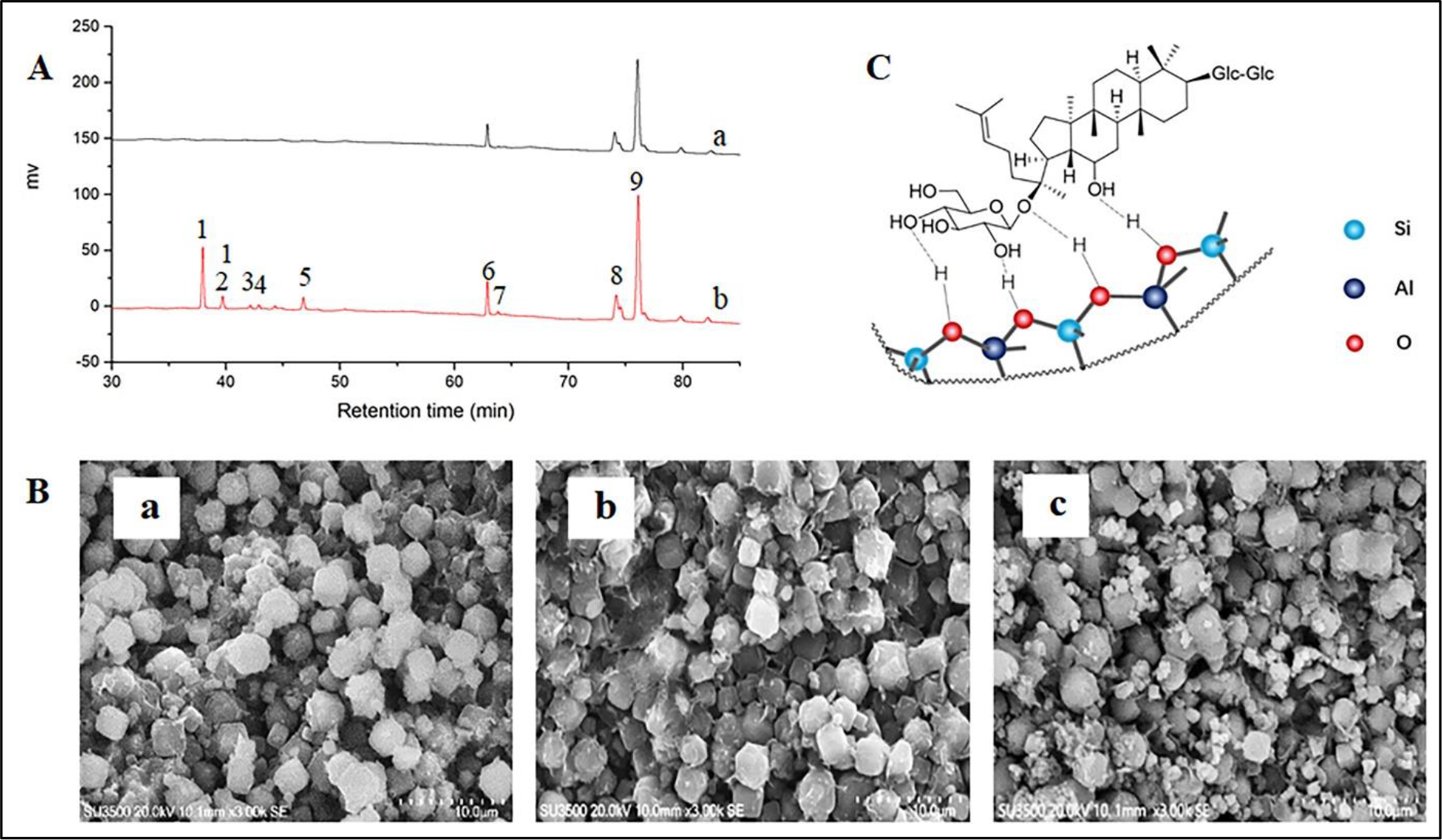
(A) A HPLC comparison chart of the supernatant and eluate of molecular sieves (a: the supernatant; b: the eluate of molecular sieves; 1: Rb1; 2: Rc; 3: Rb2; 4: Rb3; 5: Rd; 6: S-Rg3; 7: R-Rg3; 8: Rk1/Rz1; 9: Rg5); (B) A SEM image of molecular sieve zeolites (a: 4A molecular sieve; b: The PPD type saponin solution and 4A molecular sieve; c: PPD type saponin solution, 4A molecular sieve and hydrochloric acid); (C) Proposed surface and pore interaction of 4A molecular sieves with the PPD type saponin.
In order to further ascertain whether 4A molecular sieves adsorb ginsenosides, the study conducted HPLC and SEM analysis (Supplemental Figure S4, Figure 3(B)). The amount of the PPD type saponins in the supernatant and morphology of the 4A molecular sieve were analyzed for samples a (4A molecular sieve), b (a mixture of the PPD type saponin solution and 4A molecular sieve) and c (a mixture of the PPD type saponin solution, 4A molecular sieve, and hydrochloric acid). The results from HPLC analysis and SEM showed that the 4A molecular sieve hardly adsorbed the PPD type saponins in the absence of hydrochloric acid (Supplemental Figure S4-B, Figure 3(B-b)). However, the presence of hydrochloric acid led to a significant decrease in the amount of the PPD type saponins in the supernatant (Supplemental Figure S4C). The results also showed clearly that several small particles were attached to the surface of the 4A molecular sieve (Figure 3(B-c)). This showed that under the action of hydrochloric acid, a large amount of the PPD type saponins and protons were adsorbed on the surface of the molecular sieve through hydrogen bonds (Figure 3(C)). In other words, the 4A molecular sieve may act as a catalyst carrier and adsorbent in the process of acid-catalyzed conversion of PPD type saponins to the rare ginsenoside Rg5.
Furthermore, several additional experiments were conducted in order to gain insights on the reaction mechanisms (Figure 4(A)). Under the optimal conditions, the reaction for the preparation of Rg5 served as the control experiment a. In addition, ethanol, hydrochloric acid and the 4A molecular sieve were mixed and shaken. After separation, the supernatant was evaporated to dryness after which the PPD type saponin in ethanol solution was added, to make the comparative experiment b. In addition, the residue 4A molecular sieve was added to the PPD type saponin in ethanol solution, to make the comparative experiment c. As expected, the 4A molecular sieve with acid adsorption could effectively catalyze the conversion of PPD type saponin to Rg5 (Figure 4(B-c), Figure 4(C-c)). Interestingly, the catalytic effect of the evaporated supernatant was better than that of the residue 4A molecular sieve (Figure 4(B-c), Figure 4(C-c)). Under normal circumstances, the evaporated supernatant had no catalytic ability and this was verified by the control experiment d (Figure 4(B-d), Figure 4(C-d)). It can be clearly seen from residue 1 in Figure 4(A), that the supernatant contains a certain amount of veru small molecular sieves, which might have adsorbed the protons, leading to its catalytic ability. Interestingly, when the evaporated mixture of ethanol, hydrochloric acid, and the 4A molecular sieve was used as a catalyst, the highest yield of Rg5 was obtained, with the least decomposition (Figure 4(B-e), Figure 4(C-e)).

(A) A flow chart of the control experiment; (B) TLC plates of the preparation of ginsenoside Rg5 in the presence of a molecular sieve; (C) HPLC chromatograms of the control experiments. (1, Rb1; 2, Rc; 3, Rb2; 4, Rb3; 5. Rd; 6, Rg3; 7, Rk1/Rz1; 8, Rg5)
Based on these experimental results, a plausible reaction mechanism for this conversion was proposed. In the presence of hydrochloric acid, the PPD type saponins first combined with the 4A molecular sieves through hydrogen bonding to generate intermediate
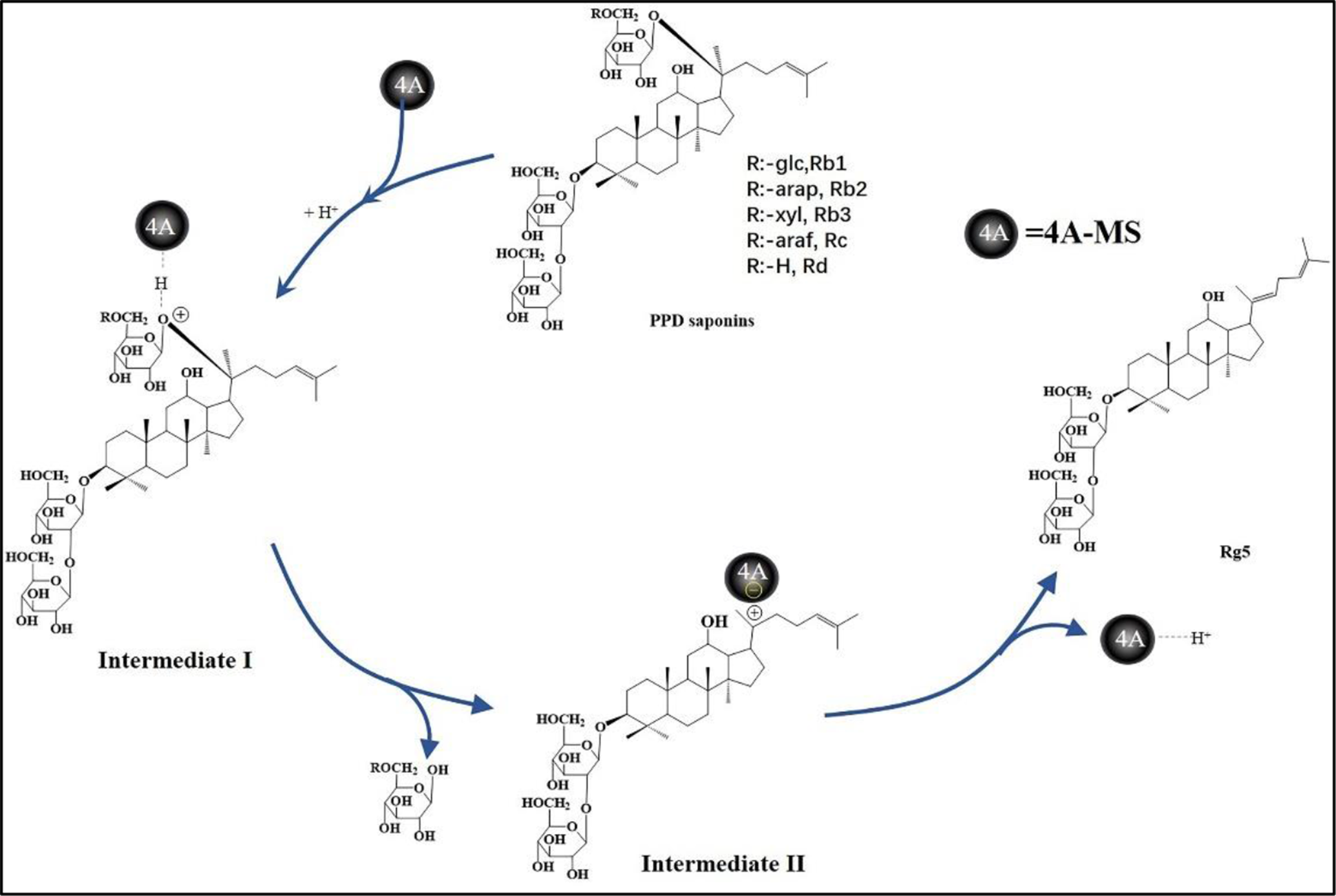
Possible reaction mechanism.
Conclusions
In summary, the study developed an effective method of synthesizing ginsenoside Rg5 through the conversion of PPD type saponins. In this conversion process, 4A molecular sieves played a vital role as catalyst carriers and adsorbents. Additionally, the strategy has the advantages of allowing for a large reaction scale, short time, less acid, and simple operation. Moreover, another important secondary ginsenoside, 20(S)-Rg3 was also produced, with high stereoselectivity. This simple methodology therefore provides an effective synthetic route for ginsenoside Rg5 for both laboratory and industrial production.
Supplemental Material
Supplementary Material 1 - Supplemental material for An Efficient Synthesis of Ginsenoside Rg5 via Conversion of PPD Type Saponins: Unusual Application of 4A Molecular Sieves
Supplemental material, Supplementary Material 1, for An Efficient Synthesis of Ginsenoside Rg5 via Conversion of PPD Type Saponins: Unusual Application of 4A Molecular Sieves by Cheng Le-qin, Ye An-qi, Zhang Hao-ran, Zhang Yue-wei and Li Ling in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by Jilin Provincial Science and Technology Development Program (20180201073YY) and Jilin Institute of Chemical Technology ([2015] No.005) in China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
