Abstract
The objective of our present study is to scrutinize the analgesic and anti-inflammatory potentials of essential oil of
Inflammation is a protective biological response to a variety of stimuli and local injury for restoring the normal tissue assembly and function. 1 The acute inflammatory process is advantageous and comprises a series of cellular responses such as the release of the inflammatory mediators and the generation of reactive oxygen species (ROS). However, excess inflammatory mediators and toxic ROS in persistent inflammation induce a chronic inflammation which can lead to numerous fatal diseases such as cancer, diabetes, atherosclerosis, rheumatoid arthritis, neurological diseases and aging. 2 Although non-steroidal anti-inflammatory drugs (NSAIDs) are commonly used for treating inflammation they may result in gastric injury, ulceration and renal damage. 3 Similarly, potent analgesic opioids are frequently associated with addiction and dependence. 4 Therefore, preference for herbal medicines is growing day by day in the treatment of pain disorders and inflammation. 5
With the advancement of computer technology, in silico approaches have been commonly applied in efforts to illuminate the pharmacological basis of the purposes of traditional therapeutic plants. 6 Virtual screening and molecular docking study help us to predict the mechanism of action of chemical compounds of relevant medicinal plants. Based on in silico results, we can execute in vivo and in vitro biological tests for corroboration. Therefore, computer-aided docking techniques will greatly increase the efficiency of evaluating the biochemical activities of medicinal plants. 7
Materials and Methods
Drugs, Chemicals, and Apparatus
Methanol was bought from SIGMA (Sigma-Aldrich, St. Louis, USA). Pentazocine and Diclofenac Na were obtained from Beximco Pharmaceuticals Ltd., Bangladesh. Heparin inj. was purchased from Rotexmedica, Germany. All the chemicals and reagents were of analytical grade.
Collection of Plant Material
Leaves of
Extraction of Essential Oil
The essential oil was obtained by hydro-distillation using a modified Clevenger apparatus coupled to a 2-L round bottom flask. A total of 1000 g of fresh plant material and 5 L of water were used for the extraction. Extraction process was performed in the Laboratory of Natural Product Research at Jahangirnagar University. The extraction was performed over an 8 hours period. The oil was transferred to black-colored vials, wrapped in parafilm and aluminum foil and stored at 4 °C until analysis. The yield (57 g) of the oil was calculated on the basis of the dry mass.
Animals and Experimental Setup
Sprague-Dawley female rats of 140‐160 g and Swiss albino female mice of 25‐30 g were obtained from Pharmacology Laboratory, Department of Pharmacy, Jahangirnagar University, and were acclimatized to normal laboratory conditions for 1 week prior to the study and were supplied with a pellet diet and water
Acute Toxicity Study
The acute oral toxicity test was performed following the guidelines of the Organization for Economic Cooperation and Development (OECD) for testing of chemicals, (Test Guideline 425) with some minor modifications (OECD 2008). Swiss albino male mice (25, 30 g) (
Evaluation of Anti–Nociceptive Activity
Acetic acid induced writhing method
The method according to Hossain et al. was employed for this test. 16 Five groups (each group having six mice) were pretreated with water, diclofenacNa (100 mg/kg) and the extract (100, 200 and 400 mg/kg). Forty-five minutes later each mouse was injected i.p. with 0.7% acetic acid at a dose of 10 mL/kg body weight. After 15 minutes of i.p. administration of acetic acid, subsequent 5 minutes period and the number of writhing responses was recorded for each animal. The mean abdominal writhing for each group was obtained. The percentage inhibition of writhing was calculated using the following formula:

Tail immersion test
The tail immersion method is widely used for the evaluation of analgesic activity (a central mechanism). In this method, the pain is induced to the animal by thermal stimulus and it is done by dipping the end of the tail into hot water.
17
In this test, we use 30 mice, which were fasted for 16 hours with water
Hot–plate test
In the hot-plate test, the mice are screened on the basis of their response when they are subjected to hot-plate. In this test, the test groups and the control group are treated as described previously at the proper dose. Here in this test the positive control received tramadol (5 mg/kg, i.p.). The pain is induced to the animals by placing the animals on hot-plate and temperature is maintained within 55 ± 0.5 °C range. 18 The response to a pain stimulus is indicated by paw licking or jumping off the plate. The response time is recorded for each group at 0, 30, 60, 120, and 180 minutes throughout the observation period. To avoid any accidental paw damage, the cut-off point of 15 s was considered. The reaction time of the test drug was compared with the control group.
Formalin-induced hind paw licking in mice
This experiment was followed by a similar procedure that was previously described by Mondal et al. 19 In the first phase, the animals were analyzed by formalin-induced licking. A subcutaneously injected 20 µL of 2.5% formalin solution (0.9% formaldehyde) made in phosphate buffer solution (PBS concentration: NaCl 137 mM, KCl 2.7 mM, and phosphate buffer 10 mM) under the surface of the right hind paw introduced to the animals. The time that the animals take licking the injected paw is considered as induction of pain. The initial nociceptive response normally peaks 5 minutes after the injection and later on 15‐30 minutes after formalin injection, representing the neurologic and the inflammatory pain response, respectively. All the mice were fasted for 24 hours before the introduction of the treatment but were allowed to access to water. A randomized 5 groups of mice, each containing 6 mice were used in this experiment. The control group received 10 mL/kg of normal saline, the standard group received diclofenac Na (100 mg/kg body weight) and the other 3 groups received 100, 200, and 400 mg/kg doses of the extract. The responses were recorded 5 minutes after (first phase) and 15‐30 after formalin injection (second phase).
Evaluation of Anti–Inflammatory Activity
Xylene induced ear edema test
Xylene-induced ear edema test was performed as it was described by Ramproshad et al. with a simple modification. 20 The positive control mice group received diclofenac Na (100 mg/kg) orally and other groups received plant extract of different (100, 200, and 400 mg/kg) concentrations. After 1 hour, each of the animals received 20 µL of xylene on the anterior and posterior surface of the right ear lobe. Here, in this case, the left ear was considered as control. The mice were sacrificed 1 hour after xylene application. After sacrificing the mice, a circular section was collected using a cork borer (which had a diameter of 3 mm) and the mass obtained using an electronic balance. The weight of edema was considered as the difference between the weight of ear treated with xylene (right ear) and weight of ear without xylene treatment (left ear). The percent inhibition can be calculated by from the following equation:

Cotton-pellet-induced granuloma model in rats
For this test, we divided the rats into 5 different groups and each of the groups having 6 rats. Inflammation was induced by the method described by Mondal et al. with a simple modification. 19 Here we used ketamine (50 mg/kg) to anesthetize the animals. The back skin was shaved and an incision was made at the lumber position followed by disinfecting with 70% ethanol. Sterilized and blunted forceps were used to make a subcutaneous tunnel and a pre-weighed cotton pellet (20 ± 1 mg) was placed at both sides in the scapular region. The control group was treated with water while the standard group was treated with diclofenac Na (100 mg/kg). The remaining 3 groups were treated with 100, 200, and 400 mg/kg of the plant extract respectively for 7 days. On the eighth day, the animals were sacrificed and the cotton pellets were removed from the back. These cotton pellets were then dried in the oven at 60 °C until the weight became stable. The net dry weight (initial- final) of the cotton pellets were determined by the following formulas:


Carrageenan-induced paw edema in rats
In this test, the acute inflammation was produced by injecting a 1% solution of carrageenan into the plantar surface of the left hind paw at a dose of 0.1 µL/gm body weight which was previously described method by Mondal et al. 19 with a simple modification. Here in this test, we used 5 groups of rats and each of the groups having 6 rats. 21 The control group received only the vehicle while the standard group received diclofenac Na (100 mg/kg, p.o.). The other 3 groups of rats were treated with different extractive concentrations of plant extract such as 100, 200 and 400 mg/kg, p.o., respectively. Thirty minutes after the administration all the 5 groups of rats were treated with a carrageenan solution. The paw volume was measured by dipping the foot into the mercury bath up to the anatomical hairline on lateral malleolus which was then compared with the control group which was treated only with a vehicle. The paw volume was measured by the digital plethysmometer (PLM 01, Orchid scientific, and Mumbai, India) which works on the principle of mercury displacement. The measurement was done as soon as possible before 1st, 2nd, 3rd, 4th, and 5th hour following carrageenan injection. The following formula was used to calculate edema inhibitory activity:

Evaluation of in-Silico Analgesic and Anti–Inflammatory Activity
In silico molecular docking analysis of the leaf essential oil of
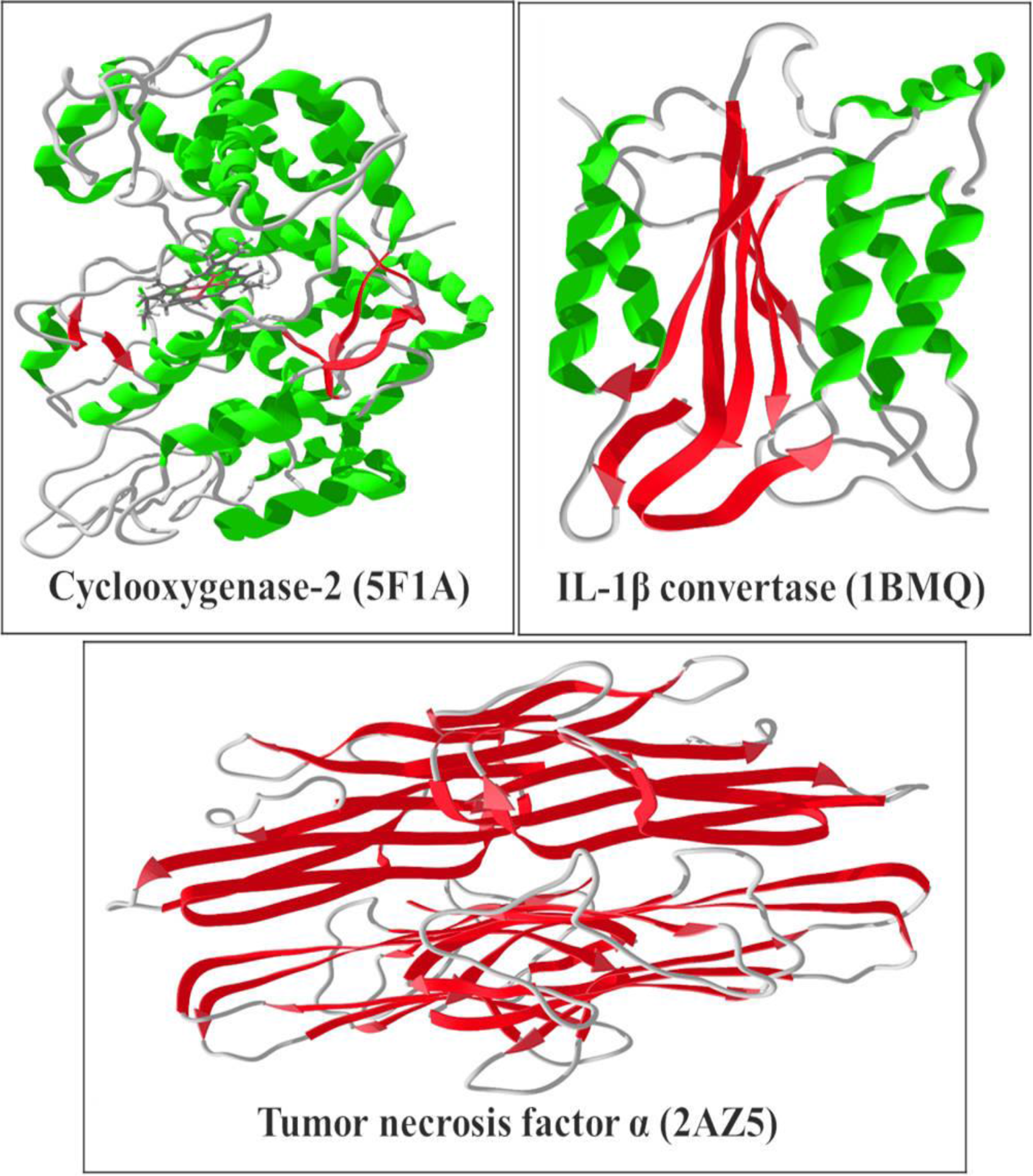
Ribbon structures of the proteins prior to docking.
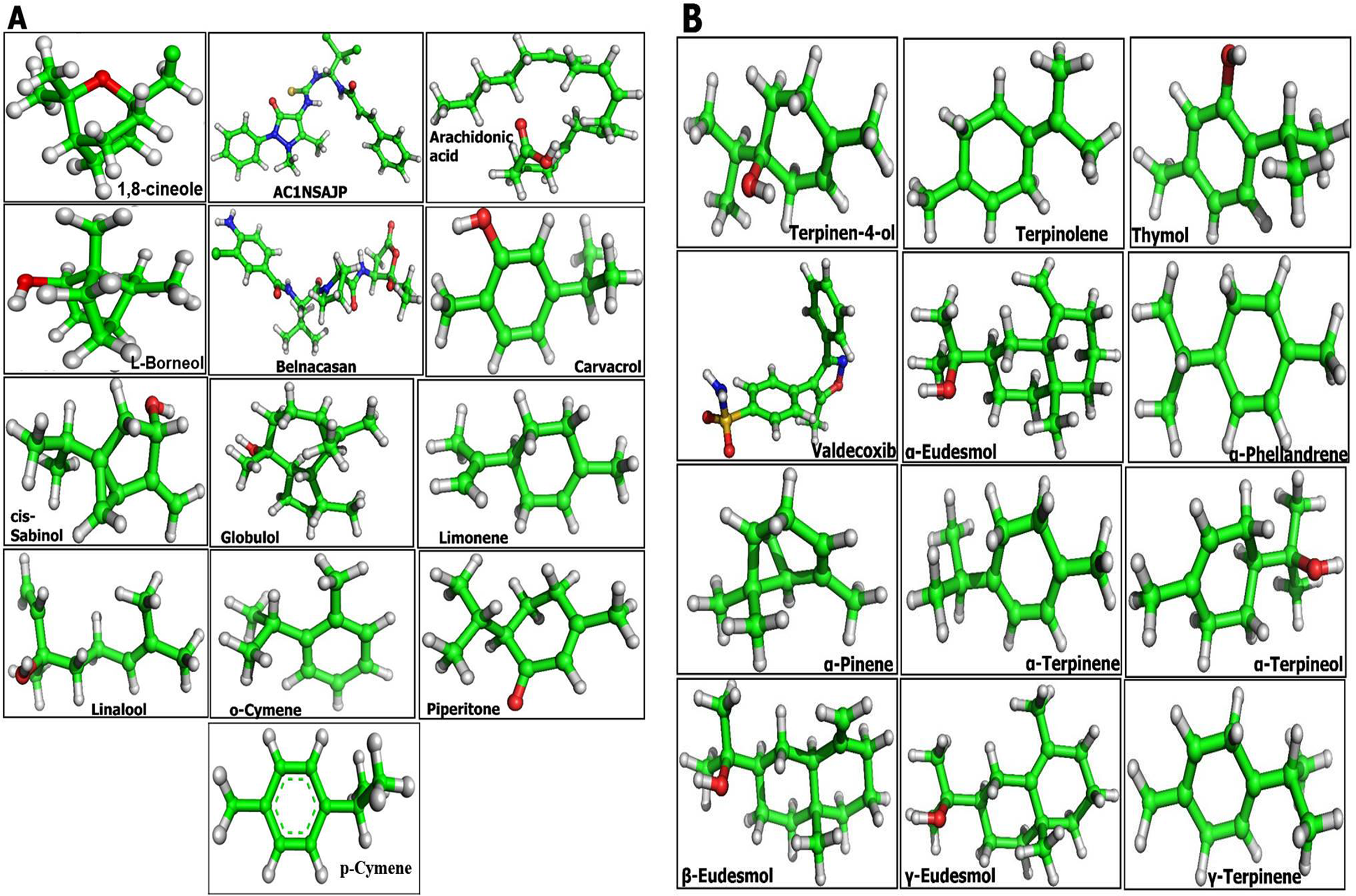
3D structures of Compounds.
Results
Acute Toxicity Study
Oral administration of the highest dose (800 mg/kg) of the leaf essential oil of
Evaluation of Anti-Nociceptive Activity
Acetic acid-induced writhing test
Table 1 shows the dose-dependent effect of
Antinociceptive Effect of Essential Oil of

Values are presented as mean ± SEM (
Tail immersion test
Table 2 indicates that the essential oil of
Antinociceptive Effect of Essential Oil of

Values are presented as mean ± SEM (
Hot–plate test
Table 3 demonstrates the anti-nociceptive activity of the essential oil of
Anti-Nociceptive Activity of Essential Oil of

Values are presented as mean ± SEM (
Formalin-induced hind paw licking in mice
Table 4 shows that the anti-nociceptive effects of
Effect of Essential Oil of

Values are presented as mean ± SEM (
Evaluation of Anti–Inflammatory Activity
Xylene-induced ear edema test
The following Table 5 presents the effects of
Effect of Essential Oil of
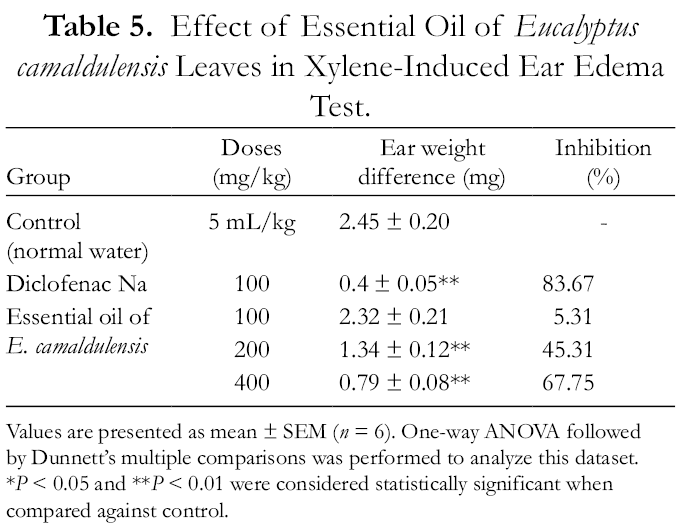
Values are presented as mean ± SEM (
Cotton-pellet-induced granuloma model in rats
The following table (Table 6) demonstrates the inhibition of granuloma formation by the leaf essential oil of
Chronic Anti-Inflammatory Activity of Essential Oil of

Values are presented as mean ±
Carrageenan-induced paw edema in rats
Anti-inflammatory activity of the essential oil of
Effects of Essential Oil of

Values are presented as mean ± SEM (
Rate of Inhibition of Rat Paw Edema (%).

Table 8 presents the rate of inhibition of edema. The highest rate of edema inhibition was 89.50% by diclofenac Na 100 mg/kg. On the other hand, 400 mg/kg dose of
Evaluation of in-Silico Anti–Inflammatory Activity
Interaction and binding affinity of compounds with COX 2 (5F1A)
We used valdecoxib (PubChem CID: 119607), arachidonic acid (PubChem CID: 444899) and 21 major chemical constituents of the essential oil of
Comparative Affinity Scores of Ligands Against Diverse Inflammatory Receptors.

aRepresents positive controls as a standard drug.
bRepresents positive controls as a standard natural substrate.
Interacting Residues of Diverse Inflammatory Receptors Against Various Ligands of

aRepresents positive controls as a standard drug.
bRepresents positive controls as a standard natural substrate. HBR represents hydrogen bonding residues. π-Int represents hydrophobic interactions. Red-colored texts indicate amino acid residues involved in the binding of standard drugs. Green-colored texts indicate amino acid residues involved in the binding of standard natural substrates. Blue-colored texts indicate amino acid residues involved in the binding of both standard drugs and standard natural substrates. Here, we only include the hydrogen bond and hydrophobic bond.
Interaction and binding affinity of compounds with TNFα (2AZ5)
Here, the binding affinities of tested compounds with TNFα (2AZ5) are displayed in Table 9. Standard drug 6, 7-dimethyl-3-((methyl-(2-(methyl-((1-(3-(trifluoromethyl) phenyl)indol-3-yl) methyl) amino) ethyl) amino) methyl)chromen-4-one (AC1NS1JP) (PubChem CID: 5331194) showed binding affinity (−6.8 kcal/mol) where TNF-alpha-IN-1(PubChem CID: 10270102) as standard natural substrate exhibited 6.4 kcal/mol affinity. β-Eudesmol (−6.8 kcal/mol), out of twenty compounds, showed a higher affinity for TNFα than that of TNF and AC1NSAJP. Additionally, γ-eudesmol (−6.5 kcal/mol) exceeded TNF in the case of binding affinity. The AC1NSAJP binding pocket was identified with the following amino acid residues: GLN61, TYR119, PRO117, TYR119, TYR115, and LEU63. However, TNF targets Tyr59 and Tyr151 of TNFα by forming π interactions, which were similar for β-eudesmol, γ-eudesmol, α-phellandrene, and terpinolene (Table 10).
Interaction and binding affinity of compounds with interleukin 1β convertase (1BMQ)
Here, we applied Belnacasan (L-Prolinamide, N-(4-amino-3-chlorobenzoyl)−3-methyl-L-valyl-N-[(2R,3S)−2-ethoxytetrahydro-5-oxo-3-furanyl]-Belnacasan) (PubChem CID: 11398092) as a standard drug for comparing the binding affinity of all the compounds of
Discussion
This study was accomplished to explore the anti–nociceptive and anti-inflammatory effects of leaf essential oil of
Owing to peripheral nociceptive sensitization, acetic acid triggered an abdominal writhing response. Acetic acid-induced writhing reflex model in mice is also related to increased levels of prostanoids in general, for example, prostaglandin E2 (PGE2) and prostaglandin F2α (PGF2α) as well as lipoxygenase products in peritoneal fluids.
23
-26
Intra-peritoneal injection of acetic acid moreover instigates the discharge of endogenous substances like bradykinin, serotonin, and histamine, which encourage the central nociceptive neurons.
27,28
Chemicals that inhibit the acetic acid-induced writhing may have an analgesic effect probably via the inhibition of prostaglandin biosynthesis, which is actually known as a peripheral mechanism of pain inhibition. The significant analgesic effects were observed for the essential oil of
A thermal nociception model like tail immersion method was used to evaluate the centrally-acting analgesic activity, which is known to elevate the neurological pain threshold of mice toward heat.
29
The tail-withdrawal response, an acute pain model is predominantly selective for centrally acting analgesics, implicating supraspinal analgesic pathways that are similar to the action of opioid agonists. The significant increase (*
Another well-known model of thermal nociception, the hot-plate test was employed to check on the possible involvement of spinal, supraspinal pathways, and μ-opiate receptor agonists in regulation (CNS modulation) of pain response.
31
Both hot plate and tail immersion tests are extensively used for evaluating central anti-nociceptive activities. Opioid agents display their analgesic effects both via supra-spinal and spinal receptors.
32
The present experiment, leaf essential oil of
The formalin-induced hind paw licking response was used as a model for evaluating analgesics.
33
Mice were subcutaneously administered with 20 µL of 2.5% formalin solution (0.9% formaldehyde) on the dorsal part of the mouse hind paw. The formalin-induced pain that was long-lasting and classified into 2 phases as follows: early phase (0, 5 min), formaldehyde directly excites nerve endings; late phase (15, 20 min), inflammatory mediators are produced and released.
33,34
Previous studies verified that bradykinin participates in the first phase, whereas histamine, serotonin, prostaglandins, NO and bradykinin were involved in the second phase of the formalin test.
35
This experimental study showed that the leaf essential oil of
Xylene-induced ear edema is a commonly used acute inflammation model. This method has been frequently used to initiate acute inflammatory response which leads to serious edematous changes and vasodilation of skin when topically applied to the surfaces of the ears of mice.
36,37
Xylene initiates the release of inflammatory mediators, which promotes vasodilation and increasing vascular permeability, and causes ear edema.
36
In our study, it was shown that the essential oil of
An excellent chronic inflammatory model, the cotton pellet granuloma in rats model, was selected to investigate chronic inflammation in the proliferative phase. This technique can be easily used for identifying diverse inflammatory responses like extravasations, the granuloma formation, and numerous biochemical exudates due to cotton pellets.
38
In our study, both 200- and 400 mg/kg doses exhibited significant reduction (
To further ascertain its anti-inflammatory activity, a carrageenan-induced paw edema test was performed. Carrageenan-induced edema is commonly used as an experimental model for acute inflammation and is proven to be biphasic where carrageenan is known to result in the step-wise discharge of the inflammatory endogenous mediators such as histamine, serotonin, and bradykinin, which are released in the initial phase of the inflammatory response, and prostaglandins, which are released in the late phase.
33,39
After carrageenan application, the paw edema was significantly inhibited at the dose 200 and 400 mg/kg in 1st, 2nd, 3rd and 4th hour where diclofenac Na 100 mg/kg (standard) also inhibited paw edema significantly in 1st, 2nd, 3rd and 4th hour and the 400 mg/kg dose of
Cyclooxygenase (COX) enzyme produces prostaglandins from arachidonic acid. Prostaglandins are important for signaling and housekeeping role in platelets, the gastrointestinal tract, lungs, and kidneys.
40,41
There are 2 isoforms of cyclooxygenase: one is cyclooxygenase-1 (COX-1) which is constitutive and another is cyclooxygenase-2 (COX-2) which is induced by cytokines and activated to cause inflammation.
42
Selective inhibitors of COX-2 are known to increase the risk of cardiotoxicity.
43
Valdecoxib is a selective COX-2 inhibitor with less effect on platelet aggregation and displays bridged gastrointestinal complications.
44,45
Arachidonic acid is a natural substrate that is metabolized to both pro-inflammatory and anti-inflammatory eicosanoids during and after the inflammatory response, respectively. For this reason, valdecoxib and arachidonic acid were selected as the positive control against the twenty chemical components present in the essential oil of

Binding site ligand-proteins.
Tumor necrosis factor-alpha (TNFα) is a critical cytokine involved in various autoimmune diseases as it plays a widespread role in the establishment and maintenance of inflammation.
49
TNFα plays a pathogenic role in many inflammatory ailments by acting as a vital controller of inflammatory pathways.
50
We selected the well-known inhibitor of TNFα namely 6,7-dimethyl-3-((methyl-(2-(methyl-((1-(3-(trifluoromethyl) phenyl)indol-3-yl) methyl) amino) ethyl) amino) methyl)chromen-4-one (AC1NS1JP) as a positive control. This is responsible for promoting disassembly of TNFα subunits.
51
On the other hand, TNF-alpha-IN-1 is a potent anti-inflammatory cytokine in autoimmune-mediated demyelination.
52
For this, we also selected a natural substrate, namely TNF-alpha-IN-1, as a positive control for comparing the anti-inflammatory activity of all compounds to present
Interleukin-1 (IL-1) is the prototypic pro-inflammatory cytokine,
55
which is divided in 2 forms IL-1α, and IL-1β and in most studies, their biological activities are indistinguishable. Interleukin 1β converting enzyme is a cytoplasmic cysteine protease that cleaves interleukin1β to its bioactive form 17-kD protein.
56,57
For this reason, interleukin 1β converting enzyme or interleukin1β convertase (IL-1BC) plays a key role in interleukin 1β-mediated inflammation. We selected a well-known inhibitor of interleukin 1β convertase (1BMQ) namely belnacasan as a positive control and compared the affinity of this inhibitor against the major components of the essential oil.
58
α-Eudesmol (−7.5 kcal/mol) showed more affinity toward interleukin 1β convertase compared to belnacasan (−7.1 kcal/mol). β-Eudesmol (−6.8 kcal/mol) also showed good docking scores, comparable to the standard drug. Belnacasan targets ARG163 and SER229 of interleukin 1β convertase by forming hydrogen bonds while showing hydrophobic interaction with ALA141, ILE152, CYS136, ILE144, VAL279, ILE155, TRP145, and PHE231. α-Eudesmol showed interactions with ILE155, PRO277, VAL279, and PHE231 by hydrophobic interactions. 1,8-Cineole targeted the maximum number of analogous amino acid residues (ILE155, CYS136, ILE144, ALA141, ILE152, and TRP145) compared to a positive control. Seo
Conclusion
The anti-nociceptive and anti-inflammatory activities of essential oil of
Footnotes
Acknowledgments
The authors would like to express their thanks to the Department of Pharmacy, Jahangirnagar University for providing necessary support to perform this study.
Statement of Human and Animal Rights
All of the experimental procedures involving animals were conducted in accordance with the Guide for the Care and Use of Laboratory Animals, Eighth Edition, which was approved by the Biosafety, Biosecurity and Ethical Committee, Jahangirnagar University, Savar, Dhaka, Bangladesh, [Approval Number: BBEC, JU/M 2018 (1)3].
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
