Abstract
Polygonum cuspidatum (PC) has been used as traditional Korean medicine to treat various diseases including asthma, hypertension, cancer, and arteriosclerosis. In this study, we assessed the antibacterial and anti-inflammatory effects of PC ethanol extract. Persistent antibacterial activity against Streptococcus mutans for up to 23 days was observed when the extract was used at a concentration of 10 mg/mL. The minimum inhibitory concentration of PC against S. mutans was 0.2 mg/mL and the minimum bactericidal concentration was 10 mg/mL. We compared the antimicrobial activities of S. mutagens cultured with or without PC. Bacterial activity was observed only in the group where RE was not added. The anti-inflammatory effect of PC on RAW264.7 cells was assessed using the MTT assay; changes in nitric oxide (NO) production and inflammatory cytokine levels (tumor necrosis factor [TNF]-α and interleukin [IL]-6) were measured in the presence of PC. In lipopolysaccharide-induced RAW264.7 cells, PC inhibited NO production by 68.6% when used at a concentration of 50 µg/mL. The expression of TNF-α and IL-6 was reduced by PC in a concentration-dependent manner. These results suggest that the ethanol extract of PC could be used as a mouthwash component with antibacterial and anti-inflammatory effects.
Human dietary patterns are becoming more diverse, and the consumption of sugars is increasing, resulting in the weakening of immune functions and an increase in oral microbe communities. 1 Dental caries, the most common oral condition, are caused by various factors such as food, bacterial interactions in the dental plaque, and saliva, often accompanied by tooth destruction. 2,3 Although there are different types of bacteria in the mouth, the microbe most commonly associated with dental caries is Streptococcus mutans, 4 a Gram-positive facultative anaerobic bacterium.
The decomposition of carbohydrates in the food, particularly glucose and fructose, results in the release of lactic acid from bacteria and can cause dental caries. 5,6 Several antibacterial agents including fluoride, penicillin, and phenol derivatives have been widely used to inhibit the growth of these bacteria, and chlorohexidine, penicillin, erythromycin, and tetracycline are currently used to treat dental caries. However, these antibacterial agents cause various adverse effects such as tooth discoloration, damage to the oral mucous epithelium, ulcers, palate disorders, attachment of calcified deposits to teeth, digestive disorders, hypersensitivity reactions, and occurrence of antibiotic-resistant bacteria. 7 -10 To overcome these limitations, studies have focused on natural antibacterial substances from plants. For example, Coptis chinensis, Sophora flavescens, 11 Galla chinensis extract, 12 bamboo charcoal, 13 Diospyros kaki peel, Momordica charantia, and Canavalia gladiata extracts 14 are active against S. mutans.
Inflammatory responses in the mouth are caused by bacteria or viruses that produce plaque in the teeth and gums, with the resulting plaque gradually calcifying to form a scale on the surface of the teeth. Plaque and scale become breeding grounds for oral bacteria, leading to inflammatory gum diseases such as gingivitis and periodontitis. 15 Lipopolysaccharide (LPS), an endotoxin that causes inflammatory reactions in the human body, causes inflammatory responses by releasing nitric oxide (NO) and pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukins (IL)−1β, IL-6, and IL-12. These cell-released substances help to activate immune-cell defense against bacterial invasion 16 ; overproduction of these factors can cause damage to normal tissues. 17
The root of Polygonum cuspidatum (PC) Siebold et Zuccarinii, family Polygonaceae, has been used for many years as a folk remedy for diuresis, and as an emmenagogue and a sedative. In Korean and Asian traditional medicine, PC has been used to treat various diseases including asthma, hypertension, cancer, and arteriosclerosis. 18
The reported chemical composition of Reynoutria includes anthraquinones such as emodin, physcion, and chrysophanol, and flavonoids such as quercitrin, isoquercitrin, and reynoutrin isolated from the leaves of R. japonica and R. sachalinensis. Malic, tartaric, and citric acids have also been reported in the fresh sprouts of these plants. 19 Anthraquinones, stilbenes (eg, resveratrol), and their glycoside piceids have been isolated from the roots of R. japonica. 20 Extracts of PC have been shown to exhibit vasorelaxant, 21 antithrombotic, 22 angiogenesis inhibitory, 23 and skin photo-aging inhibitory 24 effects. However, the activity of PC extracts on oral bacteria has not yet been reported.
This study investigated the antibacterial activity of an ethanolic extract of PC against oral bacteria and assessed the anti-inflammatory effects of the extract when added as a mouthwash component.
Materials and Methods
Plant Material
The study used the rhizome and roots of PC gathered from Omnihub (Daegu, Korea) in May 2019. A voucher specimen (8800663008915) was authenticated by the plant managing director of Skin Immunology Laboratory (Jin Tae Lee) and has been deposited at the herbarium of Skin Immunology Laboratory Gyeongsangbuk-do, Republic of Korea. All plant materials were dried for 3 days in the shade and stored at −4 °C until extraction.
Sample Extraction
Dried PC (600 g) was extracted with 70% ethanol (1:9 v/v) 3 times for 24 hours at room temperature (22, 24 ℃). The solution was passed through a paper filter (Whatman No. 2; GE, Tokyo, Japan), the extract was concentrated under reduced pressure using a vacuum rotary evaporator (N-3010; EYELA, Tokyo, Japan), and freeze-dried (MCFD8508; IlShinBioBase, Dongducheon, Korea) for 96 hours. A total of 53.8 g PC ethanol-extract powder (yield of 8.97%) was obtained. The PC ethanolic-extract powder was dissolved in deionization water for use in the experiments.
Quantification of the Major Component in PC Extract
Emodin, the active component of PC extract, was identified using a Waters ACQUITY UPLC H-Class with a C18 analytical column (Waters Corporation, MA, USA; 2.1 mm × 100 mm, 1.7 µm particle size). Water was used as mobile phase A, and acetonitrile as mobile phase B (elution conditions: 0‐5 minutes, 5% B; 5‐15 minutes, 25% B; 15‐25 minutes, 50% B; 25‐55 minutes, 100% B; flow rate, 0.2 mL/min; injection volume, 5 µL; wavelength, 280 nm; column temperature, 40 °C). Before injection, the standard and sample were dissolved in methanol and filtered through a 0.45 µm membrane filter. All solvents and mobile phases were of HPLC grade, and the presence of emodin was confirmed by matching the retention time (28.4 minutes) with that of the standard. The content of emodin was 4.1% in the 70% ethanol extract of PC (Supplemental Figure S1).
Bacteria, Cell Strain, and Reagents
Streptococcus mutans (KCTC 3065), used to evaluate the antimicrobial effect of PC, and RAW264.7 cells, used to assess the anti-inflammatory effects of PC, were obtained from the Korea Collection for Type Cultures (KCTC, Jeonseup, Korea) and the Korea Cell Line Bank (Seoul, Korea), respectively. Brain heart infusion medium for cell cultivation was purchased from BD Difco (Franklin Lakes, NJ, USA), 3-(4,5-dimethythiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and 0.4% trypan blue stain were from Sigma (St. Louis, MO, USA), protein quantification reagent and lysis buffer were from Hyclone (Logan, UT, USA), and primary mouse anti-TNF-α and goat anti-IL-6 antibodies and secondary rabbit anti-mouse IgG and mouse anti-goat IgG were from Santa Cruz Biotechnology Inc. (Dallas, TX, USA).
Measurement of Antimicrobial Activity Using the Disc Diffusion Method
The antimicrobial activity of the sample was measured using the disc diffusion method. 25,26 Briefly, one colony of the strain was inoculated into the medium, activated for 24 hours, and then cultured 3 times. The test bacteria were incubated to obtain an optical density (OD) value of 0.8 at 660 nm, resuspended at a concentration of 1 × 106 CFU/mL, smeared on agar medium, and fixed with an 8 mm paper disc. After complete absorption of the sample into the disc, the cells were cultured for an additional 24 hours in an incubator at 37 °C, and the samples were evaluated by measuring the clear zone.
Determination of the Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
The MIC and MBC were measured to confirm the antibacterial activity of the sample. 27 The sample was diluted in a liquid medium; the test-bacteria concentration was 1 × 104 CFU/mL. The growth medium and sample (500 µL each) were inoculated with the test bacteria and added to a 24-well plate; serial two-fold dilution (broth microdilution method) was used to determine the concentration range. After incubation at 37 °C for 24 hours, the turbidity of the culture medium was determined, and the samples were spread on an agar medium. After incubation for an additional 24 hours, the minimum concentration at which inhibition of the test bacteria was observed (the MIC), 28 and the minimum concentration at which the growth of the test bacteria was not observed after incubation for 24 hours in the agar medium (the MBC) were determined.
Cell Strain Culture
RAW264.7 cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco, Thermo Fisher Scientific, USA) with 10% fetal bovine serum (Introgen Therapeutics, Houston, TX, USA) and 1% penicillin/streptomycin (100 U/mL; Hyclone, GE Healthcare Life Sciences, USA). They were incubated in a humidified atmosphere containing 5% CO2 at 37 °C.
Measurement of Cytotoxicity Using the MTT Assay for Safety Evaluation
Cell viability was measured according to the method described by Mosmann. 29 RAW264.7 cells were uniformly dispensed in a 96-well plate (0.18 ml) at a density of 2 × 104 cells/well. PC was prepared at different concentrations, and 0.02 ml of the solution was added. The cells were then cultured in a 5% CO2 incubator at 37 °C for 24 hours. Next, 0.02 ml of MTT solution (5 mg/mL; Sigma-Aldrich Co.) was added, and the solution was cultured for 4 hours. The culture solution was then removed, and 0.15 ml of DMSO was added to each well. After incubation at room temperature for 15 minutes, the absorbance of the sample was measured at 540 nm using an enzyme-linked immunosorbent assay (ELISA) reader (PowerWave XS; BioTek Instruments, Inc., Winooski, VT, USA).
Measurement of NO Production
The amount of NO produced by RAW264.7 cells was measured according to the method of Green et al. 30 The cells were seeded in six-well plates at 1 × 105 cells/mL and cultured for 24 hours. After reaching 80% confluence, the cells were washed twice with PBS, cultured for 24 hours using serum-free medium, and treated with different concentrations of PC. Dexamethasone (25 µg/mL) was used as the control. After 4 hours, 1 µg/mL LPS (Sigma-Aldrich Co.) was added to all wells except the control (Nor) for 4 hours. The supernatant was collected and reacted with the same amount of Griess reagent for 10 minutes. The amount of NO produced was determined by measuring the absorbance of the sample at 540 nm using an ELISA reader.
Measurement of TNF-Α and IL-6 Production
RAW264.7 cells were cultured in six-well plates at 1 × 105 cells/mL for 24 hours, followed by treatment with PC at different concentrations (12.5, 25, and 50 µg/mL) and 1 µg/mL LPS. After 18 hours, the supernatant from each well was collected, and the levels of TNF-α and IL-6 in the supernatant were measured using ELISA kits (R&D Systems, Minneapolis, MN, USA), according to the manufacturer’s protocol.
Statistical Analysis
Statistical analyses were performed using SPSS 10.0, and significant differences were verified at a statistical significance level of P < 0.05 using t tests and analysis of variance.
Results
Comparison of the Antimicrobial Activity of PC Extracts and Other Plant Extracts Against S. Mutans (KCTC 3065)
PC extracts and 8 extracts of 3 other plants (Syzygium aromaticum, 31 S. aromaticum, 32 and ginseng berry, 33 previously reported as having antimicrobial effects, were subjected to antimicrobial activity measurement using the disc diffusion method. Only the PC extracts showed antibacterial activity, and the ethanol extract showed higher antibacterial activity than the hot-water extract. Therefore, our subsequent experiments were performed using the 70% ethanol extract of PC.
Antimicrobial Sustainability Test of PC Against S. Mutans (KCTC 3065)
PC was added to the culture medium of S. mutans (1 × 106 CFU/mL) at various concentrations at different times, and the culture broth was tested to determine the persistence of antimicrobial activity. The culture medium (control) that did not contain PC did not exhibit antimicrobial activity. However, 1 mg/mL PC showed antibacterial activity after 1 day. In the culture medium containing 10 mg/mL PC, bacterial activity was completely inhibited, and sustainability was observed for up to 23 days. In the control group, bacterial growth continued to increase over time (Supplemental Figure S2).
Determination of the MIC and MBC
The MIC of PC against S. mutans was 0.2 mg/mL and the MBC was 10 mg/mL.
Determination of the Antimicrobial Activity of Mouthwash
The antimicrobial activity of mouthwash containing 10 mg/mL PC ethanol extract was determined and compared with that without the PC extract. The components of the mouthwash solution are detailed in Table 1 Colonies of S. mutans were observed in the group without PC, but not in the one with PC (Supplemental Figure S3).
Components of the Mouthwash Solution.

Effect of PC Extract on the Viability of LPS-Stimulated Macrophages
The viability of RAW264.7 macrophages was measured in the presence of PC using the MTT assay. The cells treated with 50 and 100 µg/mL PC extract showed 80.3% and 66.1% viability, respectively, compared with control. Since low toxicity was observed with samples containing PC extract at a concentration of 50 µg/mL or less, we subsequently used this concentration (50 µg/mL) for the following analyses (Figure 1.).

Viability of RAW264.7 cells treated with Polygonum cuspidatum (PC) extract. RAW264.7 cells were treated with various concentrations (12.5, 200 µg/mL) of PC. Cell viability was measured using the MTT assay. The results are presented as mean ± standard deviation of triplicate assays.
Effects of PC Extract on NO Production
The production of NO by RAW264.7 cells in response to PC extract was determined. Treatment with LPS alone resulted in a 5-fold increase in NO expression compared with that of untreated cells (control). Dexamethasone has a strong anti-inflammatory effect and was used as a positive control. We found that NO production was significantly reduced by PC in a concentration-dependent manner (Figure 2).
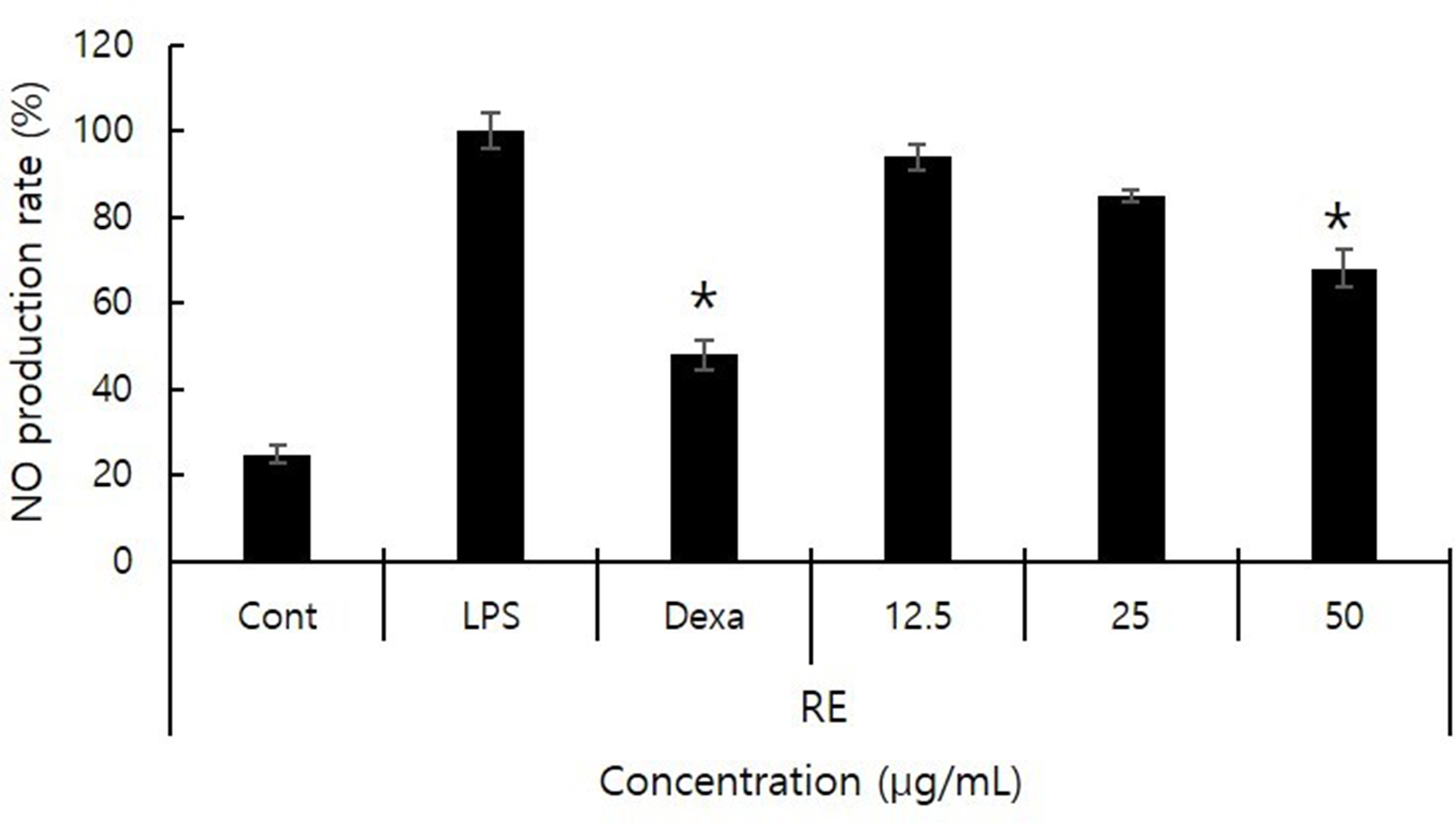
Inhibitory effects of Polygonum cuspidatum (PC) on nitric oxide production. RAW264.7 cells (1 × 105 cells/mL) were treated with 1 µg/mL lipopolysaccharide (LPS) for 2 hours, except in the Nor group. PC extract was then added to sample groups at the indicated concentrations. Cont: no LPS treatment, LPS: LPS treatment. Dexa: dexamethasone treatment. The results are presented as mean ± standard deviation of triplicate assays.
Measurement of TNF-Α and IL-6 Inhibition by PC Extract
The amount of TNF-α and IL-6 produced as mediators of the inflammatory response was measured following activation by LPS. We found that adding PC at concentrations of 12.5, 25, and 50 µg/mL decreased TNF-α and IL-6 expression in a concentration-dependent manner (Figure 3).
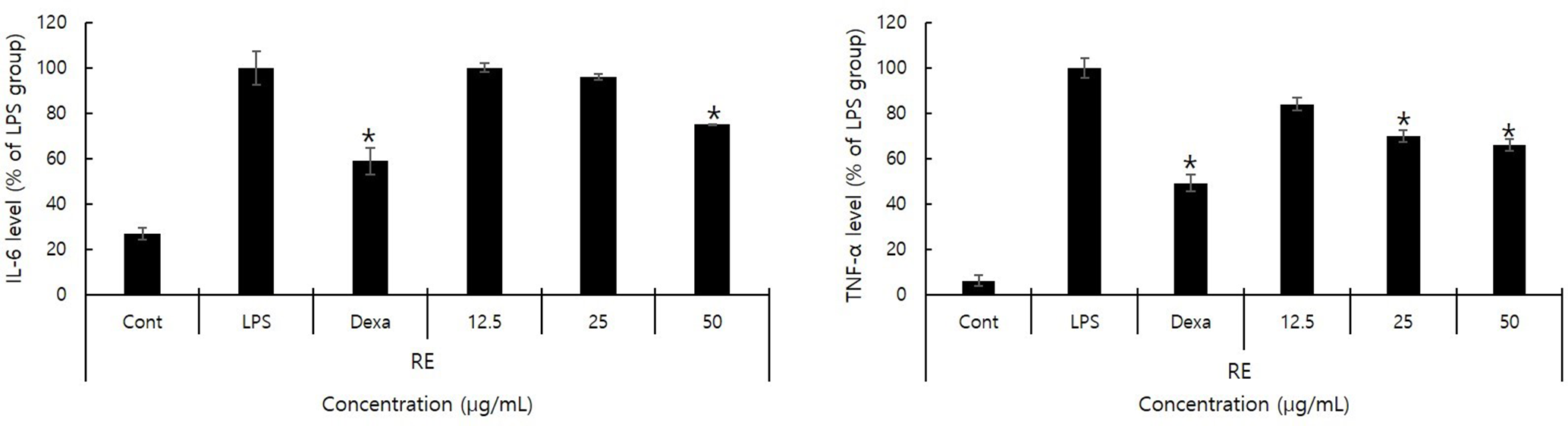
Inhibitory effects of Polygonum cuspidatum (PC) extract on the production of pro-inflammatory cytokines. The expression levels of tumor necrosis factor-α and interleukin-6 were measured after treatment with lipopolysaccharide (LPS) (1 µg/mL) for 30 minutes and the indicated concentration of PC. Cont: no LPS treatment, LPS: LPS treatment. Dexa: dexamethasone treatment. The results are presented as mean ± standard deviation of triplicate assays.
Discussion
The attitude to oral health has changed recently, and the demand for prevention, treatment, and rehabilitation of oral conditions has increased. The availability of safe and effective products containing natural antimicrobial substances while avoiding the use of synthetic preservatives has increased. 34,35 In this study, we assessed the antimicrobial and anti-inflammatory effects of PC ethanol extracts for potential applications as a mouthwash material. Our results showed that PC ethanol extract exhibited antibacterial activity against the oral bacterium S. mutans. In the culture medium, PC showed antimicrobial activity at a concentration of 1 mg/mL within 1 day, with complete inhibition of bacterial activity at 10 mg/mL. When used at a concentration of 10 mg/mL, PC extract showed persistent antibacterial activity for 23 days. The mouthwash solution containing PC also showed antibacterial activity against S. mutans. Moreover, we found that PC extract inhibited NO production and the expression of TNF-α and IL-6 in a concentration-dependent manner. Overall, these findings showed that PC ethanol extract has antimicrobial activity against the oral bacterium S. mutans via the inhibition of the production of inflammation-related factors at a safe and nontoxic concentration. These results suggested that PC extract could have applications as an antimicrobial and anti-inflammatory functional material in oral products.
The most basic way to prevent tooth decay, periodontal disease, and bad breath is by brushing the teeth. However, a busy lifestyle may reduce the frequency of teeth brushing, and there are areas where the toothbrush cannot reach. Thus, there is a need for an additional auxiliary method. Mouthwashes containing various natural products such as pine needles and green tea extracts are on the market and are used to care for oral hygiene. In this study, we confirmed that the PC ethanol extract has persistent antibacterial activity against the oral bacterium S. mutans and anti-inflammatory effects. Our PC extract contained a large amount of emodin (40.74 mg/g) that has a strong therapeutic effect on diabetes, 36 inhibitory activity against protein tyrosine kinase p65lck, anticancer activity, 37 and antibacterial activity against plant pathogens.
In summary, emodin was identified as an active ingredient in the PC extract with antibacterial and anti-inflammatory properties. The PC extract was useful as an oral product because it either inhibited or modulated the production of inflammation-related factors within a safe range of concentrations without toxicity. We suggest that PC extract can be used as an antibacterial and anti-inflammatory functional material. However, further research involving clinical trials of mouth wash containing PC extract is needed to validate these results.
Supplemental Material
Figure S1 - Supplemental material for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component
Supplemental material, Figure S1, for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component by Eun-Hee Kim, Jin-Cheon Kim, Hye Kyung Kim and Young-Ah Jang in Natural Product Communications
Supplemental Material
Figure S2 - Supplemental material for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component
Supplemental material, Figure S2, for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component by Eun-Hee Kim, Jin-Cheon Kim, Hye Kyung Kim and Young-Ah Jang in Natural Product Communications
Supplemental Material
Figure S3 - Supplemental material for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component
Supplemental material, Figure S3, for Antimicrobial and Anti-Inflammatory Effects of the Ethanol Extract of Polygonum cuspidatum as a Mouthwash Component by Eun-Hee Kim, Jin-Cheon Kim, Hye Kyung Kim and Young-Ah Jang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
