Abstract
The antimicrobial activities of (Z,E)- and (E,Z)-nepetalactones, 2 major compositional compounds from the essential oil of catnip (Nepeta cataria), were first discovered from fly larval development media studies with over 98% inhibition of bacterial growth. Further investigation demonstrated inhibition of the growth of various bacterial species of public health significance. Catnip oil showed antibacterial activity against 5 Gram-positive and 9 Gram-negative bacteria. The antimicrobial activity varied among the original essential oil from the plant and its major compositional compounds as a blended mixture or an individual compound. Growth inhibition was observed against 5 Neisseria species, with particularly strong inhibition against Neisseria sicca (with MICs ranging from 0.5 to 5 mg/mL) that provided comparable or increased levels of growth control produced by 2 antibiotics (Ceftiofur and Cephalothin). The development of plant-based antibacterial agents to prevent or delay the emergence of antimicrobial resistance in bacteria is discussed.
Natural products, such as plant essential oils, have been used throughout history for therapeutic purposes. Catnip, Nepeta cataria, is a mint herbaceous plant originating from Eurasia, North Africa, but found in North America as well. 1,2 Recently, strong antifeedant and spatial repellent activity of catnip oil were reported against various blood-sucking vectors, with a further inhibitory effect on fly larval growth from application of wax-based catnip oil. 3,4 During our laboratory bioassays of oviposition deterrence studies, we observed the catnip oil treated media with strong inhibition of larval development, compared with the control diet. This phenomenon may indicate its potential suppression of bacterial growth which is required for stable fly larvae development via feeding bacterial growth byproducts. The present study aimed to investigate whether these natural products, in essential oil forms or individual compositional compounds, would further suppress the growth of other bacteria than those living inside the fly larval media. Studies were further carried out to evaluate whether they possess similar antimicrobial activities against some pathogens of human as well as animal health significance.
Results and Discussion
Strong larvicidal activity was obtained using catnip oil wherein over 98% mortality of 3rd instar larvae was observed when larval development media was incubated with 250 mg of catnip oil (Figure 1., t = 2.10, P < 0.05). The number of colony-forming units (CFU) in the larval development media treated with catnip oil showed a significant decrease in CFU compared with those from the control (Figure 1, t = 2.07, P < 0.05). A total of 61 bacterial isolates were recovered from the stable fly larval media. Among them, significant growth reduction ranging from 76% to 99% was observed in 7 bacterial isolates identified as Bacillus cytotoxicus, B. sphaericus, B. fortis, Pseudomonas xianmenensis, Alcaligenes faecalis, and 2 strains of Myroides odoratimimus (Table 1). The majority of these identified bacterial species are associated with soil and livestock animal manure. 5 Fly larval development requires rich supplies of a bacterial community in their natural habitat, which in turn, depends upon the microbial-derived products for their food as nutrients. 6 More studies are under way to investigate which of these 6 bacterial species may play a key role either individually or in consortia in stable fly larval growth.

Inhibition of stable fly larval development from media treated with catnip oil (bar chart) and Quantitative of antibacterial activity of catnip oil. Different letters on top of bars and lines indicate significant differences (Student t test, P < 0.05).
Bacteria Identification From Stable Fly Larval Media With Significant Growth Reduction (EGR) in Media Containing Catnip Oil.

+ indicates the zone of growth inhibition. No inhibition was found from the control.
The essential oil of catnip purchased commercially contained 3 major compounds, (Z,E)-nepetalactone, (E,Z)-nepetalactone, and β-caryophyllene. The relative ratio of the 3 compositional compounds is 49:43:8, respectively. However, the concentrations of the 2 nepetalactones in commercially available catnip oils may vary based on catnip plant materials from different geographic locations, extraction methods, and different parts of the catnip plants used (flowers, leaves, stems, and seeds), as well as seasonal changes during plant collection from the same area, as reported by Schultz et al. 7 In addition to the strong antibacterial activity of catnip oil found against bacteria as shown in the stable fly larval media, its antibacterial activity was further evaluated against 5 Gram-positive and 9 Gram-negative bacteria of clinical origins (Table 2). It was noticed that the extent of growth inhibition varied with different doses tested in the bioassays. The strongest antibacterial activity was demonstrated against Neisseria subflava, followed by moderate antibacterial activities against several other Gram-negative bacteria, including Citrobacter freundii, Branhamella ovis, Aeromonas caviae, Escherichia coli O157, and Serratia marcescens. Growth inhibitions of several Gram-positive bacteria, including 3 Enterococcus species and Staphylococcus aureus, by catnip oil were also observed. Bacteria, such as Citrobacter species, have been identified from fly larval development sites (horse manure) collected from the field; among them, only C. freundii sustains stable fly development. 6 Although Aeromonas spp. have been found from horse manure, they do not support stable fly development.
Antibacterial Activity of Catnip Oil Against Selected Clinical Pathogens at 3 Dosages.

+indicates the zone of growth inhibition. No inhibition was found from the control.
Significant growth inhibition by catnip oil was found among 4 of 5 Gram-positive bacteria tested at concentrations from 5000 to 50 000 µg/mL (Table 3). The estimated minimal inhibitory concentrations (MICs) of catnip oil against Neisseria subflava were effective between 500 and 5,000 µg/mL, but the effective concentrations for M. luteus, A. caviae, and C. fruendii were 10 times higher. Comparative studies on catnip oil and its 3 compositional components against 5 Neisseria bacteria have revealed that both (Z,E)- nepetalactone and (E,Z)-nepetalactone are active agents, with a strong synergistic effect from the 2-compound mixture, as formed in the catnip essential oil (Table 4, F = 3.05‐3.48, df = 4,15; P < 0.05). Neisseria subflava is a main causative organism leading to meningitis in children and N. gonorrhoeae is responsible for the sexually transmitted infection, gonorrhea. 8,9 Neisseria (Moraxella) catarrhalis can cause infections of the respiratory system, middle ear, visual pathway and central nervous system, and joints of humans. 10 However, compared with antibiotic drugs, 11,12 MICs of catnip oil required to affect the bacterial growth were at doses of 2‐20 times higher magnitude (Table 5).
Growth Rates of Selected Pathogens Affected by Catnip Oil at 3 Dosages and Β-Caryophyllene.
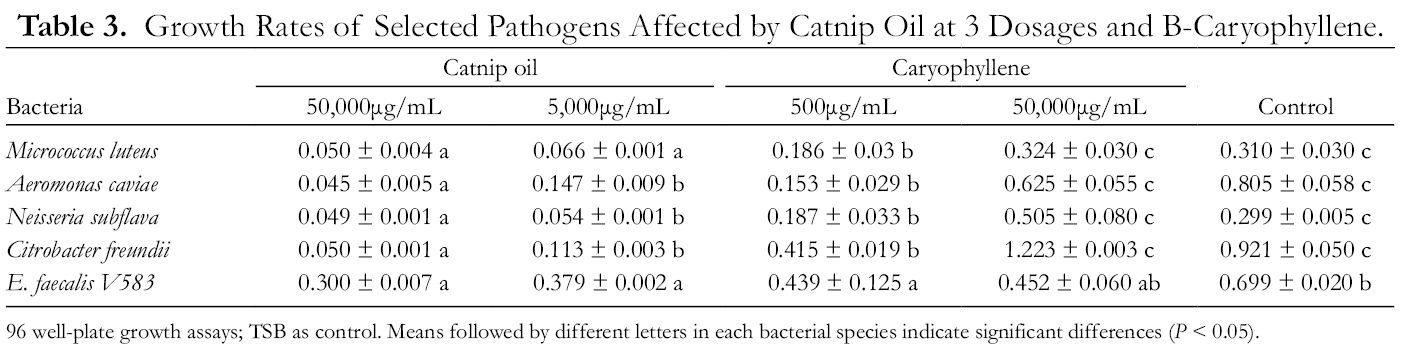
96 well-plate growth assays; TSB as control. Means followed by different letters in each bacterial species indicate significant differences (P < 0.05).
Antibacterial Activities of Catnip Oil and Its Compositional Compounds at 50,000 µg/mL Against 5 Bacteria in Neisseria.

Catnip oil and its ingredient chemicals added to disc diffusion assays; the zone of inhibition measured along the diameter including disc (Means ± SE, diameter of the disc, 6 mm). At 50 000 µg/mL, different letters followed by means indicated significant differences among catnip oil, nepetalactones and β-caryophyllene.
Comparisons of Antibacterial Activity of Selected Antibiotics and Catnip Oil Against Neisseria.
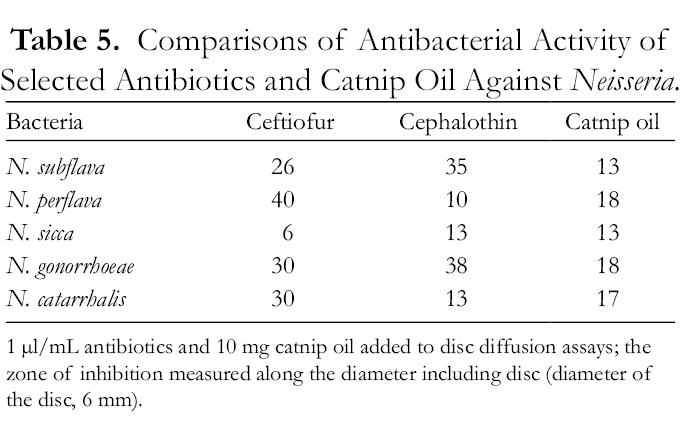
1 µl/mL antibiotics and 10 mg catnip oil added to disc diffusion assays; the zone of inhibition measured along the diameter including disc (diameter of the disc, 6 mm).
In contrast to mammalian immune systems, plants have to develop an enormous variety and quantity of small-molecule antimicrobial agents to be able to work effectively due to their generally weak response against bacteria. 8,13 Although significant antibacterial activity of catnip oil has been demonstrated, especially against Neisseria species, its effectiveness is still relatively weaker in contrast to antibiotic drugs. Plants produce over 1 00 000 small-molecule compounds exhibiting antimicrobial activity, of which many are considered to contribute to the success of plant defense mechanisms in combating pathogen infection. 8 Antimicrobial efficacy against 25 different genera of bacteria from 6 plant essential oils have been reported by Dorman and Deans. 13 Zaika proposed that Gram-positive bacteria are more resistant to properties of plant essential oils compared with Gram-negative bacteria. 14 Concurrently, the present study also demonstrated that catnip oil possesses stronger antibacterial activity against Gram-negative bacteria. Surprisingly, extremely strong antibacterial activity was observed against few Neisseria species. Till date, there is no antibiotic of plant origin available with specific cellular targets, but several antibiotics have been successfully produced by modifying chemical structures of active plant-based natural antibiotics, such as penicillin. 8 As suggested by Tiwari et al., 15 the development of novel plant-based antibiotics can reduce the selective pressure on bacterial pathogens to develop antimicrobial resistance by capitalizing upon the advantages of using various chemical strategies in plants against bacterial infection.
Conclusion
Our findings on catnip oil and its major compositional compounds (nepetalactones) as alternative antibacterial agents may provide vital information to be used not only as a larvicide to combat stable fly larval development, but also by pharmaceutical industries for novel drug discovery, especially to combat the emerging threat of multi-drug resistance. The supplementary use of plant-based antibacterial substances and drugs may provide new opportunities to treat infections caused by multi-drug resistant bacteria more efficiently and potentially delay the development of anti-microbial resistance.
Materials and Methods
Essential Oils and Their Ingredient Compounds
The catnip essential oil was purchased from Liberty Natural Products, Inc. (Oregon City, Oregon, USA). Its chemical composition was determined by gas chromatography-mass spectrometric (GC-MS) analysis via confirmed characteristic fragments and retention times from their synthetic standards. One mg of oil was dissolved in 2 ml n-hexane, and 0.5 µl of the aforementioned solution (approx. 100 ng) was injected in the injection port of a coupled HP 7890 GC interfaced to a HP 5975 Mass Selective Detector (Agilent Technologies, Inc., Santa Clara, CA, USA). The GC was equipped with a FFAP and DB-5 column (30 m × 0:25 µm ID, Agilent) with the mode of split-less injection (250 °C). The oven temperature program started at 50 °C for 3 minutes, increased to 170 °C at 5 °C/min, and then increased to 240 °C at 15 °C/min. Helium was used as the carrier gas (3 mL/min). Mass spectra were recorded from 30 amu to 550 amu with electronic impact ionization at 70 eV.
β-Caryophyllene was purchased from Sigma-Aldrich (St. Louis, MO, USA). The 2 nepetalactones (Z,E- and E,Z-) were separated using gel preparative thin-layer chromatography plates (Whatman 20 cm × 20 cm) following the method described by Peterson. 16 The solvent system used was 19 : 1 n-hexane : ethyl ether. The products were visualized under 254 nm UV light, and the silica gel was scraped off the plates and washed with diethyl ether. The ether was then removed by rotary evaporation, and the purity was checked using GC-MS analysis. The (Z,E)- and (E,Z)-nepetalactone were >95% purity. The yield was <5%. All synthetic standards were purchased from Sigma-Aldrich, with purities from 95% to 99%.
Fly Larval Development Assay, Larval Growth Inhibition by Essential Oil and Its Antimicrobial Activity
Stable flies (Stomoxys calcitrans) used for laboratory bioassays were from colonies maintained at the United States Department of Agriculture, Agricultural Research Service, Agroecosystem Management Research Unit (Lincoln, Nebraska). The flies were maintained at 24 ± 1 °C with variable humidity (60% to 70% RH, relative humidity) and a 12L:12D photoperiod. Adults were fed with citrated bovine blood (3.7 g sodium citrate/l) from a blood-soaked absorbent pad (Stayfree®, McNeil-PPC Inc., Skillman, New Jersey) placed on top of the screened cage. An oat meal diet (100 g containing 55 g of well-mixed oats, 12 g of fish meal, 22 g of vermiculite and 95 ml of distilled water in one plastic cup) was used as the larval development media.
Larval growth inhibition was assessed by placing 200 stable fly eggs on the surface of the larval development media (100 g in a 50 ml plastic cup), well-mixed with 250 mg essential oil in 5 ml of mineral oil. Cups with the same amount of media treated with 5 ml of mineral oil were used as the control. Cups were maintained in the environmental chamber at 26 ± 2 °C and 60 to 70% RH for 1 week, after which the number of third-instar larvae was counted. Larval growth inhibition was measured by counting the number of larvae harvested from the control media cup and the number of larvae surviving in the oil-treated media cup [% of Inhibition = (No. of survived larvae from the control media – no. of larvae survived from the treated media) × 100/No. of survived larvae from the control media]. Larval growth inhibition experiments were replicated at least 5 times. The stable fly larval development media treated with or without catnip oil were further extracted for colony forming units (CFU/mL) during the experiment using the Quanti-Tray/Colilert procedure to determine the total coliform concentrations in the samples.
Antimicrobial Activity of Essential Oils on Bacterial Isolates
The bacterial isolates were obtained from 2 sources including stable fly larval development media and faucets’ handles of 3 public restrooms at the East Union of the University of Nebraska. Bacterial isolates were streaked onto TSA (Tryptic Soy Agar) plates for catnip oil treatment. TSA plates with 5.67 mL/l dimethyl sulfide (DMSO), TSA plates with 5.57 mL/l DMSO and 100 mg/l catnip oil were further incubated at 37 °C for 24 hours. Antimicrobial activity of the isolate was measured according to the Poisoned Food Method used for antifungal activity of plant extracts, 17,18 and the antimicrobial effect was estimated by the following formula: Growth reduction (%) = (growth size of bacterial isolate in control – growth size in catnip oil treated media) / growth size of bacterial isolate in control × 100. Single colonies with significantly reduced growth sizes were isolated on TSA and stored at 4 °C for further DNA analysis.
For the identification of bacteria, the genomic DNA was extracted from pure cultures using a NucleoSpin Tissue kit and the 16S rRNA (16S ribosomal RNA) gene was amplified following the method described by Romero et al. 6 Sequences were determined using the same eubacterial universal primers and were analyzed for similarity to known sequences in the GenBank databases (www.ncbi.nlm.nih.gov/blast) by using the BLAST (basic Local Alignment Search Tool) program. 6
All tests for antibacterial effectiveness with catnip oil and its compositional compounds against pathogens were conducted in the Veterinary Medical Entomology Laboratory at Kansas State University and in the laboratories at the Departments of Entomology, Agronomy and Horticulture of University of Nebraska. Fresh cells of test bacterial strains were cultured on TSA (Difco ™ 2nd Edition). A bacterial suspension of OD600 0.5 (optical density at 600 nm using MacFarland standard kit) was prepared in phosphate buffered saline (PBS, MP Biomedicals) and 100 µl was spread plated on TSA. Discs (diameter of 6 mm) were prepared using sterile white filter paper and a paper punch machine. Test compounds of catnip oil, β-caryophyllene, and (E,Z)- and (Z,E)-nepetalactones were dissolved in DMSO and added to the sterile discs at different dilutions. The plates were incubated at 30 °C for 48 hours and the zone of inhibition was measured. The values are presented as mean of triplicate ±SD.
Data Analysis
Student T-test and one-way factorial analysis of variance (ANOVA) was used to analyze differences in growth or inhibition of bacteria isolate, larval development and CFU counts among essential oil-added media, DMSO/ethanol-added media, and the control. Means were compared using the least square difference (LSD) test. Values of P < 0.05 were considered significant. Analyses were performed using SAS, version 9.1 (SAS Institute Inc.).
Footnotes
Acknowledgments
We thank Dr L. Durso and J. McGhee, D. Berkebile and K. Tangtrakulwanich for their critical comments on an earlier version of the manuscript and technical assistance with this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
