Abstract
P-glycoprotein (P-gp) inhibition conduces to improving the ability of chemicals to cross through blood-brain barrier (BBB). The hook-bearing branch of Uncaria is used as a traditional herbal medicine for the treatment of hypertension, headache, stroke, and Alzheimer’s disease in recent years. In this study, the inhibitory effects of 8 alkaloids sourced from Uncaria plants on P-gp were evaluated. Meanwhile, the content of 8 alkaloids in 8 Chinese Uncaria species was quantified simultaneously by high-performance liquid chromatography analysis. Western blotting, real-time quantitative reverse transcription-polymerase chain reaction, and flow cytometry were used to evaluate the P-gp levels in MCF-7/adriamycin-resistant (ADR) cells. It was shown that hirsuteine and hirsutine, selected from 8 candidate alkaloids, could remarkably suppressed P-gp level and function in MCF-7/ADR cells when using Verapamil as positive control. By tracking their chemical distribution in 8 Chinese Uncaria species, we found that hirsuteine and hirsutine were much more abundant in Uncaria rhynchophylla and Uncaria hirsuta than in any other 6 species. And a chemical content profile of these alkaloids was plotted within 8 Chinese species.
Uncaria plants have been used to treat Alzheimer’s disease (AD) singly or formed into traditional herbal formula for decades. 1 Generally, the regular drug therapy against central nervous system (CNS) has weakness. 2 It is known that about 98% of the small molecules and almost all the large molecules are restricted to penetrate blood-brain barrier (BBB) and fail to reach brain tissues. 3 According to previous studies, several chemical components in Uncaria plants were found possessing the ability of crossing BBB and could be detected in the brain tissue of AD model mice. 4,5 In the process of crossing over BBB, p-glycoprotein (P-gp) plays an important role in efflux function. 6 Suppressing the activity of P-gp on the membrane of cerebral capillary endothelial cells could reduce drug efflux and finally give rise to the concentration of drugs in the brain. 7 The evaluation of the impact of natural compounds on P-gp function and expression will help to explain the mechanism of phytomedicine in AD treatment, even though the compounds are in a complex prescription.
MCF-7 adriamycin-resistant cell line (MCF-7/ADR) are commonly used in antineoplastic drug-resistance research. It has been proved that P-gp could be stably overexpressed in MCF-7/ADR cells. The cells can maintain the multidrug resistance (MDR) phenotype for 3 months in cultivation without adriamycin. 8 Therefore, MCF-7/ADR cells were used to evaluate the inhibitory effect of 8 Uncaria alkaloids on P-gp.
The genus of Uncaria, from the family of Rubiaceae, is composed of 34 species all over the world. There are 11 species: Uncaria rhynchophylla, U. hirsuta, U. lancifolia, U. scandens, U. macrophylla, U. homomalla, U. sessilifructus, U. laevigata, U. yunnanensis, U. lanosa, U. rhynchophylloides, and 1 variant U. sinensis distributed in China. 9 The stems and hooks of all these Chinese species can be used either as Chinese Material Medical officially (4 species are recorded in the Chinese Pharmacopoeia 2015) or as folk herbal medicine. 10 Up to date, more than 200 compounds have been isolated from the Uncaria plants. Among them, indole alkaloids have been considered as the main efficient components in the treatment of AD. 11 -16 It is noted that the most phytochemical, pharmacological, pharmacokinetic, and metabolic studies are mainly focused on rhynchophylline, isorhynchophylline, 17,18 corynoxeine, and isocorynoxeine, 19,20 while the evaluation and quantification of other alkaloids among these Uncaria species have not yet been studied systematically.
Experimental
The Inhibiting Effect on P-gp Expression and Function
Materials
Opti-MEM and fetal bovine serum (FBS) were purchased from Life Technologies (Grand Island, NY, USA). RNeasy Plus Mini Kit, Prime Script TM Master Mix, and AceQ qPCR SYBR Green Master Mix (without ROX) were obtained from Vazyme Biotech Co., Ltd. Gelatin, adriamycin hydrochloride (D1515, ADR), verapamil (V0100000, VERA), and rhodamine 123 (Rh123) were purchased from Sigma-Aldrich (St. Louis, USA). Cell Counting Kit-8 (CCK8) was purchased from QCBIO Science & Technologies (Shanghai, China). Other compounds were all in analytical grade. Standard compounds of rhynchophylline and isocorynoxeine were purchased from Beijing Zhongke Quality Inspection Biotechnology Co., Ltd. with batch numbers Lot 170319 and 170201, respectively. Isorhynchophylline, corynoxeine, hirsutine, hirsuteine, geissoschizine methyl ether, and tetrahydroalstonine were purchased from Chengdu PUSH Bio-Technology Co., Ltd., and their batch numbers were PS 11091001, PS 0251‐0020, PS 14052003, PS 0717‐0010, PS 161017‐01, and PS 0983‐0010, respectively. The purity of standard compounds is >98.0%.
Cell lines and cell culture
MCF-7 and MCF-7/ADR were obtained from KeyGen Biotech (Nanjing, China). Cells were cultured with Roswell Park Memorial Institute-1640 medium containing 10% FBS at 37 °C and 5% carbon dioxide (CO2) as an incubator. Adriamycin (1.0 µM) was added to the culture medium to maintain the MDR characteristics of the MCF-7/ADR cells.
Cells in the logarithmic growth phase were seeded in 96-well plates, adjusted to a cell density of 3 × 103 per well, and placed at 37 °C in a CO2 incubator for 24 hours. Then, the cells were treated with various concentrations of compounds, respectively. After 48 hours of incubation, 10 µL of CCK-8 was dropped into each well to culture for 4 hours. The cell viability was monitored through measurement of absorbance at 450 nm on an enzyme-linked immunosorbent assay reader (Spectra Max Plus 384; Molecular Devices, Sunnyvale, CA, USA).
Quantitative real-time reverse transcription-polymerase chain reaction
The quantitative real-time reverse transcription-polymerase chain reaction was performed as previously described. 21 Briefly, MCF-7 or MCF-7/ADR cells (1 × 106/well) were seeded in 6-well plates. The next day, MCF-7/ADR cells were treated with 10 µM of rhynchophylline, isocorynoxeine, isorhynchophylline, geissoschizine methyl ether, hirsutine, hirsuteine, corynoxeine, and tetrahydroalstonine, respectively, for 24 hours. VERA at 10 µM was used as a positive control. The Δ cycle threshold method was used for the calculation of relative differences in mRNA abundance with a LightCycler 480 (Roche Molecular Biochemicals, Mannheim, Germany). Data were normalized to the expression of glyceraldehyde 3-phosphate dehydrogenase GAPDH). The results were expressed as fold changes. The sequences of primers used were as follows: MDR1 (P-gp mRNA), 5′-CAGAGTCAAGGAGCATGGCA-3′ (sense) and 5′-TCAGAGTTCACTGGCGTTT-3′ (antisense); GAPDH, 5′-GAAAGCCTGCCGGTGACTAA-3′ (sense), and 5′-AGGAAAAGCATCACCCGGAG-3′ (antisense).
Rh123 flow cytometry assay
The function of P-gp was evaluated by examining the intracellular accumulation of Rh123 as previously described. 22 MCF-7/ADR cells were treated with or without 10 µM hirsuteine, hirsutine, or 10 µM VERA (positive control) for 1.5 hours and then incubated with 5 µM of Rh123 at 37 °C for 1.5 hours. Subsequently, the culture medium was removed, and the cells were washed twice with phosphate-buffered saline (PBS). Finally, the cells were suspended into 250 µL of PBS and followed by flow cytometry assay (FACS) to examine the green fluorescence produced. Analysis of FACS data was performed using FACSDiva (Becton & Dickinson Company, Franklin Lakes, NJ, USA) and FlowJo software (Tree Star Inc., Ashland, OR, USA).
Statistical analysis
All the experiments were performed at least 3 times unless otherwise stated. The results were analyzed using a one-way analysis of variance with Tukey’s multiple comparison test. The data are given as the mean ± SD, and P < 0.05 was considered to indicate a significant difference.
Quantitative Determination of 8 Alkaloids in Uncaria
The quantitative analysis methodology was based on the Guidelines for Analytical Methods of Drug Quality Standards (Pharmacopoeia of the People’s Republic of China 2015 edition, Appendix XIX A).
plant material
Eight species of Uncaria were collected and identified by Professor Zenglai Xu in the Institute of Botany, Jiangsu Province and Chinese Academy of Sciences. The habitats were shown in Table 1. Mature plants with 0.3-0.5 cm diameter stem were selected. The plant materials (UN201601-UN201608) were dried and deposited in the Herbarium of Institute of Botany, Jiangsu Province and Chinese Academy of Sciences. The rest selected plant materials were cleaned and tried. The part of the stem with hook was cut into 2 cm for further use.
Habitats of 8 Species of Uncaria.

Instruments and reagents
Shimadzu LC-2010 HPLC system (Japan) with a diode array detector (Shimadzu, Japan) was used for quantification. TP-040S ultrasonic cleaning apparatus (Shenzhen Jie Meng Cleaning Equipment Co., Ltd.), AB204 electronic balance (METTLER TOLEDO, Switzerland), and Digital pH meter (Shanghai INESA scientific instrument Co., Ltd.) were used. The standards of rhynchophylline, isocorynoxeine, isorhynchophylline, corynoxeine, hirsutine, hirsuteine, geissoschizine methyl ether, and tetrahydroalstonine were used as described in the Materials. Methanol (MeOH), acetonitrile (ACN), triethylamine, and acetic acid (AA) at HPLC gradient grade were purchased from National Drug Group Chemical Reagents Co., Ltd. Ammonium acetate (analytically pure) was bought from Guangdong Guanghua Sci-Tech Co., Ltd., and purified water was bought from Wahaha Co., Ltd.
Standard solutions
Stock solutions of rhynchophylline, isorhynchophylline, corynoxeine, isocorynoxeine, hirsutine, hirsuteine, geissoschizine methyl ether, and tetrahydroalstonine were initially prepared as 1.0 mg/mL in MeOH and stepwise diluted into 5 concentration levels. The solutions were stored at 4 °C as standard stock solutions.
Preparation of sample solution
The crude drug of 8 plant species was powdered and precisely weighed (1 g). Then, the powder was soaked overnight in 6.0 mL MeOH and extracted in ultrasonic bath for 30 minutes. After repeating the operation process, the extract was combined and filtrated through a 0.45-µm microporous membrane. A total of 3 duplicates of each sample were performed. The sample was preserved at 4 °C until use.
High-performance liquid chromatography
The chromatographic separations were carried out on the Agilent Eclipse Plus C18 column (5 µm, 250 × 4.6 mm) . The injection volume was set at 10 µL. The mobile phase consisted of 2 solutions, phase A: water (H2O) with 0.04% AA and phase B: ACN. The gradient profile was programmed as follows: 0 minutes 22% B, 12 minutes 25% B, 14 minutes 31% B, and 30 minutes 100% B. The chromatogram was recorded from 0 to 47 minutes. The column temperature was 25 °C, and the flow rate was 1.0 mL/min. A wavelength of 254 nm was determined for detection. The data were collected by LC solution software (Shimadzu, Japan).
Methodological validation
The linearity of the standard compounds was assessed. Standard solutions were prepared to the range of 12.29-98.33 μg/mL (rhynchophylline), 16.67-133.33 μg/mL (isocorynoxeine), 9.79-78.33 μg/mL (isorhynchophylline), 8.75-70.00 μg/mL (geissoschizine methyl ether), 11.46-91.67 μg/mL (hirsutine), 14.38-115.00 μg/mL (corynoxeine), 11.67-93.33 μg/mL (hirsuteine), and 10.83-86.67 μg/mL (isocorynoxeine). Calibration equations of every analyte were obtained by plotting the mean of peak area (y axis) vs alkaloid content concentration (x axis) within the ranges. The goodness-of-fit (GOF) was evaluated by R 2 value.
Limit of detection (LOD) and limit of quantification (LOQ) were determined by injecting 5 µL serial dilution of 6 individual solutions to HPLC. LOD and LOQ were calculated at the signal to noise (S/N) ratios of 3 and 10, respectively. 23
The intraday and interday variability were studied to assess the precision of the method. The intraday variability was determined by duplicated sample within 24 hours. Each sample was analyzed 3 times, and the relative SD (RSD) was calculated. The accuracy was assessed by measuring the mean recovery rate (%) of the spiked sample solution vs the no spiked solution.
Results and Discussion
The Inhibition of 8 Alkaloids on P-gp mRNA Levels Detected by qRT-PCR Analysis
The inhibitory effect of 8 chemicals on mRNA levels of P-gp (MDR1) was screened by real-time fluorescent quantitative PCR. The results were shown with illustration in Figure 1. P-gp mRNA level in MCF-7/ADR cells was much higher than those in MCF-7 cells. Hirsuteine (compound 3) and hirsutine (compound 4) decreased MDR1 levels markedly, the effect of which was stronger than the positive control (VERA). Besides, isocorynoxeine, corynoxeine, or isorhynchophylline suppressed P-gp mRNA levels in MCF-7 ADR cells.
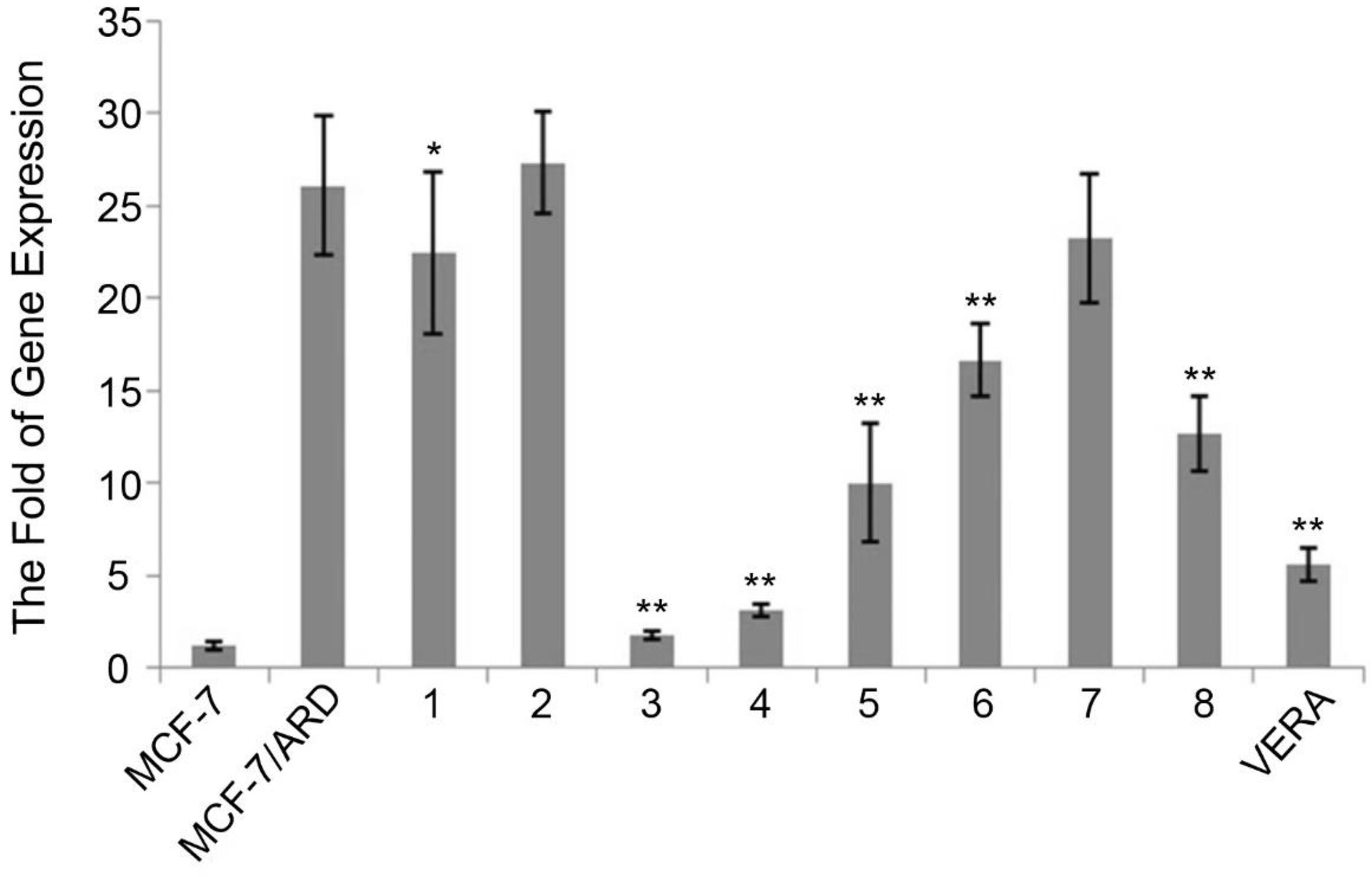
Determination of relative levels of MDR1. (1) P-gp mRNA levels in MCF-7/ADR cells showed significantly higher than that in MCF-7 cells. (2) Then, the inhibitory effect of 8 compounds (10 µM) and VERA (10 µM) on levels of MDR1 were screened in MCF-7 ADR-resistant subline. (1, MCF-7/ADR + geissoschizine methyl ether; 2, MCF-7/ADR + tetrahydroalstonine; 3, MCF-7/ADR + hirsuteine; 4, MCF-7/ADR + hirsutine; 5, MCF-7/ADR + isocorynoxeine; 6, MCF-7/ADR + corynoxeine; 7, MCF-7/ADR + rhynchophylline; 8, MCF-7/ADR + isorhynchophylline VERA. MCF-7/ADR + VERA (positive control)). Bars represent the mean ± SD, *P < 0.05, and **P < 0.01 vs. MCF-7/ADR: one-way ANOVA + Dunnett's test. ADR, adriamycin-resistant; VERA, verapamil.
The Suppression of Eight Alkaloids on P-gp Function Demonstrated by Rh123 Accumulation Assay
Rh123 accumulation assay was used to detect and confirm the inhibitory effect of compound 3 or 4 on P-gp function (VERA was also used as a positive control). As shown in Figures 2 and 3, after incubating with Rh123, the fluorescence level was lower in MCF-7/ADR cells than that in MCF-7 cells, indicating a higher P-gp activity of MCF-7/ADR subline. The Rh123 fluorescence intensity increased markedly in MCF-7/ADR cells after treating with compound 3, 4, or VERA treatment compared with the negative control (PBS). Therefore, we speculated that both compound 3 (hirsuteine) and 4 (hirsutine) displayed inhibitory effect on P-gp. The 2 compounds could be potential effective substance of Uncaria plants regarding cerebral disease treatment.

Rh123 accumulation assay (1). The abscissa was the order of magnitude, and the ordinate represented the number of cells in which the fluorescence value was in the order of magnitude, VERA was used as a positive control. ADR, adriamycin-resistant; VERA, verapamil.

Rh123 accumulation assay (2). Rh123 accumulation assay was used to detect and confirm the ability of hirsuteine or hirsutine on P-gp function (3: MCF-7/ADR + hirsuteine [10 µM]; 4: MCF-7/ADR + hirsutine [10 µM]; VERA. MCF-7/ADR + VERA [10 µM] as positive control). When MCF-7 and MCF-7/ADR cells were incubated with Rh123, the fluorescence level was lower in MCF-7/ADR cells than that in MCF-7 cells, which indicated high P-gp activity of MCF-7/ADR subline. The Rh123 fluorescence intensity markedly increased in MCF-7/ADR cells in the presence of 3, 4, or VERA. Bars represent the mean ± SD, **P < 0.01 vs. MCF-7/ADR: one-way ANOVA + Dunnett's test. ADR, adriamycin-resistant; VERA, verapamil.
Alkaloids are the most important secondary metabolites in the genus Uncaria. More than 100 indole alkaloids have been identified. It was found that among the 8 compounds in the test, hirsuteine and hirsutine, which are monoterpene indole alkaloids, were verified to be more active in P-gp mRNA inhibition. Correspondingly, isocorynoxeine, isorhynchophylline, corynoxeine, and rhynchophylline which are all monoterpene oxindole, presented less capability of suspension. Tetrahydroalstonine, with a dihydropyran heterocycle, had the lowest activity. From the perspective of structure-activity relationship, whether the formal oxidation state has influence on its inhibition activity should be taken into further investigation. 24
Quantification of 8 Alkaloids
HPLC analysis results of 8 Chinese Uncaria species and mixed reference standards were shown in Figure 4. These experiments demonstrated that the present method was selective, precise, and reproducible. Five incremental concentrations of each standard solution were injected: rhynchophylline (12.29-98.33 μg), isocorynoxeine (16.67-133.33 μg), isorhynchophylline (9.79-78.33 μg), geissoschizine methyl ether (8.75-70.00 μg), hirsutine (11.46-91.67 μg), corynoxeine (14.38-115.00 μg), hirsuteine (11.67-93.33 μg), and tetrahydroalstonine (10.83-86.67 μg). Calibration curve equations were obtained by plotting peak area vs mass. LOD and LOQ values were >0.09 µg/mL and >0.98 µg/mL, respectively (Table 2).

Chromatogram of Uncaria rhynchophylla extract (A) and 8 mixed standard (B). 1, Rhynchophylline; 2, isocorynoxeine; 3, isorhynchophylline; 4, geissoschizine methyl ether; 5, hirsutine; 6, hirsuteine; 7, corynoxeine; 8, tetrahydroalstonine.
Calibration Curves, Detection Limits, and Quantification Limits of the Analytes.
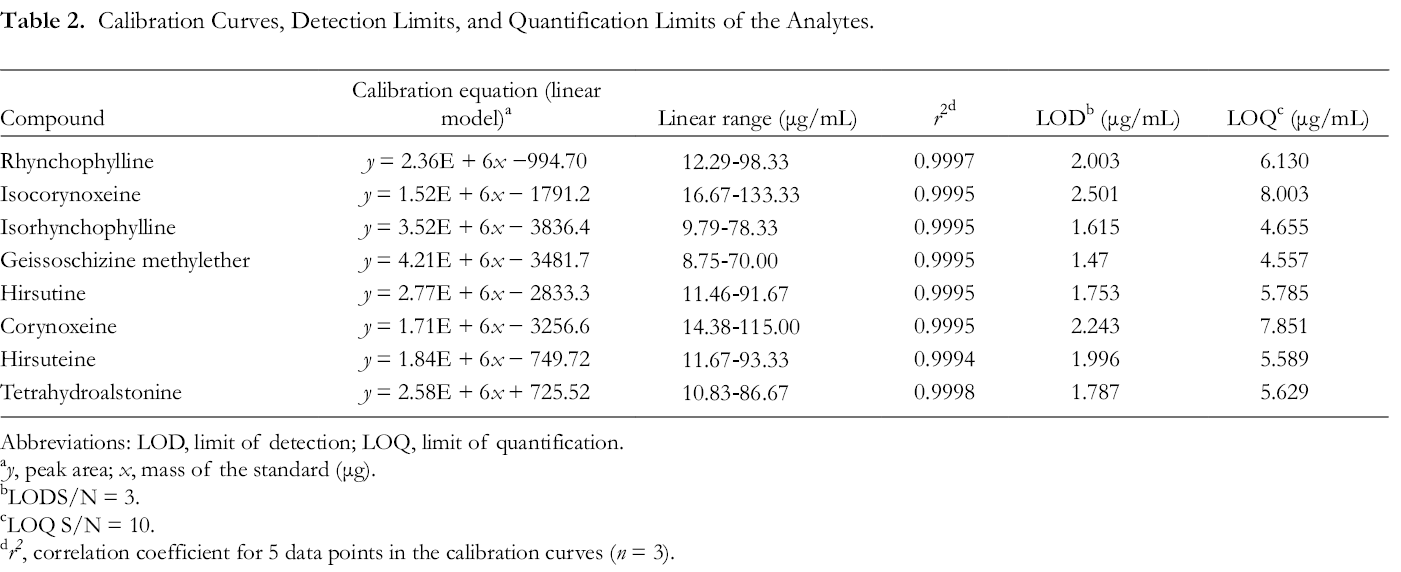
Abbreviations: LOD, limit of detection; LOQ, limit of quantification.
a y, peak area; x, mass of the standard (μg).
bLODS/N = 3.
cLOQ S/N = 10.
d r2 , correlation coefficient for 5 data points in the calibration curves (n = 3).
Intraday and interday variabilities were measured to assess the precision. In this test, sample solutions were injected 5 times to obtain RSDs. As shown in Table 3, the RSDs in intraday and interday variability were <2.39% and <2.51%, respectively. The RSD in the repeatability test was <2.64%.
Analytical Results of Intraday and Interday Variabilities and Repeatability.
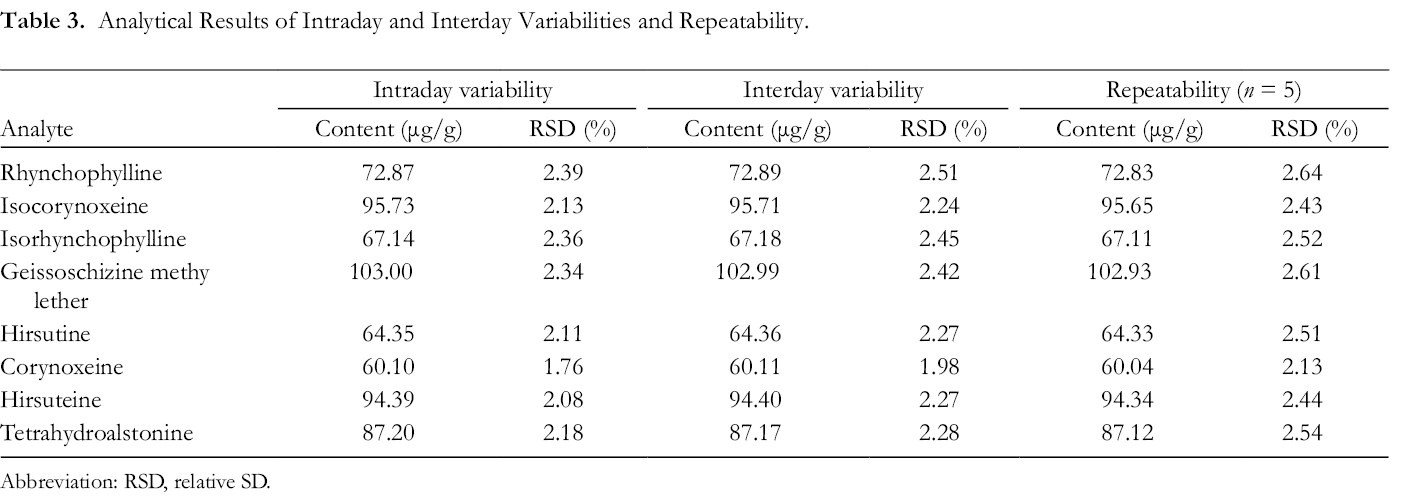
Abbreviation: RSD, relative SD.
The recovery test was evaluated by calculating the mean recovery rate (%) of each compound from the spiked extract solution vs the nonspiked extract sample. As shown in Table 4, the mean recovery rate was between 99.48% and 101.90%, and the RSD value was <3.09%.
Recovery of Each Analytes as Determined by Standard Addition Method.

Data presented as average of 3 determinations.
The contents of alkaloids from different species of Uncaria were shown in Table 5. Uncaria hirsuta had much more hirsuteine than any other species. Furthermore, this species also had the highest total content of 8 alkaloids. In a previous quantitative analysis applying liquid chromatography (LC)-mass spectrometry (MS)/MS technology, 24 both U. hirsuta and U. rhynchophylla were found rich in alkaloids. And it was reported that U. macrophylla habited in Nanning had the highest alkaloid content. Differently, according to our study, U. macrophylla collected in the Chengdu region did not contain as much alkaloids as the former 2 species. More attention should be paid to regional variation in the alkaloid content of this species. Additionally, none of the 8 alkaloids were detected in U. laevigata. This species was initially classified in 1834 and reclassified several times until 1981. The molecular identification of U. laevigata as well as other species of Uncaria showed that psbA-trnH exhibited highest interspecific divergence. The distribution of interspecific distance between ITS2 and psbA-trnH was higher than the corresponding intraspecific distance. 25 It would be meaningful to take further chemotaxonomy research, supplementing with deoxyribonucleic acid-conserved sequence analysis to verify the classification of U. laevigata.
Content of Analytes (mg/g).
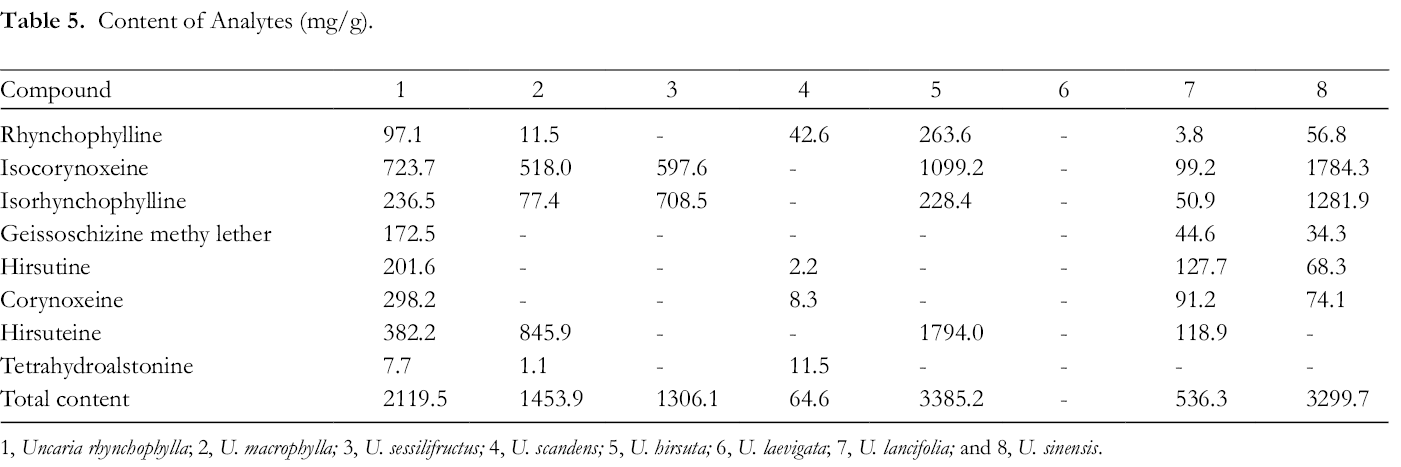
1, Uncaria rhynchophylla; 2, U. macrophylla; 3, U. sessilifructus; 4, U. scandens; 5, U. hirsuta; 6, U. laevigata; 7, U. lancifolia; and 8, U. sinensis.
Supplemental Material
Supplementary Material 1 - Supplemental material for Inhibitory Activity of 8 Alkaloids on P-gp and Their Distribution in Chinese Uncaria Species
Supplemental material, Supplementary Material 1, for Inhibitory Activity of 8 Alkaloids on P-gp and Their Distribution in Chinese Uncaria Species by Yanan Gai, Nannan Yang and Jian Chen in Natural Product Communications
Footnotes
Acknowledgment
We would like to give sincerely thanks to Prof. Zhenglai Xu for his contribution in plant material identification and preservation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Natural Science Foundation of China (Program No. 31500290) and a fund from Jiangsu Scientific and Technological Innovations Platform (Jiangsu Provincial Service Center for Antidiabetic Drug Screening).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
