Abstract
In this study, the separation of ginsenosides from the crude extract of Sanchi ginseng and American ginseng, obtained by aqueous ethanol extraction, was conducted in a simulated moving bed (SMB) with cleaning in place (CIP) design. Concentrated products with 86.2%, w/w, and 91.5%, w/w, of ginsenosides were obtained by the utilization of CIP-SMB for Sanchi and American ginseng, respectively. This study reveals that CIP-SMB could enrich ginsenosides by removing the retained impurities, which were initially extracted by aqueous ethanol and not removed by either the antisolvent precipitation for Sanchi ginseng or the ethyl acetate extraction for American ginseng. Although the ginsenosides in both ginsengs are similar, the difficulty in applying chromatography to the crude extract differs. A natural crude extract normally contains compounds with significant difference in retention. A traditional SMB with 4 sections will be, consequently, unable to continuously separate the natural products. This study develops methods for pretreating the crude extract and demonstrates the feasibility of continuously separating ginsenosides by using SMB. SMB is an effective technology for large-scale chromatography and can generally resolve solvent and energy consumption problems. This is expected to promote the industrial use of SMB for the production of concentrated ginsenosides as active pharmaceutical ingredients.
Ginseng is an effective traditional medicine in most oriental countries, especially in China, Korea, and Japan. 1 -3 Ginseng provides antioxidation ability and is used as a supplement for either preventing or as a therapeutic for hyperlipidemia, 4 activating different steroid hormone receptors for angiogenesis-related diseases, 5 improving learning and memory skills for enhancing intellectual ability, 6 and inhibiting the increase of activity of human immunodeficiency virus by saponin compounds. 7,8 Ginseng includes Panax ginseng C.A. Meyer (Asian ginseng) in Asia, P. quinquefolius L. (American ginseng) in North America, and P. notoginseng (Sanchi ginseng) in China. American and Asian ginsengs have been the most consumed varieties globally. 9 -11 Sanchi ginseng is mainly grown in southwest China. Asian ginseng, also known as red ginseng, is normally served after processing; Sanchi and American ginsengs, called white ginseng, are served without processing.
Saponins or ginsenosides are the principal active compounds in Ginseng 10 -12 ; nonetheless, the ratios of protopanaxadiol-type ginsenosides (PPDs) and protopanaxatriol-type ginsenosides (PPTs), or other markers differ from each other. 11,12 It was reported that the ginsenosides in American and Sanchi ginsengs are similar; the main ginsenosides are Rg1/Re, Rb1, and Rd. 13 -21
The extraction and isolation of ginsenosides play a pivotal role in the ginseng industry. In previous research, ginseng has been extracted by a variety of solvents, 15,22,23 including methanol, water, n-butanol-saturated water, and acetonitrile, using different methods. 24 -26 Regarding the safety for human consumption, ginsenosides are most successfully extracted with aqueous ethanol in conjunction with ultra-high-pressure extraction. 27 -29 Chromatography is normally required for the isolation of ginsenosides from the crude extract. 30 -38 In 1 study, the isolation and purification of ginsenosides was performed by organic solvents using fast-centrifugal partition chromatography. 39 Simulated moving bed (SMB) was also applied to isolate Rb1 from Sanchi ginseng 34 and separate the enantiomer of Rg3. 40 Most of the chromatographic methodologies, including SMB, were conducted by using methanol or other organic solvents, which are detrimental to humans and could lead to inappropriateness in food regulation. In this research, a 5-section SMB was utilized to separate ginsenosides from the crude extracts of Sanchi and American ginseng by using different ratios of aqueous ethanol solution as the mobile phase. The deployment of merely water and ethanol for processing is auspicious for the safety of the concentrated ginsenosides.
Traditional SMB with 4 sections does not generally conform to the separation of ginsenosides from the aqueous ethanol crude extract because polysaccharides and waxes in the crude extract will eventually plug the packed column. A pretreatment by freezing or solvent precipitation is normally performed to remove polysaccharides and waxes; nonetheless, incomplete precipitation could still result in a failure of the downstream chromatography, especially for SMB. In this study, a second eluent is applied to regenerate the solid phase. Figure 1 shows the configuration of the SMB used in this study. The SMB design has 8 columns with 5 sections. Six of the columns, C1-C6, are designed as a traditional 3-section SMB, which represents the liquid phase as not directly recycled. The 3-section design without a fourth section is meant to avoid the recycling of unretained or very weakly retained impurities in the crude extract. Two of the columns in Figure 1 are designed to regenerate the solid phase. A strong eluent is applied in the section indicated as Wash 2 to remove the very strongly retained components from the solid. After being cleaned, the solid is rinsed in the section indicated as Wash 1 by the desorbent used for C1-C6.

Chromatogram of the crude extract of Sanchi ginseng.
In Figure 2, the liquid flows from the left to the right without recycling, and the flow of solid from the right to the left with recycling is completed by periodically and simultaneously switching the ports of inlet and outlet to the next column. As in traditional SMB, the solid in sections II and III acts as a separating agent and is regenerated in section I. However, strongly retained components on the adsorbent may not be entirely removed in section I. Therefore, the solid from section I is washed by a second eluent with stronger elution power in a washing section, demonstrated as Wash 2 in Figure 2. After being regenerated by the stronger eluent, the adsorbent is rinsed by the first eluent in a section indicated as Wash 1 and recycled to section III. With this design, the strongest retained component can be eluted from the port of Wash 2 while the weakest retained component is flushed out from the port of Raffinate. Ultimately, the intermediately retained components will be eluted from the port of Extract.

The configuration of simulated moving bed with a cleaning in place design.
Material Processing and HPLC Analysis for Sanchi Ginseng
Sanchi ginseng was supplied by Yunnan Dong-Jun Pharmaceutical Group. The roots were ground to about 60 mesh, and 100 g of the powder was extracted with 500 mL of 70% ethanol under reflux for 2 hours. After filtration, water was added to the supernatant and the ethanol solution was diluted from 70% to 35%. At the same time, centrifugation was employed to remove the precipitated waxes and lipids during the dilution. The clean diluted solution then served as the feedstock solution for the down stream chromatography. After being dried, the total solid content of the feedstock solution was 62.4 g/L.
Ginsenosides R1, Rg1, Re, Rb1, and Rd were analyzed by HPLC with a Kromasil 100‐5C18 column (250 mm × 4.6 mm) and confirmed by standards supplied from Taiwan Merck Co., Ltd. A Hitachi 2130 pump at 1 mL/min and L-2455 ultraviolet (UV) detector at 203 nm were used for the analysis of the ginsenosides. The mobile phase, a mixture of water and acetonitrile, was used for gradient elution, as shown in Table 1. Figure 1 is a typical chromatogram for the feedstock solution; the gradient settings were not able to separate ginsenosides Rg1 and Re. Therefore, in this study, both ginsenosides were treated as ginsenoside Rg1 for quantitative analysis. The corresponding factors between the peak area and the concentration of ginsenosides R1, Rg1 (Re), Rb1, and Rd were calibrated as 2270, 2598, 1919, and 2632, respectively. Accordingly, the percentage by weight of total ginsenosides in the dried feedstock was 30.5%, as shown in Table 2. In addition to ginsenosides, a group of weakly retained impurities with a retention time shorter than 3.5 minutes was also found (Figure 1). These could be polar impurities extracted by the aqueous ethanol solution that had not been completely precipitated and removed during the preparation of the feedstock solution.
Gradient Settings for Analysis of Sanchi Ginseng.

The Concentration and Content of Ginsenosides in Sanchi Crude Extract.

Materials and HPLC Analysis of American Ginseng
Ginseng root hair, which is a small and hair-like structure that grows on the surface of the main roots, is from Wisconsin, USA provided by Wellhead Bio. Tech. Co., Taiwan. The root hair was ground, and 900 g of the powder was ultrasonically extracted twice with 6 L of 95% ethanol at 50 °C for 90 minutes. The use of 95% ethanol as the extraction solvent was to minimize the extraction of polysaccharides, which could plug the chromatography. After centrifugation at 10 000 rpm for 20 minutes at room temperature, the supernatant sample was vacuum dried to obtain crude extract. This was subsequently washed twice with ethyl acetate (crude extract: ethyl acetate = 1:10 w/v) to eliminate lipids and waxes. After redrying, a deoiled concrete was collected and redissolved in 50% ethanol to act as a feedstock solution for the downstream chromatography. The solid content of the feedstock solution was 4.253 g/L, and the total amount of dried crude extract from 900 g of the root hair was 68.05 g. The supernatant and the feedstock solution were filtered through a 0.45µm syringe filter and analyzed by HPLC to examine the difference in composition.
The concentration of ginsenosides in the feedstock solution was analyzed by HPLC with a Luna C18 column (4.6 mm × 250 mm, 5 µm) at a flow rate of 1.3 mL/min and a UV detector at 203 nm. Acetonitrile and deionized water were used as the elutant, and the elution gradient was set as in Table 3. The chromatograms for the crude extract and the deoiled concrete are illustrated in Figure 3. It was observed that impurities with a retention time longer than 60 minutes in the crude extract were totally removed after being washed with ethyl acetate. It was also noted that ginsenoside Re was partially lost due to the ethyl acetate extraction.
The Gradient Settings for the Analysis of American Ginseng.

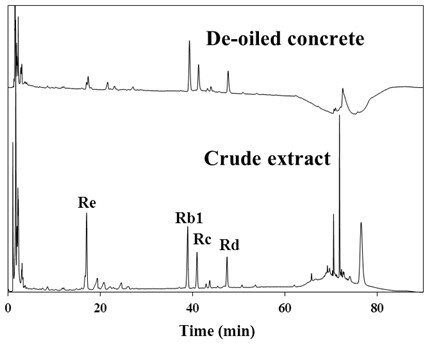
High-performance liquid chromatograms for crude extract and deoiled concrete.
The injection volume of the HPLC analysis was 20 µL, and the corresponding factors between the area and the concentration of ginsenosides Re, Rb1, Rc, and Rd were 3848, 4102, 4143, and 4970, respectively, confirmed by ginsenosides purchased from Taiwan Merck Co., Ltd. Accordingly, the concentrations of ginsenosides in the feedstock solution were 73.6, 561.4, 310.0, and 187.1 mg/L for Re, Rb1, Rc, and Rd, individually; their percentages by weight in the dried feedstock were 1.73 %, w/w, 13.20 %, w/w, 7.29 % w/w, and 4.40 % w/w, respectively.
Results and Discussion
Separation of Ginsenosides in Sanchi Ginseng
Single-column chromatography
A modified silica, Unisil 10‐100 C18 AQI (20 µm) from Suzhou NanoMicro Tech. Co., Ltd., was applied for the separation of ginsenosides from Sanchi ginseng. By packing the modified silica into a column of 1 cm in diameter and 15 cm in length, it was found that the retention and resolution of ginsenosides increased due to the water content in the aqueous ethanol solution at a flow rate of 3 mL/min. Figure 4 shows the chromatograms with different ratios of ethanol (95%) to water.

Single-column chromatography for Sanchi ginseng crude extract.
Figure 4 shows that the resolution of ginsenosides R1/Re/Rg1 was inadequate and both ginsenosides Rb1 and Rd could be eluted within 7 minutes by 60/40 EtOH/water. By increasing the water content to 35/65, the separation of ginsenosides R1 and Re/Rg1 was achieved; however, both ginsenosides Rb1 and Rd could not be eluted in 20 minutes. It was concluded that the separation of ginsenosides R1 and Re/Rg1 could only be completed with a lower content of ethanol; nevertheless, a second eluent would be required to elute ginsenosides Rb1 and Rd from the column. Although the chromatograms in Figure 4 show that ginsenosides Rb1 and Rd could be easily removed by a 60/40 ethanol solution, a 100/0 solution was used as the second eluent in this study to remove all the undetectable or undesorbed impurities in the feedstock.
SMB chromatography
Based on the triangle theory, a series of experiments with dissimilar switching times was designed and conducted at fixed flow rates: Q D = 5.0, Q E = 2.5, Q F = 0.2, Q R = 2.7 mL/min, Q W1 = 5.0 mL/min, and Q W2 = 5.0 mL/min. The separated products were analyzed by HPLC and dried to measure the total solid contents. Figure 5 shows the chromatograms of samples from the ports of Extract, Raffinate, and Wash 2 at different switching times. It was also noted that the collected samples from the ports of Wash 1 barely exhibited peaks for ginsenosides (not shown in Figure 5). This shows that the deployment of 95% ethanol as a second eluent could efficaciously regenerate the stationary phase. Figure 5 shows that, in the Raffinate outlet, the separation of weak retention impurities from ginsenosides, including R1, Rg1/Re, Rb1, and Rd, was completed in 10.5 minutes of switching time. Both ginsenoside R1 and Re/Rg1 were flushed from the port of Extract with a minute amount of Rb1, while ginsenosides Rb1 and Rd were mainly discharged from the SMB by 95% ethanol and collected at the port of Wash 2.
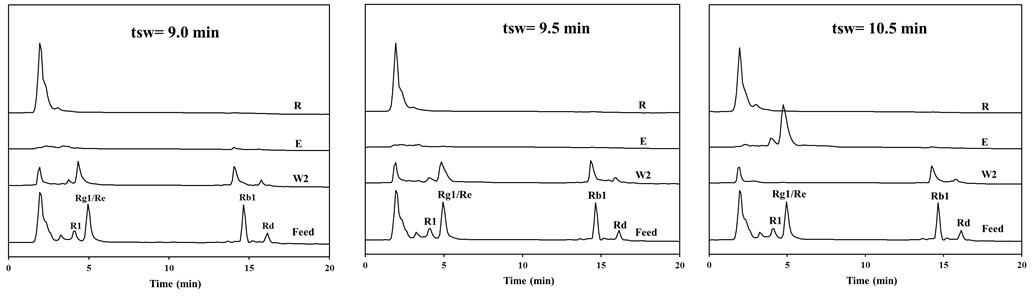
Chromatograms of separated products from simulated moving-bed experiments.
The SMB processes were generally designed and operated based on the triangle theory derived from equilibrium theory. 41 A right triangle region was formed in a plot of relative flow rate of m 2 and m 3, and the mapping of operation conditions on the phase plane should be exactly located inside the right triangle; that is, the relative flow rate in each section should satisfy the following conditions:

where K A and K B are the retention constants for weak retention components and strong retention components, m 2 and m 3 are the relative flow rate of the liquid phase to solid phase in section 2 and in section 3. The relative flow rate was generated by the following equation:
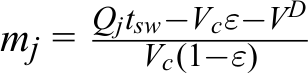
where Q j is the liquid flow rate in section j, t sw is the switching time, V C is the volume of the empty column, ε is the porosity of the packed bed, and V D is the dead volume of the SMB system.
If cleaning in place (CIP)-SMB is applied for ternary separation, an additional condition should also be satisfied 42,43 :

where K C is the strongest retention component in the feedstock, and n 2 is the number of columns in section 2.
From the single-column chromatography, it was observed that the retention times for impurity, Rg1/Re, and Rb1 (not shown in Figure 4) were 2.48, 9.72, and 33.50 minutes, respectively. Since the retention time for a nonretained compound was found as 1.65 minutes and the dead time was 0.05 minutes, the porosity of the column was calculated as 0.407. Accordingly, the retention constants for impurity, Rg1/Re, and Rb1 were computed as 0.36, 3.5, and 13.7.
By using equation (2) with the assumption of a zero dead volume, the relative flow rates in section 2 and section 3 were then determined as 2.54, 2.79, 2.72, 2.98, and 3.08, 3.73 with respect to 9.0, 9.5, and 10.5 minutes of switching time. This shows that equation (1) was fulfilled. The right side in the inequality equation (3) was solved as 3.09, 2.73, and 2.01, respectively. It was observed that only the experiment with 10.5 minutes of switching time satisfies the inequality equation (3). It was concluded that a ternary separation for the ginsenosides of Sanchi ginseng by CIP-SMB was achieved and it could be simply predicted by the triangle theory.
Table 4 illustrates the concentration and weight content of ginsenosides for separated samples with 10.5 minutes of switching time. The sample from the port of Raffinate contained only 0.2%, w/w, of ginsenoside R1, and those from the ports of Extract and Wash 2 contained 83.7%, w/w, and 89.2%, w/w, of ginsenosides, respectively. It also highlights that the sample from the port of Extract merely consisted of 2.0%, w/w, of ginsenoside Rb1 and that from the port of Wash 2 1.5%, w/w, of ginsenoside Rg1 (Re). It is, therefore, suggested that the separation of Rg1 (Re) and Rb1 was accomplished with high efficiency. With regard to Table 4, it can be concluded that 1000 kg of Sanchi ginseng could engender the production of 88.2 kg of concentrated product with 86.2%, w/w, of ginsenosides by mixing the separated product from the ports of Extract and Wash 2, as illustrated in Figure 6. The mixed and concentrated product could subsequently be applied as an active pharmaceutical ingredient.
Concentration and Percentage by Weight of Ginsenosides in Sanchi Ginseng After SMB.

Total solid content: Feed = 20.8 g/L, R = 0.807 g/L, E = 0.464 g/L, W2 = 0.187 g/L

Mass flow of ginsenosides for the extraction of Sanchi ginseng.
Separation of Ginsenosides From American Ginseng
Single-column chromatography
A modified silica, C18 AQ (40-60 µm), from Welch Co. was used for the separation of ginsenosides from American ginseng. It was found that the viscosity of the crude extract solution of American ginseng is much higher than that from Sanchi ginseng. After starting the SMB with C18 AQI as conducted for Sanchi ginseng, the back pressure of the SMB rose and the problem of plugging swiftly emerged. It was noted that the particle size of C18 AQ from Welch Co. is smaller than that of C18 AQI from NanoMicro Co. Therefore, the C18 AQ from Welch was used for American ginseng. As described, a pretreatment of ethyl acetate extraction to remove the lipids and waxes was carried out in order to resolve the plugging problem. Unfortunately, it was observed that plugging still occurred after 36 hours of operation. In addition to plugging, a white sticky precipitate was found and accumulated at the column’s frit. The sticky precipitate could instantly dissolve in the mixture of ethanol and water at 60 °C. As a result, the plugging issue could then be prevented by conducting the SMB at 60 °C, and the experiment could continuously operate for more than 2 weeks.
The C18 AQ was packed into a column of 1 cm in diameter and 15 cm in length and the column was eluted with aqueous ethanol solution at 3 mL/min. Figure 7 shows the elution of the feedstock solution with different ratios of ethanol and water at room temperature. In a previous study of Sanchi, ginsenoside Rg1, whose content was found to be higher than that of ginsenoside Re, 37 was chosen to represent the 2 ginsenosides. However, with regard to American ginseng, the ginsenoside Re content was reported to be dominant, 13 and, therefore, Re was selected to represent both ginsenosides in this American ginseng study.

Single-column chromatography at room temperature.
The SMB methodologies for Sanchi and American ginseng were designed based on the chromatograms of single columns. The majority of the ginsenosides found in both Sanchi and American ginsengs were Re/Rg1, Rb1, and Rd. However, dissimilar mobile phases were used for eluting ginsenosides in single columns, because the contents of polysaccharide and lipid in crude extracts of Sanchi ginseng and American are dissimilar. 44 These unwanted compounds affect the affinity level between ginsenosides and columns differently. In other words, ginsenosides R1 and Re/Rg1 in Sanchi ginseng were eluted by 35/65 EtOH/ water, while ginsenosides Rb1 and Rd could not be eluted within 20 minutes. Increasing the ethanol ratio to 60/40, Rb1 and Rd could be easily released (Figure 4). From another point of view, 45/55 of EtOH/H2O could totally separate ginsenosides Re, Rb1, Rc, and Rd in American ginseng; however, ginsenoside Rd was too strongly adsorbed to be eluted. By increasing the content of ethanol to 60/40, ginsenoside Rd could be eluted with inadequate resolution of ginsenoside Re. Therefore, 50/50 EtOH/H2O was selected as the mobile phase for the separation of ginsenosides from American ginseng (Figure 7).
Figure 8 shows the chromatograms for the feedstock solution and ginsenosides at 60 °C with 50/50 of EtOH/H2O. As can be seen, ginsenoside Re was merged into the first eluted peak from the feedstock, which could be regarded as the unretained impurities. As a result, ginsenoside Re could not be separated from the unretained impurities by 50/50 EtOH/H2O. Ginsenoside Re, a PPD, could also be easily and individually removed by ethyl acetate extraction, as illustrated in Figure 3. Therefore, the application of SMB in this study aimed to isolate ginsenosides Rb1, Rc, and Rd, which are categorized as PPTs.
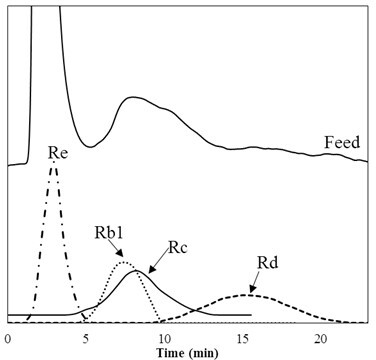
Single-column chromatography at 60°C.
SMB chromatography
Since SMB is a technology for binary separation, the separation strategy includes first, the removal of both ginsenoside Re and unretained impurities from the port of Raffinate; the remaining ginsenosides Rb1, Rc, and Rd from the port of Extract were further separated by conducting another SMB separation. A typical chromatogram for the feedstock solution, shown in Figure 1, demonstrates that the gradient setting is not able to separate ginsenosides Rg1 and Re. In addition, the separation of Re and Rg1 could not be achieved in single columns (Figure 7) with different mobile phases. Therefore, the separation of the 2 compounds could not be obtained by the mixture of ethanol and water, which are generally recognized as safe.
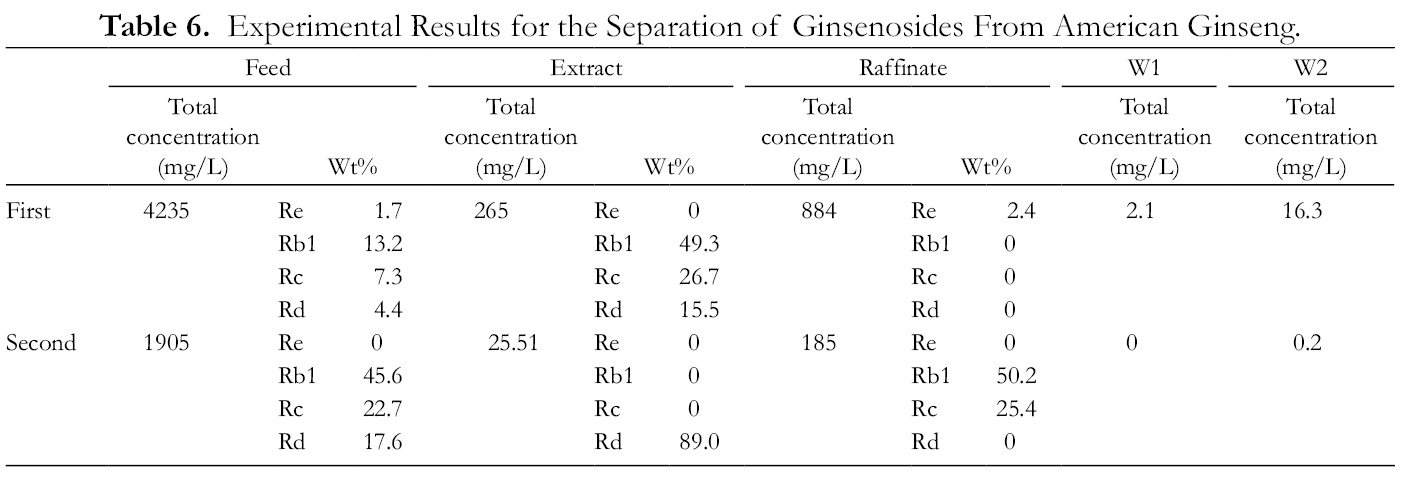
Similarly, the SMB experiments were designed based on the triangle theory, which was mentioned in the study of Sanchi ginseng. Figure 9 shows the chromatograms for separated samples from the first SMB, and Figure 10 reveals those from the second SMB. The operating conditions and experimental results are illustrated inTables 5 and 6. It is demonstrated that the solution collected at the port of Extract from the first SMB was further concentrated by rotary evaporation, and the weight fraction of each ginsenoside slightly changed. Figures 8 and 9 show that ginsenoside Re and the unretained impurities were removed in the first SMB, and ginsenosides Rd and Rb1 with Rc were separated by the second SMB. According toTables 5 and 6, the mass flow for the extraction of ginsenosides in American ginseng is summarized in Figure 11. A product with 91.5 %, w/w, of ginsenosides was obtained from the port of Extract after the first SMB separation, and a ginsenoside Rd with 89 %, w/w, was isolated after the second SMB.

Chromatograms from the first simulated moving bed separation for American ginseng.

Chromatograms from the second simulated moving bed separation for American ginseng.
Setting of the SMB Separation for American Ginseng.

Experimental Results for the Separation of Ginsenosides From American Ginseng.

Mass flow of ginsenosides for the extraction of American ginseng.
The mass flow of ginsenosides for American ginseng is illustrated in Figure 11. First, 75.71 kg of crude extract was obtained by using ethanol 95% combined with ultrasound extraction at 50 °C for 90 minutes. The crude extract was then pretreated by ethyl acetate before being dissolved in 50% ethanol to become a feedstock solution with a concentration of 4.253 g/L for the first SMB. After the first SMB, 1.97 and 53.98 kg of impurities were removed from the ports of Wash 1 and Wash 2 and Raffinate, respectively. The separated product from the port of Extract contained 91.5%, w/w, of ginsenosides.
The highest yield of ginsenosides from American ginseng was 7.51% using 70% aqueous ethanol solution, 27 and 4.41% by high-pressure water extraction at 200 MPa at 60 °C. 28 Although the extraction yields for both Sanchi and American ginsengs are low as 1.90% and 2.01%, respectively, the obtained extracts could be used for the downstream chromatography preventing clogging of the columns. Therefore, it will be worthwhile to recover the ginsenosides from the residue by using 70% ethanol to produce a polysaccharide-rich extract.
In addition, the separation of lipids from the crude extract by ethyl acetate coincided with the separation of Re. From the peak areas of the chromatogram in Figure 3, it is estimated that approximately 75% of Re would be extracted and blended in ethyl acetate. Hence, a methodology for Re recovery from ethyl acetate is indispensable to a study in the future.
In the second SMB, the separation of Rd (the strongest retained ginsenoside) from ginsenosides Rb1 and Rc was accomplished by applying the same SMB; 34.2 kg of ginsenoside Rd with 89.0%, w/w, was obtained. Additionally, 16.30 kg of a mixture of ginsenosides Rb1 and Rc with 50.2% and 25.4%, w/w, respectively, was attained. The separated products from the first and second SMBs possessed high purity and could serve as active pharmaceutical ingredients for the traditional Chinese herb industry.
Conclusion
In this study, the application of an SMB for the separation of ginsenosides from Sanchi and American ginseng was conducted and products with ginsenoside content greater than 85%, w/w, were obtained. With respect to Sanchi ginseng, a concentrated product with 86.2%, w/w, of ginsenosides R1, Rg1, Re, Rb1, and Rd was recovered by mixing the separated products from the ports of Extract and Wash 2. As for American ginseng, a concentrated product with ginsenosides Rb, Rc1, and Rd was collected at the port of Extract with 91.5%, w/w, and a ginsenoside Rd with 89.0%, w/w, was further isolated from the concentrated product.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been supported by JOPE Technology Co., Ltd. for the supply of the SMB equipment and by Wellhead Biological Technology Corp. for the provision of American Ginseng (Panax quinqueflius L.) root hair.
