Abstract
Tinea pedis (athlete’s foot) is a dermatophyte infection of the feet and is the most common type of dermatophytosis. Tenia pedis is a significant global public health problem affecting approximately 15%-25% of the worldwide population.
1
It typically involves the skin between toes but can spread to the feet, other parts of the body, and to other people.
1
Common symptoms of tinea pedis include itchy erosions and/or scales between the toes, erythema, and hyperleratotic (moccasin type), small-to-medium blisters (vesiculobullous type), or ulceration between the toes or pustules (ulcerative type).
2
Although tinea pedis is commonly caused by
Kaffir lime (
The main components of the essential oils distilled from
Microemulsions consist of a water phase, an oil phase, a surfactant, and a cosurfactant.
23
Surfactant molecules form a monolayer on the surface of an oil droplet by turning the hydrophilic part into an aqueous phase and the hydrophobic part into an oil phase, resulting in a very low value of interfacial tension at the oil and water interface.
24
Microemulsions have a particle size of approximately 10‐100 nm
25
and microemulsion drug delivery systems are transparent and thermodynamically stable.
26
Microemulsions containing essential oils improve pharmaceutical and biopharmaceutical properties of the bioactive compounds in oils. They also increase physical and chemical stability, control release, increase solubility and bioavailability, and enhance skin penetration and retention of the bioactive compounds in essential oils.
27
Panapisal et al reported that microemulsions containing Labrasol as a surfactant helped solubilize silymarin and protected it from oxidation.
28
Skin permeability and retention of
The aim of the present study was to design a microemulsion system to deliver kaffir lime oil as an alternative antifungal agent against
Materials and Methods
Materials
Plant Collection and Identification
Leaves of
Chemicals and Reagents/Drugs
The (±)-citronellal analytical standard (CAS No. 106‐23-0, Lot No. 03611 PA), clotrimazole reference standard (CAS No. 23593‐75-1, Lot No. BCBB5668), ketoconazole reference standard (CAS No. 65277‐42-1, Lot No. LRAA9173) were purchased from Sigma-Aldrich (St Louis, MO, USA). Dimethyl sulfoxide (DMSO; CAS No. 67‐68-5, Lot No. 1670195) and sodium sulfate (CAS No. 7757‐82-6, Lot No. 1601210124) were purchased from Thermo Fisher Scientific (Waltham, MA, USA). Poly-oxyethylene 80 (CAS No. 9005‐65-6, Lot No. 710860), sorbitan monooleate 80 (CAS No. 1338‐43-8, Lot No. 90710E), and propylene glycol (PG) (CAS No. 57‐55-6, Lot No. 4H0Y0B) were obtained from NSG Nam Siang Co. Ltd. (Bangkok, Thailand).
Test Organism and Culture Media
Hydrodistillation of C. hystrix Leaves
Fresh
Chemical Analysis of Kaffir Lime Oil Constituents by Gas Chromatography Coupled With Mass Spectrometry
Kaffir lime oil composition was determined by gas chromatography-mass spectrometry (GC-MS) analysis using Finnigan Trace GC ultra (Thermo Fisher Scientific, Waltham, MA, USA), and DSQ Quadrupole detector fitted with a fused silica column MEGA-5MS (30 m × 0.25 mm i.d; 0.25 µm film thickness). The GC-MS spectrum was obtained using helium as a carrier gas with a flow rate of 1.0 mL/min, split 1:100, an injector temperature of 180 °C, and a transfer line temperature of 275 °C. The column temperature progressed from 60 °C, held for 1 minute, then increased to 240 °C (at a rate of 3 °C/min). An electronic impact ionization mode at a fixed electron energy of 70 eV with ion source temperature of 200 °C was used. The acquisition mass range was 40‐650 amu with a threshold of 0 amu. The scan acquisition rate was 500 amu/sec, The chemical constituents were identified by comparing the Kovats gas chromatographic retention indices of the peaks on the HP-5MS column with the Adams EO by comparison of their MS with those stored in the MS database (NISTO5 library) and with those reported in the literature. 31,32 The alkane series used was C4-C34 alkanes. Mass spectra library and the percentage composition was computed from GC peak areas.
Preparation of Kaffir Lime Oil Microemulsion by the Phase Titration Method
Pseudo-ternary phase diagrams of kaffir lime oil, surfactant, cosurfactant, and water were constructed using a water titration method to obtain an optimal ratio and concentration of each component forming a microemulsion. The surfactant was mixed with the cosurfactant in fixed-weight ratios of 1:0, 1:1, 2:1, and 3:1. These mixtures (Smix) were then combined with kaffir lime oil at 25 °C. The ratio of oil to Smix varied from 1:9, 2:8, 3:7, 4:6, 5:5, 6:4, 7:3, 8:2, and 9:1 (v/v). Each mixture was titrated 10 times with 100 µL of ultrapure water and then followed by the addition of 1000 µL of water until the total volume was 11 000 mL under 15 seconds of vigorous stirring with a vortex mixer. The formation of a microemulsion was confirmed by visual observation for transparency. The ratios of Smix, oil, and water that gave transparent mixtures were marked as points in the phase diagram. The area covered by these points was considered as the microemulsion region. The transparency of the microemulsion was observed again after 24 hours.
Characterization of the Kaffir Lime Oil Microemulsion
Measurements including hydrodynamic diameter, PDI, and the zeta potential values of the kaffir lime oil microemulsion were carried out using a dynamic light scattering technique on a Zetasizer Nano ZS (Malvern Panalytical, Bristol, UK). For the analysis, the microemulsion was diluted 10 times with ultrapure water. The effective hydrodynamic diameter and PDI were recorded at an 173° scattering angle under 25 °C.
Determination of the Physical Stability of the Kaffir Lime Oil Microemulsion
The kaffir lime oil microemulsion was placed in light-protected glass vials and stored at 4 °C, 30 °C, and 45 °C for 28 days. At different storage times (7, 14, 21, and 28 days), the size, PDI, and zeta-potential values of the samples were evaluated via dynamic light scattering.
Broth Macrodilution Method for the Determination of Kaffir Lime Oil and Citronellal MIC for T. mentagrophytes var. interdigitale
Test Procedure
The antifungal test that followed the macrodilution broth method adapted from the United States Pharmacopeia–National Formulary turbidimetric method was performed in standard test tubes.
34
An adjusted inoculum suspension of 1 mL was added to each tube containing 1 mL of kaffir lime oil or citronellal from the 2-fold dilution series and mixed. This resulted in a 1:2 dilution of each kaffir lime oil or citronellal concentration and a 1:2 dilution of the inoculums. Therefore, the final concentrations of kaffir lime oil and citronellal in the inoculums were 0.0156%-1% v/v (0.13-8.51 mg/mL) and 0.0625%-4%v/v (0.54-34.40 mg/mL), respectively. The potato dextrose broth without kaffir lime oil was used as a negative control and the broth containing a final concentration of 1% v/v DMSO was used as a solvent control. Clotrimazole and ketoconazole at final concentrations of 500 and 10 µg/mL, respectively, were used as positive controls. The inoculated tubes were incubated at 28 °C for 7 days. Endpoint determination readings were performed visually and based on a comparison of the fungal growth in tubes containing the kaffir lime oil with the negative control. The MIC was defined as the lowest concentration at which
Agar Well Diffusion Assay
An agar well diffusion test was performed using potato dextrose agar containing 2.4 g of potato dextrose medium and 1.5 g of agar in 100 mL of purified water. Sterilization was achieved through autoclaving, after which 20 mL of potato dextrose agar was poured into the assay plate and allowed to cool. The inoculum was prepared using
Quantitative Analysis of Citronellal in Kaffir Lime Oil and Microemulsion
UV-Visible Spectrophotometric Analytical Method Development
A standard solution of citronellal at concentrations ranging from 0.2 to 10.6 mg/mL in DMSO was prepared by 2-fold dilution. The absorbance of the solutions containing citronellal was determined by UV-Visible (UV-Vis) spectrophotometry in the UV range of 200‐400 nm using DMSO as a blank. The UV spectra of citronellal at different concentrations were obtained to determine the wavelength of the greatest absorbance.
Validation of the Analytical Method
Validation of the UV-Vis spectrophotometric analytical method for citronellal was carried out based on parameters including linearity, limits of detection and quantification, accuracy, and precision. The linearity of the analytical method was verified by preparing 3 different standard solutions of citronellal (0.2-10.6 mg/mL) to plot 3 calibration curves. The linearity of the standard curve was evaluated by linear regression analysis using the least squares method. The sensitivity of the UV-Vis spectrophotometry to measure citronellal was evaluated in terms of the limit of detection (LOD) and the limit of quantification (LOQ). LOD was estimated by the lowest concentration of citronellal that could be detected, whereas LOQ was the lowest concentration of citronellal that could be determined with acceptable precision and accuracy. The LOD and LOQ were calculated using Equations (1) and (2) as follows.


where

The precision of the method was assessed by determining inter- and intraday variations. Intraday variation was ascertained by analyzing 1.328, 2.656, 5.313, and 10.625 mg/mL of citronellal 3 times in the same day. Interday precision was evaluated by analyzing 1.328, 2.656, 5.313, and 10.625 mg/mL of citronellal 3 times over the course of 3 days. The percent relative standard deviation (% RSD) of the citronella concentrations was calculated from the regression linear equation.
Determination of % Recovery of Citronellal in Kaffir Lime Oil Microemulsion
The % recovery of citronellal loaded in the kaffir lime oil microemulsion was measured by UV-Vis spectrophotometry. The kaffir lime oil microemulsion was dissolved in DMSO by adding 100 µL of microemulsion to 900 µL of DMSO. The components used to formulate the microemulsion in the absence of kaffir lime oil were diluted in DMSO and set as a blank for UV-Vis spectrophotometry. The amount of citronellal in the kaffir lime oil microemulsion was determined by measuring absorbance at a maximum wavelength of 292.4 nm using a double-beam UV-1700, UV-Vis spectrophotometer (Shimadzu Corp., Kyoto, Japan). The amount of kaffir lime oil in the microemulsion was calculated using standard calibration data. The recovery percentage of citronellal in the microemulsion was calculated using Equation (4).

Chemical Stability of Citronellal in Kaffir Lime Oil Microemulsion
The chemical stability of citronellal in kaffir lime oil microemulsion was assessed by measuring the change in citronellal concentration in the microemulsion during storage. The kaffir lime oil microemulsion was placed in light-protected glass vials and stored at 4 °C, 30 °C, and 45 °C for 28 days. At predetermined time intervals (7, 14, 21, and 28 days), the samples were collected. To prepare them for measurement, the microemulsions were diluted 10 times in DMSO (100 µL of microemulsion diluted in 900 µL of DMSO) to dissolve the citronellal. A microemulsion without kaffir lime oil was used as a blank. A calibration curve was created by dissolving the citronellal standard in DMSO in a range of 0.2‐10.6 mg/mL (

Statistical Analyses
Statistical analyses were performed using Graphpad Prism 7.0 (GraphPad Software, San Diego, CA, USA). Results are expressed as mean ± SD. Statistical evaluation of the data was performed using a one-way analysis of variance. The Newman–Keuls post hoc test was used to obtain the significance of the differences. To compare the significance of the difference between the means of 2 groups, a
Results and Discussion
Kaffir Lime Oil Distillation Yield
The essential oil yield from the hydrodistillation of
Kaffir Lime Oil Constituents Analyzed by GC-MS
Twenty-three compounds from the leaves of
Kovats Retention Index, and Percentage Chemical Composition of the Compounds Identified by the GC-MS Analysis of the Essential Oil Distilled From

Abbreviation: GC-MS, gas chromatography mass spectrometry.
aKovats retention index was identified by Adams and Davies (1990). 31,32
Previous studies have reported that citronellal was the main volatile compound found in the essential oil of kaffir lime leaves, representing 46.4%-80.04% of all the components.
36
-38
The other volatile compounds were α-pinene, camphene, β-pinene, sabinene, myrcene, limonene, (
In another study, GC-MS analysis demonstrated that kaffir lime oil was composed of at least 20 substances, with β-citronellal (46.4%) being the major component. 40 The variation in the chemical composition of the kaffir lime essential oil may be due to several factors including ecotypes and chemotypes of the plant species, and exogenous variables such as light, growing location, nature of the soil, precipitation, radiation, and temperature. 41
Construction of the Pseudo-Ternary Phase Diagram
Pseudo-ternary phase diagrams were constructed based on increases of water content and decreasing Smix contents. The effects of PG as a cosurfactant were studied by varying the ratios of Tween 80 and PG at 1:0, 1:1, 2:1, and 3:1. Figure 1 shows the pseudo-ternary phase diagrams of kaffir lime leaf oil, Smix, and water-based microemulsion systems. Based on the phase diagrams, the system composed of Tween 80/kaffir lime oil/water formed microemulsion regions from 15% to 78%, 15% to 48%, 15% to 55%, and 15% to 75% of kaffir lime oil at Tween 80:PG ratios of 1:0, 1:1, 2:1, and 3:1, respectively. These results indicate that increasing the Tween 80/PG ratio helps to increase the loading of kaffir lime oil in the microemulsion. In general, cosurfactant is added to the microemulsion to increase solubility of the drug/oil and increase the interfacial fluidity of the surfactant film on the surface of the microemulsion. 42 In addition, the surfactant and cosurfactant adsorbed at the interface may reduce interfacial energy and improve the thermodynamic stability of the microemulsion. 43 However, the concentration of the cosurfactant in a microemulsion should be minimized as it may reduce the solubility of oil in the system when the cosurfactant migrates toward the aqueous phase. In this study, the kaffir lime microemulsion could be formed with and without adding cosurfactant. Increasing the cosurfactant may lead to a reduction in the incorporation of oil in the microemulsion as the PG may migrate toward the aqueous phase upon dispersion into the aqueous media, leading to decreased oil solubility. 44

Pseudo-ternary phase diagrams of kaffir lime oil, Tween 80 and propyleneglycol, and water. Microemulsions were prepared by using a ratio of Tween 80 andpropylene glycol at (A) 1:0, (B) 1:1, (C) 2:1, and (C) 3:1.
Characterization of the Kaffir Lime Oil Microemulsion
The microemulsion containing 42.5 mg/mL (5% v/v) of kaffir lime oil was selected for further characterization, a stability test, and an antifungal activity test against
The size and PDI of the microemulsion containing 5% kaffir lime oil were within acceptable ranges. The small droplet size of the microemulsion relates to a large surface area/volume ratio and may increase drug transport into or across the skin. 45,46 The PDI is a value representing the distribution of size populations in a sample. Generally, a PDI of 0.2 or lower is considered acceptable and indicates a homogeneous population of the drug delivery system. 47 The zeta potential of the microemulsion was found to have a slightly negative charge, suggesting that the nearly neutral charge of the microemulsion was the result of the nonionic surfactant used to prepare it. The zeta potential values are in agreement with a previous report using Tween 80 and PG as a surfactant and cosurfactant in the formulation. 48,49 Kaffir lime oil microemulsion was formulated using a low concentration of surfactant (20% v/v), to minimize skin irritation. 50,51 There are several factors affecting the particle size and narrow PDI of microemulsions including the hydrophilic–lipophilic balance value of the surfactants and the type and concentration of surfactants adsorbed at the oil–water interface. Surfactants decrease the interfacial tension and stabilize the oil–water interface by reducing interfacial energy between the oil and water.
Effects of Time and Temperature on the Physical Stability of Kaffir Lime Oil Microemulsions
The physical stability of the kaffir lime oil microemulsion was evaluated by observing changes in particle size, size distribution, and zeta potential values over 28 days of storage at 4 °C, 30 °C, and 45 °C. The evaluations were carried out to uncover the optimal formulation containing 5% kaffir lime oil, 20% Smix, and 75% water. The results indicated that the colloidal stability of the kaffir lime oil microemulsion was influenced by time and storage temperature, as shown in Figure 2. The results of average size, PDI, and the zeta potential values of the microemulsion at days 0, 7, 14, 21, and 28 displayed no statistically significant differences at storage temperatures of 4 °C and 30 °C. These results show that the developed kaffir lime oil microemulsion was highly stable when stored at 4°C and 30 °C. The high stability of the microemulsion with a low zeta potential at 4 °C and 30 °C suggests a thermodynamically stable system. The zeta potential values became more positively charged when the microemulsion was stored at 45 °C for 14, 21, and 28 days. The zeta potential is an indicator of colloidal dispersion stability, and the magnitude of the zeta potential indicates the degree of electrostatic repulsion between adjacently charged microemulsion droplets in dispersion. 52 A high positive or negative zeta potential can prevent a microemulsion from aggregation and coalescence. In this study, the zeta potential of the kaffir lime oil microemulsion stored at 45 °C became more positive, which was closer to a zero value. This result suggests that microemulsions stored at higher temperatures tend to be unstable. However, aggregation and coalescence were not found in this study, as the adsorption of surfactant on the surface of the microemulsion can decrease the interfacial tension between the kaffir lime oil and the aqueous phase to a very low value, thus enhancing colloidal stability. 24 Temperature affected action over the interface and the solubility of the surfactant. Tween 80 is a nonionic surfactant that is known to exhibit temperature-dependent behavior. Increasing the temperature results in dehydration of the oxyethylene head groups of the Tween 80 molecules, leading to a loss of hydrophilicity and an increase in lipophilicity. 51 This may affect the directional interface adsorption layer on the surface of the microemulsion and the zeta potential. 53

(A)Particle size, (B) polydispersity index, and (C) zeta potential values ofmicroemulsion of kaffir lime oil after freshly prepared and stored at 4 °C, 30°C, and 45 °C for 7, 14, 21, and 28 days. Data represent mean ± SD (n=3), *indicates
The physical stability of the microemulsion at lower temperatures occurs because the surfactant and cosurfactant are adsorbed on the surface of the oil droplets and decrease the interfacial tension between oil and water to extremely low values. A surfactant has a hydrophilic polar head and a lipophilic, nonpolar tail. Therefore, when surfactant is added to oil and water, it forms a film at the oil–water interface. Cosurfactants also reduce interfacial tension. Fluidization of the surfactant film on the surface of oil droplets helps to increase the entropy of the system, hence increasing thermodynamic stability. 54 In addition, during dilution with water, the bulk concentrations of the surfactant and cosurfactant decrease their chemical potential in bulk and at the interface, thus decreasing the free energy of the system. 24
Antifungal Activity of Kaffir Lime Oil and Microemulsion Against T. mentagrophytes var. interdigitale
Kaffir lime oil and citronellal potentially inhibited the growth of

Abbreviation: DMSO, dimethyl sulfoxide.
(+) Visible growth appearance (turbid tube), (−) No visible growth appearance (clear tube).
The agar well diffusion assay was used to confirm the antifungal activity of the kaffir lime leaf oil microemulsion and exhibited a potent antifungal activity against
Antifungal Activity of Kaffir Lime Oil Microemulsion, Ketoconazole, Clotrimazole, and Microemulsion Base Against

Data represent mean ± SD from 3 experiments.
Previous studies have reported antifungal activities exhibited by citronellal against
Analytical Method of Validation for the Quantitative Analysis of Citronellal in Kaffir Lime Oil and Microemulsion
Linearity
The absorbance of the standard solutions of citronellal was determined to be in the UV range of 200‐400 nm. The

Calibration curve of citronellal at λmax of 292.4 nm. Data represent mean ± SD from three experiments.
Limits of Detection and Quantification
The LOD and LOQ of citronellal were 0.069 and 0.231 mg/mL, respectively. LOD is the lowest possible concentration of citronellal at which the UV-Vis spectrophotometer can make detections with a certain degree of confidence. LOQ is the lowest concentration of citronellal that can be reliably quantified by this method.
Accuracy
The standard addition technique for determining the accuracy of UV-Vis spectrophotometry was used in this study. A known amount of pure citronellal was added and the sample was reassayed. The accuracy of UV-Vis spectrophotometry to analyze the citronellal was determined by the recovery percentage of a known quantity of citronellal (50%, 100%, and 150% or 0.638, 1.275, and 1.913 mg/mL, respectively) spiked in a preanalyzed citronellal solution (1.275 mg/mL). The difference between the theoretical amount and the amount analytically determined in the spiked samples was expressed as % recovery. According to the results, the mean % recovery values of citronellal analyzed by the current method ranged from 104% to 110% (Table 4). Typically, the mean recovery should be within 90%-110% of the theoretical value for unregulated products. 59 Our results showed that the % recovery of citronella analyzed by UV-Vis spectrophotometry was in the acceptable range, suggesting that small changes in the oil concentration in solution could be accurately determined by this method.
Recovery Percentage of Citronellal for Accuracy Evaluation of the UV-Vis Spectrophotometry Method.
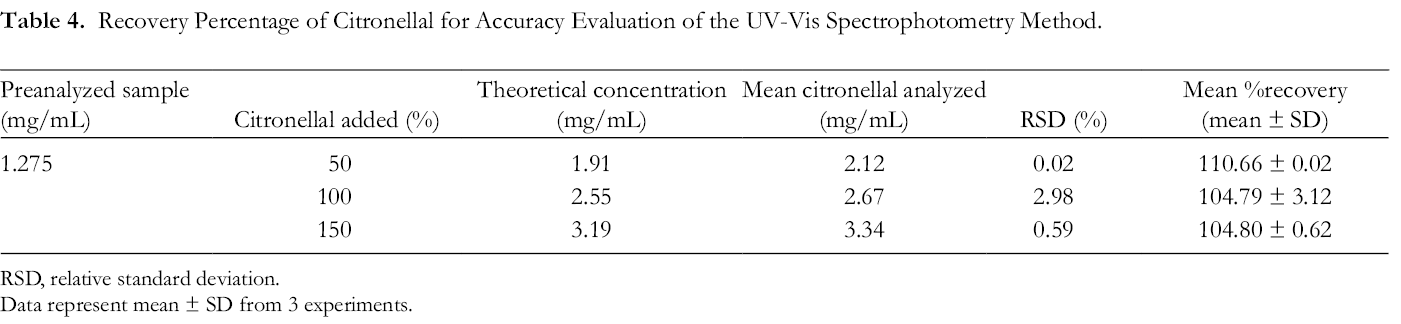
Abbreviation: RSD, relative standard deviation.
Data represent mean ± SD from 3 experiments.
Precision
Precision is a measure of repeatability among the results obtained from a series of experiments under similar conditions and aims to present the random error that may occur in an experiment. In this study, the precision of the proposed analytical method was evaluated by intra- and interday repeatability of the results following replicate absorbance measurements of the standard citronellal solution in DMSO at various concentrations. Precision was expressed in % RSD. The results showed that % RSD of both intra- and interday precision at all tested concentrations was below 2% and within an acceptable range (Table 5). 59 These results indicate that this method yielded a precise determination of citronellal.
Precision of Analytical Method for Determining Citronellal by UV-Vis Spectrophotometry.

Abbreviation: RSD, relative standard deviation.
Data represent mean ± SD from 3 experiments.
Chemical Stability of Citronellal in the Kaffir Lime Oil Microemulsion
The % recovery of citronellal in the kaffir lime oil microemulsion was 104.11% ± 4.90%, suggesting a successful loading of kaffir lime oil in the microemulsion system. The chemical stability of citronellal in the kaffir lime oil microemulsion is shown in Figure 4. The remaining percentage of citronellal in the microemulsion stored at 4 °C somewhat decreased over 28 days. The percentages of citronellal in the microemulsions stored at 30 °C and 45 °C significantly decreased with incubation time. After storage for 28 days, the amount of citronellal stored at 4 °C, 30 °C, and 45 °C was 86.36% ± 1.77%, 75.74% ± 2.99%, and 45.01% ± 2.57%, respectively. The reduction in citronellal and other chemical components in kaffir lime leaf oil microemulsions may be due to both chemical and physical degradation. The lipophilic and volatile components in kaffir lime oil are known to be susceptible to degradation by oxidation, dehydration, polymerization, isomerization, and thermal rearrangement. 60 At higher temperatures, the volatile components in kaffir lime oil microemulsions tend to vaporize more readily than the oils in microemulsions stored at lower temperatures.

Chemical stability ofcitronellal in microemulsion of kaffir lime oil. Data represent mean ± SD fromthree experiments. * indicates p < 0.05, **indicates p < 0.01, and **** indicates p < 0.0001, compared with Day 0.Statistical analysis was performed using ANOVA, followed by Newman–Keuls comparison test.
Conclusion
In this study, the chemical composition of
Footnotes
Acknowledgments
The authors gratefully acknowledge the use of the facility of the Research Center for Drug Discovery and Development, Srinakharinwirot University. This research work was partially supported by Chiang Mai University. The authors appreciate Associate Professor Dr Tapanee Hongratanaworakit for providing citronellal for use in this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Srinakharinwirot University research grant (078/2563).
