Abstract
A new neolignan, (7S,8R)-threo-1′-[3′-hydroxy-7-(4-hydroxy-3,5-dimethoxyphenyl)-8-hydroxymethyl-7,8-dihydrobenzofuran]acrylaldehyde (
Xanthium strumarium (Compositae) is an annual plant widely distributed in eastern China. The fruits of X. strumarium, named as “Cang-Er-Zi” or “Chang-Er-Zi” in China, were traditionally used to treat nasal sinusitis,
1
headache caused by wind cold,
2
urticaria,
2
and arthritis.
3
And X. strumarium also exhibited some useful medicinal properties such as diuretic,
4
anthelmintic,
5
antifungal,
6
anti-inflammatory,
7
antidiabetes,
8
anticancer,
9
and other activities. Previous chemical studies of X. strumarium revealed the presence of sesquiterpene lactone,
10
phenolic acid,
3
diterpene glucoside,
11
lignan,
2
and flavonoid.
12
However, there are not enough investigations to further study the constituents of X. strumarium. In continuation of our ongoing search for bioactive compounds from natural products, the fruits of X. strumarium were investigated. Herein we report the isolation and structural characterization of a new neolignan, together with 5 known compounds
Results and Discussion
Compound
Nuclear Magnetic Resonance Data of Compound 1 in Deuterated Methanol (1H: 500 MHz, 13C: 125 MHz).


Key heteronuclear multiple bond correlation and nuclear overhauser effect spectroscopy correlations of compound 1.
Moreover, 5 known compounds

The structure of compounds 1-
All the isolates were evaluated for their cytotoxicity against human cancer cell lines of HepG2, MCF-7, HCT-116, and SGC-7901, using cisplatin as a positive control (Table 2). The primary screening results of compounds
Cytotoxicities of Compounds 1
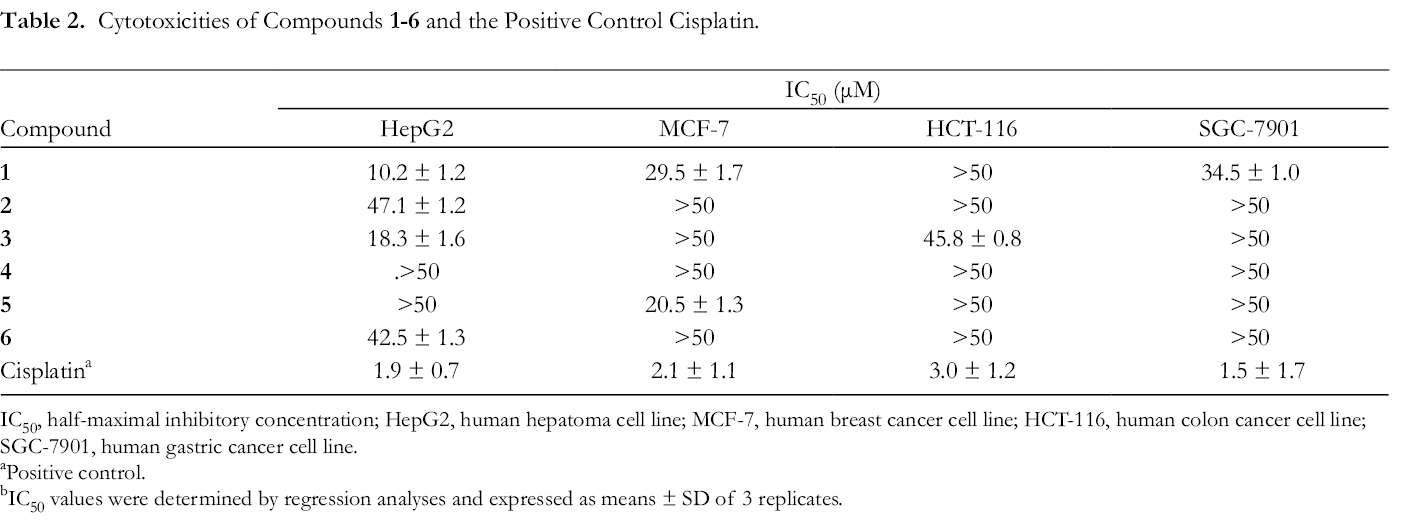
IC50, half-maximal inhibitory concentration; HepG2, human hepatoma cell line; MCF-7, human breast cancer cell line; HCT-116, human colon cancer cell line; SGC-7901, human gastric cancer cell line.
aPositive control.
bIC50 values were determined by regression analyses and expressed as means ± SD of 3 replicates.
Experimental
General
Optical rotations were determined on a Perkin-Elmer polarimeter 341 (Perkin-Elmer, Waltham, MA, USA). Ultraviolet (UV) spectra were recorded on the Jasco-UV-vis spectrophotometer (JASCO, Tokyo, Japan). Infrared (IR) spectra were recorded with a Nicolet Magna FT-IR 750 spectrometer (Nicolet, Madison, WI, USA). NMR spectra were recorded on a Bruker 500 MHz spectrometer with tetramethylsilane as an internal standard (Bruker, Karlsruhe, Germany). Mass spectra were obtained on a Q-TOF Micro LC-MS spectrometer (Waters, Milford, MA, USA). Column chromatography was conducted using silica gel 60 (200 μm particle size, Yantai Xinde Chemical Co., Ltd, Yantai, China). Thin-layer chromatography was performed with precoated silica gel GF254 glass plates (Qingdao Marine Chemical Co., Ltd). A Waters 2535 high-performance liquid chromatography (HPLC) was carried out using a 2998 Photodiode Array Detector and a C-18 column (Waters Sunfire, 10 × 250 mm, 5 µm particle size).
Plant Material
The fruits of X. strumarium were collected in Zibo, Shandong province, People’s Republic of China, and authenticated by Professor Jing Wang (Xiangya Medical College, Central South University). A voucher specimen of the plant (No. 20180923) was deposited at the Xiangya Medical College, Central South University, Changsha, People’s Republic of China.
Extraction and Isolation
Dried fruits of X. strumarium (5.0 kg) were extracted 3 times with 75% ethanol (EtOH) aqueous to give a crude extract (532.7 g). This extract was suspended in water (H2O), partitioned successively with petroleum ether (PE), chloroform (CHCl3), ethyl acetate (EtOAc) and n-butanol (n-BuOH). The EtOAc fraction (88.6 g) was subjected to a silica gel column using a gradient of dichloromethane (CH2Cl2)-methanol (MeOH) (from 100:1 to 1:1) to yield 12 fractions (Fr.1-Fr.12). Fr.4 (14.5 g) chromatographed on a silica gel column eluted with a gradient of PE-EtOAc (from 100:1 to 0:1) and was separated into 10 fractions (Fr.4.1-Fr.4.10). In addition, Fr.4.4 (2.2 g) was subjected to the RP-18 column and using the elution of MeOH-H2O (from 3:7 to 1:0) to afford 12 fractions (Fr.4.4.1- Fr.4.4.12). Fr.4.4.5 (271.5 mg) via preparative HPLC using a solvent system of 60% MeOH in H2O over 80 minutes yielded compound
(7S,8R)-threo-1′-[3′-hydroxy-7-(4-hydroxy-3,5-dimethoxyphenyl)-8-hydroxymethyl-7,8-dihydrobenzofuran]acrylaldehyde (1)
A yellow amorphous powder;
Cytotoxicity Assay
The 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay was chosen to evaluate the cytotoxicity of compounds
Supplemental Material
Supplementary Material 1 - Supplemental material for Chemical Constituents From the Fruits of Xanthium strumarium and Their Antitumor Effects
Supplemental material, Supplementary Material 1, for Chemical Constituents From the Fruits of Xanthium strumarium and Their Antitumor Effects by Chao Tong, Ri-Hui Chen, Ding-Cheng Liu, De-Sheng Zeng and Hui Liu in Natural Product Communications
Footnotes
Acknowledgements
We are grateful to the Department of Instrumental Analysis of Central South University for the measurement of the UV, IR, HREIMS, and NMR.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: the work was supported by the Project of the Hainan Province Natural Science Foundation (No. 818MS135).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
