Abstract
Triptolide (TP), a broad-spectrum antitumor drug, has very poor solubility and oral bioavailability, which limits its clinical use. Compared with conventional formulations of TP, a casein (Cas)-based drug delivery system has been reported to have significant advantages for the improvement of solubility and bioavailability of insoluble drugs. In this paper, we report the successful preparation of TP-loaded Cas nanoparticles (TP-Cas) using the self-assembly characteristics of Cas in water and the optimization of the formulation by evaluation of entrapment efficiency (EE) and loading efficiency (LE). Dynamic light scattering, transmission electron microscopy, Fourier-transform infrared spectrometry, X-ray diffractometry (XRD), and differential scanning calorimetry (DSC) was adopted to characterize the TP-Cas. Results showed that the obtained TP-Cas were approximately spherical with a particle size of 128.7 ± 11.5 nm, EE of 72.7 ± 4.7 %, and LE of 8.0% ± 0.5%. Furthermore, in vitro release behavior of TP-Cas in PBS (pH = 7.4) was also evaluated, showing a sustained-release profile. Additionally, an in vivo study in rats displayed that the mean plasma concentration of TP after oral administration of TP-Cas was significantly higher than that treated with TP oral suspension. The
With the rapid development in polymer material technology and biomedical technology, biodegradable polymers have been extensively applied in pharmaceutical and biomedical formulations. 1 Biodegradable polymers can be classified into synthetic and natural polymers depending on the source. 2 With the wide range of resources, natural polymers are generally considered much safer than synthetic ones due to their biodegradability and biocompatibility. 3
Caseins (Cass), the main components of bovine milk, are made up of 4 Cas phosphoproteins: αS1-Cas, αS2-Cas, β-Cas, and κ-Cas, which are di-block copolymers with a pronounced amphiphilic structure, 4 -6 allowing them to self-associate in water to form nanoscale core–shell nanocomposites, where the hydrophobic blocks aggregate into the core and the hydrophilic blocks form the nanocomposite shell. 7 As a food-based natural polymer, Cas exhibits many excellent properties including low cost, easy availability, nontoxicity, high stability, and self-assembly, which encourage scientists to develop Cas into an excellent carrier for drug delivery. 8,9
Drugs can be incorporated into the core of nanocomposites formed by Cas molecules through physical embedding, electrostatic interaction, and covalent bonding. 10,11 Hydrophobic chemotherapeutic agents, for example, can be entrapped and delivered into the core by either chemical bonding or physical inclusion, thereby forming an amphiphilic block copolymer nanocomposite drug loading system. 12 Penalva et al observed that the oral bioavailability of folic acid entrapped into Cas was around 52%, which is 50% higher than conventional aqueous solutions. 13 A possible reason for the enhanced bioavailability might be that folic acid was slowly released from the Cas nanoparticles under the intestinal condition, which allowed sufficient uptake of folic acid in the intestine. A similar sustained-release profile was observed in the report from Luo et al. 14 Hence, Cas is an ideal carrier to improve drug stability, dissolution, and bioavailability.
Triptolide (TP), an epoxy diterpene lactone extracted from the roots, leaves, flowers, and fruits of
Previously, we developed several drug carriers, including macromolecular carriers and liposomes, to promote the targeting profile and reduce the toxicity of TP. 24,25 In this study, we focused on increasing the solubility and dissolution rate of TP in the hope of further improving oral absorption and bioavailability. Herein, we report the production of TP-loaded self-assembled casein nanoparticles (TP-Cas) for the first time. The formulation was optimized by fine-tuning the ratio of Cas/TP using different preparation conditions, encapsulation efficiency, and drug-loading efficiency (LE) as indexes. Dynamic light scattering (DLS), Fourier-transform infrared (FTIR) spectroscopy, differential scanning calorimetry (DSC), and powder X-ray diffractometry (PXRD) were used to characterize the TP-Cas followed by transmission electron microscopy (TEM) for the morphology of the nanoparticles. Additionally, in vitro release behavior and in vivo oral bioavailability of TP-Cas were also evaluated, using TP oral suspension as the conventional comparator.
Results and Discussion
It is reported that TP, a herb-derived drug, possesses an anticancer effect on oral cancer, and in combination with other adjuvant therapy for patients with advanced oral cancer is considered a promising treatment. 26 Nevertheless, the clinical use of TP is often discouraged due to its poor water solubility and low bioavailability. Thus, how to enhance its solubility and improve bioavailability are key issues to be solved by developing new dosage forms. Various nanoparticles have been developed to load TP for the improvement of its shortcomings, including polylactic acid nanoparticles, 27 solid lipid nanoparticles 28 and lipid–polymer hybrid nanoparticles. 29 However, the high cost, limited availability, uncertain biocompatibility of the encapsulation materials, and residual organic solvent rendered them not applicable for industrial production. Conversely, Cas is a commercially available protein that is considered a generally recognized-as-safe ingredient. 30 It has desirable gelation, emulsification, and water-binding properties, showing immense applicability in drug delivery. 31 Also, Cas can self-assemble to form nanoparticles in water, where the self-assembly behavior depends mainly on temperature, ionic strength, and protein concentration. In fact, a low-cost, low-energy, and organic solvent-free nanoencapsulation method has been developed by Pan et al to improve the anticancer activity of curcumin based on Cas. It has been shown in the literature that curcumin encapsulated in Cas nanoparticles had higher dispersibility and biological activity, as well as better stability. 32,33 Turovsky et al have also developed β-Cas micelles as potential carriers, in order to improve the poor water solubility of celecoxib for oral administration. 34 Consistent with the previous study, their data showed that celecoxib-loaded micelles retained their properties after freeze-drying and rehydration, providing long-term physical and chemical stability, which was considered a promising way for oral drug delivery. Therefore, Cas was chosen as the carrier in this study to load TP by using a simple method, for the enhancement of its oral bioavailability.
The high-performance liquid chromatography (HPLC) method applied in this study for the determination of TP was validated successfully. The retention time of TP was approximately 17.0 minutes using the conditions described in the methods section.
24,25
The calibration curves used to calculate the concentration of TP in the ultrafiltrate of TP-Cas were linear over the range of 2.16-108 μg/mL, (
The results of the formulation and process variables screening for the preparation of TP-Cas are shown in Table 1. Probe ultrasonication generated better dispersion, entrapment efficiency (EE), and LE with minimal particle size than the water bath ultrasonication (sample 1–1 and 1–2). To obtain better EE and homogeneity of nanoparticles, ultrasonication was needed for the optimal formulation to provide enough energy input for the breaking of droplets into the nanometer range. For samples 2–1, 2–2, and 2–3, the EE increased with increasing carrier concentrations while the LE displayed a decreasing trend. With regard to EE of TP, 2%, w/w, Cas was found to be the best carrier concentration. The concentration of Cas was believed to be an important factor for EE and particle size of TP-Cas because of its viscosity. Cas solution of 0.5%-1.0% w/w with low viscosity did not facilitate the formation of nanoparticles, resulting in a low EE and LE of TP-Cas, whereas more concentrated Cas solutions of >2% w/w seemed more viscous to hamper uniformity of nanoparticles, resulting in an inhomogeneous particle size. The data for samples 3–1 to 3–4 showed that EE and LE were improved as the ratio of Cas/TP decreased. Once the ratio of carrier to drug (Cas/TP) exceeded 14.3:1, obvious precipitation occurred in the sample, indicating poor stability of the nanoparticles. Thus, the ratio of 16.7:1 (Cas/TP) was considered the optimal formulation. Increasing ultrasonication time resulted in increased EE and LE initially and then a decrease, suggesting the ideal ultrasonication time was 300 seconds (pulse on, 10 seconds; pulse off, 10 seconds). Therefore, the optimal formulation was as follows: 12 mg TP (dimethyl sulfoxide [DMSO] as a solvent) was dispersed in 5 mL of 2% w/w Cas solution at room temperature while being stirred using a magnetic stirrer for 10 minutes, and then ultrasonicated for 300 seconds (pulse on, 10 seconds; pulse off, 10 seconds). After lyophilization in freeze-dryer for 24 hours, TP-Cas nanoparticles were obtained with an average EE of 72.7% ± 4.7% and average LE of 8.0% ± 0.5%. Thus, TP-Cas was successfully obtained by direct addition of TP solution to the aqueous Cas solution at ambient temperature under gentle stirring.
Formulation Screening for the Preparation of TP-Cas

Abbreviation: TP-Cas, triptolide-loaded casein nanoparticles.
aIndicates that the obtained TP-Cas is unstable.
The obtained TP-Cas was redispersed into deionized water, and then a clear colloidal solution was obtained for the measurement of particle size and zeta potential. As shown in Figure 1 (A and B), the average particle size of the optimal TP-Cas was 128.7 ± 11.5 nm with polydispersity index (PDI) of 0.49 ± 0.04, and the zeta potential was −23.9 ± 1.5 mV. As indicated in Figure 1(C), TP-Cas was approximately spherical with a uniform size distribution. The surface morphology was found to be relatively smooth with no surface-adsorbed particles observed. However, the diameter of TP-Cas observed from TEM seemed smaller than the data shown in Figure 1(A). A possible reason might be due to the shrinkage of TP-Cas due to drying of the sample during preparation for microscopic analysis. Although the smaller nanoparticles can be clearly observed, the PDI value indicated the size distribution of TP-Cas was acceptable. PDI is one of the important factors in the physical stability of nanoparticles, where PDI >0.5 means the sample has a broad size distribution and may be unstable. As mentioned above, the concentration of the Cas solution might be a key to control the size distribution of TP-Cas. The zeta potential of TP-Cas was −23.9 mV, which confirmed the high stability of the TP-Cas dispersion. The negative charge of nanoparticles was probably attributed to the supramolecular structural changes of Cas. Previously, Pan et al demonstrated that the pH value of the encapsulated drug could also affect the zeta potential of nanoparticles. 33
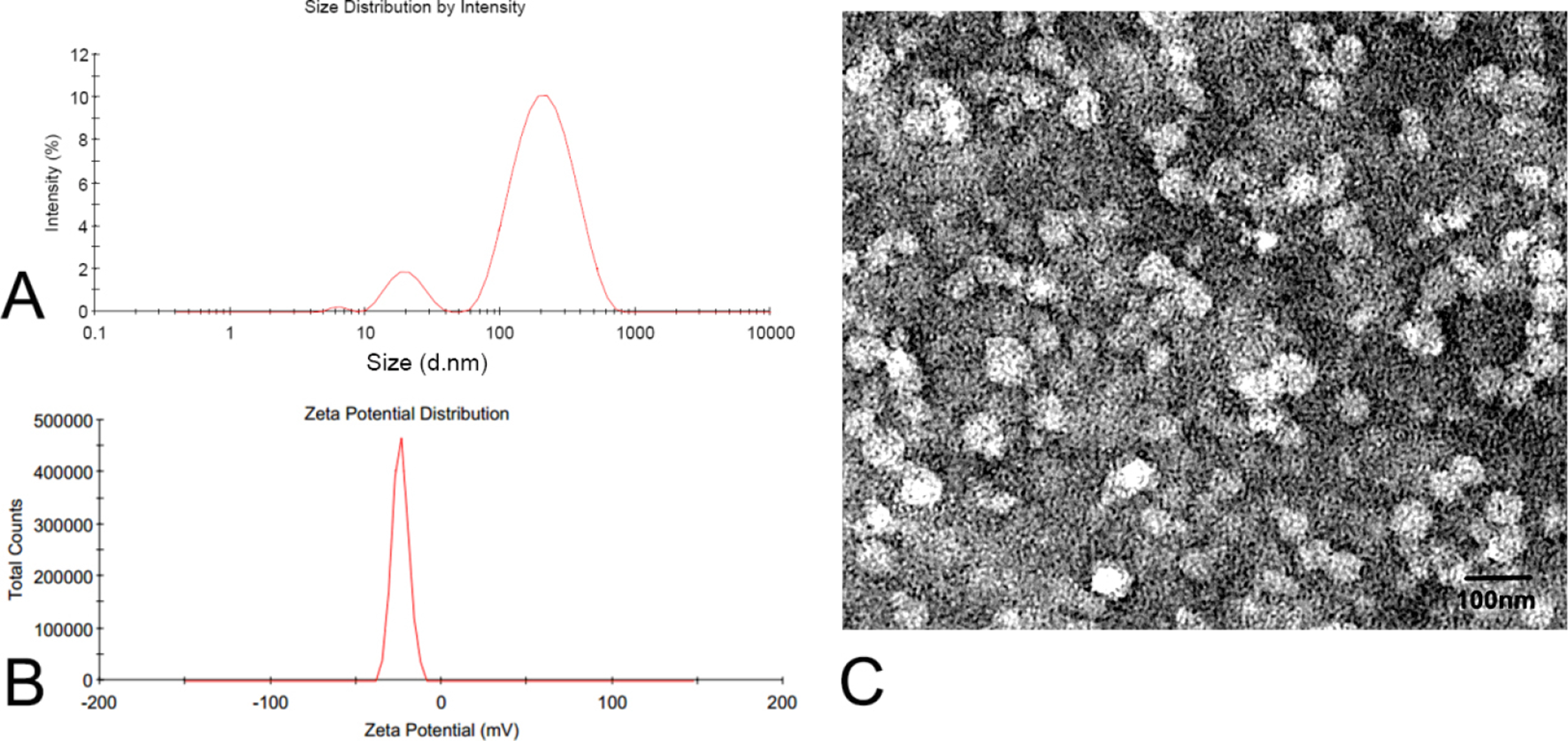
Size distribution (A), zeta potential distribution (B), and transmission electron microscopy image (C) of TP-Cas. The experiment was repeated in triplicate. TP-Cas, triptolide-loaded casein nanoparticles.
To characterize further the TP-Cas, FTIR, XRD, and DSC were adopted in this study. The FTIR spectra of TP, Cas, physical mixture (TP and Cas), and TP-Cas are presented in Figure 2. As shown in the spectrum of TP (Figure 2(A)), characteristic absorption bands at 1769 and 3455 cm−1 were detected and these are attributed to the C=O and the OH bond stretching, respectively. This is consistent with the report from Liu et al. 27 The spectrum of Cas showed some bands at 1679, 2925, 3315, and 3425 cm−1 assigned to carbonyl C=O, CH3, NH, and OH stretch vibrations, respectively (Figure 2(D)). For the physical mixture of TP and Cas, similar but wider infrared bands at 1679, 1769, 2925, 3315, and 3455 cm−1 were observed, belonging to dual feature absorption of TP and Cas (Figure 2(C)). For TP-Cas, the characteristic peaks corresponding to carbonyl C=O and OH bond stretching (broader and smoother) slightly shifted to 1771 and 3406 cm−1, respectively, which is evidence for successful entrapment of TP into the nanoparticles. This phenomenon can be explained by weak interaction between the TP and Cas. The PXRD patterns of TP, Cas, physical mixture (TP and Cas), and TP-Cas are shown in Figure 3. Among them, the strong and sharp diffraction peaks of pure TP at 2θ scattered angles 7.4°, 8.3°, and 15.3° (Figure 3(A)) indicate that TP was of typical crystalline nature, while there were only gentle and wide peaks in the Cas diffractogram (Figure 3(D)), which confirmed its amorphous nature. These diffraction peaks with less intensity in the physical mixture were considered a simple superposition of TP and Cas samples (Figure 3(B)). The lyophilized TP-Cas presented an amorphous pattern, similar to Cas, and there were some weak characteristic peaks of TP, contributed by free TP in TP-Cas (Figure 3(C)). Notably, 3 new sharp crystal diffraction peaks (27.4°, 31.7°, and 45.4°) appeared, likely owing to the formation of new crystals in the nanoparticle suspension during lyophilization. Considering the use of PBS in the preparation, these crystal diffraction peaks may be attributed to a NaCl crystal, which can be confirmed by the characteristic diffraction peaks at 27.4°, 31.7°, 45.4°, 56.4°, and 66.2° in the previous report. 35 The above results further demonstrated the conversion of TP to an amorphous form after being entrapped in Cas nanoparticles. It could be either dispersed within the nanoparticles or molecularly adsorbed on the surface of the nanoparticles. The DSC thermograms of TP, Cas, physical mixture (TP and Cas), and TP-Cas were obtained in the temperature range of 25-300 °C (Figure 4). Wide endothermic peaks appear near 40-110 °C in Figure 4B–D, which were caused by the dehydration of Cas. An endothermic peak at 236.6 °C representing the melting point of TP and another sharp exothermic peak at 286.6 °C, presumably due to the decomposition of TP, are observed in Figure 4(A). However, the characteristic peak of TP only showed an exothermic peak with low intensity at 284.7 °C in the thermogram of the physical mixture (Figure 4(B)), which may be induced by the decrease in crystallinity of TP during the grinding and mixing of TP and Cas. In the DSC thermogram of lyophilized TP-Cas (Figure 4(C)), no characteristic peak corresponding to either melting or decomposition of TP was present, revealing that TP was encapsulated in the Cas nanoparticles and converted to an amorphous state from a crystalline state.

Overlaid Fourier-transform infrared FTIR spectra: TP (A), physical mixture of TP and Cas (B), TP-Cas (C), and Cas (D). Cas, casein; TP, triptolide.

Powder X-ray diffractometry diffractograms of TP (A), physical mixture of TP and Cas (B), TP-Cas (C), and Cas (D). Cas, casein; TP, triptolide.

Differential scanning calorimetry thermograms of TP (A), physical mixture of TP and Cas (B), TP-Cas (C), and Cas (D). Cas, casein; TP, triptolide.
In vitro release behavior of TP suspension and TP-Cas in PBS (pH = 7.4) was evaluated by dynamic membrane dialysis. 36 As shown in Figure 5A, a rapid release within 0.5 hours, about 76.3%, was observed for the TP suspension, whereas only 34.5% TP was released from TP-Cas at the same time point. The release of TP from TP-Cas reached a plateau (~70%) within 12 hours, which should be contributed to the sustained-release profile of nanoparticles. Notably, the cumulative release rate for either TP suspension or TP-Cas in PBS declined slowly. This could be due to TP degradation in the release media.

In vitro and in vivo studies. (A) In vitro release profile of TP suspension (■) or TP-Cas (♦) in phosphate-buffered saline (pH = 7.4) using the dialysis bag diffusion technique. The experiment was repeated in triplicate and data are given as the mean ± standard deviation (
The calibration curves for the determination of TP level in plasma were linear over the range of 0.6-30.0 μg/mL (
Pharmacokinetic Parameters of TP After Oral Administration for Both Groups.

Abbreviations:
*
In conclusion, we successfully prepared TP-Cas using a self-assembly method for the first time based on the characteristic of Cas carrier in water. The optimal TP-Cas was characterized by DLS, TEM, HPLC, FTIR, DSC, and PXRD analysis, confirming the formation of nanoparticles and TP LE. Furthermore, in vitro release behavior showed a sustained release of TP in TP-Cas nanoparticles, which guaranteed the absorption of TP. In vivo pharmacokinetics study showed that TP-Cas enhanced the absorption, prolonged the retention time, and improved the oral bioavailability of TP. Taking the good oral safety of Cas into consideration, TP-Cas should be a promising preparation of TP for clinical application.
Experimental
Materials
TP (purity >99 %) and casein sodium salt from bovine milk (Cas) were supplied by Sigma-Aldrich Co. Ltd. (Gillingham, UK). Andrographolide (purity >98 %) was bought from Solarbio Co. Ltd. (Beijing, China), and Pepsin from Biosharp Co. Ltd. (Sichuan, China). The other chemicals were of analytical reagent grade.
Animals
Sprague Dawley rats (in male) weighing between 250 and 280 g were purchased from the Da-Shuo experimental Animal Ltd. (Beijing, China). The animals were allowed to acclimatize for a few days in environmentally controlled quarters (24 ± 1 °C, 12 hour light/dark cycle) and, unless specified otherwise, were provided with water and food ad libitum. All animal studies were approved by the Animal Ethical Experimentation Committee, College of Veterinary Medicine (SYXK (Chuan) 2014–187), and were performed according to the requirements of the People’s Republic of China National Act on the use of experimental animals.
Preparation of TP-Cas
TP-Cas was prepared as previously described by Pan et al, with minor modifications. 33 Briefly, a certain amount of TP solution (100 µL DMSO as a solvent) was dispersed in 5 mL of Cas solution (Cas was dissolved in PBS solution containing 80 mM NaCl, 5.65 mM Na2HPO4, and 3.05 mM NaH2PO4) at room temperature while being stirred using a magnetic stirrer for 10 minutes, and then ultrasonicated for a few seconds. The obtained solution was prefrozen at −80 °C for 24 hours followed by lyophilization in a freeze-dryer (LyoQuest, Telstar, Spain) under 0.4 mbar vacuum for 24 hours and a condenser temperature of −40 °C to form a powder, namely, TP-Cas. The formulation and preparation method were optimized by evaluation of EE and LE as indexes using HPLC analysis.
Entrapment Efficiency and Loading Efficiency
HPLC analysis
The HPLC (Agilent 1260, Agilent Technologies, Santa Clara, CA, USA) was equipped with an online degasser, a quaternary pump, an auto-sampler, a column thermostat, and an Agilent C18 (250 × 4.6 mm, 5 µm) analytical column for chromatographic separation. All separations were executed at 30 °C with acetonitrile:water (25:75) as mobile phase with a flow rate of 1.0 mL/min, and 10 µL sample volume. The UV detector was set at 218 nm. 25,39 Peak identification was carried out by comparing the retention times of the samples with those of reference standards.
Determination of EE and LE
An ultrafiltration method was used to determine the entrapment and loading efficiencies. The freshly prepared TP-Cas was centrifuged at 4000 rpm for 10 minutes in an ultrafiltration tube (MWCO = 3000, Millipore Corporation, Bedford, MA, USA) and then the TP encapsulated into nanoparticles remained in the ultrafiltration tube and free TP moved into the filtrate. The diluted filtrate of the centrifuged samples was injected onto the HPLC to assess the amount of non-encapsulated TP. EE and LE were determined according to the following equation. 40


Optimal Formulation of TP-Cas
The single factors of ultrasound method, carrier concentration, the ratio of Cas/TP, and ultrasound time were all optimized in this study to obtain maximum EE% and LE%. Probe ultrasonication and water-bath ultrasonication were the variables selected for this experiment, as shown in Table 1 (1–1 and 1–2). Carrier concentrations of 0.5%, 1%, and 2% w/w were also studied (Table 1, from 2–1 to 2–3). In addition, the carrier–drug ratio of 25:1, 20:1, 16.7:1, and 14.3:1 (Table 1, from 3–1 to 3–4) and ultrasonication times of 200, 300, and 400 seconds were also compared (Table 1, from 4–1 to 4–3). Thereafter, the EE and LE for each formulation were calculated using HPLC analysis.
Characterization of TP-Cas
Measurement of particle size and zeta potential
Particle size and zeta potential of TP-Cas were determined using a Malvern Zetasizer Nano ZS90 (Malvern Instruments Ltd, Malver, UK). TP-Cas freeze-dried powder was appropriately diluted with distilled water for measurement. The particle size measurement was performed using a quartz cell at ambient temperature with a scattering angle of 90° and was recorded for 180 seconds for each measurement. The zeta potential was measured using an aqueous dip cell in an automatic mode and the data were obtained from the average of several measurements.
TEM image
An S4800 transmission electron microscope (Hitachi Ltd, Tokyo, Japan) was applied to observe the morphological features of TP-Cas. Briefly, the optimal TP-Cas was appropriately diluted in water and dripped onto a dedicated copper mesh, then, negatively stained with 4% phosphotungstic acid solution for 30 seconds. After drying at ambient temperature, the particles were observed under the transmission electron microscope.
FTIR spectrometry
A suitable amount of TP, Cas, physical mixture of TP and Cas, and TP-Cas freeze-dried powder were blended with solid potassium bromide (KBr) power, respectively. Then, the blend was tableted using a single-punch tablet press and subjected to FTIR spectrometry (Perkin Elmer BX, Waltham, MA, USA). Transmittances were recorded at wave numbers of 4000-400 cm−1.
PXRD
AG-10TA PXRD (Shimadzu Corporation, Tokyo, Japan) was applied for diffraction studies. The samples, including TP, Cas, physical mixture of TP and Cas, and TP-Cas freeze-dried powder, respectively, were analyzed at a step size of 0.02° by exposing the samples to CuKα radiation (40 kV, 40 mA) and scanning continuously from 5° (2θ) to 55° (2θ).
DSC analysis
DSC1 (Mettler Toledo, Switzerland) was performed for thermal analysis. DSC thermograms of TP, Cas, physical mixture of TP and Cas, and TP-Cas freeze-dried powder were recorded by heating in a nitrogen atmosphere at a rate of 10 °C/min to analyze the change of the sample within the temperature range of 30-300 °C.
In Vitro Release
In vitro release studies were carried out in 0.05 M phosphate buffer solutions at pH 7.4 (PBS, containing 0.1% Tween 80) using dynamic membrane dialysis. One milliliter of TP-Cas was added to a dialysis bag (3500 Da) and then dialyzed against 99 mL PBS in a 250 mL beaker, which was situated in an oscillating water bath at a temperature of 37 ± 0.5 °C. Samples (1 mL) were obtained from the release medium at different time points (0.083, 0.167, 0.25, 0.5, 1, 2, 6, 12, and 24 hours) for determining the TP amount that diffused through the dialysis bag. Meanwhile, the same amount of fresh release medium preheated at 37̊ °C was added to maintain a constant volume at the above prearranged time points. Samples were analyzed by HPLC, as described previously. Cumulative drug release was calculated from the following equation. 41
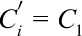

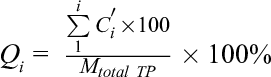
where
Oral Bioavailability Study
Ten Sprague Dawley rats (male, 250-280 g) were randomly divided into 2 groups of 5 rats each for pharmacokinetic analysis. Either TP suspension (20 mg/kg) or TP-Cas with an equivalent dose of 20 mg/kg TP was administered intragastrically by oral gavage. Blood samples were obtained (0.5 mL) from the retro-orbital plexus at different time points (0.083, 0.25, 0.5, 1, 2, 4, 6, 8, and 12 hours) after administration, followed by centrifugation at 4000 rpm (Sorvall ST16, Thermo Fisher Scientific, MA, USA) for 10 minutes to separate plasma. Andrographolide dissolved in acetonitrile (50 µL of 680 µg/mL solution), as an internal standard, and 150 µL ultrapure water were added to 200 µL of the plasma sample. Then, the mixture was extracted with 1.2 mL ethyl acetate by vortexing for 1 minute followed by centrifugation at 4000 rpm for 10 minutes, and then the upper organic phase was aspirated into a 4-mL Eppendorf tube carefully. The extraction was repeated once. All the organic extracts were pooled and dried under a gentle stream of nitrogen at 40 °C. 28 The residue was reconstituted in 100 µL methanol and centrifuged at 12000 rpm for 10 minutes after vortex-mixed. The supernatant was analyzed by HPLC as described above and the separation was performed on the C18 analytical column (4.6 × 250 mm, 5 µm) maintained at 30 °C. Mobile phase consisted of 77% water and 23% acetonitrile with a flow rate of 1.0 mL/min. 39
Statistical Analysis
The bioavailability parameters were analyzed by DAS 2.0 (BioGuider Co, Shanghai, China) computer software. Data were compared with the SPSS 15.0 statistical package. Multiple comparisons of mean values were performed by one-way analysis of variance with the Fisher’s least significant difference test applied for post hoc comparisons at 95% CI.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Guangdong Medical Research Foundation (A2018339) and Stomatological Hospital of Southern Medical University Research Training Program (PY2017017).
