Abstract
In order to expand the application of chitosan in cosmetics, it was modified with vitamin C to prepare a chitosan/vitamin C complex (CSVC complex), which was characterized using ultraviolet-visual, Fourier transform infrared, and differential scanning calorimetry techniques. Its hygroscopicity, moisture retention, antioxidant and antibacterial activities, and environmental stability were also determined. The results showed that a mass ratio of chitosan to vitamin C of 1:1 that reacted at 25℃ for 90 minutes in a medium of isopropanol–water, away from light, with the unreacted vitamin C being removed by dialysis gave a white powder (CSVC complex) after freeze-drying. The CSVC complex had good water solubility, a viscosity average molecular weight of 233.7 kDa, and a vitamin C content of 22.64%. Structure characterization confirmed that the CSVC complex was composed of the amine (C-2 position) of chitosan and the hydroxyl group (C-3 position) of vitamin C by electrostatic attraction. The complex had a significantly better scavenging effect on hydroxyl free radicals than chitosan, and the minimum inhibitory concentration against Escherichia coli was 5 mg/mL. Its moisturizing and hygroscopic properties were better than chitosan, and its stability was better than that of vitamin C. These results provide the basis for the CSVC complex as a multifunctional raw material for cosmetic use because of its antioxidant, moisturizing, antibacterial, and film-forming properties.
Chitosan and its derivatives have become a research focus for skin, hair, nail, and oral care cosmetics applications due to their excellent biocompatibility, degradation, film-forming, nontoxic, and biological activities. 1,2 Chitosan is a positively charged polysaccharide that can electrostatically interact with negatively charged keratin, which is particularly beneficial for the preparation of skin cream. 3 It was reported that dissolving chitosan in phosphoric acid and adding hydroxyapatite could synthesize a hydroxyapatite chitosan gel with sunscreen and antibacterial effects, with the nanoparticles of chitosan gel providing obvious resistance to ultraviolet (UV) radiation and having an antibacterial effect. 4 Gopal V 5 studied the antiaging ability of chitosan, collagen, and aloe gel, the results of which showed that chitosan could improve the stability of skin fibroblasts and induce local cell proliferation. The combination of chitosan and collagen could enhance the biocompatibility, adhesion, and proliferation of skin fibroblasts, and ultimately promote skin regeneration and rejuvenation. However, chitosan can form extensive intra- and intermolecular hydrogen bonds, which make it crystalline in structure and insoluble in water at and above a neutral pH. 6 Chitosan is soluble in water under acidic conditions (ie, below pH 5) because of the protonation of the free amino groups at the C2 position, which greatly limits its application in cosmetics. To overcome this limitation of water solubility, many approaches have modified the structure of chitosan: degradation to obtain chitosan oligosaccharide 7 ; chitosan derivatives obtained by acylation, esterification, and carboxylation of functional groups on the hydroxyl groups of C-3 and C-6, and the amino group of C-2. 8 For example, Seungmi Hong 9 studied the modification of chitosan with ascorbic acid at different temperatures (4-40°C), and the product was water soluble at pH 7. Hao et al 10 used laccase as a catalyst to obtain a chitosan/gallic acid derivative by combining the carboxyl group of gallic acid with the amino group at C-2 of chitosan through an amide bond. The solubility of the grafted chitosan reached 92.8% with 72.8% of the grafting rate. In order to improve the antioxidant and antibacterial properties of chitosan, Yang et al 11 studied the grafting of ferulic acid onto chitosan catalyzed by Vbb-103 recombinant bacterial laccase, which greatly improved the water solubility and DPPH scavenging rate of the grafted chitosan, as well as the antibacterial properties against Escherichia coli, Staphylococcus aureus, Bacillus subtilis, and Meyerozyma guilliermondii. Wen-hui et al 12 modified the C-2 amino group of chitosan with citric acid to prepare a water-soluble chitosan/citric acid complex. Chitosan derivatives extend and give chitosan better properties. For example, Chen Kaihang et al 13 used quaternized carboxymethyl chitosan to prepare composite silicate nanomaterials, which could be used as cosmetic ingredients to prevent skin aging; the composite materials had better moisture absorption performance, water retention, and UV protection compared with chitosan and hyaluronic acid.
Vitamin C, also known as ascorbic acid, has a strong scavenging effect on hydrogen peroxide, superoxide free radicals, and hydroxyl free radicals. It is a commonly used whitening material in cosmetics. 14 However, vitamin C is poorly tolerated by factors such as oxygen, high temperature, pH, metal ions, and strong light. 15 Adding vitamin C directly to cosmetics usually reduces the stability of the product. Pallela 16 found that chitooligosaccharide–vitamin C complex was composed of 2 or more compounds with noncovalent bonds (such as electrostatic attraction, hydrogen bond, salt bond, and water release bond). Tian 17 found that after the formation of the chitosan and vitamin C (CSVC) complex, its physical properties, such as solubility and stability, were improved, with the advantages of a simple preparation method and low cost.
In order to improve the water solubility of chitosan and the stability of vitamin C, the CSVC complex was prepared. Chitosan contains protonated amino groups (C-2 position) in acidic media, whereas vitamin C contains an acidic hydroxyl functionality (the -OH of C-3 position is the most acidic), thus allowing the formation of complexes through ionic interaction under certain conditions. The introduction of vitamin C into chitosan can destroy the hydrogen bond within and between chitosan molecules, thereby improving the water solubility of chitosan. At the same time, the chitosan polymer can protect the active group of vitamin C, the stability of vitamin C is improved, and its oxidation delayed. Taking the water solubility, vitamin C content, and yield of CSVC complex as indicators, the effects of different preparation processes on the color, water solubility, and yield of CSVC complex were studied. On this basis, the physical and chemical properties of the CSVC complex, such as hygroscopicity, moisture retention, free radical scavenging rate, antibacterial activity, and stability, were evaluated to provide an experimental basis for the application of the complex in cosmetic raw materials.
Materials and Methods
Raw Materials and Chemicals
Chitosan (light yellow, degree of deacetylation 85%-95%, viscosity average molecular weight 587.3 kDa) was provided by Ningbo Haixin Biotechnology Company. Vitamin C (L-ascorbic acid), hydrochloric acid, isopropanol, glycerin, potassium bromide, anhydrous ethanol, glacial acetic acid, sodium acetate (all AR grade), LB medium (biochemical reagent), and dialysis bag MD34 (MW8000-14000) were all purchased from Sinopharm Shanghai Chemical Reagent Co., Ltd.
Escherichia coli was provided by the microbiology laboratory of the College of Biological & Environmental Sciences, Zhejiang Wanli University.
Experimental Method
Chitosan/vitamin C complex preparation
Using acetic acid–water as the reaction medium 18 : In a 250 mL round bottom flask with 3 necks, 2 g chitosan precisely weighed by electronic balance (Germany, Sartorius, BSA224S) and 40 mL 3% (v/v) HAC solution were added, respectively, and then the chitosan solution was obtained by stirring. The flask was fitted with a nitrogen gas balloon for supplying nitrogen to remove the dissolved oxygen from the solution. At the same time, 2 g vitamin C was added to the chitosan solution and stirred at 400 rpm for 90 minutes at room temperature. The crude product was precipitated with anhydrous ethanol, and the precipitate was dissolved in deionized water to remove the unreacted chitosan; the unreacted vitamin C of the crude product was removed by dialysis. The CSVC complex was obtained by concentration and freeze-drying of the dialysate.
Isopropanol–water as the reaction medium 19 : In a 250-mL round-bottom flask with 3 necks, 2 g chitosan and 30 mL of isopropyl alcohol were added, respectively, and the flask was fitted with a nitrogen gas balloon, as above. Ten milliliters of a 20% solution (m/v) of vitamin C was added to the chitosan suspension. Under the protection of nitrogen, the reaction solution of chitosan and vitamin C was stirred at room temperature for 90 minutes. The purification procedure of the CSVC crude product was the same as above.
Single-factor experiment of CSVC complex preparation: Taking water solubility, vitamin C content, and yield of CSVC complex as indicators, the effects of the mass ratio of chitosan to vitamin C of 1:0.5, 1:1, 1:1.5, and 1:2 (fixed temperature 25°C, time 90 minutes), reaction temperatures of 25, 35, 45, and 55°C (fixed chitosan:vitamin C 1:1, time 90 minutes), and reaction times of 30, 60, 90, and 120 minutes (fixed chitosan:vitamin C 1:1, temperature 25°C) on CSVC preparation were investigated.
Vitamin C content determination in CSVC complex
Chitosan, vitamin C, and CSVC solutions were prepared by dissolving in dilute hydrochloric acid (pH 3.8). With dilute hydrochloric acid (pH 3.8) as blank, the above 3 solutions were scanned in the range of 200-400 nm using an UV-Vis spectrophotometer (China, Puxi, TU-1810). 20 The wavelength corresponding to the maximum absorption peaks of CSVC and vitamin C in their spectra was compared, and the interference of chitosan on the determination of CSVC and vitamin C was analyzed. A series of vitamin C standard solutions with a concentration of 1.0-20 µg/mL were prepared in dilute hydrochloric acid (pH 3.8). The absorbance value a of vitamin C solution was determined at 245 nm. The standard equation was A = 0.0543C + 0.0864, r = 0.9984.
Apparent solubility and molecular weight determination of CSVC complex
Apparent solubility testing: In 2 tubes containing 10 mL distilled water, 50 mg chitosan and CSVC complex were added, respectively, and the tubes were shaken vigorously at room temperature for 5 minutes. The transparency of the solution and the amount of sediment at the bottom of the tubes were observed with the naked eyes. If the solution was transparent and there was no sediment at the bottom, it meant that the sample was soluble in water.
Molecular weight: This was determined according to the Mark–Houwink formula, where
Structural characterization of CSVC complex
Fourier transform infrared (FT-IR) spectroscopic analysis 23 was carried out using an FT-IR spectrometer (Bruker, Vertex 70, Germany) over the range of 400-4000 cm−1 and recorded at a resolution of 4 cm−1. The samples were chitosan, vitamin C, chitosan + vitamin C mixture (mixed 1:1), and the CSVC complex. The powdered samples were compressed into potassium bromide (KBr) disks for the FT-IR measurements.
Differential scanning calorimetry (DSC) analysis 24 was carried out using DSC (Setaram, DSC131evo, France,). The samples were chitosan, vitamin C, mixture of chitosan and vitamin C (1:1), and the CSVC complex. The samples were weighed separately in a DSC pan (5-8 mg), sealed with a DSC lid, and then heated from 25 to 380°C at a rate of 10°C/min. Their DSC patterns were drawn under the conditions of nitrogen flow protection.
Performance testing
Hygroscopicity 25 : One gram of chitosan, CSVC, and control glycerin samples were accurately weighed into constant-weight weighing bottles, respectively, placed in drying containers containing a saturated solution of ammonium sulfate (20°C, relative humidity [RH] 82%), and weighed. The moisture absorption rates of the 3 samples were calculated at 3, 6, 12, 24, 48, and 96 hours respectively.
Moisture retention 26 : One gram of CS, CSVC, and control glycerin samples were accurately weighed into constant-weight weighing bottles, to each of which was added 0.1 g distilled water. They were then put into drying containers containing a saturated solution of calcium chloride (20°C, RH 29%). The samples were weighed at 3, 6, 12, 24, 48, and 96 hours, and their moisture retentions were calculated, respectively.
Inhibition rate to hydroxyl radical 27 : Hydroxyl radicals are produced by Fenton reaction. Chitosan and vitamin C solutions with concentrations of 0.5, 1, 1.5, 2, 2.5, and 3 mg/mL were prepared, respectively. The sample solution (1 mL), salicylic acid ethanol solution (9 mmol/L, 0.5 mL), ferrous sulfate solution (9 mmol/L, 1 mL), and hydrogen peroxide solution (9 mmol/L,1 mL) were added in turn to the test tube, and make up to 15 mL with deionized water. The test tube was placed in a 37°C water bath to react for 30 minutes, and the absorbance value (Ax ) of the solution at 510 nm was determined by an UV-Vis spectrophotometer. The H2O2 solution in the above mixed solution was replaced with 1 mL distilled water, which was recorded as Ax 0 as the background of the sample. The sample solution in the above mixed solution was replaced with 1 mL of distilled water and recorded as A 0 blank.
Antibacterial activity 28 : The activated E. coli was inoculated into the sterilized lysogeny broth (LB) medium, and the bacterial solution was placed on a shaking table (Guohua, SHA-B, China) and cultured at 37°C, 150 rpm for 8 hours.
The bacterial solution was diluted to a series of suspensions up to 100 times and the numbers were counted with a blood bacterial counting plate. The absorbance value A of the bacterial solution was measured at 600 nm. The standard equation for the number of bacteria was as follows: A = 0.0675C − 0.0458 (3-15 × 108 CFU/mL), r = 0.9915. One milliliter CSVC solution containing 1.25, 2.5, 5, and 10 mg/mL, filtered through a 0.22-µm membrane, was added to a tube containing 4 mL of LB liquid medium, and then 50 µL of bacterial solution was added to the culture for 24 hours under the above conditions; the absorbance value A of the solution was determined. One milliliter sterile water was used to replace the CSVC solution in the above mixture as the blank control.
Stability test
The influencing factor test method was used. 29 Accurately, 5 g of CSVC and vitamin C were weighed into weighing bottles, and put into a drug stability test chamber (China, Dongnan, SYW-350)at 40°C, RH 85% ± 5%, 4500 ± 500 Lx for 10 days to conduct high-temperature, high-humidity, and light influence factor tests, respectively. The appearance of the sample was observed and the content of vitamin C in the sample determined at the same time each day. The stability of the sample was indicated by the amount of vitamin C retained.
Results and Discussion
Preparation Process of CSVC Complex
Effect of reaction medium on preparation of CSVC complex
The appearance color, solubility, molecular weight, and yield of CSVC prepared in 2 kinds of reaction media are shown in Table 1.
Comparison of Chitosan/Vitamin C Complex Prepared in Different Reaction Media.


Scheme of the possible interaction between the chitosan and vitamin C.
The color and molecular weight of CSVC prepared in isopropanol–water were better than those in acetic acid–water. The reaction of chitosan and vitamin C in isopropanol–water solution belongs to the solid–liquid interface reaction. Isopropanol as a dispersion medium is conducive to the full dispersion of chitosan, while vitamin C dissolves in water to form an acid water phase, which is conducive to the continuous dissolution of chitosan at the solid–liquid interface and the reaction with vitamin C. The C2-NH2 of chitosan can be combined with C3-OH of vitamin C by electrostatic force 30 (Figure 1), which destroys the original intermolecular and intramolecular hydrogen bonds in chitosan molecules, thus improving the solubility of chitosan. However, excessive vitamin C makes the reaction medium strongly acidic, which will lead to partial degradation of chitosan, and ultimately affect the molecular weight and yield of the product. In the reaction medium of acetic acid and water, excessive vitamin C will make the molecular chain of dissolved chitosan more easily broken, which is consistent with the research results of Zoldners. 31 The main reason is that vitamin C reacts with oxygen to form hydroperoxides, which lead to the breaking of the glucoside bond in chitosan. Through comparison, isopropanol–water was selected as the preparation medium for CSVC.
Effect of preparation factors on the performance of CSVC complex
CSVC was prepared in isopropanol–water. The effect of material ratio, temperature, and time on CSVC is shown in Table 2.
Effect of Preparation Parameters on Solubility, Vitamin C Content, and Yield of Chitosan/Vitamin C Complex.

When the mass ratio of chitosan:vitamin C was 1:0.5, the CSVC product prepared was only partially soluble in water, and both the yield of CSVC and the vitamin C content in the product were lower. This was because when the amount of vitamin C was insufficient, the amino groups in chitosan were only partially protonated, which was not enough to destroy the ordered structure of chitosan, and so the solubility of the CSVC complex was limited. When the mass ratio of chitosan:vitamin C was 1:1 and above, the solubility of the obtained CSVC product was good; the vitamin C content in the product was 20.60%-22.6%, and the yield of products was 57.8%-66.3%. The significant increase in vitamin C dosage had no significant effect on the yield and vitamin C content of the product, so the mass ratio of chitosan:vitamin C for preparing CSVC was selected as 1:1.
Temperature has a great influence on the preparation of CSVC. When the preparation temperature was higher than 45°C, the color of CSVC was deep, the solubility was poor, and the vitamin C content in the product was low. This shows that vitamin C may be oxidized by high temperature without reacting with chitosan in the preparation of CSVC.
The reaction time had a major influence on the yield of CSVC and the vitamin C content in the product. The vitamin C content increased with the increase of reaction time, but the yield of CSVC increased first and then decreased. It shows that prolonging the reaction time is conducive to the generation of CSVC, but a too long reaction time may cause the chitosan molecular chain to break, which will affect the yield.
In summary, the conditions for the preparation of CSVC are isopropanol–water as the medium, a mass ratio of chitosan to vitamin C of 1:1, and the reaction carried out at 25°C and protected from light for 90 minutes. The viscosity average molecular weight of CSVC was 273 kDa. The solubility comparison between CSVC and chitosan in water is shown in Figure 2. It can be seen clearly that CSVC forms a uniform transparent pale brown-colored solution, while chitosan has deposits at the bottom of the test tube. Because of the extensive inter- and intramolecular hydrogen bonding between the layers/chains/sheets or the possible conformations of the chitosan, it does not form a mere transparent solution. However, CSVC complexes would overcome this problem to perform the function more significantly, which can greatly expand the application of CSVC in cosmetics.

Comparison of the apparent solubility of CSVC complex and chitosan in water. CSVC, chitosan/vitamin C.
Structure Analysis of CSVC
UV spectrum of CSVC
The UV spectra of chitosan, vitamin C, and CVSC showed that chitosan had no absorption peak in the wavelength range of 200-320 nm, and the absorption spectra of CSVC and vitamin C were similar, which indicates that they had similar UV absorption groups. Vitamin C has a strong absorption peak at 245 nm, which is a characteristic of its enol structure. The maximum absorption wavelength of the vitamin C enol structure in the CSVC complex was red-shifted to 247 nm, which may be due to the change in the C3-OH of vitamin C, which results in the red-shift of its enol absorption band. It shows that the C2 amino group of chitosan and the C3 hydroxyl group of vitamin C have indeed bound.
IR spectra of CSVC
The FT-IR spectra of chitosan, vitamin C, a mixture of chitosan + vitamin C, and CSVC are shown in Figure 3. The strong absorption peak of chitosan at 3428 cm−1 is the overlapping of the stretching vibration of -NH2 and the -OH groups, 2924 cm−1 is the C-H stretching vibration peak of chitosan, 1644 cm−1 is the bending vibration absorption peak of -NH2, and 1384 cm−1 is the in-plane bending vibration peak of the N-H bond. In the IR spectrum of vitamin C, the peaks at 3524, 3313, 3214, and 3028 cm−1 are ascribed to -OH groups. The 2 stretching vibration peaks at 1754 and 1674 cm−1 are the intra- and intermolecular hydrogen bonds of the lactone carbonyl group. The IR spectrum of a mixture of chitosan and vitamin C is the superposition of the IR spectra of the 2 compounds. However, it can be seen from the spectrum of the CSVC complex that the peak at 3428 cm−1 due to the overlapping of the -OH and -NH2 groups of chitosan shifted to 3418 cm−1 and became broad and blunt (weakened); the 1754 cm−1 peak of vitamin C shifted to 1721 cm−1 and the intensity decreased, indicating that the free -NH2 of the chitosan molecule was reduced after the complex was formed. A new additional peak for -NH3 + appeared at 1616 cm−1, indicating that there are electrostatic interactions between the -NH2 (C-2 position) of chitosan and the -OH (C-3 position) of vitamin C.

Fourier transform infrared spectrum: (a) Chitosan, (b) chitosan/vitamin C complex, (c) mixture of chitosan and vitamin C, and (d) vitamin C.
DSC analysis of CSVC
From the DSC spectrum of the CSVC complex (Figure 4b), it can be seen that the endothermic peak of the CSVC complex near 115°C is higher than that near 100°C of chitosan (Figure 4a), indicating that the binding ability of the CSVC complex with water is enhanced. There is no exothermic peak of chitosan at 310°C (Figure 4b) in the CSVC complex, but a new endothermic peak at 300°C in the CSVC complex, indicating that the crystal morphology of CSVC and chitosan is different. 32,33 There is no decomposition peak of vitamin C near 190°C (Figure 4d) for CSVC, but a new endothermic peak at 300°C, indicating that the formation of the CSVC complex by chitosan and vitamin C is beneficial to the stability of vitamin C.
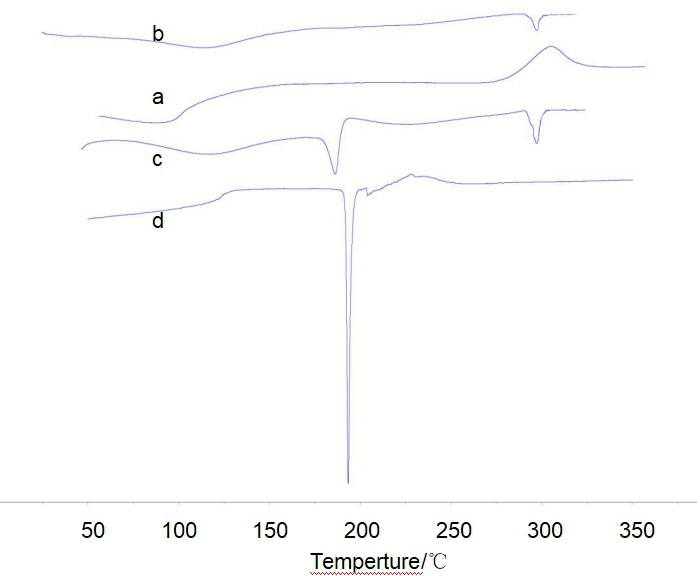
Differential scanning calorimeter spectroscopy: (a) Chitosan, (b) chitosan/vitamin C complex, (c) mixture of chitosan and vitamin C, and (d) vitamin C.

Hygroscopicity of chitosan/vitamin C complex.
The DSC spectrum of chitosan + vitamin C (Figure 4c) retains the endothermic peak of vitamin C at 190°C, while the exothermic peak of chitosan disappears at 310°C, and a new endothermic peak appears at 300°C. This shows that the mixture contains chitosan, vitamin C, and CSVC complex.
The DSC pattern of the CSVC complex is consistent with that of the chitosan/levodopa complex reported in the literature. 34
CSVC Performance
Hygroscopicity and moisture retention
Figures 5 and 6 show the results of the hygroscopicity and moisture retention properties of CSVC. With glycerol as a control, the moisture absorption and moisturizing properties of CSVC were better than chitosan. This is because the introduction of vitamin C into the chitosan molecule changes the crystal structure of the chitosan and improves the moisture retention and hygroscopicity of the chitosan.

Moisture retention of chitosan/vitamin C complex.
Anti-free radical
Radical scavenging effects of the CSVC complex were studied against the hydroxyl free radical (Figure 7).

Inhibition rate of chitosan/vitamin C complex on hydroxyl radicals.
The scavenging rate of the CSVC complex showed significant increases with increasing concentration. When the concentration was 3 mg/mL, the scavenging rate of hydroxyl radicals reached 100%. However, the scavenging effect of chitosan on hydroxyl radicals was very limited. This indicated that the synergistic effect of the new complex gained added advantage in hydroxyl radical scavenging activity. The ability of CSVC to scavenge hydroxyl radicals is mainly related to factors including active groups, intramolecular hydrogen bonding, and steric hindrance. Due to intra- and intermolecular hydrogen bonding, the -OH and -NH2 groups in the chitosan molecule are restrained, which affects the chitosan’s oxidation resistance. The introduction of vitamin C into the chitosan molecule not only introduces the enol hydroxyl group of the active group of vitamin C, but also exposes the -OH and -NH2 groups in the chitosan molecule, so that the hydrogen atom and the free radical are combined to achieve the effect of scavenging hydroxyl radicals.
Antibacterial properties of CSVC
The antibacterial activity of the CSVC solution against E. coli increased with increasing concentration. Compared with the blank control, the solutions at concentrations of 1.25-2.5 mg/mL in the CSVC group showed turbidity to varying degrees after 24 hours of culture, while the solution of the 5-10 mg/mL CSVC group was clear. From these results, the minimum inhibitory concentration of CSVC on E. coli was estimated at 5 mg/mL within 24 hours of culture.
CSVC Stability
Vitamin C was selected as a control, and the retention rate of vitamin C was used as an index to investigate the stability of CSVC. It can be seen from Figures 8 -10 that under the conditions of high temperature, high humidity, and strong light, the retention rate of vitamin C in CSVC and the control vitamin C decreased with time, but the reduction in CSVC was slower than the control vitamin C. On the 10th day of the high-temperature test, the retention rate of vitamin C in CSVC was 47.4%, and the retention rate of the control vitamin C was only 6.4%. On the 10th day of the high-humidity test, the retention rate of vitamin C in CSVC was 67.9% and that of the control vitamin C 37.7%. On the 10th day of the light test, the retention rate of vitamin C in CSVC was 31.0%, and that of control vitamin C was close to zero. This shows that the CSVC complex has a certain protective effect on vitamin C and relatively improves its stability. Experiments have shown that the factors that have the greatest influence on the stability of CSVC are in order of light >high temperature >high humidity. Therefore, CSVC should be placed in a cool and dry environment away from light.

The vitamin C retention rate of chitosan/vitamin C complex in high-temperature (40°C) accelerated test.

The vitamin C retention rate of chitosan/vitamin C complex in high-humidity (RH 85% ± 5%, 25°C) accelerated test.
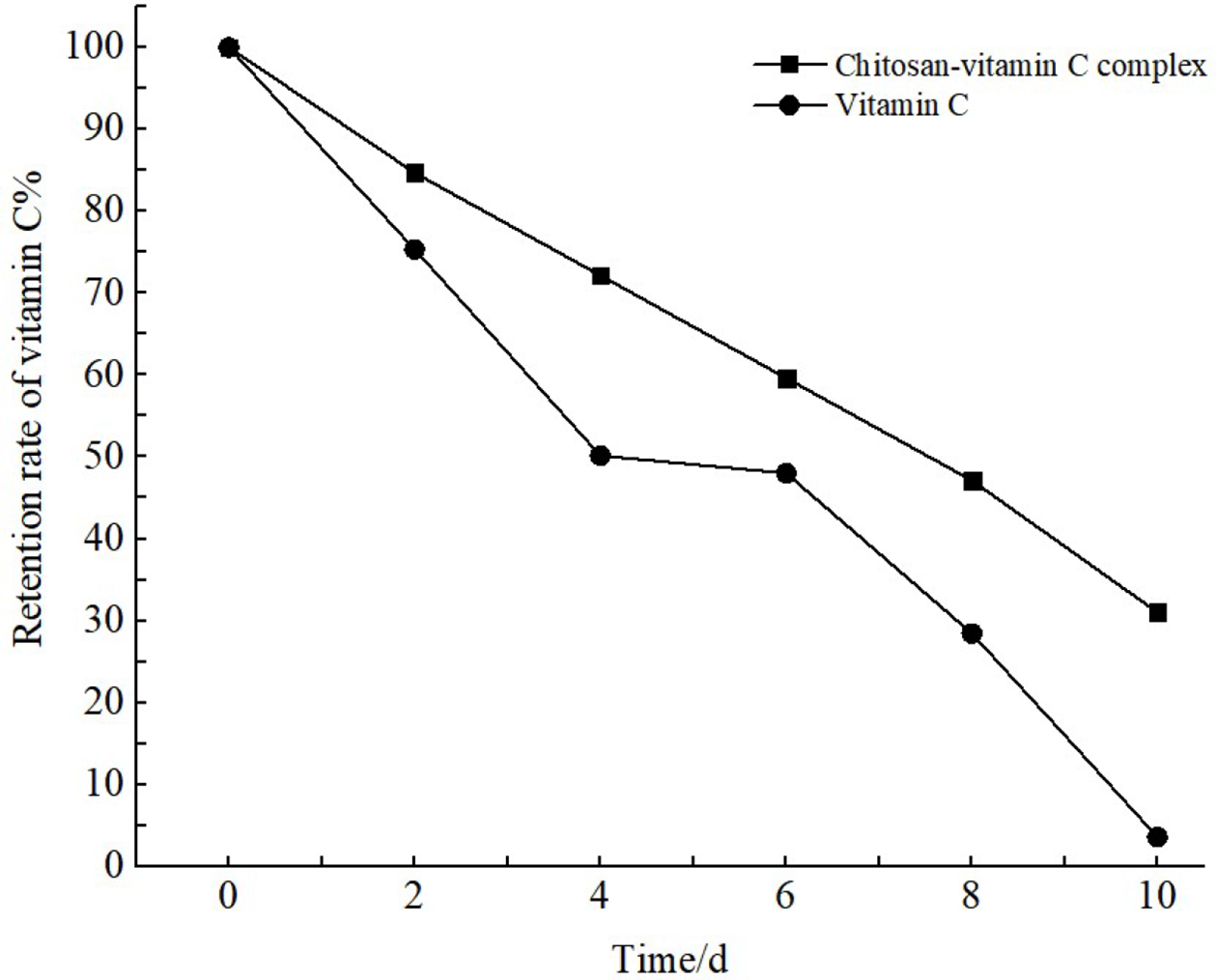
The vitamin C retention rate of chitosan/vitamin C complex in strong-light (4500 ± 500 Lx) accelerated test.
Conclusions
In the current study, a simplistic way of synthesizing CSVC complex from chitosan and vitamin C has been applied to improve the water-solubility capabilities. CSVC complex was prepared using isopropanol–water as the medium, a mass ratio of chitosan to vitamin C 1:1, and the reaction was protected from light at 25°C for 90 minutes. The CSVC complex is white in appearance, has good water solubility, has a viscosity average molecular weight of 273 kDa, contains 22.6% vitamin C, and gave a yield of 57.8%. The physicochemical properties of the complex were evaluated by UV, FT-IR, and DSC spectroscopy. The results indicate that the amino group (C-2) of chitosan and the hydroxyl group (C-3) of vitamin C have electrostatically combined to form a new reactive group -NH3 +. Compared with chitosan, the properties of CSVC complex have changed a lot due to the exposure of some -OH, -NH2 groups. The moisture retention, hygroscopicity, and scavenging ability for hydroxyl radicals of the CSVC complex are also significantly better than chitosan, and its stability is better than vitamin C. CSVC complex also retains the antibacterial, film-forming, and thickening properties of chitosan. As a cosmetic raw material, CSVC complex combines the advantages of chitosan and vitamin C, and gives chitosan new properties.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Zhejiang natural fund public welfare technology research program (LGN19C200009), National college students innovation and entrepreneurship training program 2018 (Letter No.39 [2018], 201810876026), Zhejiang province’s first class discipline "bioengineering" student innovation project (CX2019029) and Ningbo public welfare science and technology program (202002N3098).

