Abstract
The quest to determine and use polyphenolic compounds present in fruits and vegetables as natural antioxidants has recently attracted much attention due to their beneficial health effects. In this study, the total polyphenol content (TPC), total flavonoid content (TFC), total anthocyanin content (TAC), antioxidant activity (AA), and individual polyphenol components of Simon No. 1, Yuzi No. 7, Shangshu 19, and Pushu 32 sweet potato cultivars (edible parts: leaf, stalk, stem, skin, and flesh) were investigated. TPC, TFC, and TAC values ranged from 440 ± 0.17-12080 ± 0.58 CAE mg/100 g DW, 94 ± 0.08-4210 ± 0.74 QE mg/100 g DW, and 7 ± 0.01-1010 ± 0.54 CGE mg/100 g DW, respectively. Yuzi No. 7 sweet potato cultivar contained significantly higher amounts of TPC, TFC, TAC, and AA in all its edible parts, followed by Pushu 32, Simon No. 1, and Shangshu 19 in that order. Regardless of the sweet potato cultivar used, TPC, TFC, and TAC of sweet potato leaves were significantly higher than those of other edible parts. High-performance liquid chromatography revealed 19 individual phenolic compounds. In general, 3,5-di-
Keywords
Polyphenols, secondary plant metabolites widely distributed in fruits and vegetables, 1 have several reported health-enhancing properties, for example, antibiotic, antioxidant, antidiabetic, antiobesity, anticarcinogenic, and antimutagenic effects. 2 In recent decades, there has been a rise in lifestyle-related diseases due to changes in eating habits, behavior, and exercise regimes among humans. 3 This has led to an increased demand for healthy and minimally processed foods, with some health-conscious consumers opting to eat raw natural plant foods. Accordingly, natural alternatives to synthetic antioxidants have gained immense attention and research focus in recent years. 4
Sweet potato (
It has been reported that edible parts of sweet potato, such as leaves, stems, and roots, are good sources of essential amino acids, vitamins, and minerals important for animal and human nutrition. 7 A study by Sun et al 8 on the proximate composition of sweet potato leaves (SPL) obtained 16.69 ± 0.09-31.08 ± 0.09, 9.15 ± 0.49-14.26 ± 0.38, 2.08 ± 0.06-5.28 ± 0.15, 42.03 ± 0.03-61.36 ± 0.90, and 7.39 ± 0.86-14.66 g/100 g dry weight (DW) crude protein, crude fiber, crude fat, carbohydrates, and ash contents, respectively. Moreover, as attested by the nutritional quality indices, SPL was found to be an excellent source of proteins, fibers, and minerals. 8 Similarly, recent studies have underscored sweet potato as an important source of natural antioxidants, 9 and its high levels of anthocyanins make it a valuable crop in the functional food market. 10 Furthermore, the content of polyphenols in SPL was found to be higher than that of spinach and kales. 11 Increasingly, health-conscious consumers are preferring sweet potato cultivars with high levels of polyphenol compounds.
Reports suggest that considerable variation exists in the total polyphenol content (TPC), individual phenolic composition, and antioxidant activity (AA) among various cultivars of sweet potato. 7 These variations are ascribed to differences in color, variety, and environmental and processing conditions. 12 Bioactive components described in sweet potato comprise phenolic acids, such as caffeic acid and caffeoylquinic acid derivatives, including mono-, di- and tri-caffeoylquinic acids 13 ; flavonoids, such as astragalin, quercetin, kaempferol, and myricitrin 14 ; and cyanidin or peonidin-type anthocyanins. 9,15
Zengin et al 16 investigated TPC in SPL and found a range of 73.10 ± 3.75 to 89.26 ± 1.34 gallic acid equivalents (GAE) mg/g extract. Liu et al 14 recorded a 5.63 ± 0.21 quercetin equivalent (QE) g/100 g DW total flavonoid content (TFC) in SPL. Kourouma et al 17 reported 3.13 ± 0.38-9.38 ± 0.10 GAE mg/g DW TPC, 0.16-2.54 rutin mg/g DW TFC, and 0.02-1.02 ± 0.02 mg/g DW total anthocyanin content (TAC) in different sweet potato cultivars.
While several reports are available on contents of polyphenols and AA in various sweet potato cultivars, however, to date, there has not been a comprehensive study to determine the extent of variation in TPC, individual phenolic composition, and AA in all the edible parts of sweet potato. Additionally, most published reports have focused on the phenolic composition and AA in specific edible parts, that is, leaves and roots, rather than in all edible parts. A literature survey of all published work to date revealed that information on TPC, individual phenolic composition, and AA in all the edible parts of sweet potato was scarce. Therefore, due to the importance of polyphenols to human health and the ever-increasing demand for natural sources of antioxidants, we evaluated TPC, TFC, TAC, individual phenolic composition, individual flavonoid composition, individual anthocyanin composition, and AA in the edible parts (leaves, stalk, stem, skin, and flesh) of 4 selected cultivars of sweet potato. The information obtained could be useful in using sweet potato as a functional constituent in the food and pharmaceutical industries.
Results and Discussion
Evaluation of TPC, TFC, and TAC Values
As depicted in Table 1, significant differences (
Total Phenolic Content, Anthocyanin Content, Flavonoid Content, and Antioxidant Activities of 5 Edible Parts in 4 Sweet Potato Cultivars.

TPC, total polyphenol content; TFC, total flavonoid content; TAC, total anthocyanin content; FRAP, ferric reducing antioxidant potential; DPPH, 2, 2-diphenyl-1-picrylhydrazyl; ABTS, 2,2′-azino-bis (3-ethylbenzthiazoline-6-sulfonic acid); CAE, chlorogenic acid equivalent; QE, quercetin equivalent; CGE, cyanidin 3-glucoside equivalent; TE, trolox equivalent; AAE, ascorbic acid equivalent; DW, dry weight; ND, not detected; and ANOVA, analysis of variance.
Different uppercase letters showed significant differences in each group of data (
TPC values ranged from 440 ± 0.17 (obtained for flesh of Shangshu 19) to 12080 ± 0.58 (obtained for leaves of Yuzi No. 7) CAE mg/100 g DW. The TPC value for leaves of Yuzi No. 7 was 12080 ± 0.58 CAE mg/100 g DW, which was significantly higher than all the other samples (Table 1,
TFC values ranged from 94 ± 0.08 QE mg/100 g DW in the stalk of Shangshu 19 to 4210 ± 0.74 QE mg/100 g DW in the leaves of Yuzi No. 7 (Table 1). TFC values obtained for Yuzi No. 7 leaves were 4210 ± 0.74 QE mg/100 g DW, which was 1.54, 2.33, and 2.70 times higher than Pushu 32, Simon No.1, and Shangshu 19 SPL, respectively (
For TAC determination, the values obtained varied between 7 ± 0.01 CGE mg/100 g DW in flesh of Shangshu 19 to 1010 ± 0.54 CGE mg/100 g DW in leaves of Yuzi No. 7 (Table 1). TAC values obtained in our study for SPL (790 ± 0.25-1010 ± 0.54 CGE mg/100 g DW) were significantly higher than the value of 3.27 ± 0.29 mg/100 g DW reported by Su et al 19 and 9.40-227.53 mg/100 g FW reported by Jyothi et al 20 on different SPL varieties. In our study, the variation in the TAC levels between the edible parts of different sweet potato cultivars could be due to the differences in color as higher levels of TAC were achieved in the purple sweet potato cultivar (Yuzi No. 7) than in the orange and the white-colored sweet potato cultivars.
Antioxidant Activities
Table 1 presents the results obtained for ferric reducing antioxidant potential (FRAP), 2, 2-diphenyl-1-picrylhydrazyl (DPPH), and 2,2′-azino-bis (3-ethylbenzthiazoline-6-sulfonic acid) (ABTS) assays of edible parts of the 4 sweet potato cultivars. The FRAP AA varied from 26 ± 0.11 TE mg/100 g DW in the flesh of Shangshu 19 to 550 ± 0.21 TE mg/100 g DW in the leaves of Yuzi No. 7. Regardless of the sweet potato cultivar analyzed, the order of AA in different edible parts was as follows: leaves > stalk > stem > skin > flesh. In general, samples with high TPC levels exhibited higher AA and vice versa. The DPPH and ABTS assay results were also in good agreement with the FRAP results and were not examined in detail for brevity. The reported data for AA as assessed by FRAP, DPPH, and ABTS activities were significantly different. The differences in AA values could be ascribed to the individual AA measurement methods, that is, FRAP, DPPH, and ABTS assays, which measure different antioxidant effects. 21 Overall, based on our reported values for TPC, TFC, TAC, and AA, edible parts of Yuzi No. 7 sweet potato cultivar was considered as the best cultivar, suggesting a possibility of utilizing this cultivar by farmers and the food industry as a functional food.
Correlation analysis for the obtained values of TPC, TFC, TAC, and AA was done (Figure 1A–I). In this study, the correlation values for all the edible parts were measured except for SPL. This is because TPC, TFC, TAC, and AA values of SPL were significantly higher compared with other edible parts, that is, stalk, stem, skin, and flesh. Therefore, taking these correlations for SPL would result in high values that are statistically insignificant. As illustrated in Figure 1, low positive correlations were reported between TPC and DPPH (

(A) Schematic drawing showing all the five edible parts (leaves, stalk, stem, skin and flesh) of sweet potato plant. (B) Photograph of the four sweet potato cultivars (freeze dried flesh powders). Pushu 32 (skin and flesh are orange in colour), Yuzi No. 7 (skin and flesh are purple in colour), Simon No. 1 (skin and flesh are white in colour), and Shangshu 19 (skin is orange in colour while flesh is white in colour). All other edible parts, i.e., leaves, stalks, and stems, were green in colour. (C) Photograph of the four sweet potato cultivars (freeze dried leaf powders). (D) Representative photos of sweet potato anthocyanin extract.
Individual Phenolic Composition
As depicted in Table 2 and 19 individual phenolic compounds were detected in the edible parts of sweet potatoes (leaf, stalk, stem, skin, and flesh). The obtained retention times of authentic standards were used to identify the individual phenolic compounds. In addition, previously published data on retention times for phenolic acids, 23 flavonoids, 14 and anthocyanins 24 guided our study.
Contents of Individual Phenolic Compounds in Edible Parts of 4 Sweet Potato Cultivars (mg/100 g DW).

5-CQA, 5-
Different uppercase letters showed significant differences in each group of data (
Table 2 shows the detected individual phenolic acids. The most predominantly detected phenolic acids were di-, followed by mono-, and tri-
Table 3 shows the values obtained for individual flavonoids in edible parts of different sweet potato cultivars. Astragalin was the most predominant flavonoid, followed by either isoquercitrin or quercitrin, depending on the edible parts of cultivars. Yuzi No. 7, Pushu 32, Simon No. 1 and Shangshu 19 leaves showed high contents of astragalin (1729.9 ± 0.55, 1056.6 ± 0.42, 738.3 ± 0.25, and 491.3 ± 0.26 mg/100 g DW, respectively), with significant differences among them (Table 3,
Contents of Individual Flavonoid Compounds in Edible Parts of 4 Sweet Potato Cultivars (mg/100 g DW).

ND, not detected; ANOVA, analysis of variance; DW, dry weight.
Different uppercase letters showed significant differences in each group of data (
For individual anthocyanin analysis, acid hydrolysis of the samples was performed to release the major anthocyanidins (aglycons).
26
Table 4 shows 3 anthocyanidins identified as cyanidin, pelargonidin, and peonidin. Yuzi No. 7 leaves had the highest amount of cyanidin (763.1 ± 0.76 mg/100 g DW), while Shangshu 19 flesh contained the lowest amount of cyanidin (1.0 ± 0.00 mg/100 g DW) (Table 4,
Contents of Individual Anthocyanidin Compounds in Edible Parts of 4 Sweet Potato Cultivars (mg/100 g DW).
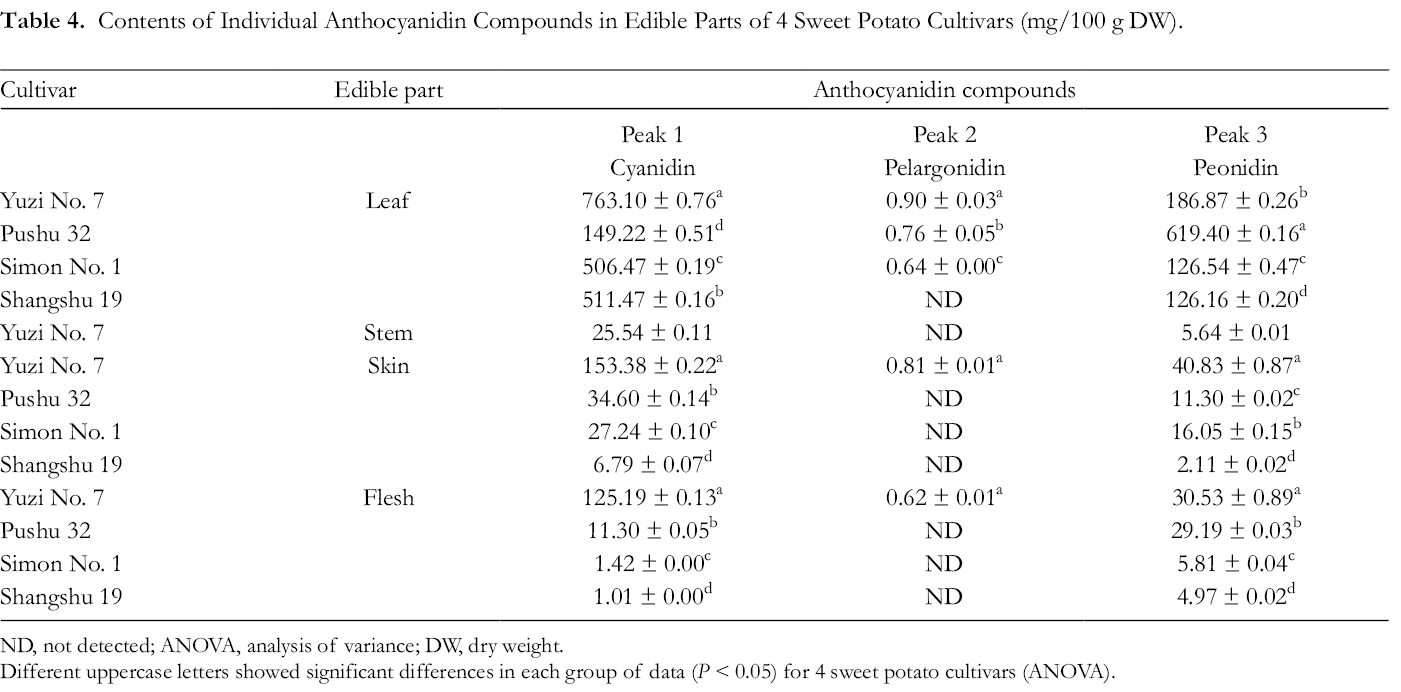
ND, not detected; ANOVA, analysis of variance; DW, dry weight.
Different uppercase letters showed significant differences in each group of data (
As observed in our study, differences in AA were noted among the different cultivars and edible parts. Sun et al 23 reported higher AA for individual phenolic compounds containing more hydroxyl groups, such as di and tricaffeoylquinic acids, than monocaffeoylquinic acids. Additionally, phenolic composition, molecular structure (number of mono-substituted or disubstituted caffeoylquinic acids), and the electron-donating abilities of molecules in sweet potato polyphenols greatly influenced their AA. 12 The aforementioned reasons, therefore, confirmed the differences in AA and phenolic composition among the different cultivars and the different edible parts as reported in this study.
In summary, this study has comprehensively explored the polyphenol abundance and AA in all the edible parts of sweet potato cultivars which are commercially available in China. To the best of our knowledge, this is the first elaborate investigation on the polyphenol content, polyphenol composition, and AA in the edible parts of sweet potatoes. TPC, TFC, TAC, AA, and individual phenolic contents displayed notable differences in all the 4 sweet potato cultivars (edible parts; leaf, stalk, stem, skin, and flesh). Overall, Yuzi No. 7 sweet potato cultivar contained significantly higher amounts of TPC, TFC, TAC, and AA in all the edible parts followed by Pushu 32, Simon No. 1, and Shangshu 19. Regardless of the sweet potato cultivar, TPC, TFC, TAC, and AAs of SPL were significantly higher than in other edible parts. Altogether, 19 individual phenolic compounds were analyzed by HPLC. In all the samples analyzed, the predominant phenolic acid, flavonoid, and anthocyanin compounds were 3,5-di-
Materials and Methods
General
Edible parts of 4 sweet potato cultivars (Simon No. 1, Yuzi No. 7, Shangshu 19, and Pushu 32) were obtained from Haileda Food Co., Ltd., Beijing, China (Figure 2A). The characteristics of the 4 cultivars were as follows: Simon No. 1 (skin and flesh white), Yuzi No. 7 (skin and flesh purple), Shangshu 19 (skin orange, while flesh is white), and Pushu 32 (skin and flesh orange). All other edible parts, that is, leaves, stalks, and stems, were green. The criteria used for choosing these specific cultivars was based on their specific applications in the food industry, that is, Simon No. 1, Yuzi No. 7, Shangshu 19, and Pushu 32 are used as a medicine, as a source of anthocyanin, in starch processing, and for fresh consumption, respectively. These 4 sweet potato cultivars were cultivated in June 2018, under identical standard conditions in an experimental farm of Haileda Food Co., Ltd in Beijing, China. The average growth temperatures were 24°C, 27°C, and 25°C for June, July, and August, respectively. The 4 sweet potato cultivars were harvested in the middle of August and separated into 5 edible portions, that is, leaves, stalk, stem, skin, and flesh. These 4 cultivars were harvested based on previous research by our research group (data not published) and other published data on polyphenol content present in fruits and vegetables at different maturity stages. 19,29 -31 The extracts from leaves, stalk, or stem were pooled from as many plants as possible (usually 10-15 plants) and combined before extraction. Also, tissues were pooled from 3-5 tubers for each skin and flesh sample. For flesh or skin extracts, whole roots of sweet potato plants were taken, their skin and flesh separated, and then combined before extraction. All independent experiments for each edible part were replicated 3 times. Finally, the samples were cleaned, freeze-dried, ground into powder, and kept at 4°C until further use.

(A) Schematic drawing showing all the 5 edible parts (leaves, stalk, stem, skin, and flesh) of sweet potato plant. (B) Photograph of the 4 sweet potato cultivars (freeze-dried flesh powders). Pushu 32 (skin and flesh are orange in color), Yuzi No. 7 (skin and flesh are purple in color), Simon No. 1 (skin and flesh are white in color), and Shangshu 19 (skin is orange in color while flesh is white in color). All other edible parts, that is, leaves, stalks, and stems, were green in color. (C) Photograph of the 4 sweet potato cultivars (freeze-dried leaf powders). (D) Representative photos of sweet potato anthocyanin extract.
Reagents
Folin-Ciocalteu reagent, 2,4,6‐tri-(2‐pyridyl)‐1,3,5‐triazine, ABTS, DPPH, 6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid (Trolox),
Polyphenol Extraction
The crude polyphenol extract was obtained according to the published procedure of Sun et al.
23
Briefly, 200 mL of ethanol (70% v/v) was used to dissolve 10 g of the sweet potato edible part powder. The contents were subjected to ultrasonic wave treatment (59 kHz) for 30 minutes at 50°C prior to centrifugation at 5000 ×
Total Polyphenol Content
TPC determination was done using Folin-Ciocalteu reagent, as outlined by Sun et al. 11 Chlorogenic acid (0.01-0.10 mg/mL) was used to calculate the amount of TPC. The results were calculated as chlorogenic acid equivalents (CAE) on a DW basis (CAE mg/100 g DW).
Total Flavonoid Content
TFC was assessed by following the colorimetric assay method published by Zhishen et al. 32 Quercetin (0.01-0.10 mg/mL) was used to determine flavonoid content. TFC was presented as quercetin equivalents (QE) per 100 g DW (QE mg/100 g DW).
Total Anthocyanin Content
TAC was evaluated based on the differential pH method as explained by Sun et al, 33 with a slight modification. Briefly, a 10 µL aliquot of sample was separately mixed with 290 µL of potassium chloride (0.025 M) and sodium acetate (0.4 M) solutions. The pH of the 2 solutions was set at 1.0 and 4.5 using potassium chloride (0.025 M) and sodium acetate (0.4 M), respectively. Absorbance readings were recorded at 520 nm and 700 nm using a spectrophotometer. TAC was calculated as cyanidin 3-glucoside equivalents using an extinction coefficient (ε) of 26 900 L/cm mol and a molecular weight of 432.
Antioxidant Activity
FRAP assay
The FRAP assay was performed as described by Benzie and Strain. 34 A standard curve made of Trolox (10-200 µg/mL) was employed as a control to determine the assay results, which are recorded as Trolox equivalents (TE) per 100 g of sample on a DW basis (TE mg/100 g DW).
DPPH Radical Scavenging Activity
The procedure used for the DPPH assay was that described by Brand-Williams et al, 35 with slight modifications. Briefly, 0.1 mM of DPPH solution (3 mL) was prepared and reacted with 200 µL of sample extract. The reaction was kept in a dark room for 30 minutes at room temperature. Absorbance readings were recorded using a spectrophotometer at 517 nm. The results were expressed as ascorbic acid equivalents (AAE) on a DW basis (AAE mg/100 g DW).
ABTS Radical Scavenging Capacity Assay
The ABTS assay followed the procedure illustrated by Thaipong et al, 36 with little modification. Briefly, ABTS solution (7.4 mM) and potassium persulfate (2.6 mM) were mixed together in equal proportions and allowed to react for 16 hours in the dark. Subsequently, the working solution obtained was diluted in ethanol to achieve an absorbance of 0.7 ± 0.05 at 734 nm before use. The activity was recorded as AAE mg/100 g DW.
Individual Phenolic Composition
Qualitative and quantitative analysis of phenolic acids by HPLC
Retention Time and Detection Wavelength of Polyphenol Compounds Found in Sweet Potato Edible Parts.

Statistical Analysis
The obtained data were reported as mean values ± SD of triplicate experiments. One-way analysis of variance followed by Duncan’s multiple range test was performed using SAS 8.1 (SAS Institute Inc., Cary, NC, USA). Significant differences between means were reported at
Footnotes
Acknowledgements
The authors are grateful to Beijing Haileda Food Co., Ltd. (Beijing, China) for supplying the sweet potato samples.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Natural Science Funding of China (31701614), the National Key R&D Program of China (2016YFE0133600), and the Earmarked Fund for China Agriculture Research System (CARS-10-B21).
