Abstract
Natural biostimulants such as plant growth-promoting rhizobacteria (PGPRs) and seaweed extracts are used in agriculture to improve crop yield in an environmentally friendly approach. However, interaction effects between different biostimulants are seldom investigated. This study aimed to determine the combined effect of PGPRs and Kelpak® (KEL), a seaweed-derived extract, on the growth and biochemical composition of Amaranthus hybridus, a traditional leafy vegetable of South Africa. A pot trial was conducted with 6 treatments—control (distilled water), Bacillus licheniformis (BL) inoculum, Pseudomonas fluorescens (PF) inoculum, 1% KEL, and combination treatments of KEL + BL and KEL + PF. These were applied 3 times to A. hybridus seedlings at 2 weekly intervals prior to harvesting after 6 weeks. Growth parameters and biochemical content were quantified in the harvested material. Kelpak® significantly improved growth (leaf number and area, shoot and root length, and fresh weight) and chlorophyll and carotenoid content. Treatments with 2 microorganisms had a significant negative effect on growth and pigment content. However, the combination treatments significantly improved growth and photosynthetic pigment content to similar levels as the KEL treatment. The mineral content was significantly increased for most minerals when treated with PF and decreased when treated with KEL and KEL + BL. Treatment with 2 bacterial strains and KEL caused a significant decrease in superoxidase dismutase activity with an additive effect when applied in combination. This suggests stress reduction in A. hybridus. Combined use of PGPR and seaweed extracts promises to be an eco-friendly strategy to improve the production and mineral content of traditional leafy vegetables such as A. hybridus.
Modern agriculture is facing emerging threats such as a rapid population increase, global warming, and environmental pollution. All these threats have impacted negatively on food production worldwide. Sustainable and eco-friendly approaches are required to address challenges of increased global food demand, decrease in arable land and resources, and several environmental pressures caused by climate change. 1,2 Macronutrients such as nitrogen (N), phosphorus (P), potassium (K), and sulfur (S) found in mineral fertilizers are important in agriculture but fertilizer overuse has also resulted in detrimental effects on the environment. Natural biostimulants provide a promising approach to reduce the reliance on synthetic agrochemicals while maintaining high productivity. 3 Natural biostimulants are defined as “any substance or microorganism applied to plants with the aim to enhance nutrition efficiency, abiotic stress tolerance and/or crop quality traits, regardless of its nutrient content.” 4 EU regulations define the specific functions of the biostimulant such as nutrient use efficiency, tolerance to abiotic stress, quality traits, and availability of locked up nutrients in the soil. 3,5 There are various categories of biostimulants, namely humic andit must be fulvic acid, animal and vegetal protein hydrolysates, seaweed extracts, silicon, and beneficial microorganisms such as arbuscular mycorrhizal fungi (AMF), N2-fixing bacteria, and plant growth promoting rhizobacteria (PGPR). 3
Microorganisms have an essential role in recycling nutrients in the soil. 6 Plants interact with a wide variety of soil-inhabiting organisms in different ways, either competitive, exploitative, neutral, commensal, or mutualistic. 7 There are an estimated 20 000 plant species that cannot survive without symbiotic associations with microorganisms. 8 These associations have been in existence for the past 450 million years when the ancestral plant lineages first colonized land. 9 The association involves the plant being a source of nutrition and a habitat for the microorganisms, and in return, the plant gets numerous benefits from the microbes such as growth promotion and stress reduction. 10
Beneficial microorganisms such as AMF and PGPR can potentially be used for crop production. They have a biofertilizer effect, especially through N2 fixation, P solubilization, and iron mobilization. Other plant growth-promoting traits are due to the presence of phytohormones, siderophores, amino acids and polysaccharides, and antimicrobial compounds. 1,11 There are a wide range of benefits on plant health that are derived from microorganisms. These include suppression of diseases, 12 priming of plant immune systems, 13 inducing systemic resistance, 14 increasing the acquisition of nutrients, 15 tolerance of abiotic stresses, 16 coping with variations in environmental conditions, 17 and establishment of mycorrhizal associations. 18 Plant-microbe interactions are of vital importance for the growth of plants under adverse climatic conditions. They should be taken into consideration when designing novel strategies targeting yield improvement and stress resistance in crop plants. 11,19
There is a great need for the production of microbial-based bioformulations that work in a complementary and synergistic manner with mineral fertilizers to increase plant performance. 1 Nutrients can limit yields of plants; for example, iron (Fe) and zinc (Zn) can be abundant in soils but not be available for crops. 20 Several strains of bacteria enhance the availability of Fe by producing siderophores (organic acids). 21 The same siderophores can also control pathogenic microorganisms by limiting the availability of Fe to the microorganisms. 21,22 For example, cotton yield was increased when PGPRs were combined with compost and NPK fertilizer 23 and long-term PGPR application in combination with compost enhanced straw biomass and grain yields of wheat. 24 The predominant plant growth-promoting bacteria are Pseudomonas and Bacillus species. 25 Rhizophagus irregularis (formerly Glomus intraradices), Bacillus amyloliquefaciens, and Pseudomonas fluorescens (PF, the most extensively studied species) 26 are usually used as commercial soil additives because of their ability to promote plant growth. 27
Natural seaweed extracts are successfully used in agriculture as biostimulants. They enhance crop productivity and reduce the use of conventional synthetic fertilizers. 28 One such seaweed extract is Kelpak® (KEL). It is made from the kelp Ecklonia maxima (Osbeck) Papenfuss that is harvested on the west coast of South Africa. 29 The growth-stimulating effects of KEL on a number of agricultural crops are well documented. 30 -33 Kelpak® is effective when applied at low concentrations, suggesting that elicitor compounds such as plant hormones are the active ingredients. Plant growth regulators, such as auxins, cytokinins, gibberellins, brassinosteroids, and abscisic acid, 34,35 polyamines (putrescine and spermine), 32 and a phlorotannin (eckol) 33 have been identified in KEL.
Plants are often exposed to adverse environmental conditions such as water deficit or excess, high intense light, low or high temperature, salinity, heavy metals, UV rays, and insect and pest attacks. 36 Such stresses induce metabolic changes that increase the production of reactive oxygen species (ROS). This causes oxidative stress which adversely affects plant growth and development. 36,37 Superoxide dismutases (SODs) are commonly occurring metalloenzymes found in plants. They provide the first line of defense against ROS toxicity and are one of the most effective components of the antioxidant defense system in plants with SOD activity increasing when the plant is exposed to oxidative stress. 36
Amaranthus hybridus L. is a naturalized traditional food crop in South Africa, referred to as “wild spinach.” The leaves are consumed as a leafy green vegetable. This drought-tolerant species grows as a weed or is planted in home gardens. It has an important role in improving food security and nutrition, especially in rural communities, as it has a high protein, iron, and calcium content as well as containing amino acids (lysine and methionine), vitamins A and C, and antioxidant compounds. 38,39 Cost-effective and easy-to-apply technology to improve its yield and nutritive value would be beneficial to small-scale farmers and households. In addition, with increased interest in organic-based agriculture and the need to minimize fertilizer use, the potential to combine application of PGPRs and plant-based biostimulants such as KEL to improve the growth, yield, and quality of the crop needs to be investigated. This study was therefore carried out to determine the interactive effect of 2 microorganisms (Bacillus licheniformis [BL] and PF) applied alone and in combination with the seaweed-based biostimulant KEL on A. hybridus L. The effect of these treatments on growth, biochemical, and nutrient composition was measured.
Results
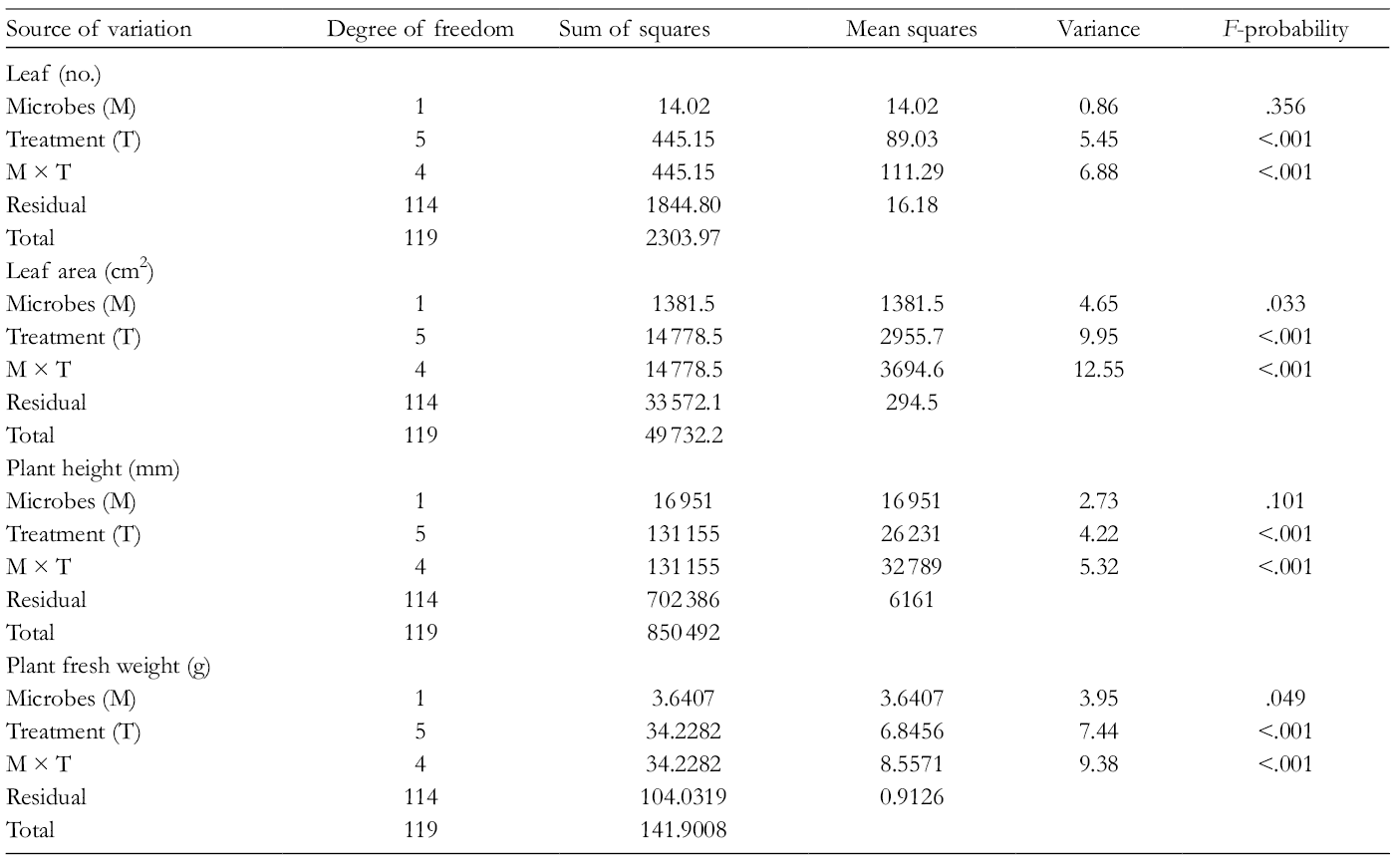
The application of microorganisms alone had a deleterious effect on the growth of A. hybridus (leaf number and area, shoot and root length, and fresh weight). Bacillus licheniformis and PF caused a significant decline in the growth of A. hybridus compared to the control plants. In contrast, KEL had a beneficial effect on plant growth with higher values recorded in these plants compared to the control treatment (Figure 1). Combined application of the microorganisms and KEL overcame the negative effect of the microorganisms with comparable growth achieved as with the KEL treatment. Kelpak® + PF showed a significant positive effect on all the growth parameters compared to the control plants. Kelpak® + BL also had a significant effect but to a lesser extent than the KEL + PF treatment (Figure 1). The results of the study are supported by analysis of variance (ANOVA) where microorganisms alone had no significant influence on the leaf number (Table 1). Only treatment (Treatment) and the interaction of microbes and treatment (Microbes × Treatment) had a significant effect on leaf number (P < .05). Microbes (M) alone, treatment (Treatment), and the combination of microbes and treatment (Microbes × Treatment) had a significant influence on leaf area (Table 1). Microbes alone showed no significant influence on the height of A. hybridus plants, while notable significant effects were observed due to treatment (Treatment) and a combination of microbes and treatment (Microbes × Treatment). The fresh weight of A. hybridus was significantly influenced by the microbes, treatment, and their interactions (Table 1).

Effects of microorganisms (Bacillus licheniformis and Pseudomonas fluorescens) BL and PF and the seaweed-derived biostimulant KEL on the growth of Amaranthus hybridus grown under greenhouse conditions. Bars (±SE) of each growth parameter with different letter(s) are significantly different according to Duncan’s multiple range test (P < .05).
Analysis of Variance for Comparing Different Treatments and Interactions for the Growth Response of Amaranthus hybridus (P < .05).
Bacillus licheniformis- and PF-treated plants had a significantly lower chlorophyll a content compared to the control plants. Kelpak®-treated plants had a significantly higher chlorophyll a content. Although not as high as the KEL-treated plants, KEL + BL- and KEL + PF-treated plants showed a significant increase in the amount of chlorophyll a compared to the plants treated with microbes alone. However, control plants were not significantly different from these treatments (Table 2). None of the treatments showed any significant influence on chlorophyll b production apart for the significant decrease in BL-treated plants (Table 2). For chlorophyll a + b and carotenoid content, the same trend was apparent as with chlorophyll a where the KEL-treated plants showed the most significant increase, BL and PF generally caused a significant decrease and the combined treatments caused increased levels, although not significantly different from the control plants (Table 2).
Effects of Bacillus licheniformis, Pseudomonas fluorescens, and Kelpak® on the Photosynthetic Pigment, Biochemical and Superoxide Dismutase Enzyme Content of Amaranthus hybridus After 6 Weeks.

BL, Bacillus licheniformis; KEL, Kelpak; PF, Pseudomonas fluorescens; SOD, superoxide dismutase; FW, fresh weight.
Mean values (±SE) in a column with different letter(s) are significantly different according to Duncan’s multiple range test (P < .05).
Bacillus licheniformis- and PF-treated plants generally had a significant decrease in total soluble protein and carbohydrate content, while the KEL treatment had no significant effect. Protein and carbohydrate levels were similar to those of the control plants for the combination treatments (Table 2).
All the treatments significantly reduced the activity of SOD compared to A. hybridus control plants with the combination treatments having the significantly lowest activity (Table 2), indicating stress reduction in the plants.
The mineral composition in A. hybridus varied with different treatments (Table 3). Plants treated with PF generally had the highest mineral content with significantly higher Ca, Mg, K, P, Zn, and Mn content and significantly lower Fe and Al compared to the control plants. N and Cu were similar to the control plants (Table 3). In contrast, KEL-treated plants had significantly higher K and P and significantly lower N, Ca, Fe, and Al compared to the control plants. Mg, Zn, Cu, and Mn were similar (Table 3). The combination treatment of KEL + BL generally decreased the mineral composition (K, P, Zn, Fe, and Al) compared to the KEL treatment with only N and Ca increasing in the combination treated plants and Mg, Cu, and Mn being similar in both treatments (Table 3).
Effects of Bacillis licheniformis, Pseudomonas fluorescens, and Kelpak® on the Mineral Composition of Amaranthus hybridus After 6 Weeks.

BL, Bacillus licheniformis; KEL, Kelpak; PF, Pseudomonas fluorescens.
Mean values (±SE) in a column with different letter(s) are significantly different according to Duncan’s multiple range test (P > .05).
Discussion
In the present investigation, KEL, a seaweed-based biostimulant, enhanced the growth of A. hybridus. The positive and significant response of A. hybridus to the application of KEL could be due to the presence of various phytohormones, polyamines, and the phlorotannin eckol that are present in KEL. 32 -35 Similarly, KEL-treated okra seedlings had a significant increase in shoot length, the thickness of stem, leaves and root numbers as well as fresh weight under deficiencies of both phosphorus and potassium. 32 The 2 rhizobacteria used in the present study had a negative effect on growth of A. hybridus. This is contrary to other reports where PGPRs elicit positive responses from the treated plants. For example, fluorescent Pseudomonas-FAP2 and BL improved growth and photosynthetic attributes in wheat 40 and PF significantly enhanced yields in rice (Oryza sativa) and improved resistance against pathogens. 41 However, not all reports on the use of PGPR are positive. For example, in a series of field trials with hybrid spruce ecotypes, seedlings were inoculated with 2 bacterial strains isolated from the seedlings. Hydrogenohaga pseudoflava increased shoot and root growth, while Pseudomonas putida had an inhibitory effect. 42 Foliar application of a seaweed extract (derived from Ascophyllum nodosum) stimulated growth and K uptake in almond trees in both K-sufficient and K-deficient conditions, while a microbial fermentation product was only effective at stimulating growth in K-sufficient conditions. 43 These examples all used commercial crops which have been extensively bred for improved productivity. Amaranthus hybridus is a “wild” plant with limited selection for beneficial traits. More promising responses may have been achieved in the present study using rhizobacteria isolated from the rhizosphere of A. hybridus rather than laboratory cultured strains which appear incompatible. More extensive screening needs to be done to find suitable PGPR strains. The combination treatments of KEL + BL and KEL + PF used in the present study resulted in a significant improvement in the growth of A. hybridus, indicating that the positive effects of KEL overrode the negative effects of the rhizobacteria.
Seaweed extracts promote the biosynthesis of chlorophyll and/or minimize its breakdown, resulting in an increase in the green color of leaves. 28 An increased chlorophyll and carotenoid content was measured in the KEL-treated A. hybridus. Increasing the chlorophyll content is advantageous to the plant since it translates into increased photosynthetic activity within the leaves. 44 Similarly, an increase in photosynthetic pigment, phytochemicals, and myrosinase activity was also observed in cabbage treated with eckol, a phenolic compound isolated from Ecklonia maxima, the seaweed from which KEL is produced. 45 Leaf color is an important parameter of quality in leafy vegetables like A. hybridus since greenness is more appealing to consumers.
There are contradictory reports on the effect of PGPR on primary metabolite content. There are positive reports of increased pigment content in plants whose roots had been inoculated with PGPRs of the genera Pseudomonas, Bacillus, and Azotobacter. 46 In contrast, primary metabolites (chlorophyll, carotenoids, protein, and sugar) and α-amylase activity in Okra were not affected by BL application despite the improved growth. 47 In the present study, chlorophyll, carotenoid, protein, and carbohydrate content decreased in A. hybridus treated with BL and PF. This may be linked to the reduced growth of the plants. This reduction in pigment, protein, and carbohydrate content was restored by KEL in the combined treatments of KEL + BL and KEL + PF.
Biostimulants are able to counteract environmental stresses such as water deficit, soil salinization, and exposure to suboptimal growth temperatures. 4,48 This results in the improvement of plant performance, increasing plant growth and productivity. 44 Kelpak® treatment as well as BL and PF treatments decreased SOD activity in A. hybridus. Combination treatments of KEL + BL and KEL + PF further significantly reduced SOD activity, indicating a synergistic effect of these treatments on the antioxidant system. Similarly, the antioxidant system of Brassica rapa was affected by KEL-treated plants having a lower ascorbic acid and phenolic content. The lower concentrations of these antioxidant compounds were attributed to the reduced need of these plants to cope with oxidative stress 49 as may be the case in the present study.
The mineral composition (Ca, Mg, K, P, Zn, and Mg) of A. hybridus increased with PF treatments. This is in agreement with other studies, eg, Bacillus subtilis and PF inoculated radish seeds (Raphanus sativus) had significant increases in P, N, K, Mg, and Ca in the roots and leaves 50 ; there was an increase in mineral elements (N, K, Ca, S, P, Mg, Fe, Mn, and Zn) in broccoli (Brassica oleracea L., var. italica) treated with PGPRs 51 ; there was a significant increase in N, P, K, Ca, and Mg in greenhouse-grown tomato (Solanum lycopersicum) plants inoculated with Pseudomonas, Azotobacter, and Azosprillum. 52 Plant growth-promoting rhizobacteria have many beneficial traits to increase the bioavailable forms of nutrients present in the soil and to enhance their absorption by plants. 1,11,53 The higher mineral content in the leaves improves the nutritional value of A. hybridus. This is an important consideration when addressing the issues of hidden hunger.
In contrast, KEL-treated A. hybridus plants only had higher P and K levels and decreased N, Ca, Fe, and Al levels. This was similar to KEL-treated B. rapa plants where P and K increased and Na decreased. 49 In contrast, higher concentrations of Ca, K, and Mg were recorded in leaves of KEL-treated lettuce 54 and high amounts of Mg, Cu, Fe, Zn, and Ni occurred in microtubers of potatoes (Solanum tuberosum) treated with KEL. 55 Mineral content was also low in the combination treatments in the present study. While the KEL treatments enhanced root growth and thus increased the absorption capacity of the plants, the increased growth would require faster nutrient utilization which could explain the lower levels recorded in the leaves of KEL-treated A. hybridus. The mineral content also varies in A. hybridus with the stage of plant development and plant age. 39 Different growth rates achieved with the various treatments may also account for the differences in mineral content.
Several experimental studies have demonstrated additive and synergistic effects of different plant biostimulants combined with PGPR to promote growth and productivity of plants. 56,57 For example, humic acid and a seaweed extract both improved growth in groundnut plants with combined treatments having a synergistic interaction resulting in a superior increase in general growth. Plant improvement was associated with the enhancement of N uptake and the subsequent synthesis of chlorophyll which increased the rate of photosynthesis, thereby triggering the movement of the photosynthates to the sinks. 58 Onion seedlings treated with humic acid and Rhizophagus intraradices inoculum resulted in a significant increase in root dry weight and carotenoids content with a further significant increase when humic acid and the mycorrhiza fungi were applied simultaneously. The increase in crop performance was attributed to an increase in the availability of nutrients caused by the synergistic action of humic acid and mycorrhizal fungi when applied in combination. 56 A microbial based biostimulant containing R. intraradices and Trichoderma atroviride applied to salinity and alkalinity stressed lettuce improved growth (root and shoot dry weight), yield, plant color (SPAD index), nutritional status and increased the photochemical activity of photosystem II and antioxidant enzyme activity (CAT and GPX). There was a synergistic effect when applied in combination with a protein hydrolysate. The beneficial effects of these biostimulants were attributed to improved rooting, enhanced chlorophyll synthesis, and increased activity of PS II. 59 Mycorrhizal fungi and humic acid had a similar synergistic effect when applied in combination on ryegrass where root biomass and chlorophyll biosynthesis were enhanced compared to either application alone. 60 A similar response occurred in micropropagated pineapple plantlets due to the synergistic action of vermicompost and PGPR, which significantly enhanced both shoot dry weight and leaf area. 57
However, not all combinations of biostimulants are synergistic. For example, in a pot trial where the hormone-producing BL and karrikinolide, a biostimulant, isolated from plant-derived smoke promoted okra growth when applied alone. Co-application had an antagonistic effect, probably due to overlapping modes of action disrupting hormone levels in the plant. 47 Pseudomonas fluorescens and BL isolated from grapevine (Vitis vinifera cv. Malbec) and smoke-water (containing karrikinolide) elicited a slight improvement in rooting of grapevine cuttings but combined treatments were antagonistic. Single treatments also enhance growth in a pot trial with combined treatments being antagonistic. Chlorophyll content and photosynthetic efficiency were not affected. These biostimulants increased the mono- and sesquiterpenes content with an additive effect with the combined treatments. These compounds are involved in the plants defense system, and thus, the treatments improved the stress tolerance of the plants. 61 Antagonistic responses were also achieved in the present study with KEL treatments improving growth and pigment content but decreasing mineral content and the bacteria inoculum inhibiting growth but enhancing the mineral content.
Conclusion
The present study demonstrated the potential benefit of applying different biostimulants to improve yield and quality of the leafy vegetable A. hybridus. The seaweed-derived extract KEL improved growth parameters and increased the pigment content, thus improving the greenness and palatability of the leaves. Plant growth-promoting rhizobacteria improved the mineral content of the leaves, thus improving its nutritional value. This is an important consideration when addressing the issues of hidden hunger that is prevalent in rural communities of South Africa. However, the bacterial strains tested in the present study inhibited the growth of A. hybridus. A screening program needs to be carried out to find a more suitable PGPR which would work synergistically with KEL. Applying suitable bacterial inoculum to the soil to maintain a threshold bacterial population may be a tool to further increase the nutritional value of A. hybridus without the high costs of fertilizer application. All treatments decreased the SOD levels, indicating reduced stress levels. The effect of these biostimulant treatments in stress conditions needs to be investigated. Combined use of PGPR and seaweed extracts promises to be an eco-friendly strategy to improve the production and mineral content of traditional leafy vegetables such as A. hybridus.
Experimental
Site of the Experiment
The experiment was carried out in a greenhouse at the University of KwaZulu-Natal (UKZN) Botanical Garden, Pietermaritzburg Campus (290°37.55′S; 300°24.13′E), South Africa.
Bacterial Inoculum
Bacillus licheniformis (ATCC 12759) and PF (ATCC 13525) were purchased from the American Type Culture Collection (ATCC). Bacterial inoculum was prepared by culturing BL and PF in 200 mL Muller-Hinton (MH) broth on an orbital shaker at 35 °C and 27 °C, respectively, for 2 days. The optical density was measured at 660 nm by spectrophotometer (Varian Cary 50 UV-Vis Spectrophotometer, Australia) to achieve uniform populations of bacteria of 108 colony-forming units (CFU)/mL. The inoculum was centrifuged at 5000 g for 10 minutes (4 °C) (Avanti J-E Centrifuge, Beckman Coulter, Inc., CA, United States). The pellet was rinsed with distilled water to remove traces of MH broth. A bacterial suspension was made using distilled water so that the absorbance value was 1.0 when measured at 660 nm.
Pot Trial
Amaranthus hybridus seeds were purchased from McDonald’s Seed Company, Pietermaritzburg, South Africa. New pots (10 cm diameter) were filled with 242 g autoclaved garden soil which comprised 80% compost, 19% bark (chipped and decomposed), 0.5% limestone ammonium nitrate, and 0.5% of a 5:7:4 NPK fertilizer (soil pH 5.8). Seeds were sown in a nursery to raise seedlings, and after 7 days (2-leaf stage), healthy seedlings were transplanted into pots (10 cm diameter). There were 3 seedlings per pot with 5 pots per treatment. Pots were arranged on a metal bench in a greenhouse with a daily maximum and minimum temperature of 22 °C ± 3 °C and 15 °C ± 2 °C, respectively, and a mid-day light intensity of 500 to 600 µmol/m2/s. A randomized pot trial was carried out in which the interactions between BL, PF, and KEL were compared to a control treatment. Seven days after transplanting, the seedlings were treated with either 10 mL bacterial inoculum per pot or 10 mL KEL (1%) per pot. This was applied to the soil around the plants. For the combination treatments, KEL (1%) was incorporated into the 10 mL bacterial inoculum. The treatments were as follows: control (distilled water), BL, PF, 1% KEL, 1% KEL + BL, and 1% KEL + PF. A second and third application of these solutions was done 2 and 4 weeks later. The seedlings were irrigated twice weekly with tap water (100 mL) for the duration of the pot trial except on the day of treatment. Plants were harvested 6 weeks after sowing. Fresh weights of roots and shoots (combined leaf and stem material) and leaf area (measured with a leaf area meter LI-31000, LI-COR Inc., Nebraska, United States) were recorded as a measure of growth. The growth measurements of 3 plants harvested from each of the 5 pots were combined to make 1 sample (n = 5). Fresh material (leaf) was randomly harvested from 5 pots to make 3 replicates of each treatment for biochemical analyses.
Determination of Total Chlorophyll and Carotenoid Content
The photosynthetic pigments [total chlorophyll (a + b) and carotenoids] were estimated by the methods of Lichtenthaler. 62 Fresh leaf material (100 mg) was ground with 5 mL ice-cold acetone (80%). The ground material was kept on ice in the dark. The resultant solution was centrifuged (Hettich Universal, Tuttlingen, Germany) at 3000 g for 10 minutes at 4°C and the volume recorded. The absorbance of the solution was read at 470, 645, and 662 nm using a UV-visible spectrophotometer (Varian Cary 50, Australia).
Chlorophyll and carotenoid content were calculated as follows:
Chlorophyll a = 11.23A662 – 2.04A645
Chlorophyll b = 20.13A645 – 4.19A662
Chlorophyll a + b = 7.05A662 + 18.09A645
Total carotenoid = (1000A470 – 1.90Chla – 63.14Chlb)/214.
Results were calculated as µg/g FW.
Protein Estimation
Total soluble protein was estimated using the Bradford method 63 with minor modifications. Samples (200 mg FW) were homogenized in an ice-chilled mortar and pestle with 6 mL ice-cold phosphate-buffer saline (PBS; 8 g NaCl [137 mM], 0.2 g KCl [2.7 mM], 1.44 g Na2HPO4 [10 mM], 0.24 g KH2O4 [1.8 mM] in 1 L distilled water [pH 7.2]). The homogenate was centrifuged at 10 000 g for 15 minutes at 4°C. Samples (100 µL crude enzyme extract) were pipetted into test tubes and made up to 1 mL with PBS. Bradford dye (1 mL) was added and the contents mixed by a vortex and allowed to stand for a few minutes. The absorbance was recorded at 595 nm against a blank. A standard curve was made using bovine serum albumin and used to estimate the total soluble protein content of the samples.
Carbohydrate Estimation
The method of Sadasivam and Manickam 64 with minor modifications was used for determining the total carbohydrate content. Leaf material (200 mg FW) was weighed and hydrolyzed in boiling water for 3 hours with 3 mL 2.5 N HCl. The hydrolyzed plant material was cooled to room temperature and further neutralized with sodium carbonate. This was added until the effervescence ceased. The volume was made up to 5 mL with distilled water and the extract centrifuged at 3000 g for 10 minutes at 4°C. Supernatant (100 µL) was taken and made up to 1 mL with distilled water. Subsequently, 4 mL anthrone reagent was added. The optical density was measured at 630 nm. A standard curve was made using 0 to 100 mg glucose and used to calculate the carbohydrate content in the samples.
Superoxidase Dismutase Activity
The SOD enzyme activity of leaf protein was assayed by measuring its ability to inhibit the photochemical reduction of nitroblue tetrazolium (NBT) using the method of Giannopolitis and Ries. 65 The reaction mixture (3 mL) consisted of 50 mmol phosphate buffer (pH 7.8), 13 mmol methionine, 75 µm NBT, 2 μmol riboflavin, 50 μmol EDTA, and 100 µL crude enzyme extract (protein). Tubes were vortexed and then subjected to illumination under 20 W fluorescent tubes. The reaction was allowed to proceed for 15 minutes after which the tubes were placed in the dark. The absorbance of the reaction mixture was taken at 560 nm. One enzyme unit was defined as the amount of enzyme which caused 50% inhibition of NBT photoreduction. The enzyme activity was expressed as U/μg FW.
Mineral Element Analysis
The nitrogen (N) content was determined by the method of Dumas 66 using a LECO-Truspec CNS analyzer. The other minerals, viz., aluminum (Al), iron (Fe), manganese (Mn), zinc (Zn), copper (Cu), calcium (Ca), magnesium (Mg), sodium (Na), phosphorous (P), and potassium (K), were determined by inductively coupled plasma-optical emission spectrometry (ICP-OES; VISTA-MPX).
For the determination of N, milled plant material was placed in an oven at 110°C overnight. The following day, samples were cooled in a desiccator for 30 minutes. The samples (0.125 g DW) were then burnt in a module consisting of an electric furnace set at 950°C.
For the determination of Al, Fe, Mn, Zn, Cu, Ca, Mg, Na, P, and K, 0.5 g milled samples were weighed and placed in crucibles (crucibles were preheated overnight at 110°C and were cooled in a desiccator and weighed) in an oven set at 110°C for 2 hours. Crucibles with the samples were placed in a furnace set at 450°C for 4 hours. After 4 hours, the furnace was opened and allowed to cool off. Subsequently, crucibles were removed from the furnace and cooled. Samples were then digested where a few drops distilled water was added, followed by 2 mL concentrated HCl. The samples were slowly evaporated to dryness in a water bath in a fume hood. Then, 25 mL freshly prepared 1:9 HCl solution was added to each sample and stirred with a glass rod. The samples were filtered through Advantec 5B: 90 mm diameter filter papers into clean sample crucibles. The filtrate was diluted with de-ionized water (ratio 5:20) and the diluted solutions analyzed for elements in the ICP-OES system. The appropriate standards were prepared by dilution of each pure element standard within the concentration range of the elements in the samples. The calibration standards were treated similar to the samples. The elemental content was calculated as a % using the dry matter and sample weight. All sample vials, crucibles, and glassware were cleaned by soaking in 10% (v/v) HNO3 and rinsed with deionized ultrapure water (Milli-Q, Millipore, Bedford, MA, United States) before use. The results were obtained from triplicate measurements.
Statistical Analysis
The quantification of all parametric data was done in replicates and the results presented as means with standard error (±). Mean value comparison was computed using one-way (ANOVA using GenStat 18th Edition. Duncan’s multiple range test was used for statistical significance (P < .05) to separate the mean values. Graphs were plotted using SigmaPlot for Windows Version 11.
Footnotes
Acknowledgments
The authors acknowledge support received from the University of KwaZulu-Natal (UKZN). The assistance offered by Mrs Alison Young (UKZN Botanical Garden Chief Horticulturist) and staff is much appreciated.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation (NRF: Indigenous Knowledge Systems, Grant UID: 93175), Pretoria, South Africa.
