Abstract
A new triterpene, named milemaronol (
The family Simaroubaceae contains 117 reported species of trees, whose characteristic is the bitter taste of their cortex. 1,2 This can be attributed to the presence of quassinoids, which is also considered a taxonomic marker of this family. 3 The genus Homalolepis Turcz., recently segregated from Simaba Aub., comprises 28 species, spread mainly in tropical South America. 2
Besides the quassinoids, other types of triterpenes, alkaloids, and other classes of compounds were also observed. 4 These compounds showed many biological activities like antibacterial, 5,6 anticancer, 7,8 antileishmanial 9 -11 ; antiviral, 12 -14 and others. 11,15 -19
Thus, in view of the high biological potential of the compounds of the Simaroubaceae, a phytochemical study of the roots of Homalolepis suffruticosa was carried out, during which a new unusual squalene triterpene, named milemaronol (
Results and Discussion
Elaboration of the MeOH and n-hexane extracts of the roots of H. suffruticosa through classical chromatographic methods resulted in the isolation of 11 compounds,

Compounds isolated from roots of Homalolepis suffruticosa (1-11) and teurilene (12).
Milemaronol (
The 13C-APT-NMR spectrum of
1H (500 MHz) and 13C (125 MHz) NMR Data of Compound 1, in CDCl3, Including HSQC and HMBC (2 J HC and 3 J HC). δ in ppm and J (in Parentheses) in Hz.

The location of these 2 C=CH bonds was established through analysis of the HMBC spectrum, which revealed cross-peaks of δ
C 131.3 (C-2) with 3H-1 (δ
H 1.69, 2
J
CH), 3H-25 (δ
H 1.64, 2
J
CH), and 2H-4 (δ
H 2.08, 3
J
CH) and δ
C 131.9 (C-23) with 3H-24 (δ
H 1.71, 2
J
CH), 3H-30 (δ
H 1.71, 2
J
CH), and 2H-21 (δ
H 2.03, 1.78, 3
J
CH). Additional HMBC couplings via 3
J
CH of CH-3 (δ
C 124.8) with 2H-5 (δ
H 2.08) and CH-22 (δ
C 124.5) with 2H-20 (δ
H 1.90, 1.75) confirmed the terminal units (Me2C=CH-CH2-CH2-5 and Me2C=CH-CH2-CH2-20). The tetrahydrofuran ring was observed based on the carbon shift of C-10 (δ
C 85.2) with cross-peak with H-7 (δ
H 3.90), 2H-9 (δ
H 2.25 and 1.48), H-11 (δ
H 3.71), and 3H-27 (δ
H 1.14). This allowed to recognize the identical unit from C-1 to C-10 of
In the region of the carbinolic carbons of
The location of this unit linked to C-11 (δ
C 75.1) was deduced by heteronuclear interaction of this nonprotonated carbon with 3H-27 (δ
H 1.14, 3
J
CH). The other terminal unit involving carbon atom C-19 was linked to CH-18 [δ
C 72.9/δ
H 3.69 (dd, J = 11.5, 3,9)] by correlation (3
J
CH) of this methinic carbon with 3H-29 (δ
H 1.31) and H-14 (δ
H 3.55). The HMBC correlation (3
J
CH) of CH-14 (δ
C 78.0) with 3H-28 (δ
H 1.20) and the 3H-28 with CH2-16 (δ
C 38.1) allowed us to postulate the presence of 2 pyran rings, 2-alkyl-octahydro-4a-methyl-6-alkylpyrano[3,2-b]pyran (
Finally, 2 tetrahydropyran rings formed by the ether bond CH-11/C-15 and CH-14/CH-18 attached to the tetrahydrofuran ring between C-10/CH-11 were established from the HMBC spectrum which exposed cross-peaks of δ
H 3.55 (H-14, dd, 7.6, 5.0) with CH-18 (δC
72.9) and CH2-12 (δC
20.3); δ
H 1.20 (3H-28, s) with CH-14 (δC
78.0), C-15 (δC
75.9), and CH2-16 (δC
38.1); δ
H 2.03 (1Ha-12) and 1.80 (1Hb-12) with CH2-13 (δC
27.2); and δ
H 3.71 (H-11, dd, 12.1, 1.9) with CH-9 (δ
C 31.2) and C-10 (δ
C 85.2). The complete assignment of the 1H and 13C signals of
The relative stereochemistry of

Important 1H-1H-NOESY correlations for milemaronol (1).
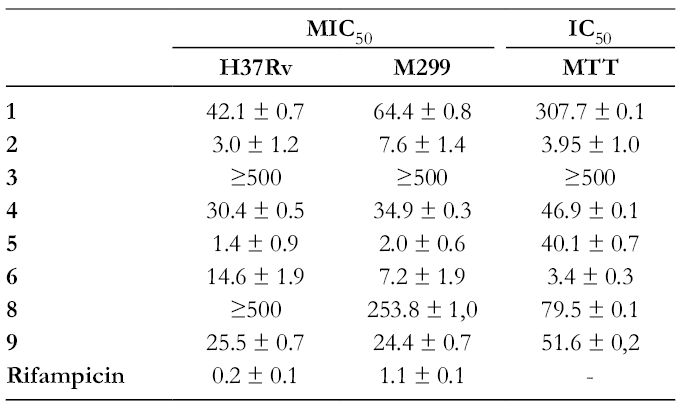
The antimycobacterial activity against Mycobacterium tuberculosis strains H37Rv and M299, and cytotoxic activities of compounds
Antimycobacterial Activities of Compounds 1
Material and Methods
General Experimental Procedures
Column chromatography (CC) was performed on silica gel 60 (0.063-0.200 mm, Merck), and n-hexane (98.5%), methanol (99.8%), ethyl acetate (99.5%), and dichloromethane (99.5%) were used as mobile phase solvents, purchased from Synth (São Paulo, Brazil). 1D and 2D NMR analysis was performed on a 500 MHz Bruker Ascend 500 NMR spectrometer operating at 500 MHz for 1H NMR and 125 MHz for 13C NMR. Deuterated chloroform (CDCl3), tetradeuterated methanol (CD3OD), and pyridine (pyridine-d 5), containing TMS (tetramethylsilane) as an internal standard, were used. HR-ESI-MS were obtained on a micrOTOF-Q II Bruker Daltonics mass spectrometer, with the use of the positive ion mode of analysis.
Plant Material
Roots of H. suffruticosa were collected in September 2017, in Araguari City, Minas Gerais. The species was identified by the taxonomist José Rubens Pirani from the Universidade de São Paulo (USP). A voucher specimen (HUENF-10840) was deposited in the herbarium of the Universidade Estadual do Norte Fluminense Darcy Ribeiro (UENF).
Extraction and Isolation
Homalolepis suffruticosa roots were dried and powdered. The extraction was performed first with n-hexane (CH, 20.5 g) and then with methanol (CM, 23.4 g). The methanolic extract (23.4 g) was fractionated by silica gel CC, with a polar gradient of CH2Cl2:MeOH, obtaining 9 fractions (CM1-CM9). CM4 was similarly rechromatographed, generating 10 fractions (CM4.1-CM4.10). Chaparrinone (
The hexane extract (CH, 20.5 g) was fractionated by silica gel CC with a gradient of hexane:acetone to yield 14 fractions (CH1-CH14). The CH5 fraction was similarly rechromatographed, yielding 9 fractions (CH5.1-CH5.9). Nilocitine (
Culture of Mycobacteria and Evaluation of Bacterial Growth
Two strains of Mycobacterium tuberculosis were used in this study (a virulent laboratory strain H37Rv, ATCC 27294 and a highly virulent Mtb strain Beijing M299, isolated from a TB patient in Mozambique), which were evaluated for virulence in a previous study. 28 Middlebrook 7H9 broth, containing 10% dextrose albumin complex (ADC), 0.5% glycerol, and 0.05% Tween-80, was used for the growth of the mycobacterial strains at 37°C, under conditions of containment of Biosafety 3. The MTT (3-[4, 5-dimethylthiazol-2-yl]-2, 5 diphenyl tetrazolium bromide) assay was performed to quantify bacterial growth, 29 using the procedures described by Ventura et al. 30 The samples were analyzed by optical density at 570 nm. For negative control, untreated bacterial suspensions were used, while for positive control rifampicin was applied.
Evaluation of Cytotoxicity by MTT Assay
RAW 264.7 macrophages were treated with the compounds at concentrations of 4, 20, 100, and 500 µg/mL. After 24 hours, the levels of cytotoxicity of the samples were assessed using mitochondrial functionality using the MTT method and compared with negative (macrophages stimulated by lipopolysaccharide - LPS) and positive (macrophages stimulated by LPS and treated with 1% Triton X-100) controls. Values are reported as mean ± standard deviation, and different groups were considered significant according to P < .001 (***), P < .01 (**), and P < .05 (*).
Supplemental Material
Supplementary Material 1 - Supplemental material for Antimycobacterial Activity of Milemarinol, a New Squalene-Type Triterpene, and Other Isolate?
Supplemental material, Supplementary Material 1, for Antimycobacterial Activity of Milemarinol, a New Squalene-Type Triterpene, and Other Isolate? by Samyra I. S. Boeno, Michel de S. Passos, Mariana Félix, Sanderson D. Calixto, Almir R. C. Júnior, Lara F. Barbosa Siqueira, Michelle F. Muzitano, Raimundo Braz-Filho and Ivo J. Curcino Vieira in Natural Product Communications
Footnotes
Acknowledgment
The authors are grateful to Fundação de Amparo à Pesquisado Estado do Rio de Janeiro, to CNPq, and to CAPES (Finance Code 001).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
