Abstract
Melanoma is the most lethal form of various skin cancers and contributes to more than 79% of all skin cancer deaths. Although there are numerous therapies available for melanoma, the high rate of recurrence in melanoma post-therapy remains a challenging issue for both patients and clinicians. Apoptosis is one of the foundations for cancer treatment as deficient apoptosis is one of the most essential reasons for the formation of tumour tissues. Shikonin (SHI), an active component extracted from
Skin cancer is one of the most prevalent types of cancer worldwide, with an estimate of more than 5 million new cases in 2019 alone (American Cancer Society 2019). Among the many forms of skin cancer, melanoma is most lethal. Despite accounting for about 4% of all skin cancer cases, invasive melanoma contributes to more than 79% of all skin cancer deaths. 1 The cost of treating skin cancer differs according to the stage of the disease as well as the country in which the treatment is received. 2 The average cost of treating melanoma is estimated at USD 3.3 billion annually in the United States. 3 Although there exist numerous viable strategies for the treatment and management of melanoma, the high rate of recurrence in melanoma post-therapy remains a challenging aspect in its treatment. 4
The selective induction of apoptosis, also known as programmed cell death, provides one of the foundations for the treatment of cancer. Dysregulation in one or more of the numerous and complex apoptotic signaling pathways is frequently evident in all forms of cancer. These defects not only suppress apoptosis in cancer cells but can also reduce their susceptibility to apoptotic signals.
5,6
Fortunately, induction of apoptosis can be achieved through multiple approaches and, therefore, is a target for the development of therapeutic strategies. In Traditional Chinese Medicine,
We have previously reported that SHI and related trigger apoptosis in hypertrophic scar-derived human skin fibroblasts (HSF). Our analysis of 8 SHI analogs identified that, in common with SHI, DO-SHI and β,β-SHI also possess viability-inhibiting ability against HSF.
14
Our review of the literature found that the effects of DO-SHI and β,β-SHI on melanoma related cells are largely unknown. Rajasekar et al
15
reported that the extract from the roots of
In this study, we aimed to determine the sensitivity of different melanoma cell lines to DO-SHI and β,β-SHI and compare responses with SHI. Our objective was to acquire a better understanding of the bioactivity of DO-SHI and β,β-SHI for potential applications as adjuvant therapies. We report that SHI, DO-SHI, and β,β-SHI effectively attenuate cell viability, inhibit cell migration, and stimulate apoptosis in human and mouse melanoma. Taken together, these data offer evidence supporting the application of SHI, DO-SHI, and β,β-SHI as novel effective treatments for melanoma.
Materials and Methods
Preparation of Shikonin Analogs
Shikonin (Sigma-Aldrich, S7576), DO-SHI (Tokyo Chemical Industry Co., Ltd., D2117), and β,β-SHI (ALB Technology Limited, RS-0026) were dissolved in dimethyl-sulfoxide (DMSO; Sigma-Aldrich, D2650) as a stock solution (10 mM) and stored away from light at −20°C until use. The chemical structures of these 3 molecules are shown in Figure 1.

Chemical structures of shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin.
Cell Culture
Human amelanotic malignant melanoma cell line A375 (CRL-1619), mouse nonmetastatic melanoma cell line B16-F0 (CRL-6322), and mouse metastatic melanoma cell line B16-F10 (CRL-6475) were purchased from American Type Culture Collection (ATCC). Ethical approval was obtained from National University of Singapore. IRB reference: B-16-135E. All cell lines were mycoplasma negative. Cells were cultured in Dulbecco’s modified Eagle’s medium containing 4.5 g/L
Cell Viability Assay
The effect of SHI, DO-SHI, and β,β-SHI on the viability of various melanoma cell lines was determined using Alamar blue (Sigma-Aldrich, R7017) assay. 20 Melanoma cells were seeded (4 × 104 cells/well) into 48-well plates and incubated for 24 hours. Culture media were then removed and replaced with DMEM containing SHI, DO-SHI, or β,β-SHI at 1, 3, and 10 µM. DMEM containing DMSO (0.01%, 0.03%, and 0.1% v/v) was also included as vehicle controls. Cells treated with fresh media were used as a positive control. After an incubation period of 48 and 72 hours, culture media from each well were aspirated followed by the addition of 300 µL of Alamar blue (6 µM). Assay plates were then incubated at 37°C for 1 hour. The fluorescence readings of each sample were measured at λex 560 nm and λem 590 nm using a SpectraMax M5 Multi-Mode microplate reader (Molecular Devices, M5).
Cell Migration Assay
Melanoma cell lines were seeded (8 × 104 cells/well) into 24-well plates and incubated for 24 hours. Culture media were replaced with fresh media containing SHI, DO-SHI, or β,β-SHI (3 µM) and DMSO (0.03% v/v). A scratch wound was made in each well using a pipette tip following a previously published protocol 21 ; images were captured using an EVOS FL Color Imaging System (Thermo Scientific, AMEFC4300) at 0, 12, 24, and 48 hours after the scratch was made.
Terminal Deoxynucleotidyl Transferase dUTP Nick End Labeling (TUNEL) Assay
Apoptosis was detected using In Situ Cell Death Fluorescent Label Solution (Roche, 11684795910) and In Situ Cell Death Fluorescent Enzyme Solution (Roche, 11684795910). The cell nucleus was stained with Prolong Diamond Antifade Mountant with 4’,6-diamidino-2-phenylindole (DAPI; Invitrogen, P36962). Briefly, cells were seeded (4 × 104 cells/well) into 24-well plates. After an incubation period of 24 hours, cells were treated with fresh medium containing SHI and SHI analogs (3 µM) and DMSO (0.03% v/v) for an additional 24 hours. Two groups of cells were left untreated as controls, and media replenished. Media were carefully aspirated and cells were fixed using 500 µL of 10% formalin (Sigma-Aldrich, HT501128) for 24 hours. The fixed cells were washed twice with Dulbecco’s phosphate buffered saline (DPBS; BioWhittaker, 17-512F) before being subjected to 300 µL of Triton X (0.1% v/v; Promega, H5142) in DPBS. Amplification Grade DNase I (Sigma Aldrich, D5307-1000) was applied to an untreated group for 5 minutes serving as the positive control. Samples were then processed for TUNEL and counterstained with DAPI; images were captured using EVOS FL color imaging system.
Fluorescence-Activated Cell Sorting (FACS)
Melanoma cell lines were seeded (6 × 104 cells/well) into 24-well plates and incubated for 24 hours. Culture media were replaced with fresh medium containing SHI and SHI analogs (3 µM) and DMSO (0.03% v/v). Cells were incubated for 24 hours and culture media were collected. Adherent cells were harvested by dissociation using TrypLE Express (Gibco, 12604013), and harvested by centrifugation with collected culture media at 1500 rpm for 5 minutes. Samples were stained with Annexin V and propidium iodide (PI) (Annexin V-FITC Apoptosis Staining/Detection Kit, Abcam, ab14085) according to the manufacturer’s instructions. Data acquisition and analysis were performed on a BD LSR II flow cytometer. Samples were measured at λex 488 nm, λem 530 nm for annexin V and λex 493 nm, λem 636 nm for PI. A total of 10 000 events were collected per sample and analyzed using BD FACSDiva Software.
Western Blot
Melanoma cell lines were seeded (8 × 104 cells/well) into 12-well plates (Thermo Scientific, 150628) and incubated for 24 hours. Cells were then treated with FBS-free culture media containing SHI and SHI analogs (3 µM) and DMSO (0.03% v/v) for another 24 hours. Whole cell lysates were collected in lysis buffer (40 µL) containing Radio immuno precipitation assay buffer (RIPA; Merck, R0278), cOmplete Protease Inhibitor Cocktail (PIC; Sigma-Aldrich, 11697498001), sodium fluoride (NaF, 10 mM; Sigma-Aldrich, 450022), and sodium vanadate (Na3VO4, 2 mM; Sigma-Aldrich, 450243). Protein quantification was performed using Bradford Protein Assay (Bio-Rad, 500-0006). Equal amounts of protein (10 µg) in each group were prepared and separated using 12% sodium dodecyl sulfate polyacryl-amide gel electrophoresis (SDS-PAGE) and then transferred onto nitrocellulose membranes. The membranes were then incubated in Odyssey blocking buffer (LI-COR Biosciences, United States) containing primary antibodies at 4°C overnight. Primary antibodies included GAPDH (Sigma Aldrich, G8795), extracellular-signal-regulated kinases 1 and 2 (ERK1/2) (Cell Signaling Technology, 9102S), phosphorylated-ERK1/2 (p-ERK1/2) (Cell Signaling Technology, 9106S), caspase 3 (Cell Signaling Technology, 9662S), p38α/β (Santa Cruz Biotechnology, sc-7972), p-p38α/β (Santa Cruz Biotechnology, sc-17852-R), calpain-1 (abcam, ab28258), and p53 (abcam, ab28). Secondary antibodies, including antimouse IgG conjugated with Alexa Fluor 790 (Invitrogen, A11371) and antirabbit IgG conjugated with Alexa Fluor 680 (Invitrogen, A-21076), were then applied as species appropriate. Images were captured and analyzed using the Odyssey infrared imaging system.
Statistical Analysis
Data are expressed as mean ± SEM, pooled from 3 independent experiments for each study parameter. Graphs were made using GraphPad Prism 6.01. Statistical analysis was performed using one-way ANOVA, Tukey’s post hoc test, and Dunnett’s test.
Results
Shikonin, DO-SHI, and β,β-SHI Decrease Cell Viability in a Dose-Dependent Manner
To determine the effect of SHI, DO-SHI, and β,β-SHI on human and mouse melanoma cell lines, they were exposed to various concentrations of these compounds (1, 3, and 10 µM). A375 exposed to SHI (3 and 10 µM), DO-SHI (1, 3, and 10 µM), and β,β-SHI (1, 3, and 10 µM) were observed to have significantly reduced cell viability after 48 and 72 hours compared with the control (

Shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin decrease cell viability in melanoma cell lines in a dose-dependent manner. (a) A375, (b) B16-F0, and (c) B16-F10. Cell viability was measured using Alamar blue assay. Data are expressed as the average percentage of the control and are pooled from 3 replicate experiments. Error bars indicate the mean ± SEM (
We determined the half-maximal inhibitory concentration (IC50, Table 1) for SHI, DO-SHI, and β,β-SHI from the Alamar blue assay data to compare the efficiency of SHI analogs on melanoma cell viability. Deoxyshikonin and β,β-SHI were found to be more potent than SHI in A375 melanoma cells. Calculated IC50 values for SHI were 4.59 ± 0.79 and 3.13 ± 0.46 µM for 48- and 72-hour treatment, respectively, whereas those for DO-SHI were 2.39 ± 0.33 and 1.81 ± 0.41 µM at 48 and 72 hours, respectively. (β,β-Dimethylacryl)shikonin was determined to be the most potent of 3 SHI analogs tested, yielding IC50 values of 1.34 ± 0.05 and 1.16 ± 0.21 µM for 48- and 72-hour treatment, respectively. The effect of all 3 treatments was markedly reduced in mouse melanoma B16-F0 and B16-F10 cells compared with human melanoma A375. Taken together, these data suggest that SHI, DO-SHI, and β,β-SHI affect cell viability of A375, B16-F0, and B16-F10 cells in a dose-dependent manner. For reasons that are not yet clear, human melanoma cells are more sensitive to the effects of DO-SHI and β,β-SHI than SHI.
IC50 of Shikonin, Deoxyshikonin, and (β,β-Dimethylacryl)Shikonin on Melanoma Cell Lines.
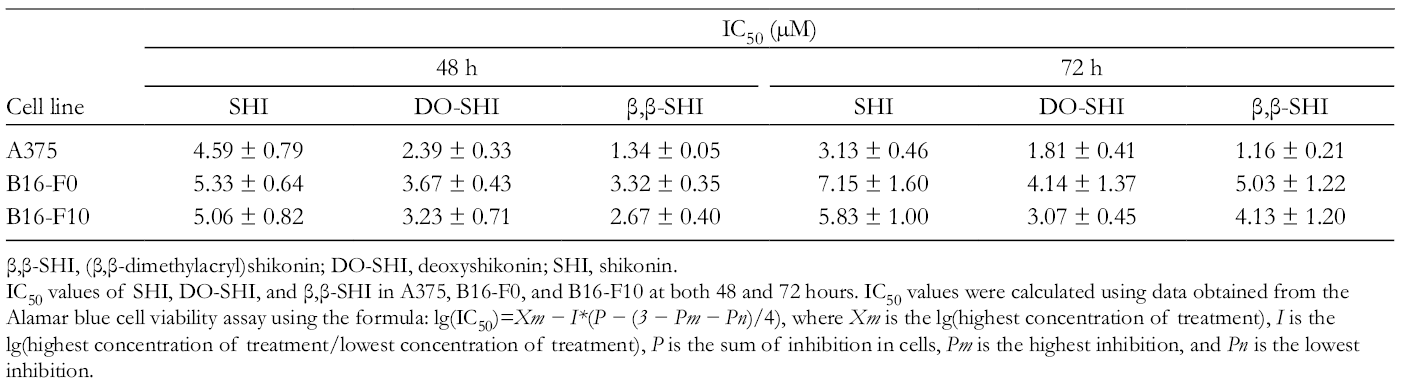
β,β-SHI, (β,β-dimethylacryl)shikonin; DO-SHI, deoxyshikonin; SHI, shikonin.
IC50 values of SHI, DO-SHI, and β,β-SHI in A375, B16-F0, and B16-F10 at both 48 and 72 hours. IC50 values were calculated using data obtained from the Alamar blue cell viability assay using the formula: lg(IC50)
Shikonin, DO-SHI, and β,β-SHI Inhibit Cell Migration
To investigate the effects of SHI, DO-SHI, and β,β-SHI on cell motility and migration, melanoma cell lines were treated with 3 µM of SHI, DO-SHI, and β,β-SHI, as well as 0.03% v/v of DMSO. As seen in Figure 3, roughly 50% of the scratched area was healed after 12 hours and more than 90% of the original wound was healed after 24 hours in the untreated control and DMSO groups in A375 and B16-F10. B16-F0 was observed to heal more slowly than A375 and B16-F10, but more than 60% of the scratched area was healed after 24 hours in the untreated control and DMSO groups. Compared with the controls, SHI, DO-SHI, and β,β-SHI almost entirely inhibited cell migration in A375 (Figure 3(a)), B16-F0 (Figure 3(b)), and B16-F10 (Figure 3(c)) cells. In particular, cell density was notably reduced in cells exposed to DO-SHI and β,β-SHI. These results demonstrate that melanoma cell migration is particularly sensitive to SHI, DO-SHI, and β,β-SHI.

Effects of shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin on cell migration in (a) A375, (b) B16-F0, and (c) B16-F10 after 12 and 24 hours. Yellow dashed lines indicate original borders made by scratch. Representative images were obtained from 3 replicate experiments. Scale bar = 0.5 mm.
Identifying SHI, DO-SHI and β,β-SHI-Induced Apoptosis Using TUNEL Assay
In order to determine if the reduced cell viability and migration that we observed is caused by increased apoptosis, we performed TUNEL assay on A375, B16-F0, and B16-F10 after exposure to SHI, DO-SHI, and β,β-SHI. As illustrated in Figure 4(a-c), green (apoptotic) cells were observed in all populations after treatment. Quantitative analysis (Figure 4(d)) was performed to determine the incidence of apoptosis stimulated by the treatment. In A375 melanoma cells, SHI induced 7.0% ± 2.9% of cells to undergo apoptosis, whereas the values for DO-SHI and β,β-SHI were 30.4% ± 3.0% and 33.1% ± 7.8%, respectively. Shikonin, DO-SHI, and β,β-SHI induced apoptosis in 2.7% ± 0.3%, 4.0% ± 0.8%, and 4.1 %± 1.0% in B16-F0, respectively (Figure 4(d)), whereas the value for SHI in B16-F10 cells was 0.8% ± 0.4% and for DO-SHI and β,β-SHI 4.1% ± 0.6% and 11.0% ± 1.3%, respectively (Figure 4(d)); a significant increase in the incidence of apoptosis in B16-F10 exposed to β,β-SHI compared with cells exposed to either SHI or DO-SHI (
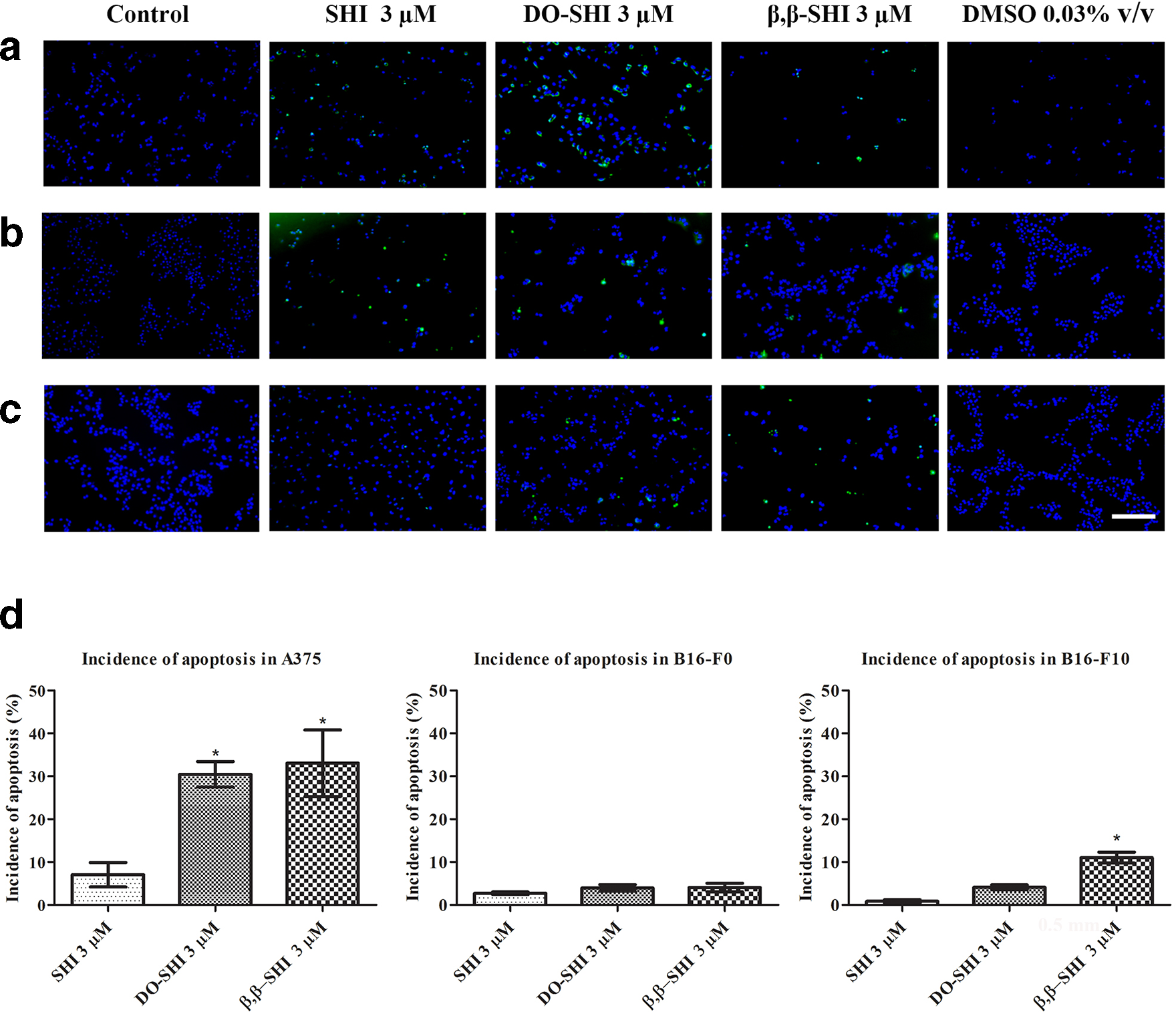
Shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin induce apoptosis in A375, B16-F0, and B16-F10. Representative images of (a) A375, (b) B16-F0, and (c) B16-F10 were selected. Images were captured using an EVOS FL color imaging system. Green indicates DNA fragmentation in apoptotic cells, while blue indicates the nuclei of both apoptotic and nonapoptotic cells. Scale bar = 0.2 mm. (d) Incidence of apoptotic cells treated with shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin was obtained by dividing the number of apoptotic cells over all cells. The data are presented as percentage of apoptotic cells from 3 replicate experiments. Error bars indicate SEM (
Shikonin, DO-SHI, and β,β-SHI-Induced Apoptosis Detected by FACS
We confirmed the ability of SHI, DO-SHI, and β,β-SHI to trigger apoptosis using flow cytometry with Annexin V and PI. 22 As shown in Figure 5(a), top left (Q1 zone), early apoptotic cells are indicated, while top right (Q2 zone) suggests late apoptotic cells. Nonapoptotic cells are separated into the bottom left (Q3 zone) containing live cells. The bottom right (Q4 zone) indicates necroptotic and damaged cells. In A375 and B16-F0 cell lines, large population of late apoptotic cells (Q2 zone) were observed after exposure to DO-SHI and β,β-SHI compared with the control, SHI, and DMSO groups. A375 populations exposed to SHI also exhibited a slightly larger late apoptotic cell subpopulation compared with the control and DMSO. Interestingly, no significant difference in the population of late apoptotic cells was detected in B16-F10 after exposure to all 3 treatments compared with the control and DMSO.
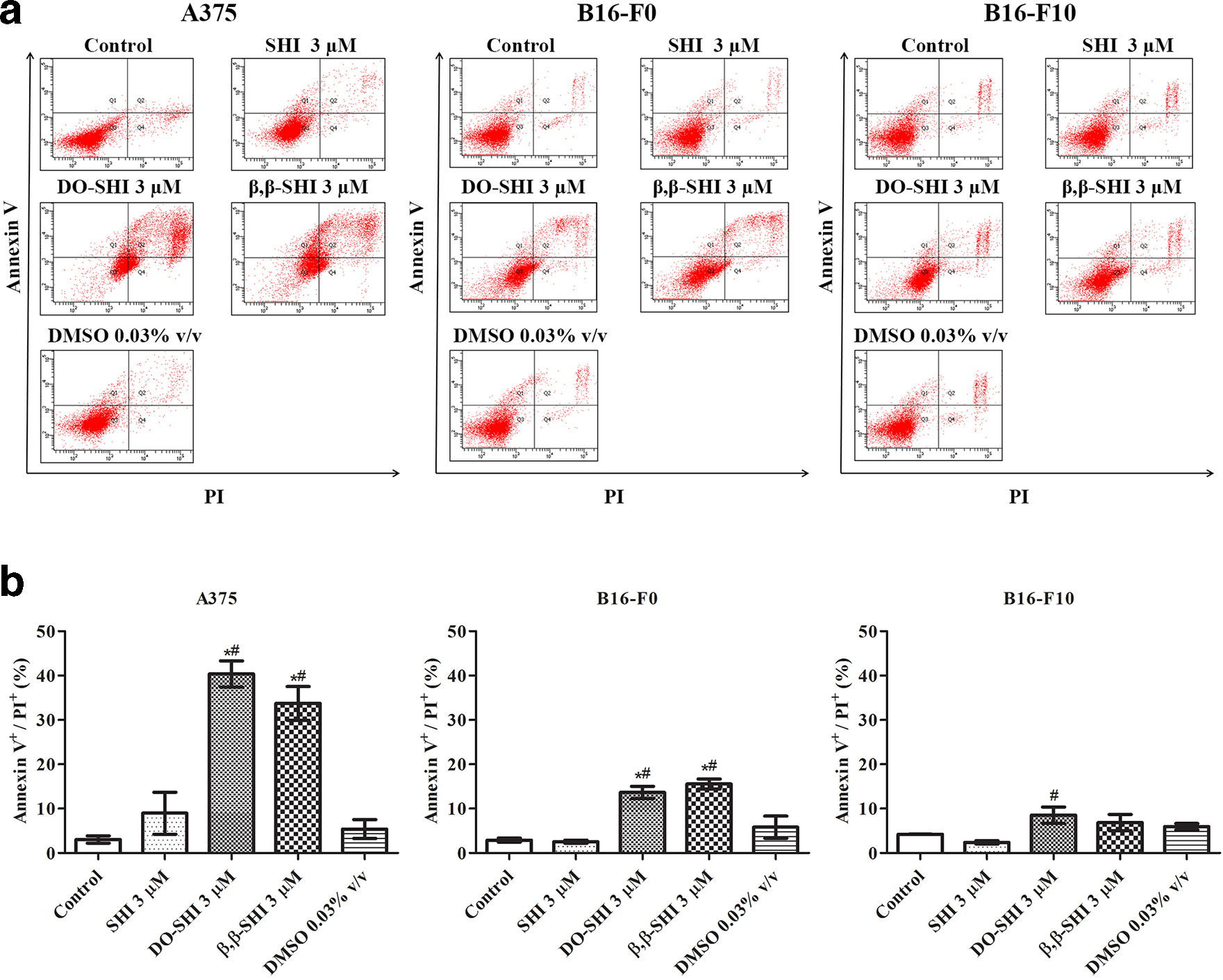
Fluorescence-activated cell sorting analysis of apoptotic cells. (a) Fluorescence-activated cell sorting analysis of Annexin V and propidium iodide stained cells following treatment with shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin for 24 hours. (b) Quantitative analysis of Annexin V+/PI+ double positive cells. Data are expressed as the average percentage of the control and are pooled from 3 replicate experiments. Error bars indicate the SEM (
We performed quantitative analysis (Figure 5(b)) using the population hierarchy data obtained from FACS. In all cell lines tested, SHI failed to increase apoptotic signals compared with the control group. However, DO-SHI and β,β-SHI achieved a significant increase of Annexin V+/PI+ double positive signals (late apoptosis) in A375 and B16-F0 compared with the control, SHI, and DMSO groups. Analysis of A375 cells treated with DO-SHI showed a double positive signal incidence of 40.4% ± 3.0%, while β,β-SHI treated A375 cells had a value of 33.7% ± 3.8% (
Shikonin, DO-SHI, and β,β-SHI Triggered Signaling Pathways
To explore the underlying mechanisms of SHI, DO-SHI, and β,β-SHI-induced apoptosis in melanoma cells, we used Western blot analysis to examine the expression of caspase 3, ERK, p-ERK, p38, p-p38, p53, and calpain-1 (Figure 6). Cleaved caspase 3, an indicator for apoptosis, was observed in A375 (Figure 6(a)) after exposure to SHI, DO-SHI, and β,β-SHI. ERK and p38 are 2 key proteins in the mitogen-activated protein kinase (MAPK) pathway, with essential roles in cell proliferation and apoptosis. 23,24 As shown in Figure 6(a), total levels of ERK and p38 were not affected in A375 after treatment; however, exposure to SHI, DO-SHI, and β,β-SHI significantly upregulated the expression of phosphorylated-ERK and p38 (p-ERK and p-p38) in A375. The expression of p53 was found to be reduced in A375 after treatment with SHI, DO-SHI, and β,β-SHI. The expression of calpain-1 was also observed to be attenuated in A375 after treatment with SHI, DO-SHI, and β,β-SHI.

Signaling pathways triggered by shikonin, deoxyshikonin, and (β,β-dimethylacryl)shikonin in (a) A375, (b) B16-F0, and (c) B16-F10. The expression of proteins was detected using the LI-COR Odyssey Fc imaging system. GAPDH was included as a loading control. Representative images of Western blots are presented. Quantitative analysis was performed using ImageJ software. Protein bands were first normalized to GAPDH and then converted to a percentage of the untreated control. Data were pooled from 3 replicate experiments. Error bars indicate SEM (
Interestingly, while the total level of caspase 3 detected in B16-F0 was found to be reduced after treatment with SHI, DO-SHI, and β,β-SHI, cleavage of caspase 3 was not detected (Figure 6(b)). In addition, while these 3 molecules were found to inhibit the expression of p53 and calpain-1 in B16-F0, no effect was detected on the expression of p-ERK and p-p38 (Figure 6(b)). In B16-F10 (Figure 6(c)), expression of caspase 3, p-ERK, p-p38, and calpain-1 was undetectable, and no effect of SHI, DO-SHI, or β,β-SHI was observed on the expression of p53 in B16-F10. These data suggest that SHI, DO-SHI, and β,β-SHI induce apoptosis in A375 via caspase 3 and MAPK signaling pathways. p53 and calpain-1 may also contribute to the induced apoptosis in A375. Similar mechanisms were observed in B16-F0 after treatment with SHI, DO-SHI, and β,β-SHI, without the activation of MAPK families. Notably, SHI, DO-SHI, and β,β-SHI may trigger cell death in B16-F10 via an alternative pathway.
Discussion
Effective clinical management of melanoma is often unsuccessful due to the high incidence of recurrence, especially among patients presenting with advanced stage melanoma. 25 The high incidence of metastasis associated with melanoma is most often seen in advanced stage melanoma. 4 Current treatments for melanoma include surgical excision, chemotherapy, radiotherapy, and innovative therapies such as BRAF and MEK inhibitors. 26,27 However, the development of resistance and the destructive side effects of current treatments are still the primary challenges of clinical management.
Shikonin is a pigment found in various plants such as
Based on the limited understanding of DO-SHI and β,β-SHI and the promising data from our recent study, we believe that SHI, DO-SHI, and β,β-SHI offer potential novel interventions for melanoma. Data from the Alamar blue assay indicate that SHI, DO-SHI, and β,β-SHI inhibit A375, B16-F0, and B16-F10 cell viability in a dose-dependent manner. IC50 values were calculated to analyze the efficiency of these species on melanoma cells. The data indicate that DO-SHI and β,β-SHI have significantly lower IC50 values compared with SHI, a finding consistent with our previous HSF study. Interestingly, DO-SHI is reported to have an IC50 more than double that of SHI, when used on cancer cells overexpressing drug-resistant genes, including P-gp, MRP1, and BCRP1, 36 suggesting that cancer with different origins responses distinct to SHI and SHI analogs.
Metastasis is the major cause of death in cancer patients. 37 Metastatic melanoma (late stage melanoma) is responsible for significantly more deaths in patients compared with nonmetastatic melanoma (early stage melanoma). 38 We observed similar capacity in the scratch assay, finding that metastatic A375 and B16-F10 migrate faster than nonmetastatic B16-F0 after the scratch-wounding. Our study is the first to report that SHI inhibits the migration of A375, B16-F0, and B16-F10 cell lines. These data offer further evidence supporting the potential use of DO-SHI and β,β-SHI as novel melanoma therapies.
The ability of SHI, DO-SHI, and β,β-SHI to induce apoptosis in human and mouse melanoma cell lines was confirmed with the data from TUNEL and FACS assays. Western blot was further used to identify the underlying pathways. Our results indicate that SHI, DO-SHI, and β,β-SHI induce the cleavage of caspase 3 in A375, accompanied by a significant reduction of total caspase 3 expression. These findings are consistent with the previously published studies. Evidence shows that caspase 3 activation is observed in SHI-induced apoptosis of human gastric cancer cells, 32 human prostate cancer cells, 31 and human breast cancer cells. 34 (β,β-Dimethylacryl)shikonin has been reported to induce apoptosis via a caspase 3-dependant pathway in human lung 39 and melanoma cells. 17 Interestingly, although a reduction of total caspase 3 was detected in B16-F0 after exposed to SHI, DO-SHI, and β,β-SHI, we did not detect cleaved caspase 3 in either B16-F0 or B16-F10. These data suggest that in contrast to human melanoma, SHI, DO-SHI, and β,β-SHI might trigger cell apoptosis in mouse melanoma cells in a caspase-independent manner.
Proteins in the MAPKs family, including ERK, JNK, and p38, are widely demonstrated to play essential roles in cell proliferation and apoptosis.
40
For example, it has been demonstrated that activation of ERK directly decreases mitochondrial respiration and membrane potential which leads to the release of cytochrome
One potential candidate is calpain-1, a calcium-dependent protease, demonstrated to induce apoptosis by triggering ER stress. 45 ER stress affects protein folding, resulting in the accumulation of unfolded proteins in ER. 46 Previous studies showed that SHI induces apoptosis in prostate cancer cell lines by activating ER stress and calpain activity. 47 Evidence also indicates that inhibition of ER stress enhanced the antitumor effect of SHI on human glioblastoma stem cells. 48 Conversely, our study suggests that the expression of calpain-1 was decreased in A375 and B16-F0 cell lines after treatment with SHI, DO-SHI, and β,β-SHI. It has been demonstrated that calpain is not necessarily associated with apoptosis, therefore the reduction of calpain-1 induced by SHI, DO-SHI, and β,β-SHI may contribute to other signaling pathways such as migration, differentiation, and cell cycle regulation. 49
As the incidence of melanoma rises, the unmet demand for effective and alternative treatment options increases. The effectiveness of treatment, patient care, invasiveness, and adverse effects are the aspects that compromise the effectiveness of current treatment options. Data generated from this study confirm that SHI, DO-SHI, and β,β-SHI inhibit cell viability, reduce cell migration, and induce apoptosis in A375, B16-F0, and B16-F10 melanoma cell lines. Deoxyshikonin and β,β-SHI were demonstrated to be more potent than the parent molecule SHI, inducing apoptosis at lower doses than SHI. In particular, β,β-SHI may be most promising among these 3 compounds exhibiting the lowest IC50 value (Table 1) against human A375 cell lines. Further validation during reformulation studies and in vivo toxicity testing will provide evidence supporting further development as a therapeutic. Taken together, data generated from this study support the application of SHI, DO-SHI, and β,β-SHI as novel alternative treatments for melanoma.
Footnotes
Acknowledgment
The authors wish to acknowledge assistance from Ms Priscilla Lim for the laboratory support; Dr Cindy Coffill for providing the A375 cell line; and Ms Alice Quek for assistance with flow cytometry.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funding from Singapore’s Agency for Science, Technology and Research “SPF 2013/004: Skin Biology Basic Research” and the “Wound Care Innovation for the Tropics” IAF-PP/2017 (HBMS) H17/01/a0/009.
