Abstract
The aim of this research was to investigate the effects of nutraceuticals including bitter melon, fenugreek, cinnamon, alpha-lipoic acid, zinc, biotin, chromium, and cholecalciferol on glycemic control, insulin sensitivity, lipid profiles, oxidative stress, and inflammatory markers in hyperglycemia. The study design was a randomized, double-blind, placebo-controlled trial. Subjects with hyperglycemia were randomly divided into 2 groups. The treatment group (n = 52) was given a nutraceutical and the control group (n = 50) was provided with a placebo for 12 weeks. Fasting blood glucose (FBG), hemoglobin A1c (HbA1C), homeostatic model assessment of insulin resistance (HOMA-IR), lipid profiles, biomarkers of oxidative stress, and inflammation were assessed before and after the intervention at 6 weeks and 12 weeks. Nutraceutical supplementation demonstrated a statistically significant decrease in FBG (13.4% and 18.9%), HbA1C (6.5% and 11.3%), and HOMA-IR (28.9% and 35.2%) compared with the placebo. Moreover, low-density lipoprotein-cholesterol (LDL-C) level was significantly reduced in the nutraceutical group (7.1% and 9.3%). Furthermore, the nutraceutical significantly decreased oxidative stress markers, oxidized LDL-C (14.8% and 18.9%) and malondialdehyde (16.6% and 26.2%) compared with the placebo. In conclusion, this nutraceutical can improve glycemic control, insulin resistance, lipid profiles, and oxidative stress markers in hyperglycemic subjects. Therefore, it has the potential to decrease cardiovascular disease risk factors. Clinical trial registration: TCTR20180907001, www.clinicaltrials.in.th.
Keywords
The IDF Diabetes Atlas reported that the global diabetes prevalence in 2019 is estimated to be 9.3% (463 million people), rising to 10.2% (578 million) by 2030 and 10.9% (700 million) by 2045. 1 The disease is accompanied by high costs mainly due to chronic complications. Diabetes and its complications have many negative effects on health. Cardiovascular disease (CVD) is the most common. Glycemic control is important for preventing diabetes and/or delaying its complications. 2 Oxidative stress and chronic inflammation are the risk factors of CVD, especially in hyperglycemic subjects. 3
The easiest way to prevent hyperglycemia is to control blood glucose levels in the normal range. Management concentrates on keeping blood sugar levels as close to normal as possible, without causing hypoglycemia. This can usually be accomplished with diet, exercise, and the use of appropriate medications. Nevertheless, the control of diabetes complication is not only possible with conventional medicine but also with alternative medicine. Nutraceuticals are promising alternative medicines that have been used for regulation of plasma glucose to prevent diabetes, in order to reduce the cost of treating diabetes and its complications. They also provide a treatment option for people who may have adverse reactions to conventional antidiabetic medications. 4 -7
Bitter melon is a tropical fruit. It has been purported to have an antihyperglycemic effect, which may serve as a treatment option for managing diabetes mellitus. 8 Fenugreek is a rich source of fiber, which has multiple benefits in patients with diabetes. Its role as an antidiabetic, by reducing fasting blood glucose levels and decreasing insulin resistance in human subjects, has been reported. 9 Cinnamon has become a natural supplement of interest because it has been provided health benefits, such as the ability to lower blood glucose and lipids. 10 Moreover, the consumption of cinnamon can reduce FBG and HbA1C among poorly controlled type 2 diabetes patients, as well as improve oxidative stress markers. 11
Alpha-lipoic acid (ALA) is a natural antioxidant. It is known to exert beneficial actions against oxidative stress caused by hyperglycemia. A recent study showed that it could help improve the glycemic status and decrease oxidative stress markers in patients with type 2 diabetes. 12 Furthermore, zinc (Zn) has the potential to affect glucose homeostasis in animals and humans and hence influence the risk of type 2 diabetes mellitus. In a meta-analysis of randomized controlled trials (RCTs), Zn supplementation exhibited a mean decrease in FBG, with a trend toward reductions in HbA1C as well. 13 Additionally, biotin has also been implicated in cellular glucose metabolism. It can stimulate beta-islet cell function and proportion in the pancreas, thereby driving the release of insulin. 14 Also, chromium (Cr) supplementation was widely adopted to assist in the management of blood glucose levels in prediabetic and diabetic individuals, in addition to supporting healthy blood lipid profiles. A previous study demonstrated that a combination of Cr and biotin could improve glycemic control by decreasing FBG and HbA1C in overweight and obese individuals with type 2 diabetes. 15 Moreover, cholecalciferol (vitamin D3) may modify the risk of type 2 diabetes mellitus. Vitamin D3 supplementation improved insulin resistance. Further beneficial effects of vitamin D3 have been observed in the form of improved inflammatory and antioxidant status, in relation to levels of respective serum protein biomarkers, as well as reduced glycation end products. 16
To date, a clinical study of a combination of bitter melon, fenugreek, cinnamon, ALA, Zn, biotin, Cr, and vitamin D3 (nutraceutical) in hyperglycemia has not been conducted. Therefore, the aim of this study was to examine the effects of a nutraceutical on glycemic control, insulin resistance, lipid profiles, oxidative stress, and inflammatory markers in hyperglycemic subjects.
Materials and Methods
Subjects and Study Design
One hundred and ten hyperglycemic subjects, aged 20-70 years, were included, and all had FBG >100 mg/dL and HbA1C >5.7%. The study was conducted at the Faculty of Public Health, Mahidol University, Thailand. Subjects were excluded if they were taking a hypoglycemic drug, had a history of chronic diseases (e.g., cardiovascular disease, liver disease, renal disease, and cancer), were using herbs or dietary supplements, smoked, or drank alcohol.
The study was approved by the Ethical Review Committee for Human Research, Faculty of Public Health, Mahidol University (Approval No. MUPH 2018-135). Furthermore, this study was conducted in accordance with the Declaration of Helsinki on human subjects. All participants were informed and gave their consent before enrolment (clinical trial registration: TCTR20180907001, www.clinicaltrials.in.th).
The study design was a randomized, double-blind, and placebo-controlled trial (Figure 1). Participants were randomly assigned to nutraceutical (Diabecure Max Biocare Pty Ltd, Victoria, Australia) (treatment group) or microcrystalline cellulose as placebo (control group). The composition of the nutraceutical is presented in Table 1. The random allocation sequence was provided by an independent consultant and was computer generated using a randomization plan from www.randomization.com with randomization in blocks of 10. A list of consecutive study numbers was generated. Treatment groups were allocated by a research assistant, but the allocation was concealed by assigning each participant with a unique number. Participants, principal investigators, and the research assistant were blinded to group allocation. Placebo tablets were manufactured to match the nutraceutical capsules in size, excipients, and appearance. The treatment group was asked to consume 2 nutraceuticals (500 mg) after breakfast and lunch daily for 12 weeks, during which time the subjects were asked to maintain their habitual diet and lifestyle. All tablets in both groups were similar in shape, size, and color. A research assistant carried out the anthropometric assessment. Dietary assessment was conducted by using a food record, and energy and nutrient intakes were calculated by using the INMUCAL-nutrient computer program (Institute of Nutrition, Mahidol University, Nakhonpathom, Thailand).

Study design and flow diagram.
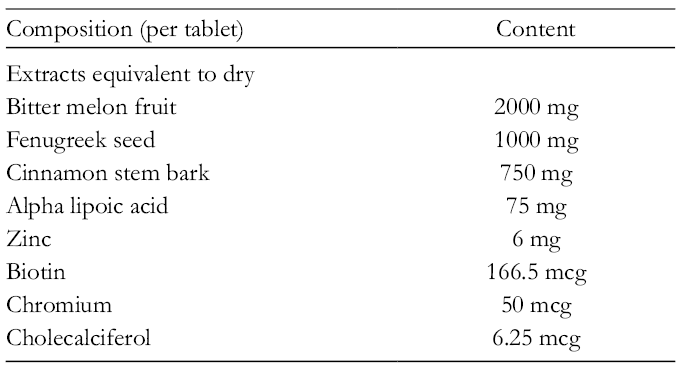
Composition of Nutraceutical.
The primary outcomes were FBG, HbA1C, and HOMA-IR. Secondary outcomes were lipid profiles (total cholesterol [TC], low-density lipoprotein-cholesterol [LDL-C], high-density lipoprotein-cholesterol [HDL-C], triglyceride [TG]), biomarkers of oxidative stress (LDL-C oxidation, malondialdehyde [MDA]), and an inflammatory marker (high-sensitivity C-reactive protein [hs-CRP]). All outcomes were assessed before and after the intervention at 6 weeks and 12 weeks.
Biochemical Analysis
Blood samples were collected by a registered nurse after an overnight fast at baseline and during and after the intervention (6 weeks and 12 weeks) for biochemical assessments. FBG was determined by the glucose oxidase method (Roche Diagnostics, Indianapolis, IN, USA). 17 HbA1c was measured by cation exchange high-pressure liquid chromatography (Bio-Rad Laboratories, Hercules, CA, USA). 18 Insulin was determined by an enzyme-linked immunosorbent assay (ELISA)-based assay kit (Roche Diagnostics, Indianapolis, IN, USA). 19 HOMA-IR (an indicator of insulin sensitivity) was calculated from fasting plasma insulin (µU/mL) multiplied by fasting plasma glucose (mmol/L)/22.5. 20 Lipid profiles were determined by an enzymatically colorimetric assay using an automated autoanalyzer (Cobas 8000, Roche Diagnostics, Mannheim, Germany). 21 Oxidized LDL-C was determined by an ELISA assay (Mercodia, Uppsala, Sweden). 22 MDA was measured by the thiobarbituric acid reactive substances assay (Oxford Biomedical Research, USA). 23 hs-CRP was determined by the latex immunoturbidimetry assay (QuikRead CRP, Orion Diagnostica Oy, Espoo, Finland). 24 All biochemical analyses were carried out at N-Health Asia Laboratory, Bangkok, Thailand, which is a medical laboratory with ISO15189:2007 certification.
Statistical Analysis
All data are presented as mean ± standard deviation (SD). An independent t-test was used to analyze the differences in weight, body mass index (BMI), % body fat, blood pressure, pulse rate, FBG, HbA1C, HOMA-IR, lipid profiles, blood urea nitrogen (BUN), Cr, aspartate aminotransferase, alanine aminotransferase, oxidative stress markers, an inflammatory marker, and total energy and nutrient intake between the treatment group and control group. A one-way repeated measures analysis of variance was used to analyze laboratory results within the group. All statistical data were analyzed by using SPSS version 18 (IBM, New York, NY, USA) for Windows. Statistical significance was considered to be P value <0.05.
Results
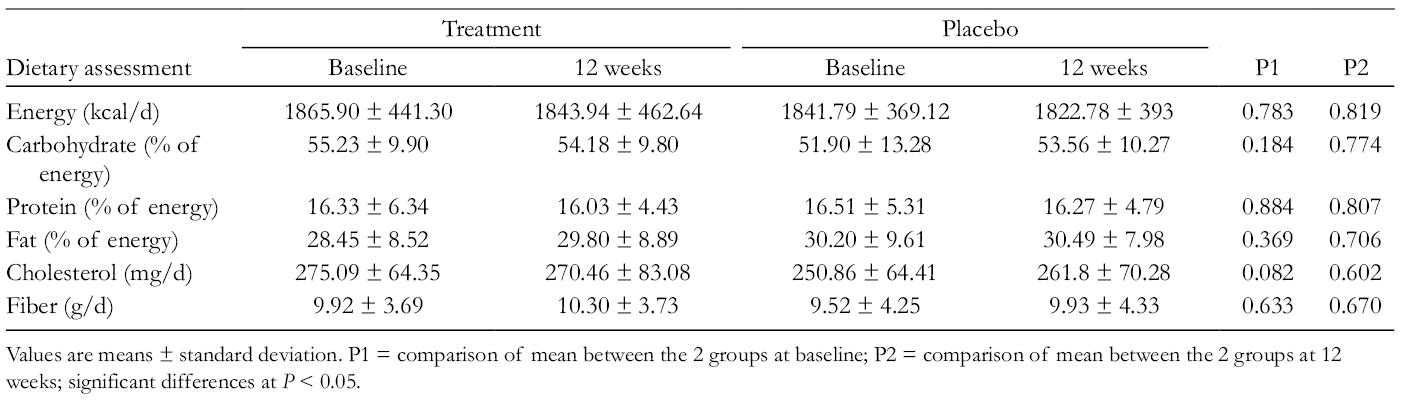
One hundred and ten hyperglycemic subjects were recruited and enrolled in this study. General characteristics and blood chemistry are presented in Table 2. Age, weight, BMI, % body fat, blood pressure, pulse rate, kidney function, and liver function test results were not significantly different between the 2 groups before and after the intervention at 6 weeks and 12 weeks. There were also no significant differences in mean daily total energy and nutrient intakes between the two groups before and after the intervention (Table 3).
General Characteristic and Blood Chemistry of Subjects.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; BUN, blood urea nitrogen; Cr, creatinine; FBG, fasting blood glucose; HbA1C, hemoglobin A1c.
Values are means ± standard deviation. Means in a row with superscript letters without a common letter differ within group; significant differences at P < 0.05. P1 = comparison of mean between the two groups at baseline; P2 = comparison of mean between the 2 groups at 6 weeks; P3 = comparison of mean between the 2 groups at 12 weeks; significant differences at P < 0.05.
Total Energy and Nutrient Intake of Subjects.
Values are means ± standard deviation. P1 = comparison of mean between the 2 groups at baseline; P2 = comparison of mean between the 2 groups at 12 weeks; significant differences at P < 0.05.
Fifty-two subjects randomly assigned to the treatment group and 50 subjects allocated to the placebo group completed the study (dropout rate = 7.27%). According to the count of the recalled capsules at the end visit, compliance was very good. The rates of tables intake were 97% and 94% in the treatment and placebo groups, respectively.
Biomarkers of glycemic control, insulin resistance, lipid profile, oxidative stress, and inflammation between the treatment and placebo groups at baseline, 6 weeks, and 12 weeks are presented in Table 4. The baseline concentrations of FBG, HbA1c, HOMA-IR, TC, HDL-C, TG, oxidized LDL-C, and hs-CRP did not differ significantly between the 2 groups. However, the baseline levels of LDL-C in the treatment group were significantly higher than in the placebo group.
Biomarkers of Glycemic Control, Insulin Resistance, Lipid Profile, Oxidative Stress, and Inflammation.

FBG, fasting blood glucose; HbA1C, hemoglobin A1c; HOMA-IR, homeostatic model assessment of insulin resistance; HDL-C, high-density lipoprotein-cholesterol; LDL-C, low-density lipoprotein-cholesterol; MDA, malondialdehyde; TC, total cholesterol; TG, triglyceride; hs-CRP, high-sensitivity-C reactive protein.
Values are means ± standard deviation. Means in a row with superscript letters without a common letter differ within group; significant differences at P < 0.05. P1 = comparison of mean between the 2 groups at baseline; P2 = comparison of mean and percentage change between the 2 groups at 6 weeks; P3 = comparison of mean and percentage change between the 2 groups at 12 weeks; significant differences at P < 0.05.
After the 6-week and 12-week intervention, the treatment group demonstrated a statistically significant decrease in FBG (13.43%; P < 0.001% and 18.87%; P < 0.001) (Figure 2), HbA1C (6.54%; P < 0.001% and 11.32%; P < 0.001) (Figure 3), and HOMA-IR (28.88%; P < 0.001% and 35.15%; P < 0.001) (Figure 4) compared with the placebo group.

Fasting blood glucose levels in treatment versus placebo.
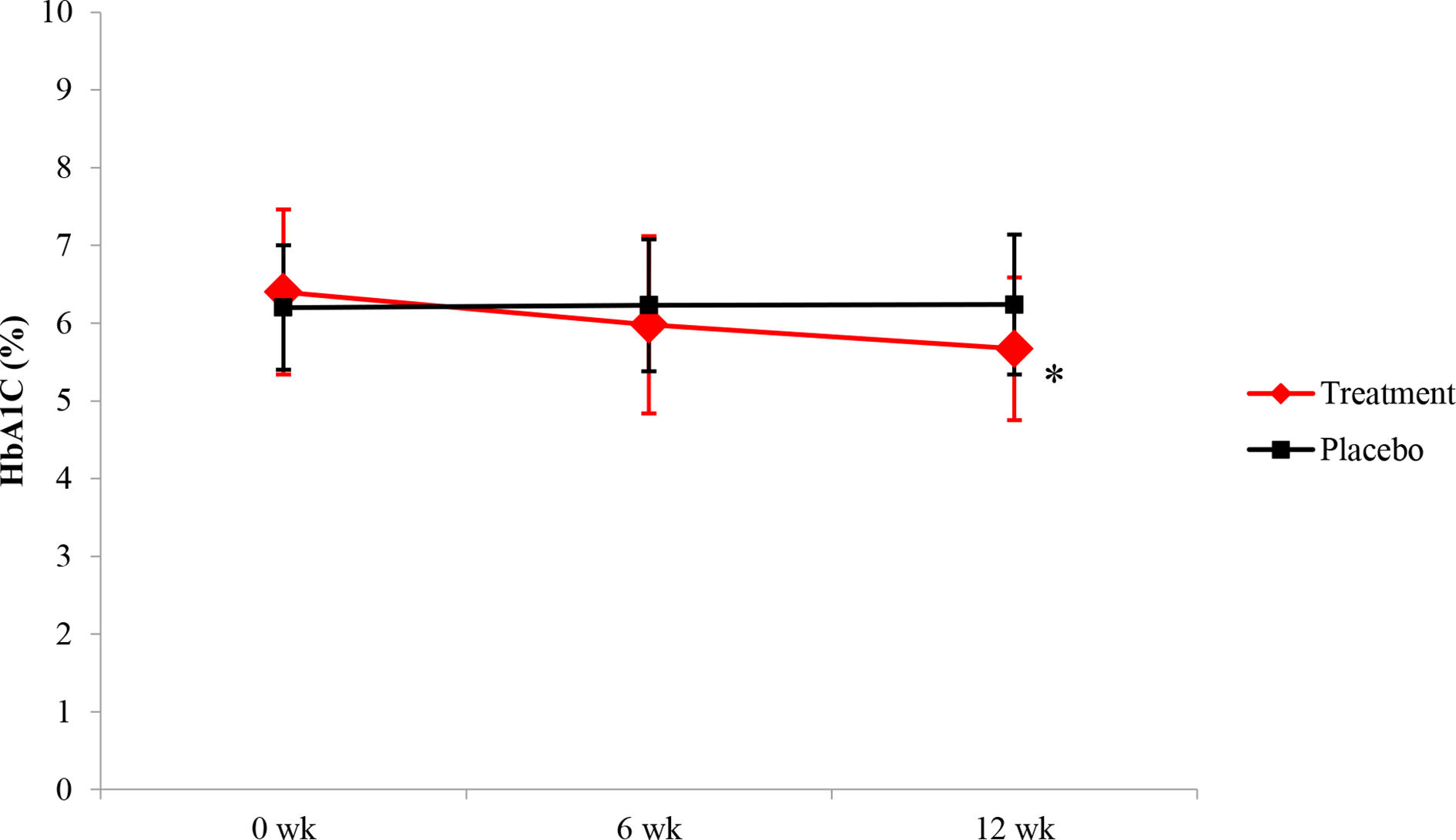
Hemoglobin A1c (HbA1C) levels in treatment versus placebo.

Homeostatic model assessment of insulin resistance (HOMA-IR) in treatment versus placebo.
Moreover, LDL-C levels were significantly reduced in the treatment group (7.12%; P < 0.001% and 9.27%; P < 0.001). Furthermore, the treatment group significantly decreased oxidative stress markers, oxidized LDL-C (14.82%; P < 0.001% and 18.86%; P < 0.001) and MDA (16.55%; P < 0.001% and 26.17%; p < 0.001), compared with the placebo group. Interestingly, in the treatment group, the percent change in TG and hs-CRP tended to decrease at week-12 (P = 0.072 and P = 0.062, relatively) compared with the placebo group.
Subjects reported side effects, which included dizziness, headache, rash, itching, nausea, excessive rectal gas, bloating, and abdominal pain resulting from the supplementation of either the nutraceutical or placebo tablets throughout the whole trial period. The results showed that 4% of subjects in the treatment group and 2% of subjects in the placebo group reported excessive rectal gas. However, there was no significant difference between the 2 groups. Moreover, the results found that 6% of subjects in the treatment group had improved bowel movements.
Discussion
Here we show that a combination of bitter melon, fenugreek, cinnamon, ALA, Zn, biotin, Cr, and vitamin D3 improves glycemic control, insulin resistance, lipid profiles, and oxidative stress markers in hyperglycemia. The results showed that the 6-week and 12-week intervention did not affect anthropometric components such as weight, BMI, body fat, and biochemical markers related to kidney and liver function (Table 2). Thus, this nutraceutical had no effects on body fat and body weight and also no adverse effects on kidney and liver function.
Our data demonstrate that a nutraceutical can decrease levels of FBG and HbA1C, which may be a result of the synergistic effects of each ingredient. Previous research has shown that bitter melon extracts promote insulin release while increasing the number of insulin-producing beta cells. 25 In addition, bitter melon extracts can enhance glucose uptake by the intestine and muscle, 26 by acting on the insulin receptor pathway. This is due in part to the insulin-mimetic effects of polypeptide-P and the hypoglycemic effects of vicine. 27 A clinical study demonstrated a statistically significant improvement in glucose tolerance in type 2 diabetics with oral bitter melon supplementation. 28 Recent research demonstrated that bitter melon can improve the outcome of an oral glucose tolerance test and decrease HbA1C levels in type 2 diabetes mellitus patients. 29
The 3 main active compounds of fenugreek that can exert beneficial metabolic effects in patients with diabetes include saponins, branched-chained amino acids, and soluble fiber. 30 In experimental animal models of diabetes, fenugreek has been shown to confer antidiabetic properties via several mechanisms. These include stimulation of β-islet cell insulin release and reduced gluconeogenesis. 4-Hydroxy-isoleucine has been shown to have insulinotropic effects in the presence of elevated blood glucose concentrations, as well as insulin-sensitizing effects in skeletal muscle and liver tissue. 31,32 It has been proposed that the soluble fiber fraction of fenugreek also supports healthy metabolic function, by slowing the rate of absorption of glucose and lipids. 31 Recently, fenugreek extract has been shown to enhance the potency of glucagon-like peptide-1 (GLP-1) by more than 40-fold. This is of relevance to diabetes, since GLP-1 is released into the bloodstream almost immediately after food consumption and stimulates glucose-dependent insulin release, thus restoring elevated glucose to normal. 33 A single-blind, randomized clinical trial has recently demonstrated that daily administration of fenugreek for 3 years significantly reduced the development of type 2 diabetes mellitus, compared with the control group, in 140 prediabetic patients. Indeed, those in the placebo group were approximately 4 times more likely to progress from prediabetes to type 2 diabetes mellitus. In addition to this, it was also shown that fenugreek was able to significantly reduce fasting and postprandial plasma glucose concentrations from baseline, which was not observed in the control group. 9 A more recent study, using fenugreek extracts, demonstrated clinical reductions in fasting and post-prandial blood glucose and HbA1C. 34
Cinnamon has been shown to stimulate glucose uptake in skeletal muscle and adipose tissue via the translocation of the insulin-dependent glucose transporter, GLUT-4. Cinnamaldehyde may also stimulate cellular glucose uptake via the GLUT-1 receptor. Importantly, some studies indicate that cinnamon can also interact with the peroxisome proliferator-activated receptor (PPAR) gamma (PPAR-γ) and alpha (PPAR-α), which are transcriptional factors involved in glucose homeostasis and adipogenesis. 35,36 Potentially, cinnamon may activate both PPAR-γ and PPAR-α that consequently would improve insulin resistance and also help maintain normal blood lipid balance more effectively than PPAR-γ activators alone. 37 In a meta-analysis of randomized clinical trials, cinnamon supplementation can decrease FBG. 10 Furthermore, recent research indicated that cinnamon intake can reduce FBG and HbA1C levels in type 2 diabetes patients. 11
An experimental study demonstrated that ALA stimulated glucose uptake to a similar extent to insulin in isolated human skeletal myocytes and adipocytes via the activation of the insulin signaling pathway. 38 In addition, ALA appears to have an insulin-sensitizing and lipid-lowering effect in obese individuals predisposed to type 2 diabetes mellitus. 39 A double-blind RCT demonstrated that ALA given as an adjunct to standard of care therapy for type 2 diabetes mellitus patients reduced FBG concentrations and HbA1C levels after supplementation. 12
Zn can potentiate glucose uptake by stimulating insulin receptor signaling in adipose tissues and muscle. 40 Zn was found to improve FBG and insulin sensitivity in prediabetics, suggesting that Zn intake can ameliorate the transition to a diabetic phenotype. 41 Also, zinc intake decreased glucose serum in patients with hypertension. 42 In a meta-analysis of 14 RCTs, Zn supplementation resulted in a reduction in FBG and HbA1C levels. 13
Biotin has been shown to increase the expression and activity of glucokinase in hepatocytes, increasing the uptake of glucose. 43 Cr also was able to alter insulin sensitivity at the cellular level in insulin-sensitive tissues. An experimental study revealed that Cr is an important co-factor for the protein apo-chromodulin. Upon binding chromium, it is converted to holo-chromodulin, which then attaches to insulin receptor to increase its own insulin binding efficiency. 44 It was predicted that biotin and Cr may synergize to create a more effective treatment for type 2 diabetes mellitus. 45 A small, open-label clinical trial demonstrated that twice daily administration of biotin and Cr in combination with a dietary education program significantly reduced HbA1c in patients with type 2 diabetes mellitus. 46 A large, multicenter, double-blind, placebo-controlled clinical trial also investigated the effects of biotin and Cr supplementation on glycemic control in 348 overweight and obese subjects with uncontrolled type 2 diabetes mellitus. It was shown that patients treated with biotin and Cr had a significant reduction in FBG concentrations. 15
Experimental evidence suggested that vitamin D3 ameliorated insulin resistance through reduction of insulin-degrading enzyme and activation of insulin receptor phosphorylation, resulting in increased insulin sensitivity. 47 A meta-analysis of 19 RCTs showed that vitamin D3 supplementation in type 2 diabetes mellitus patients can improve HbA1C and insulin resistance. 48
Furthermore, the result of this study showed that this nutraceutical can decrease LDL-C levels. Similarly, previous studies have reported that fenugreek and cinnamon improve lipid levels. Fenugreek is rich in dietary fiber. The fiber content of fenugreek has been credited with its ability to inhibit lipase enzymes in the digestive system, which is a well-established mechanism by which fiber has been shown to inhibit lipid absorption. 23 A clinical trial demonstrated that fenugreek intake significantly improved metabolic status and serum LDL-C in type 2 diabetic patients. 49 Moreover, an in vitro study indicated that the possible mechanism of the hypolipidemic effect of cinnamon is via inhibition of hepatic β-hydroxy β-methylglutaryl-CoA (HMG-Co A) reductase activity, which controls cholesterol synthesis leading to LDL-C reduction. 50 Also, clinical studies have shown that cinnamon supplementation can decrease LDL-C levels in patients with type 2 diabetes mellitus. 10,51
Oxidized LDL-C is a key factor in the arteriosclerotic process. This supplementation had a positive effect on oxidized LDL-C, which showed the potential effect of its antioxidant activity and is consistent with decreased MDA levels, a biomarker of oxidative stress. This finding was in accordance with a previous study that demonstrated the antioxidant effect of fenugreek, cinnamon, ALA, and vitamin D3. Fenugreek significantly lowered oxidized LDL-C 52 and decreased MDA levels 53 in an animal study. Moreover, cinnamon suppresses lipid peroxidation via enhancement of hepatic antioxidant enzyme activity. 50 ALA also can inhibit oxidized LDL-C and reduced the content of MDA. 54 A clinical study showed that ALA supplementation decreased oxidized LDL-C and MDA levels in obese patients with impaired glucose tolerance. 55 Also, vitamin D3 can improve antioxidant status and reduce MDA levels. 56,57
Based on our result and previously published data, we propose the following scenario by which a nutraceutical can improve glycemic control, insulin resistance, lipid profiles, and oxidative stress markers. The synergistic effects of these ingredients can increase insulin sensitivity and enhance glucose uptake, resulting in FBG and HbA1C reduction. Besides, fenugreek can decrease lipid absorption, and also cinnamon can inhibit HMG-Co A reductase resulting in decreased LDL-C levels. Moreover, the antioxidant effect of fenugreek, cinnamon, ALA, and vitamin D3 may decrease lipid peroxidation via decreased oxidized LDL-C and MDA levels.
The major strengths of this study are its randomized, double-blind controlled design, and the number of participants. The limitation of our study is that most of the study participants were female, which is less representative of the whole population. Even if most of the study participants were female, there was no significant sex difference between the 2 groups of treatment. Also, we only measured hs-CRP levels as a marker of inflammation. In a future study, other inflammatory markers such as tumor necrosis factor-α, interleukin (IL)-1β, and IL-6 in plasma should be determined.
Conclusions
In summary, a nutraceutical containing bitter melon, fenugreek, cinnamon, ALA, Zn, biotin, Cr, and vitamin D3 can improve glycemic control, insulin resistance, lipid profiles, and oxidative stress markers which are the important risk factors of CVD in hyperglycemic subjects. Therefore, this nutraceutical may be an alternative medicine for hyperglycemia.
Footnotes
Acknowledgments
The authors thank assistant professor Dr Carol Hutchinson, Department of Nutrition, Faculty of Public Health, Mahidol University, Bangkok, Thailand, for editing the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This research was funded by Max Biocare Pty Ltd, Australia.
