Abstract
Sideritis scardica is a Lamiaceae plant that is endemic to the alpine zone of the Balkan Peninsula. The tea of S. scardica has been handed down as a “tea of longevity” in the Rhodope region of Bulgaria for an unknown amount of time. In this study, we prepared a hot water extract of S. scardica (SHWE) and examined its effects on both life span and stress response in living tissue using Caenorhabditis elegans and its transgenic mutants. The life span of wild-type N2 worms was prolonged by approximately 15% at the SHWE concentration of 5 µg/mL and approximately 22% at the SHWE concentration of 50 µg/mL, as compared with the control group. The effect of SHWE on the expression of heat shock protein 16.2 (HSP-16.2) under heat stress was investigated using TJ375 worms, a transgenic mutant of C. elegans. In the TJ375 worms pretreated with SHWE, the fluorescence intensity of green fluorescent protein fluorescence, which indicates the expression of HSP-16.2, was significantly increased. In the assay using TJ356 worms, the worms pretreated with SHWE did not show the translocation of DAF-16, a forkhead transcription factor class O homolog, from the cytoplasm to nucleus under heat stress. Additionally, under heat stress, the pretreatment of SHWE improved the survival rate of GR1307 worms, a knockout mutant of daf-16. These results indicate that SHWE enhances HSP-16.2 expression through a stress-response pathway (eg, HSF-1 pathway) other than the DAF-16 pathway, resulting in a prolonged life span of C. elegans under heat stress.
Aging is a progressive and complex process that impairs vital and repair functions and causes aging-related disorders such as cardiovascular and neurodegenerative diseases and cancer. 1 Suppression of aging and aging-related degeneration would greatly contribute to improving the quality of life of humans. The major factors that cause aging and age-related disorders include stress induced by factors such as reactive oxygen species (ROS), heat shock, heavy metals, and ultraviolet light. 2 -4 The stress causes damage in biomolecules such as enzymes, proteins, deoxyribonucleic acid , and lipids. 5 Therefore, the ingestion of antioxidants and activation of a stress response pathway in vivo may reduce the stress damage and suppress the progress of aging.
Heat shock proteins (HSPs) are a group of proteins induced by ROS and heat shock and are conserved in various organisms from bacteria to humans. HSPs behave as a molecular chaperone for other cellular proteins and have strong cytoprotective effects. 6 Forkhead box transcription factors class O (FOXO) belong to a subclass of forkhead transcription factors. The mammalian genome encodes 4 FOXO proteins, while genomes of Caenorhabditis elegans and Drosophila melanogaster encode only 1 FOXO homolog, DAF-16 and dFOXO, respectively. 7 FOXO is responsible for activating genes involved in various cellular stress responses including autophagy, apoptosis, and stress resistance. 8 Therefore, it has been pointed out that increasing cellular expression of these proteins may contribute to prolonging the life span of living organisms.
Sideritis scardica (Lamiaceae family) is an endemic species in the alpine zone of the Balkan Peninsula. S. scardica is mainly used as a raw material of tea and that tea is called a “tea of longevity” in the Rhodope region of Bulgaria. 9 In addition, this plant has also been used as a traditional medicine for respiratory diseases (eg, asthma and bronchitis). 9 Because of such therapeutic effects, S. scardica is currently attracting attention as a medicinal plant. So far, it has been reported that S. scardica has antioxidant, 10,11 anti-inflammatory, 11 and antibacterial effects, 12 and the potential to improve symptoms of Alzheimer’s disease, 13 as physiological functions. However, to our knowledge, there are only a few reports analyzing the physiological functions of S. scardica in vivo, especially the effects on stress response and life span.
Caenorhabditis elegans, a non-parasitic nematode, has biological characteristics such as a short life cycle and a short life span. Furthermore, the C. elegans gene sequence has high homology with the human gene sequence, 14 so it has been used as a model organism for research on aging and life span. The aim of this study was to elucidate the antiaging effect of S. scardica. As a first step, a hot water extract of S. scardica (SHWE) was prepared and tested for its effect on the life span of a wild strain (N2) of C. elegans. As a second step, we analyzed the expression of HSP and DAF-16/FOXO using transgenic mutants of C. elegans and then investigated the bioactivities of S. scardica.
Results and Discussion
Figure 1 shows the survival curves of N2 worms cultured on modified nematode growth medium (mNGM) agar plates containing SHWE under normal culture conditions. The results indicated that the SHWE at concentrations of 5 and 50 µg/mL were capable of enhancing the survival of N2 worms at the L4 stage when compared with the control group (Figure 1). From the results of the assay, the average life span of the worms in the sample and control groups was calculated and shown in Table 1. Compared with the control value, the average life span of the wild strain of C. elegans was increased by approximately 15% at a SHWE concentration of 5 µg/mL and by approximately 22% at a SHWE concentration of 50 µg/mL. Among the tested groups, the maximum life span of N2 worms treated with 50 µg/mL SHWE was the longest, at 25 days.
HSP-16.2 is a small HSP in C. elegans, and its expression is induced by heat and oxidative stress. 15 It has been pointed out that increasing the expression level of HSP-16.2 in cells has a prolonged life span effect on C. elegans. 16 The TJ375 strain of C. elegans is a genetically modified worm with a hsp-16.2 promoter linked to a green fluorescent protein (GFP) reporter, enabling visualization of HSP-16.2 expression in response to the stress. The effect of SHWE on HSP-16.2 expression under heat stress was tested using TJ375 worms. The expression of HSP-16.2 was significantly increased in worms that were precultured on NGM agar plates containing SHWE (500 and 1000 µg/mL) compared with the expression in worms precultured on control media (Figure 2). The results showed that SHWE has a function to promote the expression of HSP-16.2 under heat stress. In order to confirm the improvement of heat stress tolerance by SHWE treatment, the survival rate under heat stress was measured using N2 worms. As a result of culturing at 35°C, N2 worms precultured on NGM agar plates containing SHWE at 500 µg/mL maintained a significantly higher survival rate than the control group (Figure 3). Six hours after the start of the experiment, there was a remarkable difference in the survival rate of the worms, and the difference between the sample and control groups was approximately 34 points. These results strongly suggest that SHWE enhances heat stress tolerance in C. elegans through activation of HSP-16.2 expression.
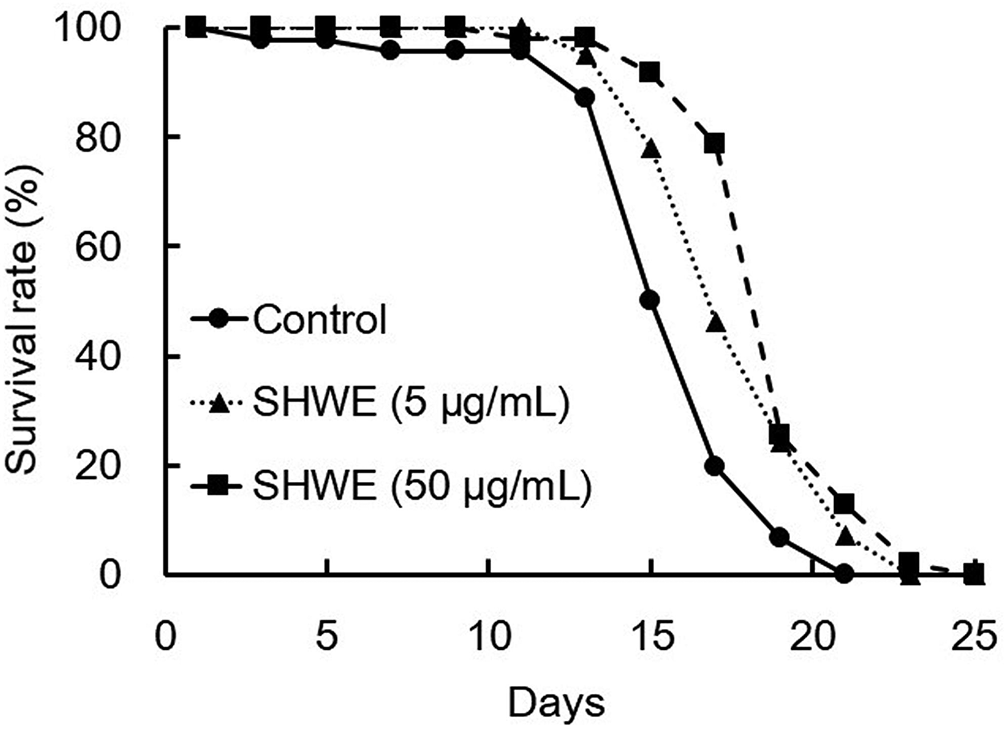
Survival curves of N2 worms treated with hot water extract of Sideritis scardica (SHWE). L4 larvae of age-synchronized N2 worms were cultured on the modified nematode growth medium agar plates containing SHWE at 25°C. The experiment was performed with 5 plates, each plate holding 10 worms. P < 0.001, compared with control.
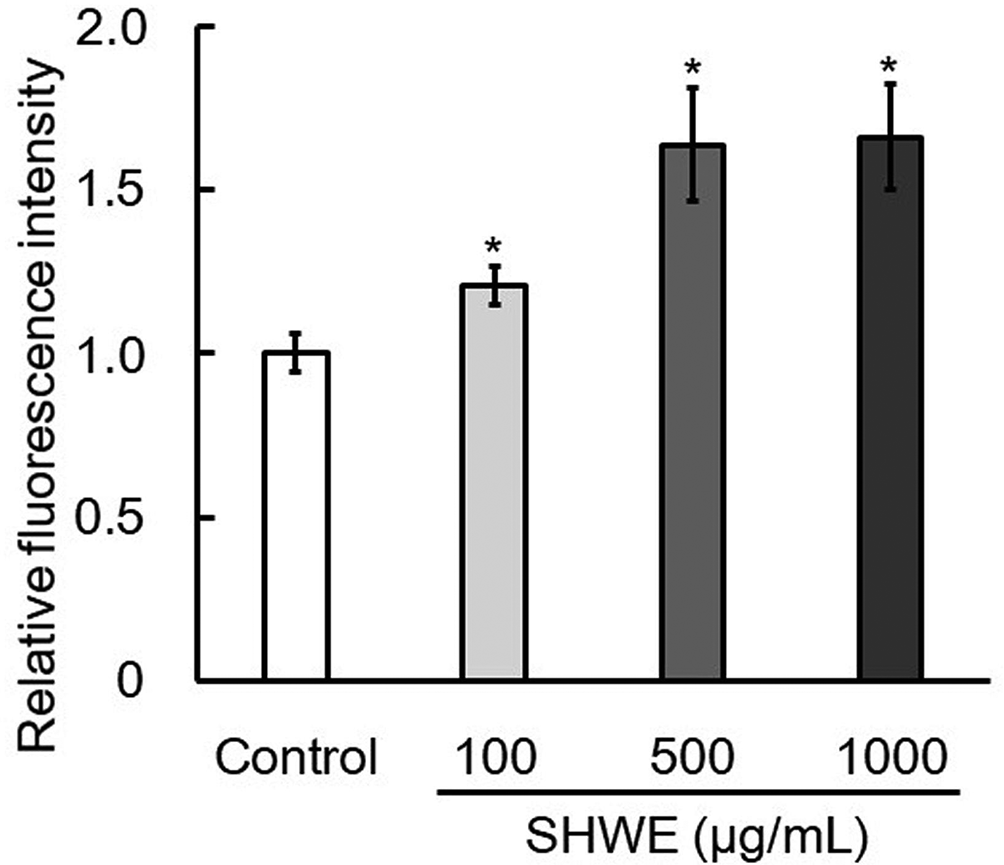
Effect of hot water extract of Sideritis scardica (SHWE) on heat shock protein-16.2::green fluorescent protein expression in TJ375 worm. L1 worms preincubated with SHWE for 72 hours at 20°C were exposed to heat stress for 8 hours at 35°C on the nematode growth medium agar plates. The results were obtained from 3 independent experiments with 10 worms in each experimental group. The relative fluorescence intensity was determined using Image J software. The data are expressed as mean ± standard error. *P < 0.05, compared with control.
Effect of hot water extract of Sideritis scardica (SHWE) on the survival rate of N2 worms under heat stress. L1 worms preincubated with SHWE for 72 hours at 20°C were exposed to heat stress for 8 hours at 35°C on the nematode growth medium agar plates. The results were obtained from 5 independent experiments with 10 worms in each experimental group. The data are expressed as mean ± standard error. *P < 0.01, compared with control.
Effect of SHWE on Life Span of C. elegans N2 Strain.

SHWE, hot water extract of Sideritis scardica.
aThe data are presented as mean ± standard error.
bMaximum life span is the survival day of the last worm alive.
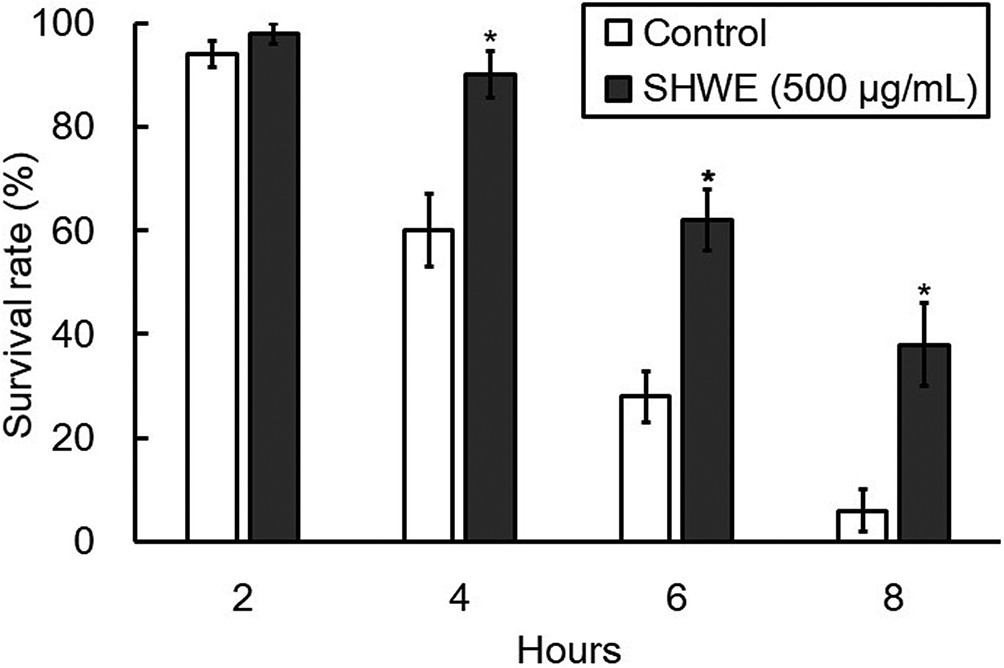
DAF-16 is the only homolog of the FOXO family of transcription factors in C. elegans and has been shown to be involved in the expression of HSP-16.2. 17,18 Normally, DAF-16 is phosphorylated by DAF-2 and AKT-1/AKT-2 in the insulin/insulin-like growth factor-1 signaling pathway and is present in the cytoplasm. 19 Under stress conditions such as heat shock, peroxidation, and starvation, phosphorylation of DAF-16 is suppressed, and as a result, dephosphorylated DAF-16 translocates to the nucleus and induces the expression of stress-responsive genes such as hsp-16.2 and sod-3. 18,20 To determine whether DAF-16 is involved in the activation of HSP-16.2 expression induced by SHWE treatment, changes in the subcellular localization of DAF-16 were analyzed using TJ356 worms, genetically modified strain of C. elegans. In this strain, a DAF-16::GFP fusion protein is constructed as a reporter to reveal the subcellular localization of DAF-16. Fluorescence microscopy revealed that in the control group, 56% of DAF-16 was localized in the nucleus under heat stress (Figure 4). On the other hand, in the worms precultured on NGM containing SHWE at 500 µg/mL, most of DAF-16 was present in the cytoplasm (Figure 4). These results suggest that the increase of HSP-16.2 expression by SHWE treatment under heat stress does not involve the activation of DAF-16 by nuclear translocation. Furthermore, a survival test under heat stress was performed using GR1307 worms, genetically modified strain in which the daf-16 gene region was knocked out. As a result of the test, the worms pretreated with SHWE significantly maintained a higher survival rate than the control group (Figure 5). The largest difference compared with the control was about approximately 40 points and was found after 6 hours. Increased HSP expression has also been shown to be mediated by activation of the transcription factor HSF-1, which is in an inactive monomeric state in unstressed cells. 21,22 These results and findings suggest that under heat stress, SWHE may promote HSP-16.2 expression through activation of HSF-1 but not the DAF-16 pathway.

Subcellular localization of DAF-16 under heat stress. L1 stage of age-synchronized TJ356 worms were preincubated with hot water extract of Sideritis scardica (SHWE) for 72 hours at 20°C and cultured on the nematode growth medium agar plates under heat stress for 25 minutes at 35°C. Each data is expressed as mean value (N = 50). No change in the subcellular localization of DAF-16 was observed in all groups pretreated with SHWE under heat stress.

Effect of hot water extract of Sideritis scardica (SHWE) on the survival rate of GR1307 worms under heat stress. L1 worms preincubated with SHWE for 72 hours at 20°C were exposed to heat stress for 8 hours at 35°C on the nematode growth medium agar plates. The results were obtained from 7 independent experiments with 10 worms in each experimental group. The data are expressed as mean ± standard error. *P < 0.05, **P < 0.01, compared with control.
This is the first report to evaluate and analyze the antiaging effect of S. scardica. Experiments with C. elegans revealed that SHWE has a prolonged life span effect and a function to enhance stress tolerance. It has been reported that plant extracts such as blueberry, apple, and green tea show life-extending effects and stress-relieving effects in experiments using C. elegans. 23 -25 In these extracts, polyphenols (proanthocyanidins) are contained as a main component, and it has been pointed out that they contribute to the antiaging effects. 23 -25 In another study, 10 it has been reported that polyphenols are contained in the water-soluble extract of S. scardica. As a result of thin-layer chromatography analysis using the Folin-Ciocalteu reagent as a spray reagent, several spots suggesting phenolic compounds were detected in SHWE (data not shown). Furthermore, some phenylethanoid glycosides and flavonoid glycosides were detected in SHWE by liquid chromatography/mass spectrometry analysis with a C18 column. The antiaging effects of SHWE revealed in this study may be due partially to the phenolic compounds contained.
Experimental
Materials
The dried plant of Bulgarian S. scardica was kindly donated by Koshin bussan. The plant used for the preparation of SHWE was pulverized via pulverizing mill (ABS-W, Osaka Chemical). The obtained powder was stored at −25°C until use. All reagents used in this study were of analytical grade.
Preparation of SHWE
Four hundred milliliter of ultrapure water was added to the dried powder of S. scardica (10 g), and the suspension was autoclaved for 20 minutes at 121°C. After paper filtration, the filtrate was centrifuged at 14 000 rpm for 30 minutes at 5°C. The supernatant was freeze-dried using lyophilizer FDU-2200 (Eyela), and the obtained lyophilized powder (1.66 g) was used as SHWE. The SHWE was completely dissolved in ultrapure water and sterilized using a filter (polar diameter: 0.2 µm, Minisart, Sartorius) for sterilization. The sterilized SHWE solution was used for all experiments.
C. elegans Strains and Maintenance
The C. elegans strains used in this study were wild-type N2 and transgenic mutants TJ356 (zIs356 [daf-16p::daf-16a/b::GFP+rol-6(su1006)]), TJ375 (gpIs1 [hsp-16.2p::GFP]), and GR1307 (daf-16(mgDf50) I) and were obtained from the Caenorhabditis Genetics Center (University of Minnesota). All the strains were cultured on nematode growth medium (NGM) agar plate with Escherichia coli OP50 as a feed and kept in an incubator at 20°C. 26 To prevent bacterial contamination, streptomycin at a final concentration of 275 µg/mL was added to the NGM agar plate. Age-synchronization of the worms was performed according to the method of Guerrero-Rubio et al. 26
Life Span Assay Using N2 Worms
L4 larvae of age-synchronized N2 worms were transferred via a platinum wire to mNGM (peptone free) agar plates containing 1 mL of SHWE solution to the final concentrations of 5.0 and 50 µg/mL. Ultrapure water sterilized by filtration was used as a control solution. The mNGM plates contained 200 µM 2′-deoxy-5-fluorouridine to prevent the growth of progeny and streptomycin to prevent bacterial contamination. The worms were cultured at 25°C, and their viability was determined every 2 days. The worms were scored as dead if they failed to respond to a gentle stimulus delivered via a platinum wire.
Assay of HSP-16.2 Expression
Each TJ375 worm L1 larvae was placed on the mNGM agar plate supplemented with SHWE to give final concentrations of 100, 500, and 1000 µg/mL, and preincubated for 72 hours at 20°C. For the control group, a medium supplemented with sterilized water was used. After that, randomly selected worms were transferred to sample-free NGM agar plates and exposed to heat stress for 2 hours at 35°C. The expression of HSP-16.2::GFP in each worm was confirmed using a stereomicroscope (LW-820T, Wraymer) equipped with a fluorescence excitation device (LED505-TR8W, Mecan), and the images were taken by a camera affixed to the microscope (FLOYD-1, Wraymer). The relative fluorescence intensity was determined using Image J software.
Assay of Subcellular DAF-16 Localization
L1 stage of age-synchronized TJ356 worms was cultured on the mNGM under the same conditions described above for 72 hours at 20°C. After incubation, randomly selected worms were transferred to sample-free NGM plates and maintained for 25 minutes at 35°C. Fifty worms per group were imaged on a fluorescence microscope (AXJ-5300TPHFL, Wraymer) with a camera affixed to the microscope (FLOYD-1, Wraymer). Distribution of DAF-16::GFP in each worm can be in the nucleus, cytoplasm, or intermediate region between the nucleus and cytoplasm. Worms were sorted and counted according to observed amounts of localized DAF-16::GFP.
Survival Assay Under Heat Stress
Age-synchronized N2 and GR1307 (DAF-16 loss-of-function mutant) worms at the L1 stage were treated with mNGM supplemented with SHWE at a final concentration of 500 µg/mL for 72 hours at 20°C. For the control group, the mNGM plate with sterilized water was used. After incubation, each strain of worm was transferred to a sample-free NGM plate and exposed for 8 hours to 35°C. The viability of worms was determined every 2 hours. The experiment was performed with 50 worms of type N2 and 70 worms of type GR1307.
Statistical Analysis
For the life span assay, the statistical significance was determined by a log-rank test. In the experiments other than the DAF-16 localization assay, the differences among groups were statistically analyzed using the t-test.
Footnotes
Acknowledgments
We thank Dr Wataru Aoki of the Graduate School of Agriculture, Kyoto University for advice on bioassays using C. elegans.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a joint research start-up promotion project with small and medium enterprises in Mie University.
