Abstract
This study presents the bioguided chemical investigation of the 80% aqueous methanol extract of Geum urbanum aerial parts. Liquid–liquid partitioning of this extract in solvents of increasing polarity combined with biological screening showed that the ethyl acetate (EtOAc) soluble fraction was the most active part of the extract. This fraction was chemically profiled by a 13C nuclear magnetic resonance (NMR)-based dereplication method, resulting in the identification of 14 compounds. The dereplication process was followed by the purification of unknown and minor compounds of the EtOAc fraction. A new glycosylated phenol, namely, 3-(3,4-dihydroxyphenyl)propyl-α-
Skin aging is a complex biological process mainly characterized by wrinkle formation, uneven pigmentation, darkening, thinning, and roughening of the skin. This could be caused by intrinsic (genetically determined) or extrinsic (mediated by exposure to ultraviolet [UV] radiation and toxic compounds) mechanisms. One of the underlying mechanisms of these processes is excessive oxidative stress, which is probably the single most harmful contributor to skin aging, leading to loss of cells and extracellular matrix degradation as the most prominent features of chronologically aged skin. Oxidative stress results from an imbalance between reactive oxygen species production and antioxidative defense. 1 Prevention of these dynamic processes is a major issue for dermocosmetics and substantial efforts are being made to discover new protective ingredients. 2,3 Plant extracts can contain a high concentration of phenolic compounds, carotenoids, terpenoids, etc., able to inhibit free radicals and therefore to slow down skin aging. 4,5 The antioxidant potency of phenolic compounds (phenolic acids, flavonoids, anthocyanidins, tannins) can be easily determined by chemical tests such as 2,2,1-diphenyl-1-picrylhydrazyl (DPPH) assay, the ferric reducing/antioxidant power (FRAP) assay, the 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay, and the cupric ion reducing antioxidant capacity (CUPRAC) assay. 4 Phenolics have also been reported as effective compounds in matrix remodeling activation through inhibition of elastases, 6 a class of proteases highly involved in aging.
In the continuation of our search for local plants with antiaging properties, 7,8 we were interested in Geum urbanum L., a perennial herbaceous plant belonging to the Rosaceae family. The genus Geum encompasses about 70 species distributed in temperate regions all over the world. Geum urbanum is native to humid woods of Europe and Asia 9 and is a common species in France, especially in cool places such as hedges and undergrowth. 10 Infusions from the roots and rhizomes of G. urbanum have been traditionally used in folk medicine in the treatment of acute diarrhea and as astringent agent for inflammations of the mucosa and gums and treatment of hemorrhoids. 9,11 Previous works have shown that plants species belonging to the Geum genus produce ellagitannins, gallotannins, flavonoids, terpenoids, and phenylpropanoids. 12 -14 Ellagitannins (tellimagrandin, stachyurin, casuarynin, gemin A, and ellagic acid derivatives), procyanidins (procyanidin B3, procyanidin C2, and catechin derivatives), gallic acid and its derivatives, steroids, and triterpenoids were isolated from the roots of G. urbanum. 11,15 -18 To our knowledge, the antioxidant and antielastase potency of G. urbanum aerial parts and their relation to chemical composition have not been reported. Recently, the radical scavenging activity of extracts and fractions obtained from aerial and underground parts of G. urbanum were investigated and the ethyl acetate (EtOAc) fractions from roots and aerial parts were found to be the most active, but only the EtOAc extract of the roots was investigated. 15
Thus, the aim of the present study was to investigate the antioxidant and elastase inhibitory activities of extracts, fractions and isolated compounds obtained from the aerial parts of G. urbanum. A 13C nuclear magnetic resonance (NMR)-based dereplication methodology combined with a bioguided fractionation and purification procedure was used to identify some of its metabolites.
Results and Discussion
An 80% aqueous methanol extract (hereafter referred to as GUM) was obtained from G. urbanum aerial parts. Its water (H2O) solution was fractionated by extraction with a series of solvents of increasing polarity, resulting in a dichloromethane fraction (DCMF), EtOAc fraction (EAF), and n-butanol fraction (n-BF). The free radical scavenging activities of GUM and obtained fractions were determined by the DPPH and hydroxyl radical assays whereas their cupric reducing capacity was evaluated by the CUPRAC assay. In addition, their ability to inhibit human neutrophil elastase activity was tested. The results are shown in Table 1. As previously described for roots and aerial parts of G. urbanum, 15 the EAF had the best antioxidant activities. n-BF exhibited the best DPPH scavenging activity (half-maximal inhibitory concentration [IC50] <12.5 µg/mL) followed by EAF and GUM (IC50 14.7 µg/mL and 21.8 µg/mL, respectively). The hydroxyl radicals scavenging activity was better for EAF and GUM (IC50 142.7 µg/mL and 174.5 µg/mL, respectively). For all extracts, a substantial cupric ion reducing capacity was observed (IC50 <2.0 µg/mL), but lower than the one of trolox, used as a positive standard. In addition, a very good elastase inhibitory activity was observed for DCMF and n-BF (IC50 17.6 µg/mL and 14.3 µg/mL, respectively), but EAF showed the best activity (IC50 6.0 µg/mL), identical to quercetin used as a positive standard. The EAF having interesting biological activities, in both antioxidant and anti-elastase assays, was chemically investigated through a bioassay-guided isolation strategy in order to tentatively determine the active constituents.
Antioxidant and Elastase Inhibitory Activities of Extracts, Fractions and Compounds Isolated From Geum urbanum.

CUPRAC, cupric ion reducing antioxidant capacity; IC50, half-maximal inhibitory concentration; DCMF, dichloromethane fraction; EAF, ethyl acetate fraction; n-BF, n-butanol fraction.
a% Inhibition at 200 µg/mL
bUsed as positive control.
The major compounds of EAF were identified using a dereplication method
22
combining centrifugal partition chromatography (CPC), NMR analysis, clustering of NMR peak emergence profiles, and database search, without purification of individual components. The CPC fractionation of EAF was performed with the biphasic solvent system methyl tert-butyl ether (MtBE)/acetonitrile (CH3CN)/H2O (3/3/4, v/v/v) to afford 11 subfractions (EAF1–11). After 13C NMR analyses of EAF1–11, all spectra of the fraction series were processed and submitted to hierarchical clustering analysis (HCA) for the recognition of the similarity between emergence profiles of 13C NMR peaks throughout the fractionation process. In this way, 13C NMR signals belonging to the same compounds were grouped to build “chemical shift clusters” represented in the heat map drawn in Figure 1. As a result, 13 major chemical shift clusters corresponding to the major metabolites of the EAF (Figure 1), colored in yellow, were revealed by the heat map. An in-house database containing predicted chemical shift values of natural metabolites was used to identify the metabolites from the chemical shifts within clusters. Cluster 1 in fraction EAF10 was assigned to saccharose and β-fructose. The identification of saccharose and β-fructose was easily confirmed by checking heteronuclear single quantum coherence (HSQC), heteronuclear multiple bond correlation (HMBC), and correlation spectroscopy (COSY) data of fraction EAF10 and by comparison with literature data.
23
Similarly, clusters 2–11 were identified as ellagic acid and gallic acid
24
(clusters 2, 2′, and 2″; fraction EAF6), a mixture of 6-O-E-caffeoyl-β-
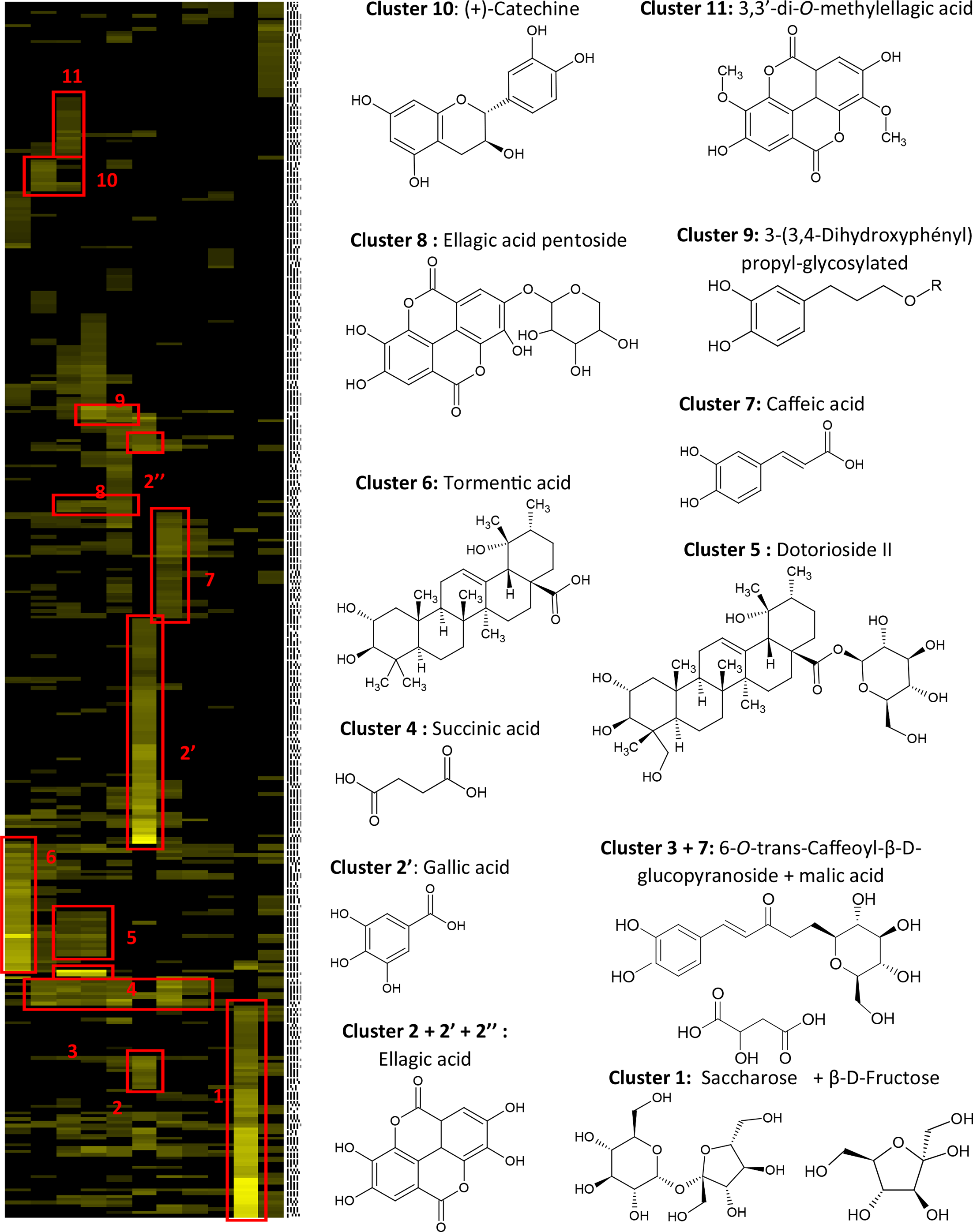
13C nuclear magnetic resonance (NMR) chemical shift clusters obtained by applying hierarchical clustering analysis on ethyl acetate fraction on centrifugal partition chromatographyfractions from Geum urbanum.
Fractions EAF1–7 containing the phenolic compounds identified by dereplication were further screened for their antioxidant and elastase inhibitory activities. As shown in Table 1, all tested fractions presented very good antioxidant activities, revealed by DPPH and CUPRAC assays. EAF1–7 showed also a significant elastase inhibitory activity, in particular EAF1–4 (IC50 2.1 µg/mL, 2.9 µg/mL, 5.1 µg/mL, and 8.0 µg/mL, respectively). The most active fractions EAF2–4 and EAF7 were further purified using semipreparative high-performance liquid chromatography (HPLC), to identify the compounds responsible for the activities, and afford an undescribed glycosylated phenol (

Chemical structure of compound 1 isolated from Geum urbanum aerial parts.
Compound
Isolated compounds
Conclusions
The present study reports for the first time the isolation and identification of 19 secondary metabolites including ellagic tannins, ellagic acid derivatives, phenolic acids, glycosides derivatives, and triterpenes from aerial parts of G. urbanum. Our studies demonstrated that EAFs were characterized by the highest antielastase and antiradical activities among all prepared fractions. For this reason, we isolated individual constituents of the EAF fraction and evaluated their contribution to the observed activities. We isolated and identified a new glycosylated phenol, together with 6 compounds.
From a chemotaxonomic viewpoint, catechin (
Regarding biological activities, 4 subfractions, essentially containing polyphenols, exhibited a significant elastase inhibitory activity and, among the identified compounds, phenolics
Experimental
General Experimental Procedures
The 1-dimensional (1D) and 2D-NMR spectra were recorded at 298 K in methanol-d 4 or dimethyl sulfoxide (DMSO-d 6) on a Bruker AvanceIII-600 spectrometer (Karlsruhe, Germany) (1H at 600 MHz and 13C at 151 MHz) equipped with a 5 mm TCI cryoprobe. The Bruker TopSpin 3.6 software was used for NMR data acquisition and processing. HR-ESI-MS spectra were recorded on a Micromass Q-TOF micro instrument (Manchester, UK). Mass spectra were recorded in the negative ionization mode in the m/z range 100-2000, with a mass resolution of 20 000 and an acceleration voltage of 0.7 kV. Semipreparative HPLC purification was carried out on a Dionex apparatus equipped with an ASI-100 automated sample injector, an STH 585 column oven, a P580 pump, and a UVD 340S diode array detector, all driven by the Chromeleon software version 6.8. Analytical HPLC experiments were performed using a Thermofisher Ultimate 3000 (Thermo Fischer Scientific, Villebon-sur-Yvette, France), equipped with a 4-way pump LPG 3400 SD, an automatic injector WPS 3000 SL, a UV/visible diode array detector 3000, and the Chromeleon software version 6.8. A prepacked C18 column (Interchim, 250 × 10 mm, 5 μm) was employed for semipreparative HPLC. The mobile phase was composed of H2O acidified with trifluoroacetic acid (TFA; 0.025%) and CH3CN with a flow rate of 5 mL/min, and the chromatograms were monitored at 205, 254, 300, and 360 nm. A prepacked C18 column Uptisphere Strategy C18 (Interchim, 250 × 4.6 mm, 5 μm) was used for analytical HPLC and the mobile phase contained H2O with TFA (0.025% v/v) and CH3CN. A gradient elution method was applied from 5% to 80% of CH3CN in 30 minutes with a flow rate of 1 mL/min, and the chromatograms were monitored at 205, 254, 300, and 360 nm. Thin-layer chromatography (TLC) was carried out on silica gel 60 F254 precoated aluminum plates (0.2 mm, Merck), using CHCl3/MeOH/H2O (14/6/1, v/v/v) as mobile phase. The compounds were visualized under UV light (254 and 366 nm) after high-performance TLC (HPTLC, CAMAG TLC Visualizer 2) and sprayed with 50% sulfuric acid followed by heating. A FLUOstar Omega spectrophotometer (BMG LABTECH) was used for absorbance measurement in antioxidant assays. An Infinite F200 PRO spectrofluorimeter (Tecan, Lyon, France) was used for measuring fluorescence during antielastase assays.
Plant Material
The aerial parts of G. urbanum L. (Rosaceae) were collected in Châlons-sur-Vesle (Northeastern part of France: 49°2167′N, 4°05′E) in July 2016, authenticated by Dr Abdulmagid Alabdul Magid and dried at room temperature. A voucher specimen (MA-GU-2016-07) was deposited at the Herbarium of the Botanic Laboratory-Faculty of Pharmacy, University of Reims Champagne-Ardenne.
Extraction and Isolation
The dried and powdered G. urbanum aerial parts (300 g) were macerated in MeOH/H2O (4/1, v/v, 3 × 3 L, 24 hours) at room temperature. The macerate was concentrated to about 1 L at 40°C under vacuum. An aliquot of the aqueous solution (100 mL) was evaporated to dryness to obtain the GUM extract (2.6 g). The aqueous solution (1 L) of GUM was successively extracted with dichloromethane (3 × 750 mL), EtOAc (3 × 750 mL), and n-butanol (3 × 750 mL), then dried under reduced pressure to yield DCMF (6.1 g), EAF (6.2 g), and n-BF (24.2 g) extracts, respectively, and a water-soluble part (29.2 g).
Centrifugal Partition Chromatography
CPC fractionation was carried out on a lab-scale FCPE300 column of 303 mL inner volume (Rousselet Robatel Kromaton, Annonay, France) made of 7 circular partition disks and engraved with a total of 231 partition twin cells (≈1 mL per twin cell). The liquid phases were pumped by a KNAUER Preparative 1800 V7115 pump (Berlin, Germany). The column was coupled online with a UVD 170 s detector set at 210, 254, 280, and 366 nm (Dionex, Sunnivale, CA, USA). Fractions of 20 mL were collected by a Pharmacia Superfrac collector (Uppsala, Sweden). The solvent system was MtBE/CH3CN/H2O (3/3/4, v/v/v). The column rotation speed was set at 1200 rpm and the flow rate at 20 mL/min. EAF (1.5 g injected mass) was dissolved in 15 mL of a mixture of both lower phase (10 mL) and upper phase (5 mL). The upper phase of the biphasic solvent system was pumped for 85 minutes in the ascending mode. The stationary phase was then extruded by pumping the organic phase in the descending mode at 20 mL/min. Fractions of 20 mL were collected over the whole process. All fractions were analyzed by TLC and HPLC and then pooled to yield fractions EAF1–11.
NMR Analysis and Dereplication of the Metabolites
The structures and names of metabolites already reported in the genus Geum (n = 50) were collected from articles available in the scientific literature. The predicted 13C NMR chemical shifts of each one were then stored into a local database already comprising 3038 structures of natural compounds (NMR Workbook Suite 2012, ACD/Labs, Ontario, Canada). In the second step, all CPC fractions were dried under vacuum and an aliquot (up to 20 mg when possible) was dissolved in 600 µL of DMSO-d 6 and analyzed by 13C NMR. A standard zgpg pulse sequence was used with an acquisition time of 0.9 seconds and a relaxation delay of 3 seconds. For each sample, 1024 scans were added to obtain a satisfactory signal-to-noise ratio. The spectral width was 240 ppm and the receiver gain was set to the highest possible value. Spectra were then manually phased, baseline corrected, and calibrated on the central resonance of DMSO-d 6 (δ 39.8 ppm). The last step consisted of the binning of all 13C NMR signals followed by the visualization of the whole dataset as a heat map. For this purpose, the absolute intensities of all 13C NMR signals detected in the spectra of the fraction series were automatically collected and each resulting peak list was stored as a text file. The binning step was performed by a locally developed computer script written in Python language. Its principle was to divide the 13C spectral domain (from 0 to 240 ppm) into chemical shift intervals (or bins) of identical width (Δδ = 0.2 ppm) and to associate the absolute intensity of each peak to the corresponding bin. The resulting table was imported into the PermutMatrix version 1.9.3 software (LIRMM, Montpellier, France) and submitted to HCA for data visualization. The chemical shift clusters regrouped with the HCA were then submitted to the database search for compound identification. Additional 1D and 2D NMR experiments (1H NMR, HSQC, HMBC, and 1H-1H-COSY) were recorded and analyzed in order to confirm the structures of the identified compounds.
HPLC Purification of EAF
Fraction EAF2 was subjected to semipreparative HPLC using the gradient system (15%–40% CH3CN, 30 minutes) to yield compounds
3-(3,4-Dihydroxyphenyl)propyl-α-L -Rhamnopyranoside (1 )
Yellowish-brown solid; 1H NMR (600 MHz, CD3OD, supplemental data, supplemental figure S1) 3.28 (1H, m, H-1a), 3.57 (1H, m, H-1b), 1.76 (1H, quin, J = 6.9 Hz, H-2a), 2.50 (1H, qd, J = 7.3 Hz, H-3a), 2.52 (1H, qd, J = 7.2 Hz, H-3b), 6.65 (1H, d, J = 2.0 Hz, H-2′), 6.77 (1H, d, J = 8.2 Hz, H-5′), 6.59 (1H, dd, J = 8.2,2.0 Hz, H-6′), 4.53 (1H, d, J = 1.9 Hz, H-1″), 3.69 (1H, dd, J = 3.4,1.9 Hz, H-2″), 3.55 (1H, dd, J = 9.4,3.4 Hz, H-3″), 3.26 (1H, t, J = 9.4 Hz, H-4″), 3.48 (1H, m, H-5″), 1.14 (3H, d, J = 6.2 Hz, H-6″) ; 13C NMR (125 MHz, CD3OD, supplemental data, supplemental figure S2) 66.2 (C-1), 31.1 (2), 31.2 (3), 134.7 (1′), 116.4 (C-2′), 143.7 (C-3′), 141.6 (C-4′), 116.3 (C-5′), 121.0 (C-6′), 100.3 (C-1″),70.9 (C-2″), 71.0 (C-3″), 72.6 (C-4″), 68.4 (C-5″), 16.6 (C-6″) ; HR-ESI-MS (negative-ion mode) m/z: 313.1284 [M − H]− (calculated for C15H21O7, 313.1287).
DPPH Radical Scavenging Activity
Extracts, fractions, and compounds
Hydroxyl Radical Scavenging Activity
Extracts and fractions were evaluated for their hydroxyl radical scavenging activity at various concentrations, ranging from 1330 to 41.56 µg/mL, according to a recently published procedure. 8
Power Cupric Ion Reducing (CUPRAC) Assay
Extracts and fractions were tested for their cupric ion reducing ability. The cupric ion reducing activity (CUPRAC) was determined according to a published method. 49 Samples were prepared at concentrations of 572, 286, 143, 71.5, 35.75, 17.87, and 8.94 µg/mL and dissolved in H2O/DMSO (9/1, v/v). A volume of 45 µL of each diluted sample was added to a premixed reaction mixture containing cupric chloride (CuCl2; 90 µL, 10 mM), freshly prepared neocuproine (90 µL, 7.5 mM) dissolved in distilled H2O and ethanol in proportion 8/2 (v/v), and NH4Ac buffer (90 µL, 1M, pH 7.0). Similarly, a blank test mixture was prepared by adding a sample solution (45 µL) to the premixed reaction mixture (270 µL) without CuCl2. The reaction proceeded for 30 minutes at room temperature on a 96-well microplate and the absorbance was then read at 450 nm. Ascorbic acid, quercetin, and trolox were used as positive controls. The power cupric ion reducing was calculated as follows: [1 – A 0/(A 1 – A 2)] × 100, where A 0 is the absorbance of the control (without sample), A 1 is the absorbance in the presence of the sample, and A 2 is the absorbance of the blank. All the tests were conducted in triplicate and IC50 were determined by interpolation of concentration versus inhibition curves obtained by an MSExcel calculation sheet.
Elastase Enzyme Assay
Extracts and fractions were evaluated for their ability to inhibit human leukocyte elastase (HLE) (Merck Biosciences). Tests were performed in 96-well microplates precoated with 1% bovine serum albumin. HLE (0.8 µM) was incubated for 1 hour at 27°C in Tris buffer (50 mM Tris–hydrochloric acid pH 7.5 containing 500 mM sodium chloride) containing 0.1–1000 µg/mL of the tested sample. The pure solvent was used as a control sample. The assay was initiated by adding HLE fluorogenic substrate MeOSuc-Ala-Ala-Pro-Val-AMC (λ exc = 380 nm/λ em = 460 nm) at a final concentration of 80 µM. The rate of substrate cleavage was measured in triplicate for each sample concentration with 1 measurement per minute for 60 minutes. HLE activity was calculated according to: % HLE activity = (Slopesample × 100)/slopecontrol, where slopesample and slopecontrol are the slope of the curve in the graph of fluorescence intensity versus time. The IC50 values were calculated by non-linear regression analysis with the Graphpad software (La Jolla, USA).
Supplemental Material
Supplementary material - Supplemental material for Investigation of Antioxidant and Elastase Inhibitory Activities of Geum urbanum Aerial Parts, Chemical Characterization of Extracts Guided by Chemical and Biological Assays
Supplemental material, Supplementary material, for Investigation of Antioxidant and Elastase Inhibitory Activities of Geum urbanum Aerial Parts, Chemical Characterization of Extracts Guided by Chemical and Biological Assays by Marie Schmitt, Abdulmagid Alabdul Magid, Jean-Marc Nuzillard, Jane Hubert, Nicolas Etique, Laurent Duca and Laurence Voutquenne-Nazabadioko in Natural Product Communications
Footnotes
Acknowledgments
Dr Marie Schmitt gratefully acknowledges ICMR and MEDyC laboratories as well as URCA PlAneT platform (University Reims Champagne-Ardenne) for giving the possibility to perform all the necessary manipulations for the realization of this publication.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors are grateful to Grand Est region in France and EU-program FEDER for their financial support of this thesis CHAVIC project.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
