Abstract
Pseudomonas bacteria are entomopathogenic that can naturally infect and kill insects upon ingestion. The insecticidal and plant growth-promoting roles of the bacteria were assessed by applying Pseudomonas IUK001 to insects and plants. The culture extract (CE) of IUK001 at the concentration of 1 mL/cm3 of an artificial diet was used as a treatment for Galleria mellonella larvae. The CE was heat stable at 70°C for 30 minutes and proteinase-K stable, showing 100% and 90% insecticidal activity against G. mellonella larvae. Gas chromatography–mass spectrometry (GC–MS) analysis revealed the insecticidal compounds, for example, trans-cinnamic acid, ornithine, and cyclo (
Keywords
Pseudomonas spp. are Gram-negative bacteria residing in diverse ecological positions such as water, soil, plant surfaces, and sometimes which penetrate into epidermal tissues of plants. 1 Some Pseudomonas spp. produce insecticidal toxins and cause mortality in a wide variety of insects, particularly at their larval stages. 2 In vitro and in vivo investigations of Pseudomonas bacteria revealed that the insecticidal activity is not confined to proteinaceous toxins only. 3 Genomic analysis of Pseudomonas bacteria has revealed that there are number of genes associated with the generation of secondary metabolites causing toxicity in insects. 4 Pseudomonas species such as P. aeruginosa and P. entomophila are well-known entomopathogenic bacteria and their pathogenicity has been documented against 3 different orders including Lepidoptera, for example, Bombyx mori and Galleria mellonella. 4 A wide host range of the Pseudomonas spp. makes them a promising model for the study of host–pathogen interactions and for the development of biocontrol agents against insect pests. 5 Regardless of the insecticidal activities related to toxins synthesized by Pseudomonas spp., they have yet to be exploited as biocontrol agents on a commercial level. 1 The reason for this is that the effects of Pseudomonas on plants have been inadequately investigated. 6 It is important to explore the impacts of Pseudomonas spp. on plant development prior to proposing it as a biocontrol agent.
The application of biocontrol agents as a substitute for agrochemical pesticides has been considered a feasible solution to get rid of agricultural pollutants. 7 Agrochemicals are being utilized as often as possible in numerous parts of the world to annihilate pests. In spite of the fact that they reduce the loss of economically important crops by controlling the phytopathogens, the utilization of agrochemicals poses extreme ecological risks, for example, leaching of nitrates and other compounds into the underground water table. 8 These agrochemicals are regularly non-degradable and accumulate in the food chain and could be a serious health hazard to the consumers. 9 An alternative strategy is the control of plant pests and diseases through biological processes, such as the use of entomopathogenic bacteria that either confine or antagonize other plant pathogenic microorganisms. The application of alternative strategies helps us to reduce the usage of agrochemicals in agriculture. 10
Some species of Pseudomonas have contributed to human societies due to their capabilities of producing different types of secondary metabolites. 9 These secondary metabolites have attracted much attention as they possess diverse bioactivities such as antibacterial, antifungal, antitumor, and antioxidant. 11 Achieving the dual effects of development in plant growth and pesticide potential of a microbe is an ideal strategy to control pests, as well as to improve crop yield. 11 Based on their insecticidal and plant growth-promoting effects, microbes like Pseudomonas can be used as biocontrol agents, which function as plant strengtheners, phytostimulators, and biopesticides. 6 The present study is an initial phase to analyze the plant development and insecticidal effects of Pseudomonas.
Materials and Methods
Isolation and Culture of Bacterial Strain
The bacterial strain was isolated from Calotropis procera collected from Jeddah, Kingdom of Saudi Arabia. Roots of the plants were cut off and surface sterilized using 2% sodium hypochlorite (NaClO) for 1 minute, followed by washing with autoclaved distilled water. Samples were placed on Luria Bertani (LB) agar plates and incubated at 28°C. The endophytic bacterial colonies which appeared on the plates were identified based on morphological characteristics. The insecticidal activities were assessed using G. mellonella larvae, and IUK001 was selected based on the best insecticidal activities.
Insect Breeding
The artificial medium was made by mixing autoclaved rice bran (600 g), wheat bran (600 g), water (175 mL), glycerol (250 mL), yeast extract (4.5 mg), CaCO3 (2 mg), and honey (600 mL). Finally, vitamin B complex (600 mg) was added to the autoclaved mixture. Eggs of G. mellonella in butter paper were added into 150 g of the medium and incubated at 25 ± 2°C. The small larvae were then transferred to a larger container containing a large amount of medium.
Insecticidal Bioassay Against G. mellonella Larvae
IUK001 was cultured in 50 mL LB broth (1% sodium chloride, 0.5% yeast extract, 1% tryptone) and incubated for 7 days at 30°C. The supernatant containing secondary metabolites, including insecticidal toxins, was collected after centrifugation of the culture at 10,000× g for 10 minutes. The insecticidal bioassay was accomplished by adding 1 mL/cm3 of an artificial diet to the first instar G. mellonella larvae. The supernatant of Escherichia coli DH5α culture was used as the control, and the experiment was repeated 3 times. The culture extract (CE) of IUK001 was exposed to heat treatment, ranged from 20°C to 100°C for 30 minutes, to analyze the effects of heat on the insecticidal activity. Furthermore, the CE was subjected to heat treatment at 70°C from 0 to 100 minutes to discover the association of heat treatment and time periods. After heat treatment, CE was mixed with artificial diet at a concentration of 1 mL/cm3 and was administered to larvae to investigate the insecticidal activity. In addition, proteinase-K was applied to affirm the fate of the toxins; if they were proteinaceous or non-proteinaceous. The CE was treated with 10, 20, 30, 40, and 50 mg/L of proteinase-K (10, 20, 30, 40, and 50 mg/L), and each treated sample was added into the diet of larvae to determine the insecticidal activity. Larvae, after the treatments, were incubated at 25 ± 2°C with 50% ± 5% relative humidity, and the mortality rate was assessed at an interval of 12 hours. Three replications (15 larvae/replication) were used per assay, and the experiment was repeated 3 times.
Identification of Isolate Using 16S DNA
Total genomic DNA (gDNA) of the isolate was extracted using a standard gDNA extraction protocol. 12 Universal 16S ribosomal DNA (rDNA) primers (27F and 1492R) were used to amplify the DNA using a polymerase chain reaction (PCR). Total PCR mixture of 50 µL/tube consisted of 1× PCR buffer, 0.2 pmol of each primer, 0.2 mM deoxynucleoside triphosphates, 2 mM magnesium chloride, and 5 U of Taq DNA polymerase (Takara, Japan). The PCR reaction consisted of 30 cycles; each cycle consisting of 96°C for 30 s, 54°C for 30 s, and 72°C for 1 minute. The initial denaturation at 96°C and a final extension at 72°C were extended for 5 minutes. PCR products were purified using a purification kit (purification kit, Solgent, Daejeon, Korea) and sequenced through the Sanger sequencing technique (Solgent, Daejeon, Korea). The sequence was analyzed through BLASTN available on NCBI, and the phylogenetic tree was constructed based on the similarity of nucleotide sequences using the neighbor-joining method in Mega 7.0 program. 13
Antibacterial Assay Using TTC
A serial dilution microplate colorimetric assay using 2, 3, 5-triphenyl tetrazolium chloride (TTC) was used to assess the minimum inhibitory concentration (MIC) of the bacterial extract. The bacterial strains were cultured in LB broth at 37°C for 24 hours. The IUK001 culture (1 L) grown at 37°C for 48 hours was extracted with 100% methanol (MeOH), freeze dried, and subsequently dissolved in MeOH at a concentration of 1 mg/mL. The TTC (5 mg) was dissolved in 1 mL of sterile distilled water and filtered through a 0.22 µm screen. The antibacterial activity was determined by adding 100 µL of IUK001 extract into 900 µL bacterial culture. The negative control consisted of 900 µL of the cultures to which was added 100 µL MeOH. Samples were incubated at 37 ± 2°C for 6 hours, and then 100 µL TTC solution was added. Afterward, the samples were incubated for 30 minutes more and then centrifuged for 10 minutes at 10,000× g. The pellets were suspended in 100 µL ethanol (50%) and absorption was noted at 510 nm. Living bacteria change TTC to formazan, which produces a red color. The microbial growth inhibition (%) was determined using the formula
Bioassay on Rice and Canola Plants
The CE was applied to plants to assess the effects of IUK001 on their health and growth. Rice (Oryza sativa L.) cultivar “Dongjin-beyo” was selected for the experiment. Seeds were treated with 0.1% NaOCl for 30 s followed by 75% ethanol for 1 minute for surface sterilization. Subsequently, the seeds were rinsed 5-7 times with deionized autoclaved water to remove traces of disinfectants. Seeds were germinated at 28°C on sterilized filter paper, placed in a Petri dish, soaked with autoclaved water, and incubated at room temperature. Equally germinated seeds were transferred into 0.8% agar (w/v) medium in pots (22 × 15 × 7 cm). The plants were grown in a growth chamber at 30°C for 16 hours (20,000 lx, light intensity) and 20°C for 8 hours per day, and the relative humidity was 70% ± 5%. The CE was prepared by culturing IUK001 in 1 L of LB broth and was incubated at 28 ± 2°C for 7 days. The culture broth was subjected to centrifugation at 10,000× g at 4°C for 15 minutes, and the supernatant was filtered through 0.45 µm filter and lyophilized using a freeze dryer. The lyophilized contents were added to distilled autoclaved water and 10 mL solution was applied to the plants. The experiment was conducted in the controlled environment of a growth chamber at 25 ± 5°C for the day and 20 ± 5°C for the night with 75% humidity for an interval of 15 days after treatments. The experiment consisted of 10 plants per treatment and 3 replications were used.
In the second phase of bioassay, Canola (Brassica napus L.) seeds were used for the experiment. The seeds were treated with 1.0% NaOCl and then washed with distilled to remove all traces of NaOCl. The sterilized seeds were then germinated in Petri dishes with autoclaved filter paper impregnated with 1.5 mL deionized distilled water and were incubated for 48 hours at room temperature. Equally germinated seeds were transferred onto 0.8% agar medium in Petri dishes, which were placed vertically at a 25° angle to let the root grow geotropically along the agar surface. The seedlings were incubated in a growth chamber at 30°C for 16 hours day (16,000 lx, light intensity) and 20°C for 8 hours night and the relative humidity was 70% ± 5%. The seedlings were treated with CE to determine the effects of IUK001 on roots and biomass of the plants. Deionized distilled water was used as the negative control. The experiment included 10 plants per treatment with 3 replications.
Quantitative Analysis of IPA and IBA by GC–MS
Culture broth of IUK001 (50 mL), incubated at 28 ± 2°C for 7 days on the shaker at 200 ± 10 rpm, was centrifuged for 10 minutes at 10,000× g to obtain the supernatant. This was filtered using a 0.45 µm cellulose filter and the filtrate was acidified using 1 N HCl to pH 2.8. The acidified solution was extracted 3 times using 2× volumes of . Organic layers, obtained after 3 rounds, were combined and subjected to vacuum drying at 45°C. The dehydrated contents were resuspended in 5 mL 100% MeOH and then evaporated in a rotatory evaporator at 45°C. The dried residues were resuspended in 0.5 mL EtOAc and subjected to chromatography using a polyvinylpyrrolidone column. A gradient of n-hexane–EtOAc (1:1, 1:4, 1:9 [v/v]) was used for elution. Indole positive fractions, screened through Salkowski’s assay, were combined together, dehydrated in a rotary evaporator at 45°C under vacuum and dissolved in 50% MeOH. A Sephadex LH-20 column (3 × 70 cm) was used to elute the active fractions with 50% MeOH at a rate of 0.5 mL/min. The eluent was dehydrated using a rotary evaporator at 45°C under vacuum and the dry residues were dissolved in 0.1 M acetic acid (5 mL); 1 mL was injected into the GC–MS. The remaining 4 mL was fractionated by high-performance liquid chromatography (HPLC; Varian, Inc., Palo Alto, CA), equipped with a C18 column. The solvent system consisted of 80% solvent A (1.0% acetic acid) and 20% solvent B (100% MeOH) at a rate of 0.5% mL/min. The detection range for indole compounds was between 254 nm and 280 nm and the expected indole fractions were collected and methylated with diazomethane; excess was evaporated through a sharp stream of oxygen-free nitrogen. The sample was subsequently dissolved in EtOAc prior to analysis through GC equipped with an MS using selected ion monitoring (SIM; 6890N network GC system and 5973 network mass selective detector; Agilent Technologies, Palo Alto, CA, USA). A 1 µL sample was injected into the GC–MS and the oven temperature was set at 60°C for 1 minute and then with a gradual rise at a rate of 15°C/min up to 200°C and then finally to 285°C at the rate of 5°C/min; the head pressure of helium was maintained at 30 kPa. The GC was linked with a mass-selected detector with an interface and ionizing voltage of 70 eV. Confirmation of the indole compounds was carried out by comparison of the retention time with pure indole-3-butyric acid (IBA) and indole-3-propionic acid (IPA) standards.
Statistical Analysis
Means and standard deviations of the values were calculated using MS-EXCEL (version 2007; Microsoft Inc., Redmond, WA, USA). The data were analyzed statistically for standard deviation using Graph-Pad online software. The mean values were compared using Student’s t-test with P < 0.05.
Results
Metabolite Profiling and Insecticidal Bioassay
IUK001 was cultured for 7 days at 28 ± 2°C in 250 mL of LB media and the extract was assessed through GC–MS (Table 1). In addition, the extract was bioassayed against larvae of G. mellonella to determine the toxicity of the extract. Figure 1(a) reveals that CE caused 100% mortality in larvae within 36 hours. In addition, paralysis symptoms appeared in the larvae after 12-18 hours of CE being added to the feed.

Insecticidal bioassay of the culture extract of IUK001 on Galleria mellonella larvae. (a) Mortality (%) caused by culture extract of IUK001 (closed circle) and culture extract of Escherichia coli DH5α (open circle) used as control supernatant. (b) Rate of mortality of culture extract after heat treatment from 20°C to 100°C for 30 minutes. (c) Toxicity caused by culture extract treated at 70°C from 0 to 100 minutes. (d) Rate of mortality of culture extract after proteinase-K treatment against G. mellonella larvae. All the experiments were repeated at least 3 times to calculate the means ± standard deviation. Each experiment was monitored for 3 days.
GC–MS Profiling of Compounds Present in the Culture Extract of IUK001.

Note. The identified constituents are listed in their order of elution. RT indicates the retention time of the compound.
In order to eliminate protein toxins, the extract was heat treated for 30 minutes from 20°C to 100°C. Figure 1(b) shows that CE treated at 70°C caused 100% mortality in G. mellonella larvae, but the mortality dropped to 65% and 40% when the temperature was increased up to 80°C and 90°C, respectively. The CE was also treated at 70°C from 0 to 100 minutes to investigate the effects time had on insecticidal activity. The level of insecticidal activity remained up to 90% after 60 minutes of heat treatment; however, a gradual decrease was observed as the time elapsed. Furthermore, after 80 and 100 minutes of heat treatment, the insecticidal activity was maintained at 70% and 60%, respectively, in comparison with the control (Figure 1(c)).
Besides the heat treatments, proteinase-K treatments were applied in the concentration range between 10 and 50 mg/L. Proteinase-K confirmed the non-proteinous status of the toxins present in the CE of IUK001. Figure 1(d) shows that there was a negligible effect of proteinase-K treatment and up to 90% of the insecticidal activity was preserved at all the applied concentrations of proteinase-K.
Antimicrobial Activity
The CE of IUK001 was used to evaluate its antimicrobial activity against 7 bacterial strains using TTC. The antibacterial activity of the EC was assessed quantitatively by determining the percent inhibition of bacterial growth as compared with the control. Table 2 shows the MIC and IC50 values of the EC against the tested bacterial strains. The lowest MIC and IC50 were 8 ± 1.8 and 4.6 ± 0.6 reported, respectively, against Enterobacter cowanii and the highest were 28 ± 0.8 and 18.9 ± 0.7 reported against Staphylococcus aureus.
Antibacterial Activity of Extract of IUK001 Using Chromogenic Reagent 2,3,5-Triphenyltetrazolium Chloride.

MIC, minimum inhibitory concentration; IC50, half-maximal inhibitory concentration.
The potency was assessed by MIC and IC50 Values of extract in comparison with control. The resultant IC50 values are means ± standard deviation of repeats (N = 3).
Prescreening of Indole Compounds
A colorimetric approach known as Salkowski’s assay was exercised to screen out the auxins (indole derivatives) produced by IUK001 in the culture broth. IUK001 produced indole compounds, predominantly auxins, in high concentration in the culture medium (Figure 2(a)).
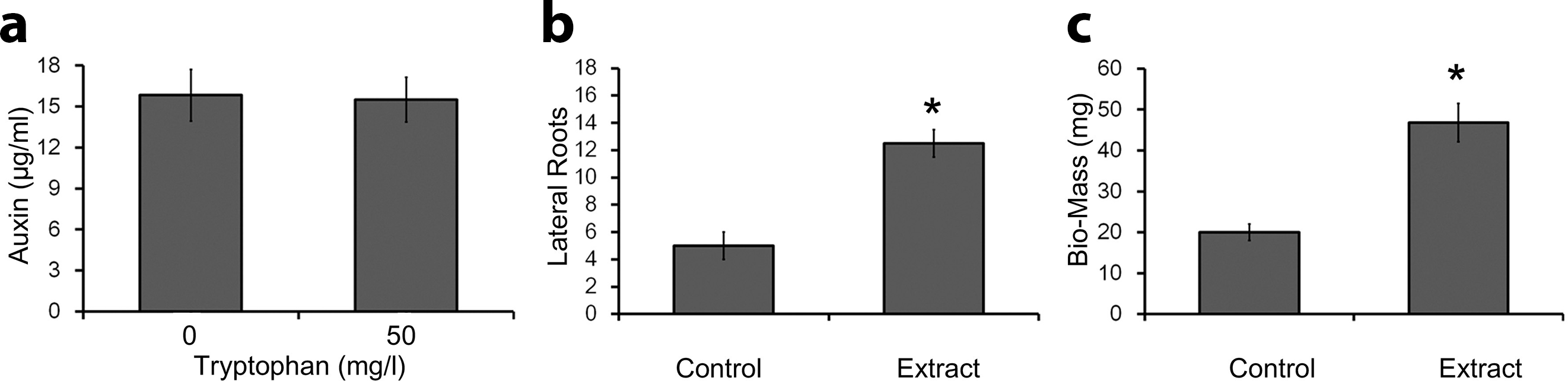
Calorimetric assessment of auxin from culture extract of IUK001 and root growth assessment on canola seedlings. (a) Estimation of auxin produced by IUK001 in culture broth supplemented with 0.0 and 50.0 mg/L of
Effects of Auxins on Root Dynamics
The CE of IUK001 was applied to plant roots in order to determine their growth dynamics. Figure 2(b) shows that lateral root growth was significantly induced after treatment with the CE of IUK001. In addition, the number of the lateral roots and their proliferation were prevailing as compared with control plants. In the course of the experiment, no significant effects of the extract were observed on the growth of primary roots, but lateral roots extensively proliferated after the treatments, indicating the characteristic feature of auxins. The biomass of the seedlings was also significantly increased as the addition of the lateral roots dominantly added to the biomass of the seedlings (Figure 2(c)).
Identification of Bacterial Isolate
The identification of Pseudomonas sp. IUK001 was based on 16S DNA sequencing. The query coverage was 99% and the nucleotides sequence showed more than 94% sequence similarity with Pseudomonas sp. of strains SB 980, SB 832, and SB 531. The phylogenetic analysis showed the relationship of IUK001 with Pseudomonas sp., shown in the phylogenetic tree (Figure 3). The sequence was submitted to Gen-Bank under the accession number MK521428.

Phylogenetic assessment based on 16S rDNA gene sequences from IUK001 and related strains of bacteria. CLUSTAL-W was used for sequences alignment and the phylogenetic tree was constructed through MEGA6 software by using the Neighbor Joining method.
Effects of IUK001 on Plant Health and Growth Dynamics
The effects of IUK001 on plant growth and development were determined through a bioassay of rice plants. These were treated with the EC of IUK001 and the results revealed that plant length of the treated plants was significantly increased (P < 0.05) as compared with the control (Table 3). These results also showed that the application of the EC increased the root length of rice plants significantly (P < 0.05) compared with the control. In addition, chlorophyll contents and biomass of the plants were also enhanced significantly (P < 0.05) in the treated plants in comparison with the un-inoculated control plants.
Effects of IUK001 Culture Extract on Growth Attributes of “Dongjin-Beyo” Rice Seedling.

CC, chlorophyll content; FB, fresh biomass; DB, dry biomass.
Values are expressed as mean ± standard deviation with n = 3.
An asterisk (*) represents significant differences in each column, compared with control; Student’s t-test was applied to calculate the significant variations (P < 0.05).
Identification of IPA and IBA by GC–MS Analysis
HPLC and GC–MS have been used widely for the precise measurement of metabolites, including auxins. During analysis, IPA and IBA were fractionated by HPLC, and reproducibility of the retention times of the 2 auxins was investigated with repeated injections of the standard. Under the optimum GC–MS conditions, the full scan mass spectra of the analytes were recorded for an initial trial and then analyses were done by acquiring compound-specific molecular ions in SIM to increase the signal-to-noise ratio of the MS experiment. GC analysis imparted a couple of peaks for IPA, 1 for the purified sample and the other for the internal standard at the same retention time (12.8 minutes). Similarly, the retention time for IBA peaks (standard and sample) was 14.8 minutes. The retention time of both standards and samples at a single point revealed that the samples were highly homogenized and purified. Further, the IPA and IBA were quantified on the basis of the peak area ratios of purified IPA and IBA to the internal standard.
The individual auxins from the ethyl acetate extract of the culture medium of IUK001 were determined. The GCs of purified IPA and IBA fall at 12.80 and 14.80 minutes, respectively, coincide with their respective internal standard. Quantification of endogenous IPA and IBA was accomplished with GC–MS–SIM. The retention time was determined with standards to calculate the Kovats retention index, while the quantification was based on the peak area ratios of extracts to standards. The major fragment ions were located at m/z 144 and 116 for IPA, and 116, 158, and 184 for IBA. The MS peak at m/z 116 represents the indole group, and the presence of the peak in the results indicates that the indole group and peaks represent the specificity of IPA and IBA.
Discussion
Pseudomonas spp. have been reported as entomopathogenic against various genera of insects including Lepidoptera (eg, B. mori, G. mellonella, Anopheles gambia, Drosophila melanogaster) and Coleoptera (eg, Sitophilus oryzae). 1 Genome analysis of Pseudomonas species showed that the genome is impregnated with a wide array of genes encoding insecticidal toxins, as well as other critical secondary metabolites. 2 A number of studies reported antifungal, antibacterial, and insecticidal activities in bacteria. 14
Bacteria are well-known sources of different bioactive metabolic compounds. GC–MS profiling of CE has shown different insecticidal compounds, for example, trans-cinnamic acid, ornithine, and cyclo (
The EC of IUK001 was evaluated against 7 bacterial strains and the results revealed significant activity against all the test strains. The antibacterial property of the microbe is actually an indirect mechanism of plant protection assessed by microbes. 22 The microbes act as a biocontrol agent, constraining the growth of phytopathogens, that is, bacteria and fungi through the production of antibiotics. 23 A number of antimicrobial metabolites produced by bacteria have been an alternative option to overcoming the increasing levels of drug resistance in pathogenic microbes causing different diseases. 24 Veselova et al 1 demonstrated that CF of Pseudomonas chlororaphis showed antibacterial activities against Bacillus subtilis and Staphylococcus aureus. A large array of antibiotic metabolites have been identified and reported from different natural sources, including Pseudomonas spp., showed strong bacteriostatic and fungistatic activities. 1
Besides, antibiotic and insecticidal activities, the results of the present study showed that IUK001 treatment enhanced plant length, chlorophyll content, and biomass of the rice plants as compared with the control. The effect of application of CE to the apex of the rice seedlings was promotive, suggesting the presence of bioactive secondary metabolites.
25
The secretion of bioactive metabolites by microbes in their culture broth exerts greater benefits to crop improvement and hence enhance the yield.
25
Previous studies showed that the application of bioactive CE to various vegetable and horticultural crops is good for improving plant growth and development.
25
-27
Plant bioassay was conducted in 0.8% agar medium to avoid the effects of other components such as soil and nutrients, in order to measure the sole effect of the microbial extract on the plants.
28
Previous studies have explained the mechanism of enhancement of plant growth resulting from the plant–microbe interaction.
1
Growth enhancement of the plant could be attained by the direct plant microbe and/or could also be attained indirectly by antagonistic effects against plant pathogens.
29
The CE of IUK001 was analyzed for bioactive auxins, which are potential plant growth promoters. Concentrations of the auxins produced in LB broth were unaltered by supplementation with
Conclusion
The discussion is concluded by findings of dual effects of IUK001. Due to such dual effects, IUK001 can be a strong candidate to be exploited as a multidimensional factor in agriculture. Insecticidal compounds such as cinnamic acid, ornithine, and cyclo (
Footnotes
Acknowledgments
This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah under grant no. G-431-130-39. The authors, therefore, acknowledge with thanks DSR for technical and financial support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Deanship of Scientific Research (DSR) at King Abdulaziz University, Jeddah under grant no. G-431-130-39.
