Abstract
Though natural polysaccharides commonly show antioxidant activities, the current research on the isolation of polysaccharides from Galla Turcica and their antioxidant activities still remain as an ongoing challenge. In this work, response surface analysis was employed to optimize an ultrasonic-assisted extraction method for polysaccharides of Galla Turcica. Their antioxidant and free radical scavenging activities were then evaluated using 2,2-diphenyl-1-picrylhydrazyl, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid), total antioxidant activity, and iron ion reduction assays. Moreover, the protective effects of polysaccharides of Galla Turcica were determined on human embryonic kidney fibroblast 293 and hepatoma 7721 cells by cell proliferation assay. Overall, the key parameters of Galla Turcica polysaccharides extraction were optimized as crushing degree 100 mesh, ultrasonic time 50 min, and materials–liquid ratio 1:50. The isolated polysaccharides presented dose-dependent antioxidant and free radical scavenging effects in vitro. It also demonstrated an effective protective effect for human cells under oxidative damage. The results firstly determined the antioxidant activities of polysaccharides from Galla Turcica, thus providing a new natural resource for future investigation and development of the polysaccharides-based antioxidant drugs, health products, or additives.
Galla Turcica, known as Turkish gallnuts, is formed on the tree Quercus infectoria Olivier parasitized by the gall wasps, Cynips gallae-tinctoriae Olivier. Galla Turcica is a dark green or brown sphere with a diameter of about 1-2.5 cm. Its surface is quite smooth but has tubers on the upper part. 1 In the traditional medicine, Galla Turcica is mainly used for the treatment of inflammatory diseases in Turkey and far eastern countries such as Thailand, Korea, and China. 2 Its anti-inflammatory activities were proved to be related to the inhibition of interleukin (IL)-6 and prostaglandin E2 (PGE2) release. 3 Moreover, Galla Turcica was also claimed to be used as an ingredient in many prescriptions for the treatment of oxidant damage-associated symptoms in traditional Chinese medicine (TCM) encyclopedia. 4
Based on its empirical uses for oxidative damage diseases, such as cancers, cardiovascular diseases, degenerative diseases, and metabolic diseases, Galla Turcica recently creates a great interest in the scientific community toward its antioxidant and free radical scavenging potentials. Gallic acid, ellagic acid, β-sitosterol, amentoflavone, and purpurogallin were most frequently investigated and considered as the antioxidant compounds of Galla Turcica. Their protective role in preventing various oxidative damage diseases have been widely reported toward the various application potentials in recent years. 5 -9 However, as the complex plant–insect symbiont, bioactive processes during galls formation must generate an abundance of bioactive metabolites in Galla Turcica. The understanding of its phytochemical knowledge thus clearly needs further complement.
Polysaccharides are biological macromolecules with immune regulation, antitumor, antivirus, antiradiation, anti-inflammation, antifatigue, and antiaging effects, which are related to their antioxidant properties. 10 -13 The polysaccharides isolated from natural products have been reported to be able to increase antioxidant enzyme activity, eliminate free radicals, inhibit lipid peroxidation, and protect the biofilm. 14 They thus have been identified as the main antioxidant ingredient of many herbal medicines. 15 -18 However, the antioxidant effects of Galla Turcica polysaccharides were barely reported. To underpin a fundamental understanding of bioactive materials and antioxidant activities of Galla Turcica and support its ongoing clinical uses in China, the aim of the study was to develop and optimize an ultrasonic-assisted extraction method by response surface analysis for polysaccharides isolation from Galla Turcica. Nevertheless, the antioxidant and free radical scavenging activities of extracted polysaccharides were also determined in vitro, especially on human cell models.
Materials and Methods
Plants and Cell Lines
Galla Turcica was purchased from Hao Yi Sheng Chinese herbal medicine store, Jinghua Street, Luoyang. The identity of original medicine was kindly confirmed as Galla Turcica by Professor Pu Liu at the School of Chemical Engineering and Pharmaceutics, Henan University of Science and Technology. A voucher specimen has been deposited in natural products research laboratory, School of Chemical Engineering and Pharmaceutics (access number: JGS2019-007).
The human embryonic kidney fibroblast cells 293 and human hepatoma cells 7721 were purchased from Beinglay Biotech (Wuhan, Hubei, China). Cells were grown in Dulbecco’s modified Eagle medium supplemented with 10% fetal bovine serum, 100 µg/mL penicillin, and 100 µg/mL streptomycin in a 5% carbon dioxide humidified incubator at 37°C. Experiments were performed when cells were approximately 80% confluent.
Polysaccharides Isolation
The polysaccharides of Galla Turcica were extracted using the water–alcohol precipitation method. 19 The dry herb was minced to the proposed crushing degree (60, 80, 100, or 120 mesh). The samples were then extracted with distilled water at a proposed ratio (1:10, 1:20, 1:30, 1:40, or 1:50) in the ultrasonic cleaning machine (150 W) for proposed minutes (10 minutes, 20 minutes, 30 minutes, 40 minutes, or 50 minutes) at 60°C. The samples were then centrifuged at 4000 rpm/min for 10 minutes and the supernatant was collected. The supernatant was mixed with absolute ethanol to achieve the final ethanol concentration at 80%. The mixture was standstill for 12 hours and centrifuged to precipitate the polysaccharides. The isolated polysaccharides were washed with distilled water and dried at −30°C under reduced pressure for 15 hours to produce the crude polysaccharides of Galla Turcica.
Response Surface Analysis
According to the results of single-factor test, factor A (crushing degree: 60, 80, and 100 mesh), factor B (ultrasonic extraction time: 20, 30, and 40 minutes), and factor C (materials–liquid ratio: 1:20, 1:30, and 1:40 g/mL) were selected for response surface analysis. In the response surface analysis of Design-Expert 8.0.6, Box-Behnken was selected to perform the experimental design with 3 factors and 3 levels. The coding and actual values were shown in Table 1, and the extraction rate was the response value.
Experimental Factors and Levels in Response Surface Analysis.

DPPH Free Radical Scavenging Assay
Polysaccharides and vitamin C were prepared at a concentration of 0.01, 0.03, 0.06, 0.12, 0.18, and 0.24 mg/mL. The 8.2 mg 2,2-diphenyl-1-picrylhydrazyl (DPPH) was dissolved in 250 mL absolute ethanol to produced 0.08 mmol/L DPPH working solution. The 3 mL DPPH working solution was mixed with 1 mL sample. The mixtures were then incubated at room temperature for 30 minutes and the absorbance was measured at 517 nm. DPPH free radical scavenging rate = (A 0 − (A S − A C))/A 0× 100%. A 0 is the absorbance of DPPH working solution + absorbance of distilled water; A S is the absorbance of DPPH working solution + absorbance of samples; A c is the absorbance of distilled water + absorbance of samples.
ABTS Free Radical Scavenging Assay
Polysaccharides and vitamin C were prepared at a concentration of 0.01, 0.02, 0.03, 0.06, 0.12, and 0.24 mg/mL. The 0.8 mL of 7.4 mmol/L 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) solution and 0.8 mL of 2.6 mmol/L potassium peroxodisulfate were mixed and reacted in dark for 12 hours. The prepared mixture was then diluted 50 times using absolute ethanol to produced ABTS working solution. The 2.4 mL ABTS working solution was mixed with 0.6 mL sample. The mixtures were then shaken for 6 minutes in dark and measured the absorbance at 734 nm. ABTS free radical scavenging rate = (A 0 − (A S − A C))/A0 × 100%. A 0 is the absorbance of ABTS working solution + absorbance of distilled water; A S is the absorbance of ABTS working solution + absorbance of samples; A c is the absorbance of distilled water + absorbance of samples.
Total Antioxidant Activity Test
Polysaccharides and vitamin C were prepared at a concentration of 0.06, 0.12, 0.18, 0.24, 0.30, and 0.36 mg/mL. The stock solution of phosphorus molybdenum reagent (0.6 mol/L sulfuric acid, 28 mmol/L sodium phosphate, and 4 mmol/L ammonium molybdate) was also prepared. One milliliter of each sample was mixed with 3.0 mL of phosphorus molybdenum reagent solution. The mixtures were placed in a 95°C water bath for 90 minutes. The samples were cooled down until room temperature and then measured the absorbance at 625 nm. The distilled water was used as a blank and vitamin C as positive control.
Iron Ion Reduction Assay (FRAP)
Polysaccharides and vitamin C were prepared at a concentration of 0.02, 0.04, 0.06, 0.08, 0.10, and 0.12 mg/mL. The ferrous sulfate (FeSO4) solution was also prepared at a concentration of 400, 200, 100, 50, and 25 μmol/L. Tripyridyl triazine (31.233 mg) was dissolved in 40 mL of 10 mmol/L HCL to produce 10 mmol/L 2,4,6-tris(2t-pyridyl)-s-triazine (TPTZ) solution. Then, 2.5 mL of 10 mmol/L TPTZ solution, 2.5 mL of ferric chloride hexahydrate, and 25 mL of 0.3 mmol/L natrium aceticum buffer solution were mixed to produce fluorescence recovery after photobleaching (FRAP) working solution. The 3.6 ml FRAP working solution (37°C) was mixed with 0.4 mL samples. The mixtures were shaken and the absorbance was measured at 593 nm. Series concentrations of FeSO4 solution were used to produce the standard curve, and then the FeSO4 concentrations were determined in the samples as FRAP values representing the antioxidant ability.
Cell Proliferation Assay
Cell suspension (200 µL) was seeded at a concentration of 1.5 × 104 cells/mL in a 96-well plate. After overnight incubation, hydrogen peroxide (H2O2) (1000, 10, and 0.1 µg/mL) and serial dilutions of polysaccharides (1000, 500, 100, and 10 µg/mL) were added in 10 µL aliquots. After 6 and 24 hours of incubation, 20 µL of MTT solution (5 mg/mL) was added and incubated at 37°C for 4 hours to develop color. The optical density (OD) was measured at 590 nm using a Multiskan Spectrum Reader. The cell survival rate was determined using cell survival rate = sample OD/control OD × 100%.
Statistical Analysis
The data were shown as mean ± standard deviation. Statistical comparison among treatments was carried out using one-way analysis of variance. The statistical significance between control and sample groups were calculated by the Student’s t-test. Data were taken as significant where P < .05.
Results
Optimization of Polysaccharides Isolation From Galla Turcica
In the single-factor experiment, crushing degree, ultrasonic time, and materials–liquid ratio were selected to be the parameter optimized during the polysaccharides isolation of Galla Turcica. As shown in Figure 1, the extraction rate of polysaccharides increased at first and then decreased at the different crushing degrees, ultrasonic time, and materials–liquid ratios. The extraction rate reached the top value at 100 crushing meshes, 40 minutes, and ratios of 1:30, respectively. The 80, 100, and 120 meshes; 30, 40, and 50 minutes; and ratios of 1:30, 1:40, and 1:50 were chosen to be used in the following response surface analysis.

Single-factor experiment of polysaccharides isolation from Galla Turcica (P < .001).
As shown in Table 2, the parameters selected above were used to perform the response surface analysis using Box-Behnken of Design-Expert 8.0.6. The results obtained were then merged and optimized to produce the formula:
Experimental Scheme and Results.


As shown in Table 3, the model P value was .001 and the missing P value was .358, which indicated that the model was fully fitted and could well describe the relationship between each factor and the response value. The determination coefficient R 2 of the regression equation above was 0.9435. The corrected correlation coefficient R adj2 was 0.8709, which showed that the variability of the test data could be explained by this regression model. Therefore, the model could be used to predict the extraction rate of Galla Turcica polysaccharides.
Variance Analysis.

*P < .05 significant; **P < .01 highly significant.
Moreover, the P value of primary items A and C was less than .05 with a significant difference. The P value of quadratic term item AC and C2 was less than .05 with a significant difference. The P value of primary item B and quadratic term item A 2 was highly significantly different. The P value of quadratic term item AB, BC, and B2 was not significantly different. The results showed that the extraction rate of Galla Turcica polysaccharides was not determined by a single factor but by a variety of interactive factors. According to their P values, it could be concluded that the influence levels were ratio of materials–liquid (C) > ultrasonic time (B) > crushing degree (A).
As shown in the interaction diagram (Figure 2), the AB factor could produce the optimal value under the experimental conditions, and the AC factor and the BC factor could also simulate the maximum value in this range. The optimum extraction conditions for polysaccharides of Galla Turcica were determined using the regression model above as follows: crushing degree 100 mesh, ultrasonic time 50 minutes, and materials–liquid ratio 1:50. The predicted value of the extraction rate under those conditions was 0.64%. The actual value was then determined as 0.63%, which accurately verified the feasibility of the model.
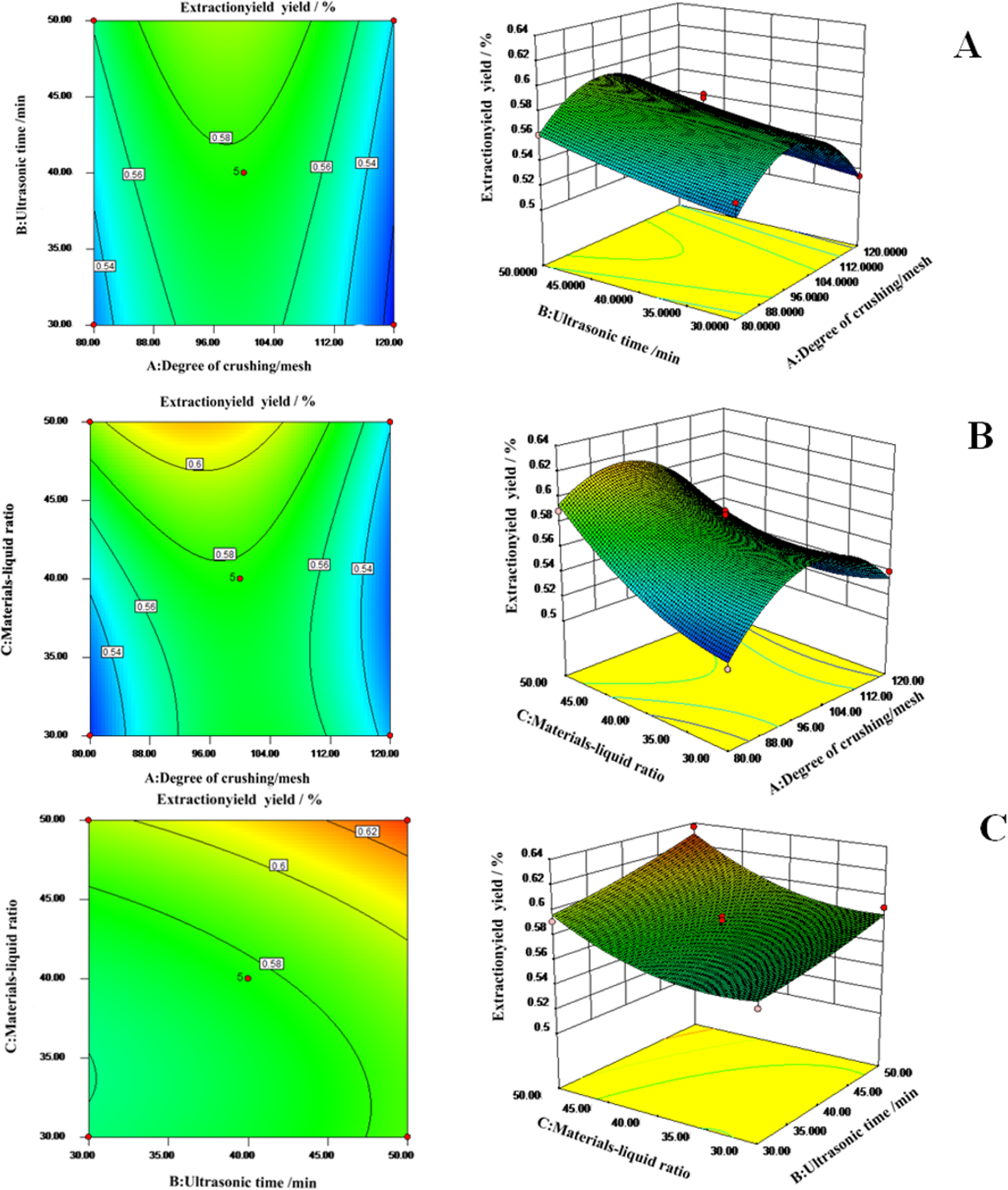
Effects of (a) degree of crushing/mesh and ultrasonic time; (b) degree of crushing/mesh and material–liquid ratio; and (c) ultrasonic time and material–liquid ratio (c) on extraction yield.
Antioxidant and Free Radical Scavenging Activities of Galla Turcica Polysaccharides Through Chemical Analysis
As shown in Figure 3(a, b), the polysaccharides of Galla Turcica presented strong dose-dependent effects for both DPPH and ABTS free radical scavenging. The maximum DPPH and ABTS clearance rates were over 85% and 88% when the concentration of polysaccharides reached 0.06 and 0.01 mg/mL, respectively. Moreover, polysaccharides of Galla Turcica demonstrated a stronger free radical scavenging effect than that of vitamin C against ABTS in the test. It could clear over 88% of free radicals at a concentration of 0.01 mg/mL, while vitamin C cleared only 36% at the same concentration (Figure 3(b)). Compared with vitamin C, the antioxidant activity of Galla Turcica polysaccharides were relatively weak in the total antioxidant assay. At the dose of 0.36 mg/mL (maximum value tested), the antioxidant effects of polysaccharides were about 25.9% of those of vitamin C (Figure 3(c)). In the iron ion reduction assay, both vitamin C and Galla Turcica polysaccharides demonstrated the effective reductive ability of ferrous ions in a dose-dependent manner. The reduction ability of polysaccharides was significantly higher than that of vitamin C solution. The reduction amount of ferrous ion in vitamin C solution could reach 17.40 μmol/L, and the reduction amount of polysaccharides could reach 153.51 μmol/L at the lowest concentration of the experimental range (Figure 3(d)).
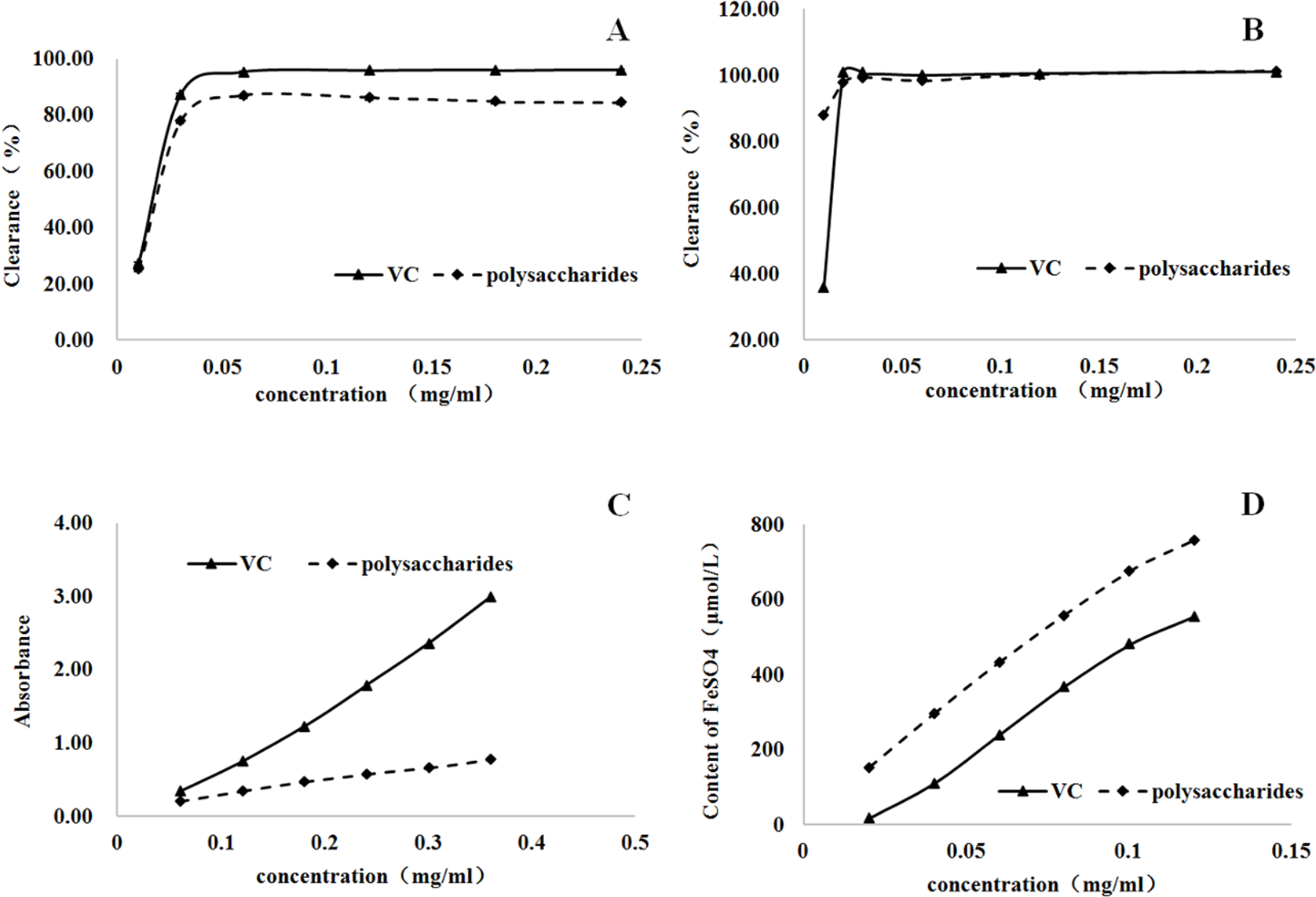
Antioxidant and free radical scavenging activities of Galla Turcica polysaccharides (P < .001). (a) 2,2-Diphenyl-1-picrylhydrazyl assay; (b) 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) assay; (c) total antioxidant activity test; and (d) iron ion reduction assay (fluorescence recovery after photobleaching).
Taken together, polysaccharides of Galla Turcica presented nearly similar free radicals scavenging effects as vitamin C. It also showed the reductive ability of ferrous ions which was stronger than that of vitamin C. However, its antioxidant effect was not as strong as that of vitamin C.
Cellular Antioxidant Activities of Galla Turcica Polysaccharides
As shown in Figure 4, polysaccharides of Galla Turcica presented effective protection against H2O2-induced oxidative damage on the human embryonic kidney fibroblast cells 293 and human hepatoma cells 7721 through a dose-dependent manner after 6 and 24 hours of treatment.

Cellular antioxidant activities of Galla Turcica polysaccharides; (a) 293 cells, 6 -hour treatment; (b) 293 cells, 24-hour treatment; (c) 7721 cells, 6-hour treatment; and (d) 7721 cells, 24-hour treatment.
In both tested 293 and 7721 cells, the higher concentration of the H2O2 could lead to the lower survival rate of the cells. After polysaccharides were added, the cell survival rate significantly increased. At the maximum concentration of polysaccharides (1000 µg/mL), the 293 cell survival rate reached 51%, 85%, and 93% in 1000 µg/mL, 10 µg/mL, and 0.1 µg/mL H2O2 treatment groups at 6 hours, respectively (Figure 4(a)). At 24 hours, these numbers were 85%, 81%, and 80% in 1000 µg/mL, 10 µg/mL, and 0.1 µg/mL H2O2 treatment groups, respectively (Figure 4(b)).
In 7721 cells, polysaccharides of Galla Turcica showed greater antioxidant activities as well. The higher concentrations could achieve a higher survival rate of cells. At the concentration of 1000 µg/mL, polysaccharides could protect 93%, 92%, and 93% cells out of oxidative damage by 1000 µg/mL, 10 µg/mL, and 0.1 µg/mL H2O2 treatment groups at 6 hours, respectively (Figure 4(c)). These protective effects could last at least 24 hours. The cell survival rate could be maintained at 77%, 70%, and 80% in 1000 µg/mL, 10 µg/mL, and 0.1 µg/mL H2O2 treatment groups at 24 hours, respectively (Figure 4(d)).
Discussion
Natural polysaccharide isolation optimized by response surface analysis has been exhaustively reported and reviewed elsewhere. 20 -22 In the single-factor isolation tests, crushing degree of raw materials, ultrasonic time, and materials–liquid ratio were determined as the key factors. In the current study, it was found that the smaller size of Galla Turcica could enhance the extraction rate by increasing the contact area between herbal powders and extracting liquid. However, size materials smaller than a certain degree (100 mesh in this case) may lead to the accumulation and caking of raw materials, thus decreasing the extraction rate. Similarly, the higher materials–liquid ratio enhances the mutual contact and extraction between materials and solvent. The extraction rate reached the top when the materials–liquid ratio increased to 1:30 and roughly remained at the same level. Moreover, ultrasound time also aided the extraction of polysaccharides. But the extensive period of ultrasonic concussion may risk the destruction of polysaccharide macromolecule. 23 In this study, the constructed regression model accurately predicted the extraction rate of polysaccharides. The actual extraction value was validated as 0.63%, which was closed to the predicted value (0.64%) to a great extent with a fractional error of less than 5%.
The chronic oxidative damage, caused by excessive free radicals, has been exemplified to increase the risk of diseases such as cancers, type 2 diabetes mellitus, coronary artery disease, atherosclerosis, and neurodegenerative disease. 24,25 In this study, the antioxidant and free radical scavenging activities of polysaccharides from Galla Turcica were found to be as strong as those of vitamin C and much stronger than those of many natural polysaccharides for the first time. To achieve over 80% DPPH radical scavenging rates, Lilium davidii, Hovenia dulcis, and Viscum coloratum polysaccharides should reach 2, 4, and 6 mg/mL, respectively. 26 -28 However, the concentration of polysaccharides from Galla Turcica only needs as low as 0.06 mg/mL to achieve a similar effect. To achieve 60% ABST radical scavenging rates, the concentration of polysaccharides was 2 mg/mL and 4 mg/mL for H. dulcis and L. davidii, respectively. 27,28 And only 0.01 mg/mL Galla Turcica polysaccharides could eliminate about 88% ABST radicals.
High levels of free radicals could initiate lipid peroxidation, cause oxidative damage to cellular proteins, membrane lipids, and nucleic acids, resulting in cell injury. 29 In this study, the protective effects of Galla Turcica polysaccharides under oxidative damage were firstly determined on human embryonic kidney fibroblast cells and human hepatoma cells in vitro. Under strong oxidant agent and H2O2 treatment, Galla Turcica polysaccharides could effectively increase cell survival compared with cells without protection. And this protection could last at least 24 hours since the survival rates of cells remained from 6 hours to 24 hours. The antioxidant pharmacological mechanisms of natural polysaccharides have been related to the regulation of various signaling pathways through a wide range of enzymic and non-enzymatic antioxidants. Therefore, further research was highly suggested to investigate the roles of polysaccharides of Galla Turcica in the regulation of those potential targets, such as superoxide dismutase, catalase, malondialdehyde, reduced glutathione, glutathione peroxidase, and glutathione reductase. 30 -32 Taken together, the work determined polysaccharides as a bioactive antioxidant of Galla Turcica on both chemical and cellular models for the first time.
Conclusion
This study discovered the antioxidant activities of polysaccharides from Galla Turcica for the first time that could be beneficial for future investigation and development of polysaccharide-based drugs, health products, or additives. An ultrasonic-assisted method of polysaccharide extraction was developed, and the key parameters were optimized as crushing degree 100 mesh, ultrasonic time 50 minutes, and materials–liquid ratio 1:50. The isolated polysaccharides presented dose-dependent antioxidant and free radical scavenging effects in DPPH, ABTS, total antioxidant, and iron ion reduction assays. Moreover, it also demonstrated an effective protective effect for human cells under oxidative damage. This work also demonstrated the potential of response surface analysis as a tool for extraction optimization of bioactive compounds from herbal extracts efficiently and accurately. This study will help the researcher to uncover the critical areas of bioactive constituents from gallnuts medicine that many researchers were not able to explore. Thus, a new theory on the development of gallnuts polysaccharide-based medicine against oxidative damage diseases may be arrived at.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
