Abstract
Several natural drugs (termed prodrugs) when administered orally undergo transformation by intestinal bacteria, producing metabolites, which may be more active than the parent compound. Mangiferin (
Many natural products are available worldwide as potential cancer chemoprotective agents and are present in vegetables, fruits, plant extracts, and herbs. The xanthone glycoside mangiferin (1,3,6,7-tetrahydroxyxanthone-C2-beta-
Between 5% and 7% of drugs approved worldwide are reported to be or are classified as prodrugs. 11 Several glycosides of natural products are prodrugs and an example are the sennosides, which have been used for many years as laxatives. Sennosides taken orally are hydrolyzed by the colonic microflora to liberate the active rhein anthrones. 12 The intestinal microflora produces a variety of glycosidases, and therefore, a number of synthetic glycosides have been examined as potential colon-specific delivery systems, in particular corticoids. A similar strategy was applied to the colon-specific delivery of corticosteroids used to treat inflammatory bowel disease. 11,13 Davis 14 reported that all drugs which are absorbed into the bloodstream are subjected to hepatic metabolism. Most administered drugs are rapidly and completely absorbed in the upper gastrointestinal tract and therefore do not come into contact with the bacterial flora of the large bowel. However, Biopharma is providing an increasing number of new drug candidates that have low solubility, low permeability, or both. Drugs that display these properties enter the large bowel and are subjected to metabolism by the indigenous bacterial flora therein and the metabolites may be absorbed by the colonic mucosa.
Several researchers have reported that the bioavailability of orally administered mangiferin is very low,
15,16
and in rats, it has been shown to be as low as 1.2%,
16
but there is no data on blood levels of its major metabolite norathyriol in humans. Pure norathyriol is extremely expensive, and therefore, we sought a local source for the production of sufficient material to assess the chemopreventive capacity of this compound against human cancer cell lines. Human intestinal bacteria are reported to deglycosylate
Brief Profiles of the Human Volunteers (

BMI, Body mass index.
Fecal inocula of 1 of the volunteers (V1) consistently degraded mangiferin (
Following isolation and purification of
The data shows that the transformation of mangiferin to bacterial metabolites was extremely variable even in this very small pilot study using a fecal matrix donated by 3 male volunteers. V1 metabolized mangiferin (Figure 1) extremely rapidly with over 80% conversion to metabolites in 12 hours and was virtually complete at 120 hours. With V2, metabolism (Figure 2) was incomplete at 120 hours and reached a maximum of 32% at 48 hours with no increase thereafter. On the other hand, the metabolism of mangiferin was not detected with fecal inocula from V3 and mangiferin remained unchanged after 120 hours. The data for the time-course experiments are shown in Figure 2.

Transformation at 120 hours of mangiferin (1) to norathyriol (2) and homomangiferin (3) by volunteer 1.

Transformation of mangiferin to norathyriol by human fecal matrix from volunteer (V) 1 to V3.
These results were extremely reproducible when conducted in triplicate and on 3 different occasions. Therefore V1, V2, and V3 can be considered as ultrarapid, intermediate, and non-metabolizers of mangiferin, respectively.
20
The major metabolite in the fermentation broths produced by V1 and V2 was initially tentatively identified as norathyriol (
High Performance Liquid Chromatography–Electrospray Ionization-Mass Spectrometry Data in Negative Ion Mode of Mangiferin (I) and Its Bacterial Metabolites, Norathyriol (
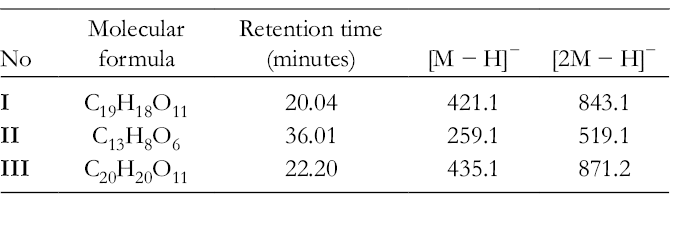
The ultraviolet spectra of mangiferin (
Fermentation of mangiferin with the ultrarapid metabolizer V1 was repeated 10 times, and following fractionation of the fermentation broths by SPE on C18 columns, fractions enriched in the metabolites were subjected to semipreparative HPLC. The yield of metabolites

1H nuclear magnetic resonance (NMR) spectrum at 600 MHz of mangiferin (
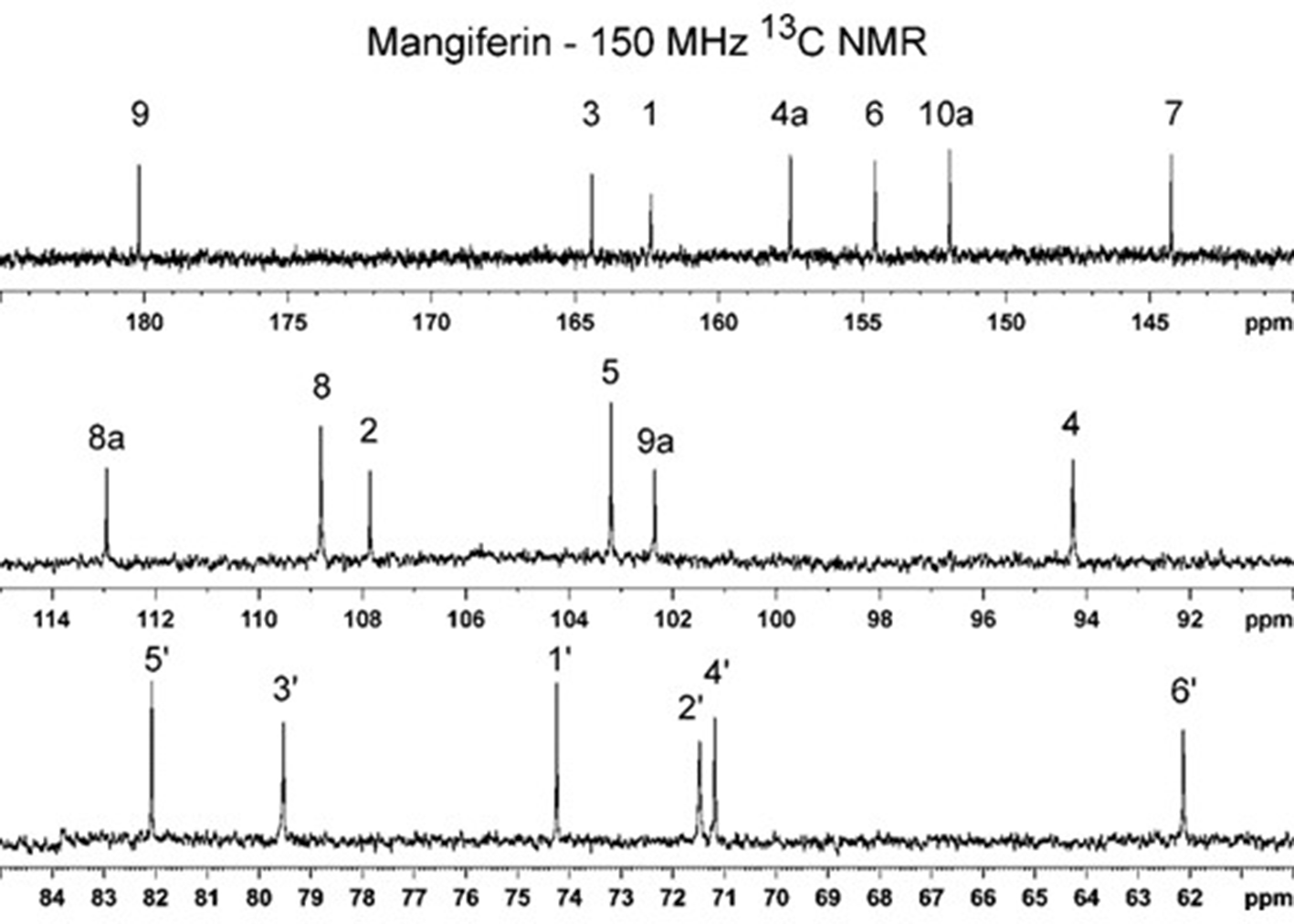
13C nuclear magnetic resonance (NMR) spectrum at 150 MHz of mangiferin (
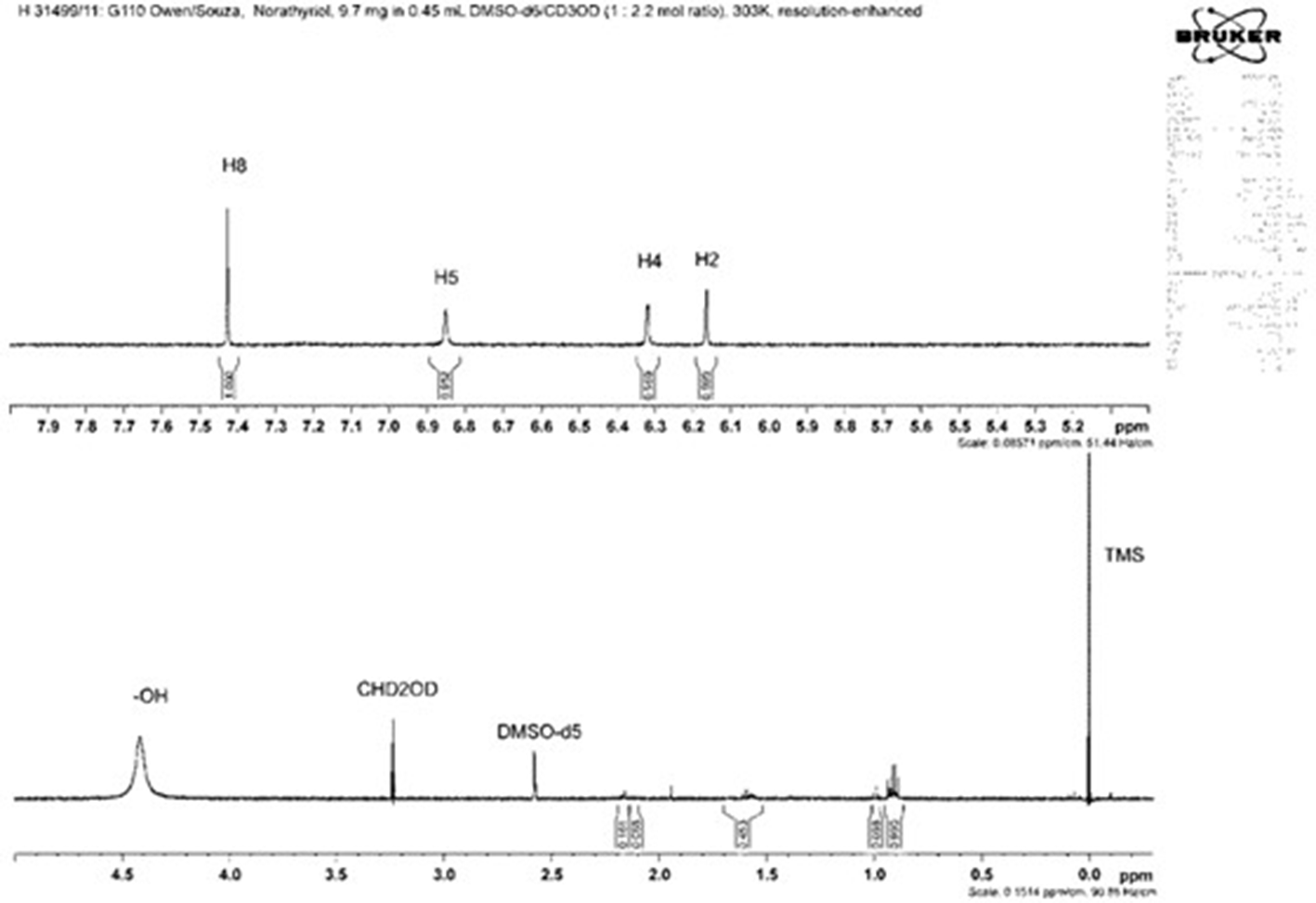
1H nuclear magnetic resonance spectrum at 600 MHz of norathyriol (

13C nuclear magnetic resonance spectrum at 150 MHz of norathyriol (
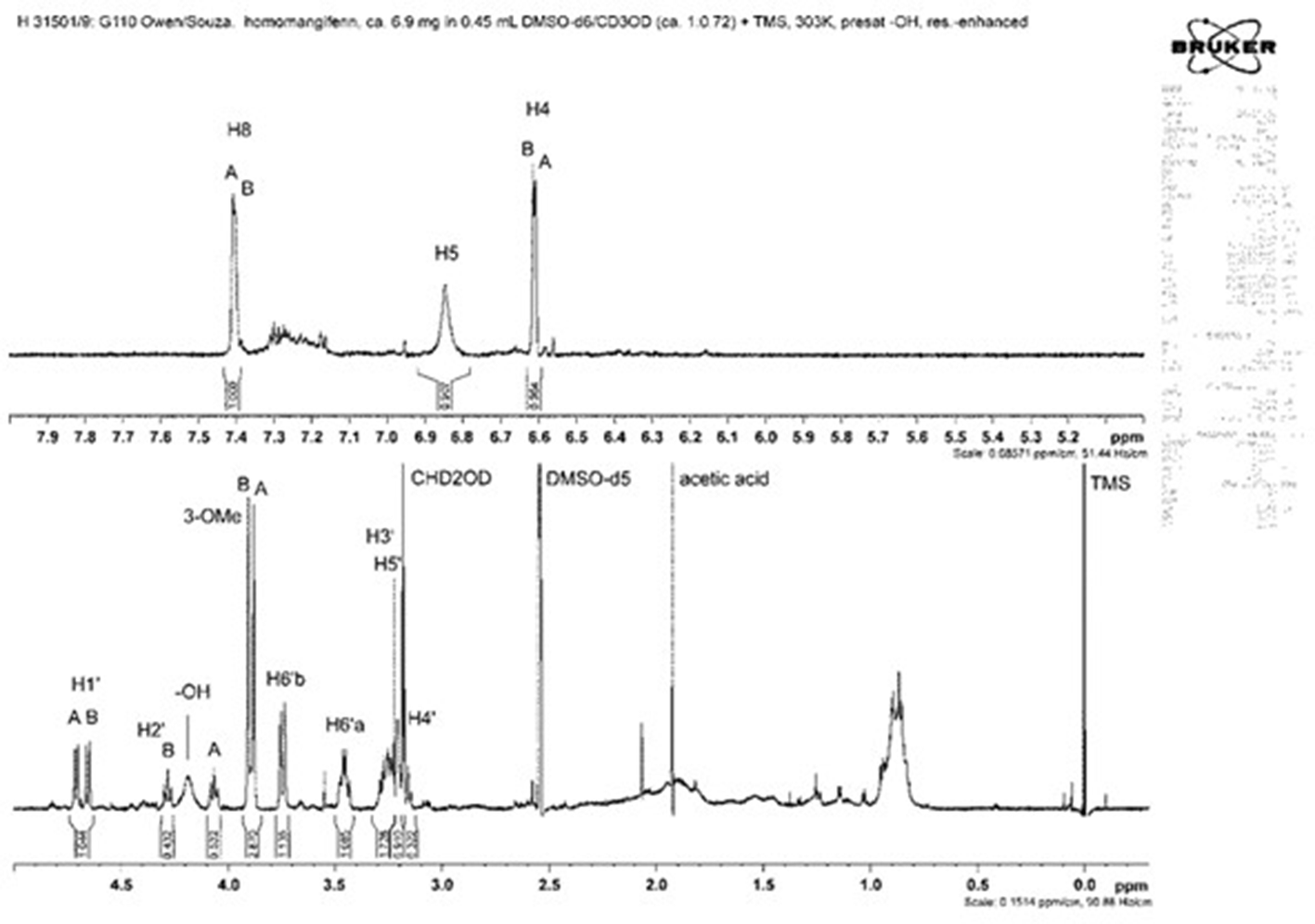
1H nuclear magnetic resonance spectrum at 600 MHz of homomangiferin (
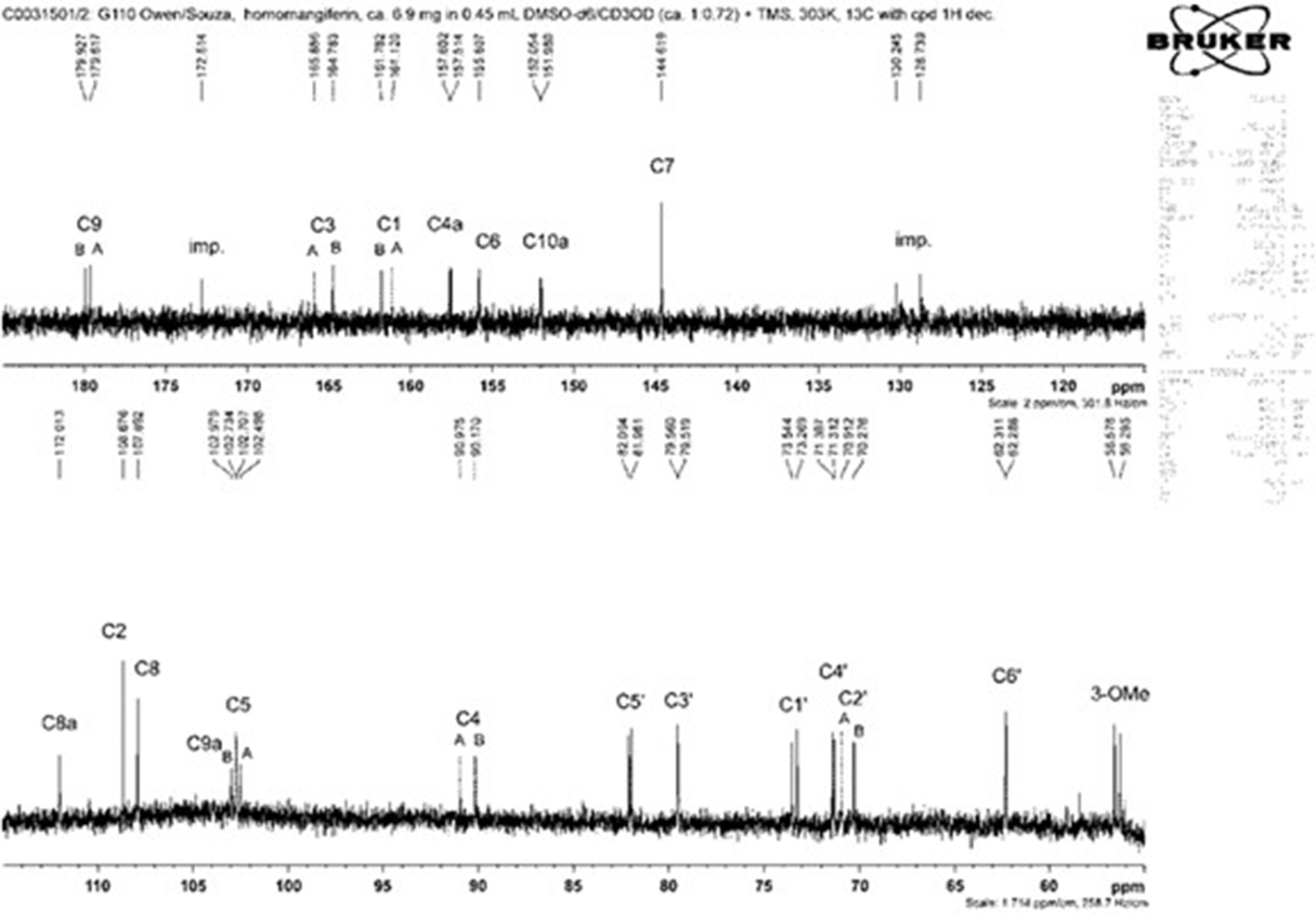
13C nuclear magnetic resonance spectrum at 150 MHz of homomangiferin (
The 1H and 13C NMR data for mangiferin (
The structures of mangiferin and its bacterial metabolites are depicted in Figure 9. The fermentation data for V1 is very similar to the in vitro data of Huang et al
23
who reported that after 12 hours of incubation with human intestinal bacteria, extensive conversion of mangiferin to norathyriol occurred. The bacterial species of the intestinal microflora responsible for deglycosylation of mangiferin has been identified as

Structures of the substrate mangiferin (
There is only 1 report of a pharmacokinetic study on mangiferin in human plasma. A detailed study was conducted by Hou et al 15 in which human volunteers were administered a single dose of mangiferin (0.1, 0.3, and 0.9 g, respectively, to 3 groups of 7 volunteers) with 200 mL of water. Analyses of plasma samples by HPLC-ESI-MS revealed maximal concentrations of mangiferin at 1 hour of 19.94 ± 3.47, 34.70 ± 6.83, and 38.64 ± 6.75 ng/mL with increasing dose, which is equivalent to 47.25, 82.23, and 91.56 nM. At this time point, these values represent 0.10%, 0.06%, and 0.02% of the administered doses of mangiferin. They concluded that the pharmacokinetics of mangiferin in plasma were not linear. However, no attempt was made to evaluate the presence of possible glucuronide or sulfate derivatives in plasma, feces, or urine.
As yet, there are no reports on the plasma concentrations of the major metabolite of mangiferin, namely, norathyriol in human plasma. However, an intervention study was conducted by Bock et al,
24
with mature female pigs administered 74 mg/kg of mangiferin/day in the form of a
The data reported by these authors show that approximately 80% of the administered dose of mangiferin was unaccounted for, based on the ratio of intervention and biospecimen levels. In a further study, aimed at the identification of further bacterial metabolites of norathyriol, Bock et al 25 detected 4 phenolic acid metabolites ostensibly derived from mangiferin in pig feces, but they were below the limits of quantitation. The fate of the remainder is as yet unknown. Fermentation studies with human bacterial microflora of norathyriol are underway in our laboratories to establish whether or not complete metabolism through to CO2 is evident.
In conclusion, mangiferin can be fermented in vitro to a major (>95%) metabolite namely norathyriol by the fecal matrix. Norathyriol is less effective as an antioxidant compared with the substrate mangiferin (Table 3) but has a far greater (49%; 13.0 µg/mL) anticarcinogenic capacity, as tested against colon and lung cancer cell lines. Screening of the general human population, with regard to the efficacy of the fecal metabolome to produce norathyriol from mangiferin, in tandem with its bioavailability, is therefore warranted.
Antioxidant Capacites of Mangiferin (I) and Norathyriol (

DPPH, 2,2'-diphenyl-1-picrylhydrazyl ; FRAP, ferric reducing antioxidant power; ORAC, oxygen radical absorbance capacity; IC50, half-maximal inhibitory concentration; EC50, half-maximal effective concentration.
Experimental
Reagents and Materials
Mangiferin was purified and characterized as described by Barreto et al,
2
and an authentic standard was also obtained from Extrasynthese (Lyon Nord, Genay, France) for comparison. Acetic acid, acetonitrile, brain heart infusion broth (BHI), K2HPO4, KH2PO4 were obtained from Merck (Darmstadt, Germany); methanol from Carl Roth (Karlsruhe, Germany); Duran bottles,
Cell Cultures
The A240286S (non-small lung cancer adenoma) and Caco-2 (colon cancer) cell lines utilized in this study were obtained from the in-house collection of the German Cancer Research Center (Heidelberg, Germany).
Stool Sampling
Stool samples from each volunteer were collected on the same day. Aliquots (approximately 500 mg) prepared in an atmosphere of nitrogen were stored at –80°C for use in the in vitro experiments.
Anaerobic Fermentation of Mangiferin
The BHI broth was prepared by dissolving the powdered material in double distilled water at a concentration of 37 g/L fortified with the reducing agents sodium formaldehyde sulfoxylate (0.3 g/L) and
HPLC–ESI-MS
HPLC–ESI-MS was conducted on an Agilent 1100 HPLC, coupled to an Agilent single quadrupole mass-selective detector (HP 1101; Agilent Technologies, Waldbronn, Germany). Chromatographic separation of methanol extracts was conducted using a C18, Gemini reverse-phase (5 µm), column (250 × 4.6 mm I.D.) Phenomenex (Aschaffenburg, Germany). The mobile phase consisted of 2% acetic acid in doubly distilled water (solvent A) and acetonitrile (solvent B) with the following gradient: initially 95% A for 10 minutes; to 90% A in 1 minutes; to 80% A in 9 minutes; to 60% A in 10 minutes; to 40% A in 10 minutes; to 0% A in 5 minutes; and continuing at 0% A until completion of the run. Detection of phenolic compounds was by means of UV absorbance (A) at 257, 278, 320, and 340 nm at 30°C. Mass spectra in negative ion mode were generated under the following conditions: fragmentor voltage, 100 V; capillary voltage, 2500 V; nebulizer pressure, 30 psi; drying gas temperature, 350°C; and mass range, 100-1500 D. Instrument control and data handling were by means of an HP Chemstation operating in the Microsoft Windows software environment.
Chromatography on C18 SPE Columns
Fermentation of mangiferin with the ultrarapid metabolizer V1 was repeated 10 times and completed fermentation broths were placed in 50 mL Falcon tubes and spun at 5000 rpm for 15 minutes. The supernatants were decanted and applied to C18 columns for subfractionation. The columns were preconditioned with methanol (50 mL) and double distilled water (50 mL) and were not allowed to dry. Elution was performed with solvent mixtures containing increasing concentrations of methanol (0%, 5%, 25%, 50%, and 100%) in 2% acetic acid, resulting in 5 fractions. The solvent was removed on a freeze drier (Christ, Gefriertrocknunsanlagen, Osterode, Germany). Individual phenolic compounds in relevant fractions were purified by semipreparative HPLC for structure elucidation by spectroscopic analyses.
Semipreparative HPLC
Semipreparative HPLC was conducted on an HP 1100 liquid chromatograph (Agilent Technologies, Waldbronn, Germany) fitted with an Agilent C18 reverse-phase Zorbax Eclipse Plus Phenyl Hexyl column (250 × 10 mm I.D.) under the conditions described by Owen et al 26 but at 30°C.
Nano-ESI-MS
Mangiferin and the bacterial metabolites were analyzed as described by Owen et al 27 after purification by semipreparative HPLC.
NMR Spectroscopy
NMR spectra for all compounds were recorded on a Bruker Avance II(III) spectrometer equipped with a 5 mm inverse(normal)-configuration probe with triple(z)–axis gradient capability at a field strength of 14.1(9.4) T operating at 600–1(440.1) and 150.9(100.6) MHz for 1H and 13C nuclei, respectively, at 303 K. The xanthones were poorly soluble in CD3OD (99.8% D), typically 3–10 mg in 4.0 mL, so that a 3:1 mixture of CD3OD/dimethyl sulfoxide-
MTT Assay
The cytotoxicity of the xanthones (
Antioxidant Assays
The antioxidant assays were conducted exactly as described by Maia et al. 29
Statistics
The concentration of xanthones producing 50% cytotoxicity (IC50) against the cancer cell lines was determined using the Table Curve program (Jandel Scientific, Chicago, IL, USA).
Supplemental Material
Supplementary material - Supplemental material for Transformation of Mangiferin to Norathyriol by Human Fecal Matrix in Anaerobic Conditions: Comprehensive NMR of the Xanthone Metabolites, Antioxidant Capacity, and Comparative Cytotoxicity Against Cancer Cell Lines
Supplemental material, Supplementary material, for Transformation of Mangiferin to Norathyriol by Human Fecal Matrix in Anaerobic Conditions: Comprehensive NMR of the Xanthone Metabolites, Antioxidant Capacity, and Comparative Cytotoxicity Against Cancer Cell Lines by José R. R. Souza, Maria Teresa Salles Trevisan, Judith P. A. Feitosa, Nágila M. P. S. Ricardo, William E. Hull, Gerhard Erben, Gerd Würtele, Andrea Breuer, Eva Frei, Cornelia M. Ulrich and Robert W. Owen in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The authors should like to express their thanks for financial support from the CNPq, CAPES/DAAD Sandwich Doctorate program (Brazil).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
