Abstract
Ten compounds were separated and purified by chromatographic methods from the 70% ethanol extract of the leaves of Eleutherococcus sessiliflorus (Rupr. & Maxim.). Their structures were identified according to their physicochemical properties and the spectral data of dihydromyricetin (
The leaves of Eleutherococcus sessiliflorus (Rupr. & Maxim.) S.Y.Hu, commonly known as Ciwujia, have been used in traditional Chinese medicine for the treatment of rheumatism. 1 It has been reported that E. sessiliflorus is rich in flavonoids, 2 terpenoids, 3 -5 and xylogens. 6 As a continuation of our phytochemical investigations, 6 flavonoids, 3 phenolic acids, and 1 triterpenoid were isolated from the n-butanol solvent fraction of the leaves of E. sessiliflorus. Herein, we report the isolation, structural elucidation, and cytotoxic activity of the compounds.
Results and Discussion
The 70% ethanol extract of the dried leaves of E. sessiliflorus was subjected to multiple chromatographic steps over silica gel, Sephadex LH-20 and recycling preparative high-performance liquid chromatography (HPLC). Ten compounds were obtained: dihydromyricetin (

Structures of compounds 1
The Eleutherococcus Maxim. (Acanthopanax [Decne. Et Planch] Witte) is a genus of 38 species growing in Eastern Asia, from the Himalaya to Vietnam, and from Northeastern Russia to North Philippines, with 18 of them coming from China.
17
The main chemical substances are eleutherosides and flavonoids. The roots of Eleutherococcus senticosus are a source of flavonoids (hyperin, rutin, afzelin, quercetin, naringenin, and kaempferol), phenylpropionic acids, triterpenic acids, and anthocyanins.
18
Compounds
The in vitro cytotoxic activities of all compounds are shown in Table 1. The 6 flavonoids, the 3 phenolic acids, and the triterpenoid isolated from the leaves of E. sessiliflorus showed weak cytotoxicity against the A549 cell line.
Cytotoxicity Data of Isolated Compounds 1
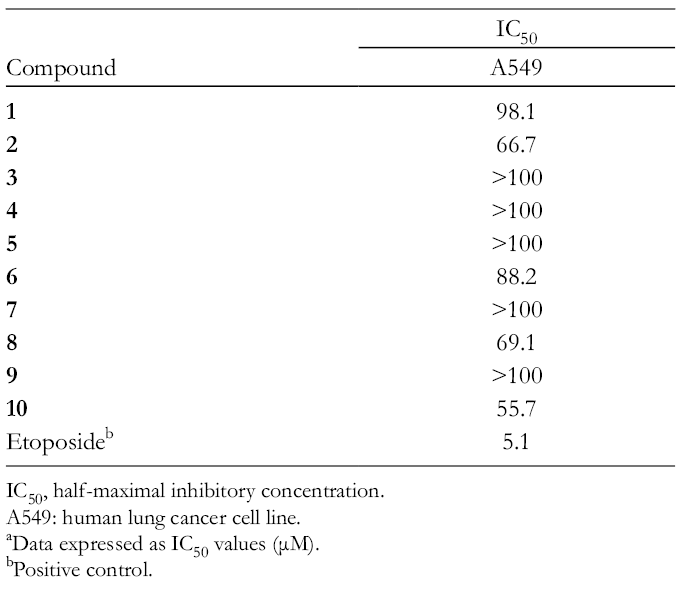
IC50, half-maximal inhibitory concentration.
A549: human lung cancer cell line.
aData expressed as IC50 values (μM).
bPositive control.
Experimental
General Experimental Procedures
The NMR spectra were measured in methanol-d 4, on a Bruker ARX-400 or AV600 instrument with tetramethylsilane as an internal standard. The circular dichroism spectrum was tested using the JASCO pu-2080 spectrometer. Infrared spectra were taken on a Bruker IFS-55 infrared spectrophotometer with a KBr disk. Optical rotations were measured on a Peking-Elmer 241 MC Spectropolarimeter at 20°C. ESI-MS spectra were recorded on Waters Quattro micro API LC/MS/MS spectrometer (Waters) with C18 reversed-phase column (HS12S05-2546WT 5 µm; 250 × 4.6 mm [YMC, Japan]; 25°C). HPLC was performed on JAI LC9103 recycling preparative HPLC (Japan Analytical Industries) equipped with JAIGEL-ODS-AP-P column (YMC-Pack ODS-AQ 15 µm; 500 × 20 mm [YMC, Japan]; 25°C) using a JAI refractive index detector and a JAI UV-3702 detector with MultiChro 2000 workstation. Thin-layer chromatography (TLC) was performed on precoated GF254 plates (Merck) and detected by spraying with 10% sulfuric acid followed by heating. The mobile phase for TLC was eluted with dichloromethane (CH2Cl2)–methanol (MeOH) (100:5).
Plant Material
The leaves of E. sessiliflorus were collected in October 2017 at Dandong, Liaoning, China, and authenticated by Professor Mingyan Jiang (The School of Pharmacy, China Medical University). A voucher specimen has been deposited in our laboratory (voucher No. MY-2017–012).
Extraction and Isolation
The leaves of E. sessiliflorus (7 kg) were extracted 3 times (3 × 30 L) with 70% (v/v) aqueous ethanol under reflux at 80°C to give 500 g of crude extract. The crude extract was suspended in 2 L of water. The suspension was successively partitioned with ethylacetate (3 × 2 L) and n-butanol (BuOH) (3 × 2 L). The n-BuOH soluble fraction (80 g) was subjected to silica gel column chromatography (CC), eluting with gradients of CH2Cl2–MeOH (100:1 2500 mL; 100:3 2500 mL; 100:7 3000 mL; 100:15 2500 mL; 100:30 2500 mL; 100:70 1500 mL) on silica gel column (column dimension: 6.0 × 80 cm) to give 6 fractions, Fr. A–F (2.3 g, 5.1 g, 10.9 g, 5.1 g, 3.3 g, and 0.2 g), which were detected by TLC. Fraction A was purified by silica gel CC (column dimension: 2.5 × 40 cm) with CH2Cl2–MeOH (300:1 800 mL; 150:3 1000 mL; 100:1 1000 mL) to afford 230 mg of Fr.A-1. Fr. A-1 was then subjected to HPLC with MeOH–water (H2O) (60:40) as eluent and each subfraction was further purified on preparative HPLC with MeOH–H2O (60:40) to yield
Cytotoxicity
Human lung cancer cell lines (A549) were provided by the American Type Culture Collection (ATCC). The cells were cultured in medium (RPMI1640 for A549) supplemented with 10% heat-inactivated fetal bovine serum and antibiotics antimycotics (100 units/mL penicillin G sodium, 100 µg/mL streptomycin, and 250 ng/mL amphotericin B). The cells were incubated at 37°C and 5% carbon dioxide in a humidified atmosphere. Etoposide (Sigma, purity >98%) was used as a positive control. Cell viability was determined by the sulforhodamine B protein staining method. Cells were seeded in 96-well plates and incubated for 24 and were fixed (for zero day controls) or treated with test compounds for 72 hours. All compounds were solved in dimethyl sulfoxide (final concentration of 0.1% [v/v]), stored at −20°C and diluted to the desired concentration (0.01, 0.1, 1, 10, 100 µM) in normal saline immediately prior to each experiment. Each concentration was tested thrice. At least 3 experiments were performed. After incubation, cells were fixed with 10% trichloroacetic acid, dried and stained in 0.4% sulforhodamine B in 1% acetic acid solution. Unbound dye was washed and stained cells were dried and dissolved in 10 mM Tris (pH 10.0). Absorbance was measured at 515 nm and cell proliferation was determined as follows: cell proliferation (%) = (average absorbancecompound – average absorbancezero day) / (average absorbancecontrol – average absorbancezero day) ×100%. GI50 values were calculated by nonlinear regression analysis using the Table Curve 2D software (Version 5.01, Systat Software Inc., CA, USA).
Conclusions
Ten compounds were separated and purified by chromatographic methods from the 70% ethanol extract of the leaves of E. sessiliflorus. Four compounds were isolated from the genus Eleutherococcus for the first time. All compounds exhibited weak cytotoxicity against the A549 cell line. Our findings can enrich the phytochemical content of genus Eleutherococcus.
Supplemental Material
Supplementary material - Supplemental material for Chemical Constituents of Eleutherococcus sessiliflorus (Rupr. & Maxim.)
Supplemental material, Supplementary material, for Chemical Constituents of Eleutherococcus sessiliflorus (Rupr. & Maxim.) by Ye Ma, Donghu Zhang and Mingyan Jiang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
