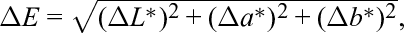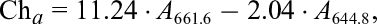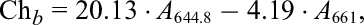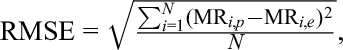Abstract
Lemon thyme contains several bioactive health-promoting compounds of high antioxidant capacity—such as polyphenols, carotenoids, and chlorophyll—which may undergo degradation during drying in incorrect processing conditions. This work is aimed at evaluating the impacts of different drying techniques and parameters on the chemical and physical properties of lemon thyme. In the experiment, lemon thyme leaves are dried using two traditional hot-air methods, convective drying (temperature 70°C or 80°C, air velocity 2 m/s), and fluidized bed drying (temperature 70°C or 80°C, air velocity 5–10 m/s); as well as two innovative drying methods, microwave-assisted fluidized bed (MFB) drying (microwaves at a power of 240 or 440 W, air temperature 40°C, air velocity 5–10 m/s) and microwave-vacuum (MV) drying (microwaves at a power of 240 or 440 W, under vacuum at 4–6 kPa). The kinetics of water delivery are described using the Page Model. After drying, the nutritional composition of the material is analyzed: polyphenol (total content and polyphenols profile by ultra-performance liquid chromatography), carotenoid, and chlorophyll total content, as well as antioxidant capacity (2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt [ABTS+] and ferric reducing antioxidant power [FRAP]). Color changes (L*, a*, b*, ΔE) are also analyzed using the visible effects of lemon thyme pigment disintegration. It is found that the MV drying method is the best for lemon thyme preservation, as it has the shortest process time and the highest retention of polyphenols (78.90%-82.14%), chlorophylls (51.54%-52.68%), antioxidant capacity (47.83%-48.63% of ABTS+ and 20.85%-45.45% of FRAP), and presented the least color change (ΔE = 25.57-28.32). For preserving carotenoids, the most protective method is MFB (retention 68.46%-70.61 %).
Edible plants containing biologically active compounds can be used as ingredients in functional or beneficial foods. 1 Thyme (Thymus Lamiaceae) is a perennial, green, and delicate herb with small and simple leaves, which is common all over the world. 2 In the food industry and gastronomy, different types of thyme have been widely used to enrich the taste and aroma of food; for example, Thymus vulgaris for meat and vegetable dishes, or Thymus citriodorus for fish dishes, desserts, ice cream, chewing gum, and teas. 3 The chemical compounds present in thyme include phenols, terpenes, terpene alcohols, ketones, aldehydes, ethers, and esters. 4 Many of these compounds have been reported as strong antioxidants, positively affecting human health by protecting cells against oxidative stress and even preventing the development of coronary heart disease, cancer, and neurodegenerative diseases such as Alzheimer’s or Parkinson’s disease. 5 Furthermore, thyme leaves are a wealthy source of flavonoids, tannins, phenols, vitamins, and various other important products of secondary metabolism. 3 The chemical composition of thyme depends on various agronomic factors, as well as the processing method and storage conditions used. 6,7
Lemon thyme (T. citriodorus) has a delightful, citrus-sweet rose flavor and aroma, generated mainly from geraniol, nerol, and 2 products of geraniol oxidation; terpene aldehydes collectively known as citral: geranial (E-citral) and neral (Z-citral). 8 Lemon thyme has been shown to have strong bactericidal properties connected strictly with these aromatic components, 9,10 but other bioactive herb ingredients, such as phenolic compounds, carotenoids, and chlorophyll have also been reported to be beneficial for human health. 7 Plant polyphenols, such as phenolic acids, flavonoids, curcuminoids, stilbenes, and lignans, are especially important and have multiple applications against a number of human diseases. 11 Dietary intake of polyphenols and carotenoids has been associated with a decreased risk of chronic diseases, including several types of cancer, type 2 diabetes, cardiovascular diseases, and age-related macular degeneration. 12,13
Although fresh lemon thyme has been widely recommended as a very precious accompaniment to many dishes, its tendency to perish makes the dehydrated variety more popular and convenient. Preservation by drying is carried out in order to stabilize the biologically active substances during the storage of the dehydrated plant, 11 which entails negative changes in the structural properties of the plant tissues and its sensorial characteristics, as well as the degradation of the sensitive chemical components. 12,13 A proper drying technique results in the protection of the chemical composition 11 and inhibits degrading enzymes, with the low-water content and low-water activity helping to counteract microbial spoilage. 13 Among the drying techniques commonly used for plant preservation, one of the most popular is convective drying (CD); however, due to the relatively high temperature and long dehydration process, degradation of valuable nutritional components may occur. 12 Intensification of heat and mass transfer during hot-air drying can be achieved by the fluidized bed (FB) method, where air flows through the drying chamber and keeps the particles moving. Another method of facilitating water evaporation and shortening the length of the process may be through combining the FB approach with microwave heating (MFB) or combining microwave heating with a vacuum (MV). Little has been reported in the literature about how different drying methods affect the nutritional properties of lemon thyme. Therefore, the aim of this study is to evaluate the influence of the parameters of the selected drying methods on the physical and chemical properties of lemon thyme.
Materials and Methods
Material
The material used in the experiments was lemon thyme (T. citriodorus L.) cultivated at the Faculty of Life Sciences and Technology at the Wroclaw Environmental and Life Sciences University, Wrocław, Poland. Average-sized leaves were separated from the stem just before processing.
Drying
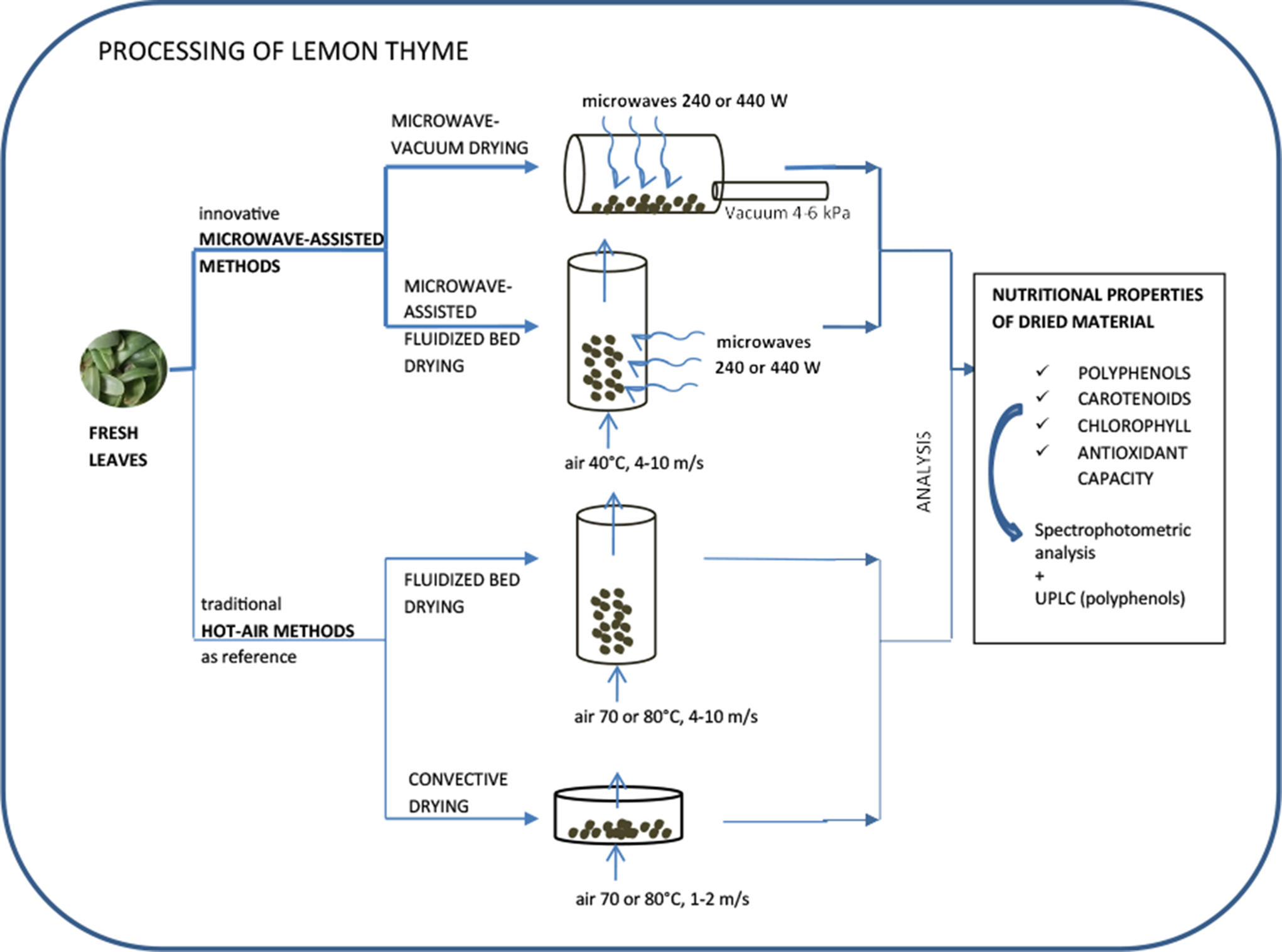
Drying was carried out using two innovative methods: microwave-assisted fluidized bed (MFB) drying, microwave-vacuum (MV) drying, and two traditional hot-air methods: convective drying (CD) and FB drying. Experiments were performed using the equipment of the Institute of Agricultural Engineering, Wroclaw University of Environmental and Life Sciences, Wroclaw, Poland.
MFB was conducted in an experimental stage MP20 14 consisting of the following elements: a fan, a heater, a cylindrical polycarbonate drying chamber, and magnetrons. Portions of fresh material (150 g) underwent drying at temperatures of 40°C, with a drying airspeed of between 4 and 10 m/s and a microwave power of 240 or 440 W. Wooden inert balls of diameter 3 ± 0.5 mm were used, in a 1:1 mass ratio, in order to obtain steady fluidization.
MV was conducted in a Plazmatronika SM-200 dryer 15 consisting of a glass cylindrical drying chamber, magnetrons, and a vacuum pump, at a microwave power of 240 or 440 W and a pressure of 4–6 kPa. Portions of fresh material (60 g) underwent drying in the cylindrical drying chamber at a rate of 6 rotations per minute.
CD was conducted in a chimney dryer 15 consisting of a fan, a heater, and steel drying baskets (50 g of leaves in each basket), at a temperature of 70°C or 80°C and with an airspeed of 2 m/s.
FB was conducted in an experimental stage 16 consisting of a fan, a heater, and a Plexiglas drying chamber, where portions of fresh material (150 g) underwent drying at temperatures of 70°C or 80°C with a drying airspeed of 5–10 m/s. Polypropylene inert balls of diameter 3 ± 0.5 mm were used, in a 1:1 mass ratio, in order to obtain steady fluidization.
Dehydration was carried out until a consistent water content was achieved. The drying effects were noted at intervals of 5–20 minutes, depending on the method used. Each test was repeated twice.
The drying processes were undertaken while controlling the loss in weight, which enabled the adaptation of a model that adequately describes the drying kinetics. In order to present the kinetics, it was necessary to calculate the relative water content, according to the formula:
where MR is the relative water content (-),u o is the initial water content (g H2O/g dm), and u r is the final water content (g H2O/g dm).
In order to assign the drying curves, the Page Model (Equation 2), which has been verified as the most frequently used mathematical model of plant drying kinetics, 17 was used:
where a, k, and n are function coefficients, and τ is the drying time [min].
The curve regression analyses, along with process rate calculations, were carried out using the Microsoft Excel 2007 software. After selecting the coefficients k and n, the drying effects were graphically presented as MR = f (τ).
Dry Matter Content, Water Activity, and Color Measurements
Both the fresh and the final dried product were evaluated on the basis of chosen quality factors: dry matter content, water activity, and color parameters. Fresh product characteristics were made for evaluating the range of changes after the drying process.
The dry matter content was appointed a gravimetric method, 18 using a vacuum oven SPT-200 (ZEAMiL, Horyzont, Cracow, Poland) at a temperature of 60°C for 24 hours.
The water activity was measured using the AquaLab TDL Dew Point Water Activity Meter 4TE ± 0.003 (AquaLab, Warszawa, Poland) at a temperature of 25°C ± 2°C.
The color parameters were evaluated using the Minolta CR-400 colorimeter (Minolta Conica, Japan) using the CIE L*, a*, b* system by the design coordinates (L*, lightness; a*, red-green axis; and b*, blue-yellow axis) and the standard illuminant D65. On the basis of the L*, a*, b* coordinates, ΔE between the dried and fresh material was calculated according to the following formula:
where ΔE is the total color difference, ΔL* is the lightness difference between fresh and dried lemon thyme, Δa* is the greenness difference between fresh and dried lemon thyme, and Δb* is the yellowness difference between fresh and dried lemon thyme.
Chemical Composition and Antioxidant Capacity
Total polyphenols, chlorophylls, and carotenoids
The total content of polyphenols was determined using the Folin–Ciocalteu colorimetric method (Shimadzu UV-2401 PC spectrophotometer, Osaka, Japan). The absorbance was measured at a wavelength of 765 nm. The result was expressed in terms of gallic acid. 19
The chlorophyll (evaluated as the sum of chlorophyll a and b) and total carotenoid content were determined using the colorimetric method described by Nawirska-Olszańska et al. 20 using a UV-160A spectrophotometer (Shimadzu, Osaka, Japan). A total of 8 mL of 100% acetone was added to 0.3 g portions of ground fresh or dried material and left for 24 hours at room temperature with ultrasound. Absorbance coefficients were measured at wavelengths of λ = 661.6, λ = 644.8, and λ = 470 nm. Then, the content of chlorophylls (a and b) and carotenoids in lemon thyme were calculated using the following formulas:
where Ch a is the chlorophyll a content, Ch b is the chlorophyll b content, Ca is the carotenoid content, А λ is the absorbance at the corresponding wavelength λ, and the values 11.24, 2.04, 20.13, 4.19, 1.9, and 63.14 are molar absorptivity coefficients.
Identification of polyphenols by the UPLC mMethod
The extract for analysis was prepared as described by Kolniak–Ostek and Oszmiański. 21 Identification of polyphenols was conducted using an ACQUITY ultra-performance liquid chromatography (UPLC) system, consisting of a binary solvent manager (Waters Corp., Milford, MA, USA), an UPLC BEH C 18 column (1.7 µm x 100 mm, Waters Corp., Milford, MA, USA), and a Q-Tof Micro mass spectrometer (Waters, Manchester, UK). The 10 µL sample was injected and elution was completed in 15 minutes with a sequence of linear gradients and isocratic flow rates of 0.42 mL/min. A strong solvent (100% acetonitrile solution) and a weak solvent (10% acetonitrile–water solution) were run through the column. Phenolic compounds were separated by the gradient elution method using 4.5% formic acid and acetonitrile as solvents A and B, respectively. The column temperature was 90°C. The retention times of individual compounds were compared with standard retention time. The compounds were identified at λ = 360 nm (flavonol glycosides), λ = 320 nm (phenolic acids), and λ = 280 nm (flavan-3-ols and hydroquinones). Standard curves were developed on the basis of chlorogenic acid, neochlorogenic acid, cyaniding-3-O-glucoside and rutinoside of different concentrations between 0.05 and 5 g/L. The results of measurements repeated in duplicate were analyzed using the Empower 3 software and presented in mg/100 g dm.
Antioxidant capacity
The antioxidant capacity was determined by the colorimetric method. First, 50 mL of ethanol was added to 0.5 g portions of ground fresh or dried material and agitated for 24 hours at room temperature. Then, the sample underwent centrifugation at 5000 rpm for a duration of 15 minutes at 20°C. The antioxidant capacity was tested using the 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt (ABTS+) and ferric reducing antioxidant power (FRAP) methods (Shimadzu UV-2401 PC spectrophotometer, Osaka, Japan).
The ABTS+ method (radical cation scavenging assay, or Trolox equivalent antioxidant capacity—ABTS) was performed as follows. The solution was diluted with redistilled water until it reached a saturation of 0.700 at a wavelength of λ = 743 nm. Then, 3 mL of ABTS+ was added to 30 µL of the supernatant solution. After 6 minutes, the absorbance measurement was carried out. 22
In the FRAP method, 3 mL of the FRAP solution (TPTZ 10 mmol/L, FeCl3 × 6H2O 20 mmol/L, and redistilled water in a 10:1:1 vol ratio) was added to 1 mL of the diluted sample. The absorbance measurement was taken after 10 minutes at a wavelength of λ = 593 nm. 23
The analyses were performed in duplicate, and results were presented in mmol Trolox equivalent/100 g dm.
Statistical Analysis
The statistical analysis was carried out using the Statistica 10.0 software (StatSoft, Tulsa, OK, USA). The data were recorded as means ± standard deviation and analyzed using Excel 2007. Analysis of variance (ANOVA) was performed by ANOVA procedures. Significant differences (P ≤ 0.05) between the mean values of chemical characteristics and color were determined by Tukey’s multiple range test.
The degree of adaptation of the model of lemon thyme drying was based on the determined values of the RMSE, χ 2, V e, and R 2. These factors were calculated based on the following formulae:
where RMSE is the root mean square error, χ2 is the test reduction coefficient, V e is the residual variance coefficient, R 2 is the determination coefficient, MR i,p is the calculated value of relative water content, MR i,e is the experimental value of relative water content, N is the number of observations, n is the number of parameters in the model equation, S e is the standard deviation of the residual component, Y t is the arithmetic average of the y variable, SSM is the sum of squares for the model, SST is the total sum of squares, y r is the actual value of the dependent variable (measured), y t is the expected value of the dependent variable (based on the regression model), and y ś is the arithmetic average of the actual dependent variable.
Results and Discussion
Processing of plants by drying leads to a significant reduction in their content of bioactive components, but the degree of destruction depends on the applied drying techniemical Composition and Antioxidant Capacityque and parameters used. 12 In the current study, two conventional hot-air drying methods—CD and FB drying—were compared with two innovative drying methods—MFB drying and MV drying. The comparison was made on the basis of drying kinetics, color changes, and selected nutritional properties of the dried lemon thyme (ie, chemical composition and antioxidant capacity).
Drying Kinetics
Lemon thyme drying proceeded with the intensity, depending on the technique and parameters used, and lasted from 20 minutes (MV) to 8 hours and 20 minutes (CD). In the current study, for the description of the drying kinetics, the Page Model (Equation 2) was chosen on the base of popularity 17 and presented acceptable results, as good fitting the experimental data was obtained for all methods applied (Table 1). As the parameter a in the Page Model, which determines MR at = 0 (ie, the beginning of the drying process), was equal to 1 for all methods, it has been omitted in the further analysis.
Analysis Results of the Effects of Drying Lemon Thyme, Page’s Model.

RMSE, root mean square error; V e, residual variance coefficient; k, drying constant (min-1); n, function coefficient; χ 2, test reduction coefficient; R 2, determination coefficient.
For all the examined drying methods, the values of RMSE, V e, and χ 2 were relatively low (RMSE = 0.009751-0.027992, V e = 3.9-8.7, and χ 2 = 0.000142-0.001235), whereas R 2 reached values above 0.992727. This proves that the model described the empirical points accurately. According to other authors, this model is also suitable for displaying different plant drying effects, such as those for T. vulgaris, 24 green tea, 25 lavender flowers, 26 and dill. 27
CD was the most time-consuming method of all the methods used in this study (Figure 1). The analyzed material released moisture very slowly; however, raising the temperature of the air from 70°C to 80°C improved the dehydration dynamics and led to a shortening of the time consumed from 8 hours and 20 minutes to 6 hours and 40 minutes. This finding was similar to the observation made for lavender flowers dried using CD by Łyczko et al 26 : raising the drying temperature by 20°C reduced the process time nearly by half. The model parameters k and n determined the drying speed and, during CD of lemon thyme, the parameter k decreased along with the temperature (from 1.3300 to 1.1582), whereas the parameter n rose (from 0.0008 to 0.0043; see Table 1).

Drying kinetics of lemon thyme using the convective drying (CD), fluidized bed (FB) drying, microwave-assisted fluidized bed (MFB) drying, and microwave-vacuum (MV) drying methods.
An increase in the temperature of the air caused a significant shortening of the drying time during FB as well. Drying processes carried out at temperatures of 70°C and 80°C, lasted 1 hour and 10 minutes and 35 minutes, respectively (Figure 1). Therefore, increasing the drying temperature by 10°C reduced the time required to dry the material by half. Similar conclusions were made by Surma et al. who analyzed the drying effects of red pepper. 28 They conducted FB at a temperature of 70°C, which lasted approximately 340 minutes, and at a temperature of 90°C, which lasted approximately 110 minutes. In the current experiment, the parameter k in the Page Model increased with an increase in air temperature (from 1.1178 to 1.5953), while the parameter n decreased (from 0.0352 to 0.0156). This means that the parameter k influenced the kinetics of FB stronger than the parameter n (Table 1).
MFB drying, carried out at a temperature of 40°C and with a microwave output of 240-440 W, lasted approximately 2.5 hours (Figure 1). In this instance, using a higher microwave output did not necessarily generate a significant reduction in the duration of the drying process. This phenomenon can also be observed when analyzing the changes of the constants in the Page Model: the parameter k rose with increasing temperature (from 1.1400 to 1.1576) and the constant n increased slightly (from 0.0130 to 0.0137) (Table 1). A similar correlation between microwave power and the process time was observed in studies involving the MFB drying of apples 29 and soya. 30
MV drying was the fastest of all the tested methods for lemon thyme dehydration. Moreover, it was found that the microwave power had a strong impact on the drying kinetics and the time of the process. The desired dried matter was obtained after 36 minutes for the 240 W output, and after 20 minutes for the 440 W output (Figure 1). A similar finding was described by other authors during MV drying of oregano, where increasing the microwave output from 240 to 480 W resulted in a reduction of the process time from 24 to 15 minutes. 31 Function coefficients in the Page Model varied with the change of microwave level, where the parameter k decreased (from 1.5708 to 1.5356) and the parameter n increased (from 0.0124 to 0.0432; Table 1). This indicates that, for the MV of lemon thyme, the parameter n had a stronger influence on the kinetics than the parameter k.
Analyzing both microwave-assisted drying methods of lemon thyme, it can be seen that microwaves are an effective source of heating when drying the material when used in a vacuum system, but not when used in a fluidized one.
Dry Matter Content, Water Activity, and Colour Changes
The color of biological materials is strictly connected with the dry matter content and water activity; parameters that characterize the stability of the dried product. While biological material can be treated as effectively dried when the water content is ≤12%, 11 in fact, only a low water activity of A w < 0.6 can prevent the multiplication of microorganisms and protect against physical and biochemical changes. 32 The dry matter of fresh lemon thyme was 5.35% and, after dehydration, its content ranged between 88.54% and 94.95% (Table 2), which indicates that all samples were properly dried.
Dry matter content (dm), A w, and colour indicators (L*, a*, b*, and ΔE*) of lemon thyme: fresh or dried using the CD, FB drying, MFB drying, and MV methods.

CD, convective drying; FB, fluidized bed; MFB, microwave-assisted fluidized bed; MV, microwave-vacuum; A w, water activity.
Values are mean ± standard deviation, n = 3; in columns, values followed by different letters (A, B, C,…) were significantly different (P < 0.05), according to Tukey’s test.
Among the samples obtained, the highest moisture content was measured after MFB and the lowest after MV. These effects might be connected with the observed deformations of the thyme leaf tissue, which probably disrupted dehydration during MFB, and with the puffing mechanism (rapid simultaneous evaporation of surface and deep water 33 caused by the vacuum), which may have facilitated dehydration in MV. This phenomenon has been also observed during the MV drying of green tea leaves. 25 Water activity in the dried thyme was below 0.6, which ensures microbial safety; however, the samples obtained after MFB at 240 W were characterized by the highest water activity (0.5753), while the lowest water activity was noted after FB at a hot-air temperature of 80°C.
The drying process caused a significant change in the color of lemon thyme, depending on the drying method and the process parameters. All drying methods led to the darkening of leaves, measured as a decrease of the L* coordinate, except for CD at 80°C, for which the level stayed the same as that of the fresh material. In CD and FB, the value of the coordinate L* decreased along with an increase in temperature; whereas, in MV and MFB, the value of L* increased along with an increase in microwave power. The darkening of green leaves processed by drying has been connected with browning reactions (the enzymatic browning reactions or non-enzymatic Maillard reactions) attributed to the degradation of green pigments. 34 Confirmation of this phenomenon can be also seen in the loss of greenness observed in dried lemon thyme, as indicated by a significant increase of the a* value, compared with fresh thyme. In particular, the smallest a* increase was noted during MV with a microwave output of 440 W, and the largest one was observed during MV at a microwave output of 240 W. A reduction in the value of the b* parameter, indicating loss of yellowness in the thyme leaf color, was noted in all the dried samples, where CD at a temperature of 70°C resulted in the highest reduction of the b* color coordinate. Opposite observations were made by other authors during celak (Thymus daenensis subsp. daenensis) drying, where air drying caused much lower changes in the yellow color than microwave drying. 35 However, microwave drying led to obtaining dried celak of the highest color quality, which was consistent with the results obtained by the authors in the presented experiment. In the case of the ΔE parameter (total color difference, Equation 3), it was observed that the color of all samples subjected to dehydration differed significantly from the color of the fresh material. According to interpretation recommended by the International Commission of Illumination (CIE), 36 a value of ΔE > 3.5 means “a clear color difference”. The highest ΔE between the dried and fresh material was recorded for MFB at a microwave output of 240 W (33.97), while the lowest was with MV at a microwave output of 440 W (25.57).
Nutritional Properties of Lemon Thyme
The drying affected the bioactivity of the output materials, which was evaluated as the stabilities of the main valuable nutritional compounds. It was found that all tested drying methods caused significant reductions in polyphenol, chlorophyll, and carotenoid contents in lemon thyme, but the level of chemical destruction was dependent on the conditions and parameters of dehydration (Table 3).
Total content of polyphenols, chlorophylls, and carotenoids, as well as antioxidant capacity (ABTS+, FRAP) of lemon thyme leaves: fresh or dried using CD, FB drying, MFB drying, and MV methods.

CD, convective drying; FB, fluidized bed; MFB, microwave-assisted fluidized bed; MV, microwave-vacuum; ABTS, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) diammonium salt; FRAP, ferric reducing antioxidant power .
Values are mean ± standard deviation, n = 3; in columns, values followed by different letters (A, B, C,…) were significantly different (P < 0.05), according to Tukey’s test.
Polyphenols
The phenolic components of plants are very sensitive to high temperatures. Several studies have confirmed that the drying process diminishes these bioactive components. 11 -13,37 All drying methods used in the current study were destructive for polyphenols in lemon thyme, but the highest polyphenol loss was observed when using CD, with reduction of polyphenols by 80.47% at 70°C and by 74.68% at 80°C. Thus, higher temperatures contributed to better retention of polyphenols. The obtained results were in contrast to results obtained by Lahnine et al, 38 who also dried thyme (Thymus satureioides) at 70°C and 80°C using a solar dryer; they reported a decrease of total polyphenol contents along with an increase in temperature. In the studies conducted at a temperature of 35°C by Piekut et al, 39 lower polyphenol decrease was observed (22.85%), but the experiment lasted a very long time—20 hours. Such a low temperature of drying (25°C-32°C, 10 days) is also recommended by other researchers since a considerable increase of total phenolics in oregano (by half) and in paper mint (+32%) was obtained. 40
The dried lemon thyme obtained from the FB method lost 73.37% of its phenolic compounds at a temperature of 70°C and 56.44% at 80°C. It was observed that when a higher temperature was applied, higher retention of polyphenols was achieved. These results correspond to those obtained for the CD drying of lemon thyme, indicating that the time of processing and, thus, time of contact with oxygen, affected polyphenol retention stronger than the temperature.
MV drying of lemon thyme was the most protective method for polyphenols, resulting in a loss of 21.10% for a microwave output of 240 W and 17.87% for 440 W. In this sense, lower microwave power and longer time implied more exposure to oxygen, accounting for the lower phenolic content. This phenomenon has also been reported by Cao et al 41 during MV drying of litchi fruits, but their explanation was the protective influence of reducing sugars.
A more detailed analysis of polyphenols in lemon thyme (UPLC) revealed 16 polyphenolic compounds, identified at wavelengths λ max 280-360 nm (Table 4). In fresh lemon thyme, 7 phenolic acids or derivatives, 6 flavan-3-ols, and 3 flavone glycosides were identified, and all of them were previously detected in thyme species. 42 -44
Profile of phenolic compounds (Rt and content) in lemon thyme: fresh or dried using CD, FB drying, MFB drying, and MV methods.
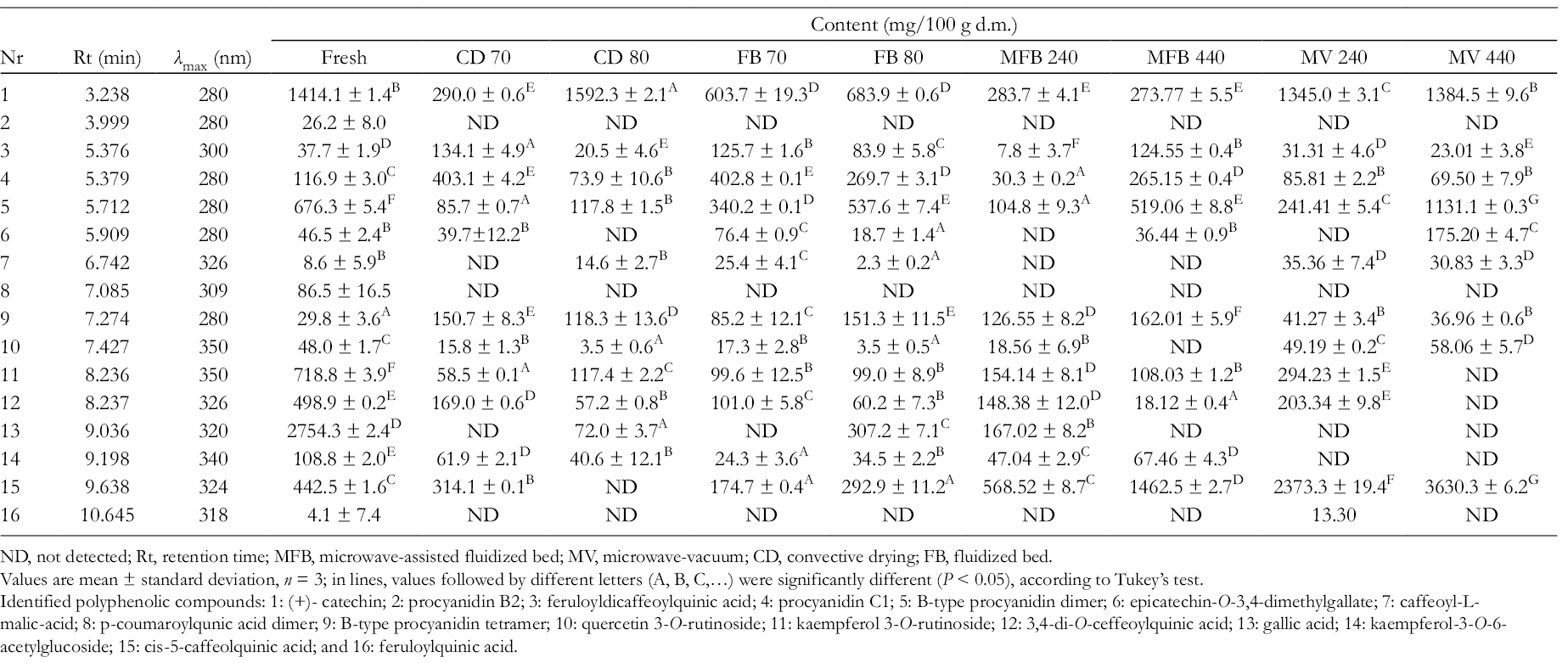
ND, not detected; Rt, retention time; MFB, microwave-assisted fluidized bed; MV, microwave-vacuum; CD, convective drying; FB, fluidized bed.
Values are mean ± standard deviation, n = 3; in lines, values followed by different letters (A, B, C,…) were significantly different (P < 0.05), according to Tukey’s test.
Identified polyphenolic compounds: 1: (+)- catechin; 2: procyanidin B2; 3: feruloyldicaffeoylquinic acid; 4: procyanidin C1; 5: B-type procyanidin dimer; 6: epicatechin-O-3,4-dimethylgallate; 7: caffeoyl-L-malic-acid; 8: p-coumaroylqunic acid dimer; 9: B-type procyanidin tetramer; 10: quercetin 3-O-rutinoside; 11: kaempferol 3-O-rutinoside; 12: 3,4-di-O-ceffeoylquinic acid; 13: gallic acid; 14: kaempferol-3-O-6-acetylglucoside; 15: cis-5-caffeolquinic acid; and 16: feruloylquinic acid.
Drying processes caused the reduction, conversion, disintegration, or deletion of phenolic components, depending on the method and parameters used. The influence of temperature and microwave power on the content of identified polyphenols was significant.
The influence of temperature on the content of identified polyphenols was significant but ambiguous in different drying methods.
It was observed that an increase of drying temperature and microwave power caused a decrease in feruloyldicaffeoylquinic acid, procyanidin C1, and 3,4-di-O-ceffeoylquinic acid content; whereas, to the contrary, caused also an increase in the content of (+)-catechin, B-type procyanidin dimer, and cis-5-caffeolquinic acid. The amount of epicatechin-O-3,4-dimethylgallate, caffeoyl-l- malic-acid, B-type procyanidin tetramer, quercetin 3-O-rutinoside, and kaempferol 3-O-rutinoside did not depend on the drying parameters. Procyanidin B2, p-coumaroylqunic acid dimer, and feruloylquinic acid were not detected in the dried material, meaning that they had collapsed or been transformed into other compounds during dehydration.
In the case of CD, the content of 8 polyphenols decreased regardless of the temperature (B-type procyanidin dimer, epicatechin-O-3,4-dimethylgallate, quercetin 3-O-rutinoside, kaempferol 3-O-rutinoside, 3,4-di-O-caffeoylquinic acid, gallic acid, kaempferol-3-O-6-acetylglucoside, and cis-5-caffeolquinic acid). On the contrary, the content of B-type procyanidin tetramer increased in both temperatures. The amount of feruloyldicaffeoylquinic acid and procyanidin C1 increased in material dried at 70°C and decreased at 80°C, compared with the fresh one, while in case of (+)- catechin, the relation between the amount and the temperature was opposite.
FB drying carried out in both temperatures resulted in a decrease in the content of 7 phenolics compounds ((+)- catechin, B-type procyanidin dimer, quercetin 3-O-rutinoside; kaempferol 3-O-rutinoside; 3,4-di-O-caffeoylquinic acid, kaempferol-3-O-6-acetylglucoside, and cis-5-caffeolquinic acid). At the same time, an increase in the content of 3 phenolic compounds was noted (feruloyldicaffeoylquinic acid, procyanidin C1, and B-type procyanidin tetramer). The amount of epicatechin-O-3,4-dimethylgallate and caffeoyl-l-malic-acid increased at 70°C and decreased at 80°C, compared with the reference.
MFB, regardless of the microwave power used, caused a decrease in the content of 8 components ((+)- catechin, B-type procyanidin dimer, epicatechin-O-3,4-dimethylgallate, quercetin 3-O-rutinoside, kaempferol 3-O-rutinoside, 3,4-di-O-caffeoylquinic acid, gallic acid, kaempferol-3-O-6-acetylglucoside), whereas the amount of 2 components, B-type procyanidin tetramer, and cis-5-caffeolquinic acid, rose after drying by this method. Caffeoyl-l-malic-acid was not detected in material dried by MFB, while higher microwave power induced growth of feruloyldicaffeoylquinic acid and procyanidin C1.
MV of lemon thyme caused a significant increase of 4 phenolic components (caffeoyl-l-malic-acid, B-type procyanidin tetramer, quercetin 3-O-rutinoside, and cis-5-caffeolquinic acid) and inconsiderable decrease of 3 components ((+)-catechin, feruloyldicaffeoylquinic acid, and procyanidin C1). In the case of B-type procyanidin dimer and epicatechin-O-3,4-dimethylgallate, the higher microwave power led to better retention, whereas kaempferol 3-O-rutinoside and 3,4-di-O-ceffeoylquinic acid underwent decomposition. Regardless of the microwave power used, gallic acid and kaempferol-3-O-6-acetylglucoside were not detected in the dried thyme.
On the basis of phenolic profiling of lemon thyme (qualitative and quantitative), we can suggest microwave-assisted methods as being more protective than hot-air methods.
Chlorophylls
The drying of green plants can potentially cause degradation of green pigments. Every method used in this experiment resulted in a total chlorophyll content decrease. However, in microwave-assisted drying methods, retention of chlorophyll was higher than that in hot-air methods. The best technique for preserving chlorophylls was MV (reduction by 47.32%-48.46%), while the worst method was FB at 70°C (reduction by 86.81%). The positive influence of microwaves on chlorophyll content has been also reported in cases of oregano (Origanum vulgare L.) and basil (Ocimum basilicum L.) during microwave-assisted CD. 45
Degradation of chlorophylls a and b during thermal processing has been shown to produce color changes in green vegetables (broccoli and spinach), from the bright green of the chlorophyll a and b to the olive brown of pheophytins a and b. 46
Carotenoids
The influence of microwaves on carotenoid content during the drying of lemon thyme was noted. Usage of the MFB drying method allowed preservation of the highest content of total carotenoids (reduction by 29.39%-31.54%), whereas the other methods and parameters tested in presented experiments were more destructive for carotenoids: reduction by 83.74% and 86.18% in the cases of CD at 70°C and MV at 240 W, respectively. However, the reduction of carotenoid content has also been observed by other authors during the CD of oregano and melissa (reduction by 50% and 45%, respectively). 24 The phenomenon of the protective influence of microwaves only during MFB on carotenoid content has not been observed in the literature, but Sanchez et al 46 noted an increase in carotenoid contents during drying of broccoli and spinach in high pressure (625 MPa) and high temperature conditions (70°C and 117°C).
Antioxidant capacity
Analysis of antioxidizing properties performed using the ABTS+ method demonstrated that MV drying is the most suitable method for preserving thyme (48.63%), compared with the other drying techniques investigated. The application of milder drying parameters was associated with a higher reduction in the antioxidizing potential. In MV, the differences between the results for particular microwave outputs were minor; however, in each instance, a process time reduction was essential for preserving the highest possible material output activity (Table 4). A significantly higher degree of antioxidizing capacity was observed in lyophilized blueberries, where the activity was reduced by approximately 19.5% 47 . On the other hand, dried peaches analyzed by Güçlü et al 48 (depending on the variety chosen) resulted in only 29.23%-38.27% reduction of their output potential. Reduced antioxidizing compound content has been shown in CD thyme, which lost approximately 92.39% of its output potential. 39 Siddhuraju 49 conducted CD of Tamarindus indica and reported retention of approximately 22.49% of its antioxidizing potential.
Analysis using the FRAP method showed that MV drying using a microwave output of 440 W was the best method for thyme processing. Using this method, it was possible to retain 45.45% of the antioxidizing activity of fresh lemon thyme. Better results have been obtained for beans dried together with acid-washed sea sand on an open heating plate at a temperature of 125°C for 25 minutes, which reduced the fresh plant’s antioxidizing potential by approximately 9%. 49 The highest antioxidizing activity losses were observed for CD and FB, using a drying air temperature of 70°C. Increasing the temperature or microwave output resulted in shortening the process time, while simultaneously retaining a higher antioxidizing potential. Studies conducted using soy, by Niamnuy et al, 50 showed the same correlation, where the antioxidizing activity retention in preserved plant material for each of the drying methods depended on the drying agent and temperature used. Drying methods in a fluidized bed, using heated steam in a fluidized bed, and hot air vibration drying in combination with infrared were employed. For each of these methods, higher temperatures enabled a higher antioxidizing potential. A similar tendency was observed in the results obtained following the analysis of lemon thyme. This indicates that the major parameter which impacts the retention of valuable properties of the material is the process time.
Conclusions
The results showed that all examined drying methods can be used to obtain dried lemon thyme with physical and chemical properties typical of dried herbs. The drying method and process parameters had a significant influence on the changes in drying effects and nutritional value. Drying caused degradation of all measured bioactive compounds as well as the antioxidant capacity of lemon thyme leaves (polyphenols by 17.87% to 80.47%, carotenoids by 29.39% to 86.18%, chlorophylls 13.77% to 47.32%, ABTS+ 7.64% to 48.65%, and FRAP 20.80% to 45.33%). Furthermore, the longer the dehydration process proceeded, the higher the bioactive compound degradation effect was. Therefore, harder conditions (ie, higher microwave power or temperature level) which caused a shortening of the drying process provided higher retention of chemical components. Microwaves can be used as an effective source of energy during the drying of lemon thyme when combined with a vacuum; the combination of microwaves and fluidization proved to be an ineffective and destructive method.
Dried lemon thyme of the highest nutritional value (ie, with the lowest polyphenol, chlorophyll, and antioxidant capacity degradation, and the most beneficial phenolic profile) was obtained by MV drying at a microwave power of 440 W.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article was funded by Wroclaw University of Environmental and Life Sciences, Poland; grant number D2010/0016/18.