Abstract
A novel alkaloid scaffold was designed through scaffold-hopping strategy based on the active pyrazines alkaloid isolated previously. A total of 25 derivatives were synthesized based on this scaffold and evaluated for their antitumor activities. Among all these tested compounds,
Cancer is one of most serious public health problems representing the leading cause of morbidity and mortality worldwide, which causes great social and economic burdens. 1 Natural products have been considered as important sources for drug discovery, which could be developed for the treatment of various diseases. “Fu zi,” also named Radix Aconiti Lateralis Praeparata, is the lateral root of Aconitum carmichaelii, which is widely distributed in Sichuan Province of China and is often used as agents toward various diseases. 2 -4
Our previous study on “Fu zi” resulted in the isolation of 7 nonditerpene alkaloids, among which 2 new pyrazines (Figure 1, compounds
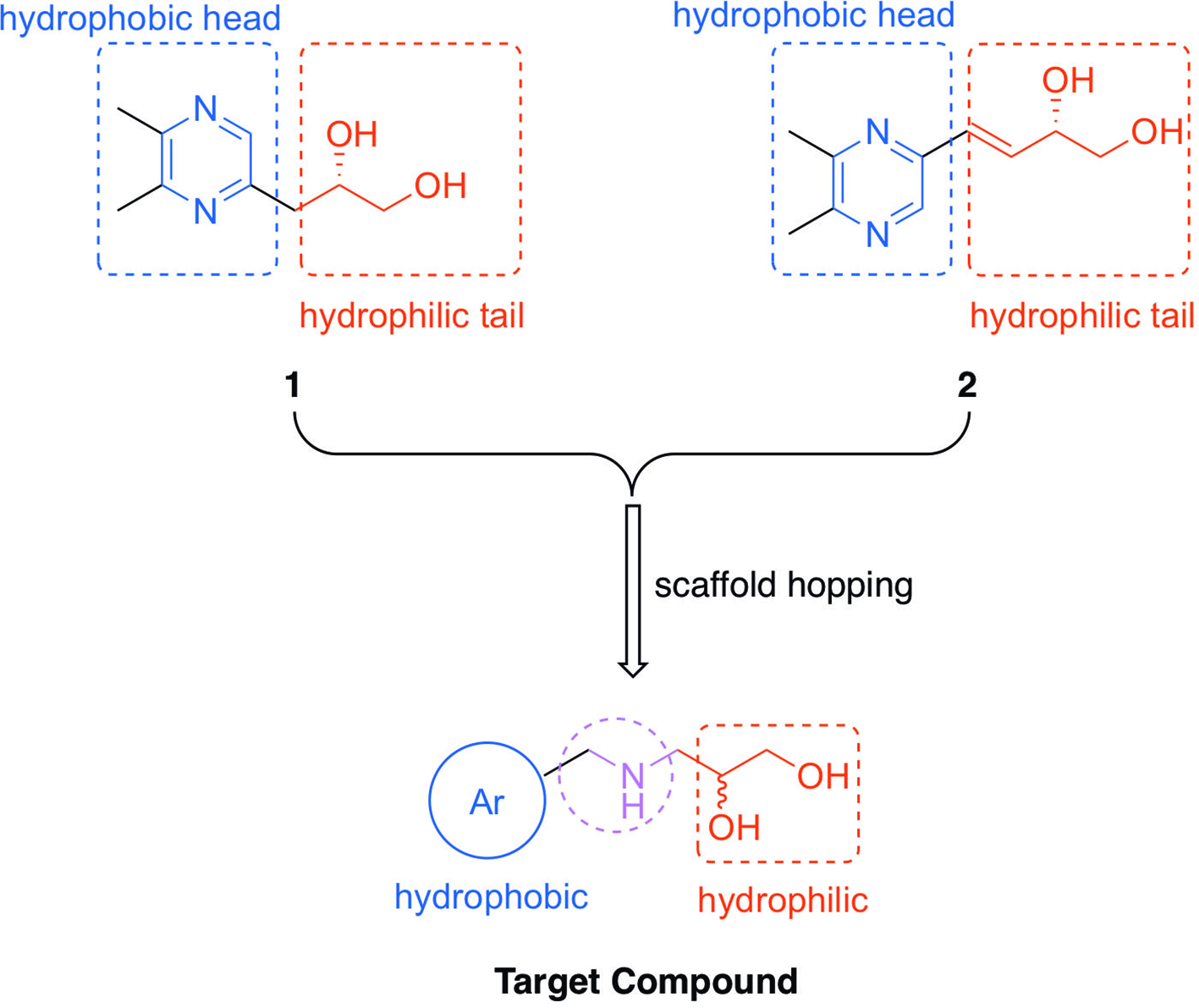
Design of the target compound based on the lead compounds through scaffold hopping.
The starting materials were (S)-3-aminopropane-1,2-diol (

Synthetic route of compounds 6a-6p. Reagents and conditions: (a) DCM, r.t. 3 hours. (b) NaBH4, DCM, r.t. 3 hours.
Derivatives

Synthetic route of compounds 9a-9i. Reagents and conditions: (a) DCM, r.t. 3 hours. (b) NaBH4, DCM, r.t. 3 hours.
All these target compounds were evaluated for their antiproliferative activities against tumor cell lines, including H460 cells (human nonsmall-cell lung cancer cells), TMD-8 cells (human diffuse large B-cell lymphoma cells), and MV4-11 cells (biphenotypic B myelomonocytic leukemia cells), in vitro by 3-(4, 5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. Each experiment was repeated 3 times. The results of these studies are summarized in Table 1.
Antitumor Activity Evaluation of Synthesized Compounds.

aThe data are expressed as the mean ± SD. All experiments were independently performed at least 3 times.
bUsed as a positive control.
As illustrated in Table 1, the S-configuration compound
In conclusion, 25 new pyrazines alkaloid derivatives were successfully synthesized and evaluated cytotoxic activity against 3 cancer cell lines. The results indicate that some of these derivatives possess at least moderate cytotoxic activity. Among all these tested compounds,
Experimental
General
The reactions were monitored by TLC with silica gel GF254 plates, which were visualized by ultraviolet (UV) light (254 nm). 1H Nuclear Magnetic Resonance (NMR) spectra were measured on a Bruker 600 and 400 MHz spectrometer at 25°C, and referenced to Me4Si. Chemical shifts are reported in ppm (δ) using the residual solvent line as an internal standard. Splitting patterns are designed as s, singlet; d, doublet; t, triplet; m, multiplet. NMR spectra were taken in Dimethylsulfoxide (DMSO) with Tetramethylsilane (TMS) internal standard on a Bruker SF-400 spectrometer (operating frequency 400 MHz). Starting materials and solvents were purchased from common commercial suppliers and were used without further purification.
General Procedure for Preparation of Conjugates Between Triterpenoid and 1,2-Diaminobenzene 6a-6p and 9a-9i
(S)-3-((3,4-Dimethylbenzyl)amino)propane-1,2-diol (6a)
(S)-3-Amino-1,2-propanediol (1.0 g, 11 mmol) was dissolved in chloroform (30 mL) and methanol (10 mL), and 3,4-dimethylbenzaldehyde (1.33 mL, 10 mmol) was added to the solvent and stirred for 1 hour at room temperature. Sodium borohydride (378.4 mg, 10 mmol) was added and the mixture was stirred for 12 hours at room temperature. After the reaction was complete, the solution was removed under reduced pressure and the residue was purified with column chromatography (DCM/CH3OH = 30:1) to give compound
(S)-3-((3-Methoxybenzyl)amino)propane-1,2-diol (6b)
Light yellow oil. Yield = 27.8%. C11H17NO3. 1H NMR (400 MHz, DMSO-d 6) δ 7.27-7.22 (m, 1H), 6.89 (m, 2H), 6.81 (dd, J = 8.2, 2.8 Hz, 1H), 3.80 (m, 5H), 3.69 (dd, J = 11.4, 3.6 Hz, 1H), 3.57 (dd, J = 11.4, 4.8 Hz, 1H), 2.83 (dd, J = 12.2, 3.8 Hz, 1H), 2.71 (dd, J = 12.2, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 159.75, 140.72, 129.56, 120.57, 113.93, 112.63, 70.15, 65.46, 55.21, 53.64, 51.54. HR-ESI-MS (positive ion mode): m/z 212.1268 [M+H]+ (calcd for C10H14ClNO2, 211.1208).
(S)-3-((2-Chloro-4-fluorobenzyl)amino)propane-1,2-diol (6c )
White solid. Yield = 37.1%. C10H13ClFNO2, mp 66.0 to 66.5°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.55 (dd, J = 8.5, 6.6 Hz, 1H), 7.38 (dd, J = 8.9, 2.6 Hz, 1H), 7.20 (td, J = 8.5, 2.6 Hz, 1H), 4.57 (d, J = 4.7 Hz, 1H), 4.49 (s, 1H), 3.76 (s, 2H), 3.55 (m, 1H), 3.32 (m, 2H), 2.59 (dd, J = 11.7, 4.3 Hz, 1H), 2.44 (dd, J = 11.7, 7.2 Hz, 1H), 2.10 (s, 1H). 1C NMR (100 MHz, DMSO-d 6) δ 162.40, 134.93, 133.54, 131.61, 116.80, 114.60, 71.01, 64.94, 52.60, 50.15. HR-ESI-MS (positive ion mode): m/z 234.0686 [M+H]+ (calcd for C10H13ClFNO2, 233.0619).
(S)-3-((3,4-Dichlorobenzyl)amino)propane-1,2-diol (6d)
White solid. Yield = 26.2%. C10H13Cl2NO2, mp 83.1 to 83.3°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.59 (d, J = 1.8 Hz, 1H), 7.55 (d, J = 8.2 Hz, 1H), 7.31 (dd, J = 8.2, 2.0 Hz, 1H), 4.54 (d, J = 4.4 Hz, 1H), 4.47 (s, 1H), 3.70 (s, 2H), 3.54 (m, 1H), 3.33 (m, 2H), 2.55 (dd, J = 11.8, 4.6 Hz, 1H), 2.40 (dd, J = 11.8, 7.2 Hz, 1H), 2.19 (s, 1H). 1C NMR (100 MHz, DMSO-d 6) δ 142.95, 131.23, 130.66, 130.17, 129.31, 128.64, 71.01, 64.96, 52.45, 52.17. HR-ESI-MS (positive ion mode): m/z 250.0389 [M+H]+ (calcd for C10H13Cl2NO2, 249.0323).
(S)-3-((4-Chlorobenzyl)amino)propane-1,2-diol (6e)
White solid. Yield = 15.4%. C10H14ClNO2, mp 77.4 to 77.9°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.39 (m, 4H), 4.53 (brs, 1H), 3.70 (s, 2H), 3.55 (m, 1H), 3.31 (m, 2H), 2.57 (dd, J = 11.7, 4.5 Hz, 1H), 2.42 (dd, J = 11.7, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 137.36, 133.00, 129.62, 128.67, 70.20, 65.43, 58.16, 53.06, 51.54, 18.35. HR-ESI-MS (positive ion mode): m/z 216.0777 [M+H]+ (calcd for C10H14ClNO2, 215.0713).
(S)-3-((3-Chloro-4-fluorobenzyl)amino)propane-1,2-diol (6f )
White solid. Yield = 17.3%. C10H13ClFNO2, mp 76.1 to 76.6°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.53 (d, J = 7.8 Hz, 1H), 7.36-7.30 (m, 2H), 4.55 (brs, 1H), 3.69 (s, 2H), 3.54 (m, 1H), 3.32 (m, 2H), 2.55 (dd, J = 11.8, 4.4 Hz, 1H), 2.40 (dd, J = 11.8, 7.2 Hz, 1H). 1C NMR (100 MHz, DMSO-d 6) δ 157.65, 139.48, 130.22, 128.85, 119.57, 116.97, 70.97, 64.97, 52.42, 52.16. HR-ESI-MS (positive ion mode): m/z 234.0680 [M+H]+ (calcd for C10H13ClFNO2, 233.0619).
(S)-3-((3,5-Di-tert-butyl-2-hydroxybenzyl)amino)propane-1,2-diol (6g )
Light yellow oil. Yield = 19.5%. C18H31NO3. 1H NMR (400 MHz, DMSO-d 6) δ 7.07 (d, J = 2.3 Hz, 1H), 6.86 (d, J = 2.2 Hz, 1H), 3.87 (m, 2H), 3.58 (m, 1H), 3.32 (m, 2H), 2.64 (dd, J = 11.8, 4.4 Hz, 1H), 2.45 (dd, J = 11.8, 7.2 Hz, 1H), 1.35 (s, 9H), 1.23 (s, 9H). 1C NMR (100 MHz, CDCl3) δ 153.01, 136.01, 130.11, 129.19, 125.41, 124.96, 69.67, 67.78, 65.82, 65.07, 58.92, 58.32, 55.87, 54.29, 52.06, 34.34, 30.31, 18.40. HR-ESI-MS (positive ion mode): m/z 310.2384 [M+H]+ (calcd for C18H31NO3, 309.2304).
(S)-3-((Pyridin-4-ylmethyl)amino)propane-1,2-diol (6h )
Light yellow oil. Yield = 22.9%. C9H14N2O2. 1H NMR (400 MHz, DMSO-d 6) δ 8.49 (d, J = 6.0 Hz, 1H), 7.35 (d, J = 5.8 Hz, 2H), 3.77 (s, 2H), 3.57 (m, 1H), 3.34 (m, 2H), 2.60 (dd, J = 11.2, 4.8 Hz, 1H), 2.44 (dd, J = 11.2, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 149.84(2C), 148.87, 122.99(2C), 69.87, 65.56, 52.63, 51.84. HR-ESI-MS (positive ion mode): m/z 183.1122 [M+H]+ (calcd for C9H14N2O2, 182.1055).
(S)-3-(((5-(4-Nitrophenyl)furan-2-yl)methyl)amino)propane-1,2-diol (6i)
Brown solid. Yield = 34.6%. C14H16N2O5, mp 111.8 to 112.6°C. 1H NMR (400 MHz, DMSO-d 6) δ 8.27 (d, J = 9.0 Hz, 2H), 7.91 (d, J = 9.0 Hz, 2H), 7.24 (d, J = 3.4 Hz, 1H), 6.49 (d, J = 3.4 Hz, 1H), 4.60 (s, 1H), 3.81 (s, 2H), 3.61-3.51 (m, 1H), 3.42-3.27 (m, 2H), 2.67 (dd, J = 11.8, 4.4 Hz, 1H), 2.51 (m, 1H). 1C NMR (100 MHz, DMSO-d 6) δ 157.58, 150.39, 146.00, 136.64, 124.88, 124.08, 111.63, 110.37, 70.92, 64.95, 56.50, 52.58, 46.28, 19.01. HR-ESI-MS (positive ion mode): m/z 293.1135 [M+H]+ (calcd for C18H31NO3, 292.1059).
(S)-3-(((1-Methyl-5-nitro-1H-imidazol-2-yl)methyl)amino)propane-1,2-diol (6j )
Yellow oil. Yield = 39.4%. C8H14N4O4. 1H NMR (400 MHz, DMSO-d 6) δ 8.01 (s, 1H), 4.61 (s, 1H), 3.91 (s, 3H), 3.87 (s, 2H), 3.51 (m, 1H), 3.38-3.24 (m, 2H), 2.62 (dd, J = 12.0, 4.4 Hz, 1H), 2.45 (dd, J = 11.8, 7.4 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 131.70, 70.24, 65.12, 58.41, 51.92, 46.00, 33.26, 18.41. HR-ESI-MS (positive ion mode): m/z 231.1095 [M+H]+ (calcd for C8H14N4O4, 230.1015).
(S)-3-((3-(4-Methoxyphenoxy)benzyl)amino)propane-1,2-diol (6k )
Yellow oil. Yield = 32.7%. C17H21NO4. 1H NMR (400 MHz, DMSO-d 6) δ 7.26 (t, J = 7.8 Hz, 1H), 7.05-6.93 (m, 5H), 6.91 (s, 1H), 6.76 (dd, J = 8.2, 2.0 Hz, 1H), 4.54 (s, 1H), 3.75 (s, 3H), 3.67 (s, 2H), 3.57-3.47 (m, 1H), 3.32 (m, 2H), 2.56 (dd, J = 11.8, 4.6 Hz, 1H), 2.41 (dd, J = 11.8, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 158.75, 155.95, 149.91, 141.18, 129.75, 122.24, 120.90(2C), 117.37, 116.25, 114.90(2C), 69.98, 65.50, 55.66, 53.48, 51.56. HR-ESI-MS (positive ion mode): m/z 304.1512 [M+H]+ (calcd for C17H21NO2, 303.1471).
(S)-3-((Quinolin-3-ylmethyl)amino)propane-1,2-diol (6l )
Yellow oil. Yield = 16.6%. C13H16N2O4. 1H NMR (400 MHz, DMSO-d 6) δ 8.80 (s, 1H), 8.01 (d, J = 8.0 Hz, 1H), 7.95 (s, 1H), 7.68 (d, J = 8.0 Hz, 1H), 7.60 (t, J = 8.0 Hz, 1H), 7.45 (t, J = 8.0 Hz, 1H), 3.84 (d, J = 2.8 Hz, 2H), 3.60 (dd, J = 7.6 Hz, 1H), 3.52 (dd, J = 7.6 Hz, 1H), 3.42 (s, 1H), 2.66 (m, 2H). 1C NMR (100 MHz, CDCl3) δ 151.19, 146.96, 135.12, 132.24, 129.36, 128.64, 127.83, 127.64, 126.85, 70.43, 65.28, 51.73, 51.21. HR-ESI-MS (positive ion mode): m/z 233.1279 [M+H]+ (calcd for C13H16N2O2, 232.1212).
(S)-3-((3-Nitro-4-(pyrrolidin-1-yl)benzyl)amino)propane-1,2-diol (6m )
Brown solid. Yield = 33.0%. C14H21N3O4, mp 77.2 to 77.4°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.71 (d, J = 2.0 Hz, 1H), 7.50 (dd, J = 8.4, 2.2 Hz, 1H), 7.24 (d, J = 8.4 Hz, 1H), 4.54 (d, J = 4.4 Hz, 1H), 3.68 (s, 2H), 3.53 (m, 1H), 3.39-3.24 (m, 2H), 2.96-2.88 (m, 4H), 2.55 (dd, J = 11.8, 4.6 Hz, 1H), 2.40 (dd, J = 11.8, 7.2 Hz, 1H), 1.59 (d, J = 5.0 Hz, 4H), 1.52 (s, 2H). 1C NMR (100 MHz, CDCl3) δ 146.18, 142.27, 133.32, 132.16, 125.56, 121.04, 70.21, 65.45, 52.99, 52.47, 51.50, 25.94, 23.99, 18.37. HR-ESI-MS (positive ion mode): m/z 296.1510 [M+H]+ (calcd for C14H21N3O4, 295.1532).
(S)-3-((Pyrazin-2-ylmethyl)amino)propane-1,2-diol (6n )
Brown oil. Yield = 35.2%. C8H13N3O2. 1H NMR (400 MHz, DMSO-d 6) δ 8.70 (d, J = 0.9 Hz, 1H), 8.60-8.55 (m, 1H), 8.52 (d, J = 2.4 Hz, 1H), 4.61 (brs, 1H), 3.89 (s, 2H), 3.57 (m, 1H), 3.33 (m, 2H), 2.66 (dd, J = 11.8, 4.3 Hz, 1H), 2.59-2.44 (m, 1H). 1C NMR (100 MHz, CDCl3) δ 154.71, 144.26, 143.35, 70.41, 65.26, 58.18, 52.36, 51.88. HR-ESI-MS (positive ion mode): m/z 184.1070 [M+H]+ (calcd for C8H13N3O2, 183.1008).
(S)-3-((3,4-Difluorobenzyl)amino)propane-1,2-diol (6o)
Light yellow oil. Yield = 30.6%. C10H13F2NO2. 1H NMR (400 MHz, DMSO-d 6) δ 7.44-7.31 (m, 2H), 7.21-7.15 (m, 1H), 4.58 (brs, 1H), 3.73 (s, 2H), 3.56 (m, 1H), 3.33 (m, 2H), 2.59 (dd, J = 11.8, 4.4 Hz, 1H), 2.43 (dd, J = 11.8, 7.4 Hz, 1H). 1C NMR (100 MHz, DMSO-d 6) δ 151.03, 148.60, 139.24, 124.85, 117.47, 117.12, 70.90, 64.94, 52.36, 52.24. HR-ESI-MS (positive ion mode): m/z 218.0975 [M+H]+ (calcd for C10H13ClFNO2, 217.0914).
(S)-3-((Pyridin-2-ylmethyl)amino)propane-1,2-diol (6p )
Yellow oil. Yield = 23.8%. C9H14N2O2. 1H NMR (400 MHz, DMSO-d 6) δ 8.51 (d, J = 1.8 Hz, 1H), 8.44 (dd, J = 4.8, 1.6 Hz, 1H), 7.73 (d, J = 7.8 Hz, 1H), 7.34 (dd, J = 7.6, 5.0 Hz, 1H), 4.58 (brs, 1H), 3.74 (s, 2H), 3.55 (m, 1H), 3.32 (m, 2H), 2.58 (dd, J = 11.8, 4.4 Hz, 1H), 2.43 (dd, J = 11.8, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 158.75, 149.12, 136.91, 122.73, 122.32, 70.30, 65.28, 54.55, 51.89. HR-ESI-MS (positive ion mode): m/z 183.1125 [M+H]+ (calcd for C9H14N2O2, 182.1055).
(R)-3-((3,4-Dimethylbenzyl)amino)propane-1,2-diol (9a)
Yellow oil. Yield = 38.7%. C12H19NO2. 1H NMR (400 MHz, DMSO-d 6) δ 7.02 (d, J = 8 Hz, 1H), 7.01 (s, 1H), 6.97 (d, J = 8 Hz, 1H), 4.24 (brs, 3H), 3.77 (m, 1H), 3.64 (m, 2H), 3.47 (m, 2H), 2.58 (m, 2H), 2.20 (d, J = 4 Hz, 6H). 1C NMR (100 MHz, CDCl3) δ 136.73, 136.04, 135.67, 129.78(2C), 125.81, 69.96, 65.39, 53.27, 51.35, 19.75,19.43. HR-ESI-MS (positive ion mode): m/z 210.1473 [M+H]+ (calcd for C12H19NO2, 209.1416).
(R)-3-((2-Chloro-4-fluorobenzyl)amino)propane-1,2-diol (9b)
White solid. Yield = 33.7%. C10H13ClFNO2, mp 82.0 to 82.3°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.26 (d, J = 12 Hz,1H), 7.04 (d, J = 8 Hz, 1H), 6.88 (t, J = 16 Hz, 1H), 3.78 (m, 2H), 3.72 (m, 1H), 3.55 (m, 2H), 3.02 (brs, 3H), 2.65 (m, 2H). 1C NMR (100 MHz, DMSO-d 6) δ 162.41, 134.89, 133.55, 131.63, 116.80, 114.60, 70.99, 64.94, 52.59, 50.13. HR-ESI-MS (positive ion mode): m/z 234.0682 [M+H]+ (calcd for C10H13ClFNO2, 233.0619).
(R)-3-((3,4-Dichlorobenzyl)amino)propane-1,2-diol (9c )
White solid. Yield = 29.5%. C10H13Cl2NO2, mp 81.7 to 81.9°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.59 (d, J = 2.0 Hz, 1H), 7.56 (d, J = 8.2 Hz, 1H), 7.32 (dd, J = 8.2, 2.0 Hz, 1H), 4.55 (s, 1H), 3.70 (s, 2H), 3.53 (m, 1H), 3.40-3.24 (m, 3H), 2.55 (dd, J = 11.8, 4.6 Hz, 1H), 2.40 (dd, J = 11.8, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 139.68, 132.47, 131.17, 130.47, 130.10, 127.58, 70.24, 65.39, 52.65, 51.51. HR-ESI-MS (positive ion mode): m/z 250.0383 [M+H]+ (calcd for C10H13Cl2NO2, 249.0323).
(R)-3-((4-Chlorobenzyl)amino)propane-1,2-diol (9d)
White solid. Yield = 21.6%. C10H14ClNO2, mp 79.9 to 80.7°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.28 (d, J = 8 Hz, 2H), 7.22 (d, J = 8 Hz, 2H), 3.74 (m, 3H), 3.63 (m, 1H), 3.52 (m, 1H), 3.27 (brs, 3H), 2.68 (m, 2H). 1C NMR (100 MHz, CDCl3) δ 137.78, 133.00, 129.62(2C), 128.67(2C), 70.22, 65.46, 53.07, 51.55. HR-ESI-MS (positive ion mode): m/z 216.0776 [M+H]+ (calcd for C10H14ClNO2, 215.0713).
(R)-3-((3-Chloro-4-fluorobenzyl)amino)propane-1,2-diol (9e)
White solid. Yield = 18.8%. C10H13ClFNO2, mp 75.8 to 76.2°C. 1H NMR (400 MHz, DMSO-d 6) δ 7.37 (dd, J = 2, 5.2 Hz, 1H), 7.17 (m, 1H), 7.09 (t, J = 16 Hz, 1H), 3.78 (m, 1H), 3.76 (m, 2H), 3.72 (m, 1H), 3.58 (m, 1H), 2.80 (dd, J = 4, 12 Hz, 1H), 2.70 (dd, J = 8.0, 12.0 Hz, 1H), 2.60 (brs, 3H). 1C NMR (100 MHz, DMSO-d 6) δ 157.67, 139.27, 130.20, 128.83, 119.58, 116.98, 70.91, 64.96, 52.38, 52.11. HR-ESI-MS (positive ion mode): m/z 234.0681 [M+H]+ (calcd for C10H13ClFNO2, 233.0619).
(R)-3-((3,5-Di-tert-butyl-2-hydroxybenzyl)amino)propane-1,2-diol (9f )
Light yellow oil. Yield = 16.9%. C18H31NO3. 1H NMR (400 MHz, DMSO-d 6) δ 7.22 (d, J = 2.4 Hz, 1H), 6.86 (d, J = 2.4 Hz, 1H), 4.28 (brs, 2H), 3.98 (s, 2H), 3.91 (m, 1H), 3.70 (m, 1H), 3.57 (m, 1H), 2.76 (m, 2H), 1.41 (s, 9H), 1.28 (s, 9H). 1C NMR (100 MHz, DMSO-d 6) δ 155.11, 139.66, 134.89, 123.46, 122.89, 121.94, 70.59, 64.73, 53.20, 51.84, 34.87, 34.20, 32.03(3C), 29.97(3C). HR-ESI-MS (positive ion mode): m/z 310.2381 [M+H]+ (calcd for C18H31NO3, 309.2304).
(R)-3-((Pyridin-4-ylmethyl)amino)propane-1,2-diol (9g )
Yellow oil. Yield = 18.1%. C9H14N2O2. 1H NMR (400 MHz, DMSO-d 6) δ 8.55 (m, 2H), 7.25 (m, 2H), 3.83 (m, 3H), 3.74 (m, 1H), 3.61 (m, 1H), 2.78 (m, 2H). 1C NMR (100 MHz, CDCl3) δ 149.74(2C), 148.97, 123.04(2C), 70.02, 65.51, 52.60, 51.84. HR-ESI-MS (positive ion mode): m/z 183.1115 [M+H]+ (calcd for C9H14N2O2, 182.1055).
(R)-3-(((5-(4-Nitrophenyl)furan-2-yl)methyl)amino)propane-1,2-diol (9h )
Brown solid. Yield = 37.2%. C14H16N2O5, mp 115.5 to 116.1°C. 1H NMR (400 MHz, DMSO-d 6) δ 8.26 (d, J = 9.0 Hz, 2H), 7.91 (d, J = 9.0 Hz, 2H), 7.23 (d, J = 3.4 Hz, 1H), 6.48 (d, J = 3.4 Hz, 1H), 4.59 (s, 1H), 3.80 (s, 2H), 3.59-3.50 (m, 1H), 3.33 (p, J = 10.8 Hz, 3H), 2.66 (dd, J = 11.8, 4.4 Hz, 1H), 2.50 (q, J = 7.6 Hz, 2H). 1C NMR (100 MHz, DMSO-d 6) δ 157.58, 150.39, 146.01, 136.64, 124.88, 124.08, 111.63, 110.37, 70.91, 64.95, 56.50, 52.58, 46.28, 19.00. HR-ESI-MS (positive ion mode): m/z 293.1140 [M+H]+ (calcd for C18H31NO3, 292.1059).
(R)-3-((3-Fluorobenzyl)amino)propane-1,2-diol (9i)
Light yellow oil. Yield = 31.4%. C10H14FNO2. 1H NMR (400 MHz, DMSO-d 6) δ 7.35 (td, J = 8.0, 6.4 Hz, 1H), 7.20-7.14 (m, 1H), 7.04 (td, J = 8.4, 2.2 Hz, 1H), 4.58 (s, 1H), 3.74 (s, 2H), 3.56 (s, 1H), 3.33 (s, 2H), 2.58 (dd, J = 11.8, 4.4 Hz, 1H), 2.43 (dd, J = 11.8, 7.2 Hz, 1H). 1C NMR (100 MHz, CDCl3) δ 164.17, 142.02, 130.05, 123.75, 115.09, 114.21, 70.23, 65.42, 53.24, 51.55. HR-ESI-MS (positive ion mode): m/z 200.1060 [M+H]+ (calcd for C10H14FNO2, 199.1009).
Antitumor Activity
Antitumor activity assays were carried out by MTT method on H460 cells (human nonsmall-cell lung cancer), TMD-8 cells, and MV4-11 cells (biphenotypic B myelomonocytic leukemia cells). Cells were plated into 96-well plates at a density of 1 × 104 cells per well in 100 mL of medium and grown for 48 hours. The cells were then exposed to the tested compounds at different concentrations (0.025-20 μM) for 48 hours. A 0.5% MTT solution was added to each well. After further incubation for another 4 hours, formazan formed from MTT was extracted in 150 µL of DMSO for 15 minutes standby. Absorbance at 570 nm was then determined on a microplate reader. In brief, the mean percentage of cell survival rates relative to that of untreated cells was estimated from the data of 6 individual experiments.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Chun-Hui Project from Ministry of Education of China (No. Z2017062), the Open Research Subject of Key Laboratory of Food Biotechnology of Sichuan province of China (No. szj2016-021), and the Center of Comprehensive Health Management (No. szj2017-043).
