Abstract
This study aimed to investigate the effect of Cordyceps militaris extract on the proliferation and apoptosis of carboplatin- resistant SKOV-3 and determine the underlying mechanisms for overcoming carboplatin resistance in human ovarian cancer. We cultured the carboplatin-resistant SKOV-3 cells in vitro until the exponential growth phase and then treated with different concentrations of C. militaris for 24, 48, and 72 hours. We performed cell proliferation assay, cell morphological change assessment using transmission electron microscopy, apoptosis assay, and immunoblotting to measure the protein expression of caspase-3 and -8, poly (ADP-ribose) polymerase (PARP)-1, B-cell lymphoma (Bcl)-2, and activating transcription factor 3 (ATF3)/TP53 signaling-related proteins. As a result, C. militaris reduced the viability of carboplatin-resistant SKOV-3 and induced morphological disruptions in a dose- and time-dependent manner. The gene expression profiles indicated a reprogramming pattern of the previously known and unknown genes and transcription factors associated with the action of TCTN3 on carboplatin-resistant SKOV-3 cells. We also confirmed the C. militaris-induced activation of the ATF3/TP53 pathway. Immunoblotting indicated that cotreatment of C. militaris and carboplatin-mediated ATF3/TP53 upregulation induced apoptosis in the carboplatin-resistant SKOV-3 cells, which are involved in the serial activation of pro-apoptotic proteins, including Bcl-2, Bax, caspases, and PARP-1. Further, when the ATF3 and TP53 expression increased, the CHOP and PUMA expressions were upregulated. Consequently, the upregulated CHOP/PUMA expression activated the positive regulation of the apoptotic signaling pathway. In addition, it decreased the Bcl-2 expression, leading to marked ovarian cancer cells sensitive to carboplatin by enhancing apoptosis. We then corroborated these results using in vivo experiments. Taken together, C. militaris inhibits carboplatin-resistant SKOV-3 cell proliferation and induces apoptosis possibly through ATF3/TP53 signaling upregulation and CHOP/PUMA activation. Therefore, our findings provide new insights into the treatment of carboplatin-resistant ovarian cancer using C. militaris.
Ovarian cancer is the sixth most common cancer and the seventh most common cause of cancer-related deaths among women worldwide. Worldwide, it is the leading cause of gynecological cancer death and the fourth leading cause of cancer death in women. 1 Approximately 70% of patients are diagnosed at advanced stages (III and IV), and the 5-year survival rate for these patients is only 30%. 2 The standard treatment involves the partial removal of tumors followed by a chemotherapeutic scheme based on platinum drugs, including carboplatin and taxanes. 3 Carboplatin treatment of cancer cells induces apoptosis, a highly regulated cell death program. Despite an initial clinical response in most patients (70%-80%), recurrence frequently occurs with acquired resistance to carboplatin especially in ovarian cancer. 4 The various management and treatment strategies of ovarian cancer have improved over the past 20 years. The development of an antiangiogenic treatment for chemotherapy backbone therapy at the first recurrence 5 and annexation of an anti-poly (ADP-ribose) polymerase (PARP) maintenance treatment for platinum-sensitive relapsed patients 6 have yielded clinical improvements. Further, combination chemotherapy for the treatment of women with platinum-sensitive/resistant epithelial ovarian cancer has been developed and was evaluated in multiple phase III studies. However, there is still a crucial need to develop new therapies that can manage carboplatin or multidrug resistance that ultimately occurs for most patients with ovarian cancer. Tumor cells can resist apoptosis by increasing the expression of proteins blocking pro-apoptotic pathways among other processes. 7 Therefore, overcoming the complicated mechanisms of cancer resistance in relation to antitumor drugs, and survival and activating cancer cell death through apoptosis are the focus of the current trends in cancer research and drug development.
Cordyceps militaris is a parasitic mushroom that has been traditionally used as a herbal medicine in Korea and China to enhance longevity and vitality. 8,9 The few well-known active ingredients from this mushroom include cordycepin, cordycepic acid, sterols (ergosterol), nucleosides, and polysaccharides. 10 Cordyceps militaris has been reported to exert immunomodulatory, anti-inflammatory, antimicrobial, and antitumor effects, although the primary pharmacological activity slightly varies depending on the main ingredients in its extract. 11,12 Both in vivo and in vitro experiments have demonstrated the antiproliferative and apoptotic activities of C. militaris extract (CME) against human tumor cell lines, including H460, RKO, PC-3, MDA-MB-231, and HepG2 cells. Cordyceps militaris extract exhibited antitumor effects mainly through the induction of apoptosis, inhibition of angiogenesis, and suppression of invasion and metastasis in cancer cells. 13 -15 Cordyceps militaris has recently received considerable attention as a potential source of anticancer drugs. However, the molecular mechanism underlying the inhibitory effects of C. militaris on tumor cell growth remains unclear. In this study, we tested the in vitro cytotoxicity of C. militaris in carboplatin-sensitive ovarian cancer cell lines (SKOV-3) and carboplatin-resistant SKOV-3 cell lines.
First, to study the chemoresistance in human ovarian adenocarcinoma, we established carboplatin-resistant ovarian adenocarcinoma models on SKOV-3 cell lines. Thereafter, we applied carboplatin on parental SKOV-3 continuously for approximately 6 months and then maintained it continuously in the presence of cisplatin at the new IC50 concentrations for another 6 months. In the present study, we investigated the anticancer effect of C. militaris on carboplatin-resistant SKOV-3 human ovarian cancer cells. First, we performed a cell counting kit (CCK)-8 assay to determine the effect of carboplatin on the cell viability carboplatin-resistant and original parental SKOV-3. As shown in Figure 1(a), the carboplatin-resistant SKOV-3 cells exhibited significant resistance to carboplatin compared with the corresponding SKOV-3 cells.

Evaluation of carboplatin resistance in human ovarian cancer cells. (a) Establishment of carboplatin resistance of SKOV-3 cells. Carboplatin-resistant SKOV-3 and SKOV-3 cells were treated with 5, 10, 20, 50, 100, and 200 µg/mL of carboplatin for 24, 48, and 72 hours. The viability of 2 cell lines was estimated using the cell counting kit-8 assay. The experiment was performed in triplicate. Carboplatin-resistant SKOV-3 significantly was established resistance to 50 µg/mL carboplatin. (b) Morphological changes of carboplatin-resistant SKOV-3 treated with carboplatin compared with control (vehicle). Microscopic images of A549 treated with Cordyceps militaris for 48 hours and 72 hours. Magnification ×400. The morphological characteristics of carboplatin-resistant SKOV-3 in control, 50 and 100 μg/mL carboplatin were little differed. Each point represents the mean ± standard deviation of 3 independent experiments. Statistical significance was considered that * means P < 0.05, ** means P < 0.01, *** means P < 0.001, and ns means nonsignificance.
Remarkably, chemoresistance to carboplatin in the SKOV-3 cells decreased the apoptosis rate and upregulated the IC50 values. In contrast, the original parental cells increased the apoptosis rate and additionally decreased the IC50 values. In addition, the analysis of the morphological changes showed that the carboplatin-resistant SKOV-3 cells did not differ significantly from the controls (Figure 1(b)). Taken together, we confirmed that we successfully established a carboplatin-resistant SKOV-3 cell line.
And then, we confirmed the cytotoxic effects of C. militaris on carboplatin-resistant SKOV-3 proliferation and the cells were directly treated with 0, 10, 50, 100, 200, 500, or 1000 µg/mL of C. militaris for 24, 48, and 72 hours. First of all, C. militaris inhibited the growth of cells in a dose- and time-dependent manner. Treatment with 500 and 1000 µg/mL of extract for 48 and 72 hours resulted in the inhibition of approximately half of the carboplatin-resistant SKOV-3 cell population (Figure 2(a)). In addition, 50 µg/mL of carboplatin considering the IC50 concentration was coadministered with C. militaris to determine whether C. militaris increases the drug sensitivity of resistant SKOV-3 to carboplatin. The cell viability assays showed that C. militaris treatment significantly enhanced the carboplatin-induced cell death in carboplatin-resistant SKOV-3. The anticancer effect of C. militaris appeared more in the cotreatment with carboplatin than in C. militaris treatment alone (Figure 2(a)). In addition, the morphologies of the treated and untreated cells were compared under a light microscope to observe the effects of C. militaris on carboplatin-resistant SKOV-3. The morphology of the carboplatin-resistant SKOV-3 cells drastically changed after treatment with 500 µg/mL of C. militaris for 24 and 48 hours (Figure 2(b)). In the C. militaris/carboplatin cotreated resistant SKOV-3 cells, the cell body began to collapse significantly even at 100 µg/mL of C. militaris. Thus, we found that C. militaris was an effective adjuvant therapeutic drug for carboplatin-based chemotherapy in carboplatin-resistant SKOV-3 cells.

Cordyceps militaris reverses carboplatin resistance and decreases cell proliferation of chemoresistant SKOV-3 cells. (a) Carboplatin-resistant SKOV-3 were exposed to 0 (vesicle), 25, 50, 100, 200, 500, and 1000 µg/mL C ordyceps militaris extract or cotreated with 50 µg/mL carboplatin and Cordyceps militaris for 24, 48, and 72 hours before estimation of cell number using the cell counting kit-8 assay. The experiment was performed in triplicate. Cordyceps militaris significantly inhibited cell proliferation of carboplatin-resistant SKOV-3 and upregulated sensitivity to carboplatin. (b) Morphological changes of carboplatin-resistant SKOV-3 were treated with Cordyceps militaris alone or cotreated with 50 µg/mL carboplatin and Cordyceps militaris compared with control (vehicle). Magnification ×400. The statistics demonstrated that the percentage of the cells mainly represents treated cells, which was apparent when the percentage of control cells markedly decreased. Data are presented as means ± standard deviations from triplicate experiments. Statistical significance was considered that * means P < 0.05, ** means P < 0.01, *** means P < 0.001, and ns means nonsignificance.
Whether C. militaris increases the apoptosis rate in carboplatin-resistant SKOV-3 cells, we proceeded the apoptosis analysis via propidium iodide (PI)/Annexin V staining. To investigate the establishment of carboplatin-resistant SKOV-3 and apoptotic effect of C. militaris on carboplatin-resistant SKOV-3, the cells were analyzed with the Annexin V- and PI-stained cells using flow cytometry after the 24- and 48-hour treatment with the control and 0 (negative control), 100, 500, and 1000 µg/mL of C. militaris. The assay was performed to evaluate how far cancer cell death was induced by C. militaris. In addition, the apoptosis rate increase was also investigated by cotreatment with C. militaris and carboplatin to confirm the upregulation of the sensitivity of carboplatin by C. militaris. The relative proportion of nonviable cells was quantitatively measured as those at the early stage of apoptosis (Annexin V-stained, nondisrupted cells) or as those entering the late stage of apoptosis (disrupted or lysed cells). Compared with the control group, apoptosis was not induced in the 50 and 100 µg/mL carboplatin-treated cells, and the analysis found that carboplatin resistance was established in SKOV-3 (Figure 3(a)). The flow cytometry analysis showed that C. militaris-induced apoptosis in the carboplatin-resistant SKOV-3 cells reported a reduction in the viable fraction at 1000 µg/mL of C. militaris for 48 hours from 92.30% to 69.89%, rapidly increasing the apoptotic stage from 5.36% to 29.56%. In the C. militaris and 50 µg/mL carboplatin cotreatment, C. militaris significantly enhanced the apoptotic effect of carboplatin (Figure 3(a)). Taken together, these results demonstrate that adjuvant treatment with C. militaris could reverse the resistance of SKOV-3 cells to carboplatin-induced apoptosis. In addition, to confirm the development of apoptotic bodies after C. militaris treatment and to visualize the ultrastructural changes in the carboplatin-resistant SKOV-3 apoptotic cells, we used transmission electron microscopy (TEM) (Figure 4). The apoptotic bodies were observed in the cells treated with 500 µg/mL of C. militaris; these bodies were spherical protuberances containing fragmented and segregated chromatin clumps, which separated from the cell surface (Figure 4(b)). The C. militaris-exposed cells showed damaged mitochondria and autophagosomes containing dense organelles a day after treatment (Figure 4(b)). In addition, combination treatment of C. militaris and carboplatin induced more apoptosis changes mainly including cytoplasmic shrinkage, blebbing of the plasma membrane, chromatin condensation, and the formation of apoptotic bodies (Figure 4(c)). This result suggested that C. militaris was an effective adjuvant therapeutic drug for carboplatin-based chemotherapy in carboplatin-resistant SKOV-3 cells. Conversely, the untreated carboplatin-resistant SKOV-3 cells had intact plasma membranes and ordered chromatin (Figure 4(a)).

C ordyceps militaris increases the apoptosis rate and sensitivity to carboplatin in carboplatin-resistant SKOV-3 cells. Flow cytometry analysis was performed with carboplatin-resistant SKOV-3 cells after treatment with the indicated concentrations of (a) carboplatin alone or (b) Cordyceps militaris alone or (c) in combination with 50 µg/mL of carboplatin for 24 and 48 hours. The cells were stained using Annexin V-fluorescein isothiocyanate Apoptosis Detection Kit and the apoptosis array was determined by Guava system (Millipore). The results are expressed as the percentage of early and late apoptotic cells, which increased as the percentage of live cells markedly decreased. The experiment was performed in triplicate.
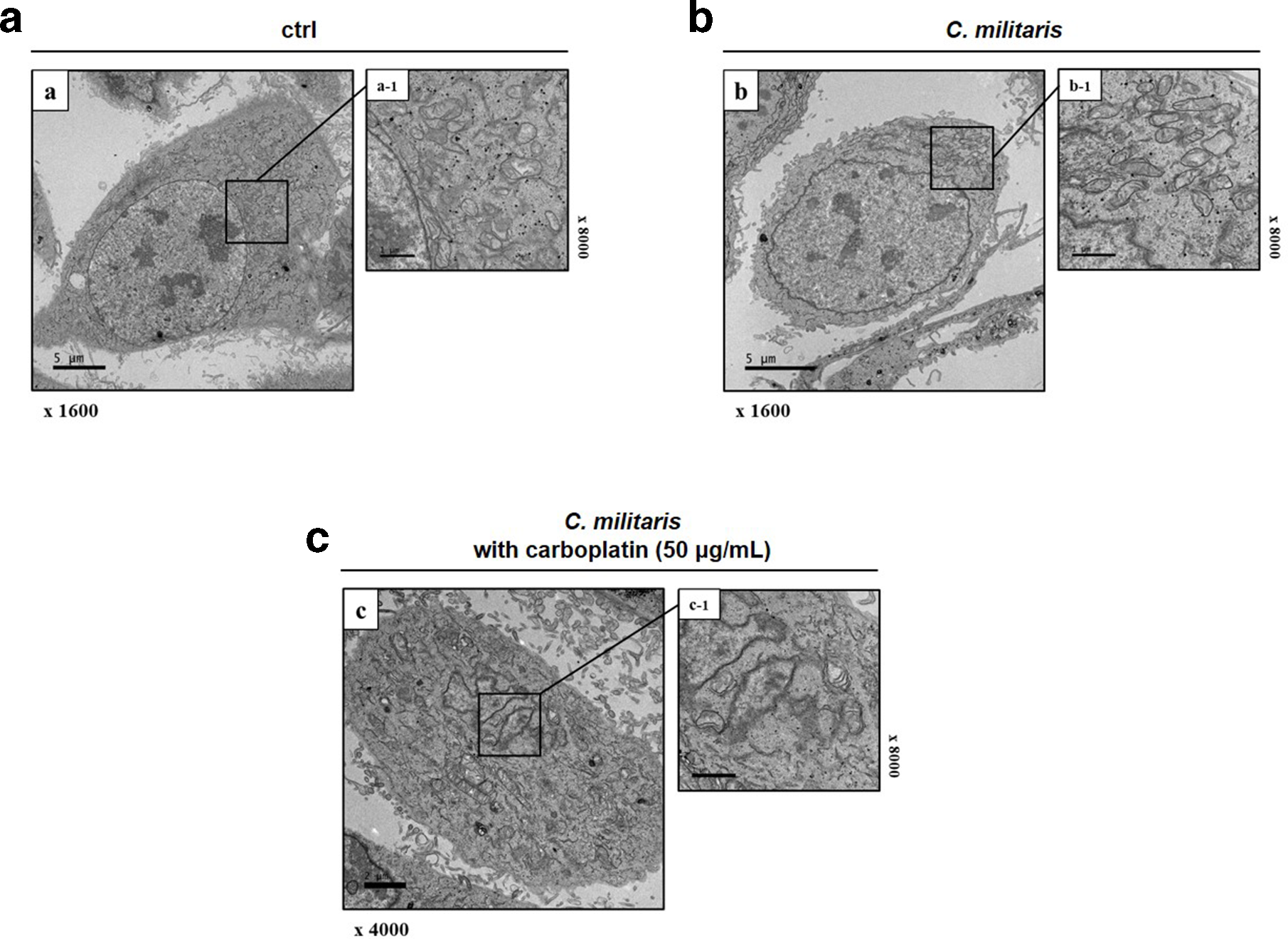
Detection to the morphological ultrastructural appearance of apoptotic bodies through transmission electron microscopy for identification that Cordyceps militaris reverses carboplatin resistance. (a) Untreated carboplatin-resistant SKOV-3 and these cells were incubated with (b) Cordyceps militaris at 500 µg/mL, or (c) in combination with 50 µg/mL of carboplatin for 48 hours analyzed the production of apoptotic bodies by transmission electron microscopy. The typical apoptotic bodies in Cordyceps militaris-treated carboplatin-resistant SKOV-3 were spherical protuberances containing fragmentation and segregation of chromatin clumps separated from cell surface compared with untreated carboplatin-resistant SKOV-3. Mitochondrial disruptions, autophagosomes, and autolysosome were more detected in Cordyceps militaris with 50 µg/mL carboplatin-exposed cells. Representative images are shown.
Carboplatin is a representative platinum-based chemotherapeutic drug. It exhibits effective activity against various solid tumors, especially ovarian cancers. 16 The majority of patients with ovarian cancer respond to initial therapy with tumor cytoreductive surgery and platinum-based chemotherapy. 17 However, approximately 70% of patients with advanced-stage cancer will develop recurrent cancer and eventually succumb to recurrent disease typically characterized by administration of multiple drugs. 18,19 Further, the carboplatin resistance of ovarian cancer cells has a very low activity of apoptotic signaling caused by carboplatin treatment. In the present study, our established carboplatin-resistant SKOV-3 cells showed resistance to carboplatin treatment with significant low induction of the apoptosis pathway. Therefore, increasing the sensitivity of carboplatin-resistant SKOV-3 cells to apoptotic signaling may represent an effective strategy against drug resistance.
Additionally, identification of natural products to overcome drug resistance represents another important strategy, as drug resistance can lead to treatment failure in clinical settings and remains an obstacle in cancer therapy. 20 Management of drug resistance is important toward successful chemotherapy. There are many reports that natural products can overcome cancer cell drug resistance, which deserves sharing with scientific and industrial communities. 21,22 Considering the complexity of various medicinal herbs, application of artificial intelligence may promote the development of anticancer drugs from many medicinal herbs. 23
As mentioned earlier, C. militaris has been known to have various biological functions including antitumor, antiviral, antioxidant, and anti-inflammatory activities. A few studies have reported the anticancer activity of C. militaris. 15,16,24 In our previous study, we investigated the anticancer effect of C. militaris on human ovarian cancer and nonsmall-lung adenocarcinoma cells. We found that C. militaris reduced the viability and migration activities, indicative of its potential ability to mediate apoptosis. In addition, apoptosis was induced in human ovarian cancer and nonsmall-lung adenocarcinoma cells by C. militaris, which was related to the suppression of the nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) signaling pathway by downregulating the tumor necrosis factor-α (TNF-α) signaling pathway in SKOV-3 ovarian cancer cells. NF-κB activation has also been identified as a key mechanism of chemoresistance. 25 The NF-κB activity inversely correlated with the cellular sensitivity to chemotherapy in carcinoma cell lines. Chemoresistant cells showed an increased expression of NF-κB compared with their matched drug sensitive parent, implicating NF-κB as a potential mediator of acquired drug resistance. 26 Therefore, based on our previous findings, in vitro and in vivo studies were conducted to determine whether C. militaris can reverse chemoresistance to carboplatin and induce cell death in carboplatin-resistant SKOV-3.
Using microarray analysis, we examined the C. militaris-induced apoptotic gene and protein expression pathways in carboplatin-resistant SKOV-3. To investigate the expression of the genes affected by C. militaris, microarray analyses of the C. militaris (500 µg/mL)-treated resistant SKOV-3 ovarian cancer cells were conducted. Among the 46 735 genes assayed, 15 338 genes were expressed in the CME-treated cells. Among 4108 genes, C. militaris treatment upregulated and downregulated 1953 and 1842 genes, respectively, as compared with the observations reported in the untreated controls at 24 hours. From the gene expression array data, we clustered the significantly affected core apoptosis-related genes (Figure 5(a)). Genes that were upregulated or downregulated by more than 2-fold following C. militaris treatment were categorized as “significant” in data mining. Biologically relevant features were constructed using the Excel-based differentially expressed gene analysis program. The lists of the 3-fold upregulated and downregulated cell death-related genes in the C. militaris-treated/carboplatin-resistant SKOV-3 cells were uploaded to the Multiple Experiment Viewer tool for heat map and hierarchical cluster analyses (Figure 5(a)). The heat maps and hierarchical clusters demonstrated 37 affected genes after CME treatment. Twenty-one of these genes were found to be downregulated, and 16 genes showed an upregulated expression (Figure 5(a)). As shown in Figure 5(a), the heat map and hierarchical cluster analysis demonstrated that C. militaris induced apoptosis in carboplatin-resistant SKOV-3 via upregulation of ATF3 and TP53 expression. Based on these results, protein-protein interaction and gene ontology analyses were performed using the STRING database. Fifteen genes, including those encoding ATF3 and TP53, were found to interact with each other (Figure 5(b)). Pathway analysis comparing between the nontreated and C. militaris-treated resistant SKOV-3 cells revealed the involvement of all related proteins in the regulation of the apoptotic process (GO: 0042987; false discovery rate P = 7.34-13: ATF2, ATF3, ATF4, Bax, B-cell lymphoma (Bcl)-2, caspase-3, caspase-8, FAS, MAPK8, MDM2, MDM4, TNFRSF10A, TNFRSF10B, and TP53), especially the positive apoptotic signaling pathway (GO: 0043065; false discovery rate P = 5.52-13: ATF2, ATF3, ATF4, Bax, Bcl-2, caspase-3, caspase-8, FAS, MAPK8, TNFRSF10A, TNFRSF10B, and TP53) (Figure 5(c)). In the Kyoto Encyclopedia of Gene and Genome analysis, related genes were involved in apoptosis, p53 signaling pathway, hepatitis B, platinum drug resistance, and TNF signaling pathway (Figure 5(c)). Thus, we hypothesized that C. militaris induces ATF3/TP53 signal transduction pathway-mediated apoptosis of chemoresistant ovarian cancer cells. We found that apoptosis of carboplatin-resistant SKOV-3 induced by C. militaris was associated with the alternation of ATF3 and TP53 expression. In other researches, ATF3 is well recognized as a stress-responsive protein. 27

Systematic analysis of gene expression alternation to identify the biomarker induced by Cordyceps militaris in carboplatin-resistant SKOV-3. (a) Hierarchical gene clustering was performed with the TM4 Microarray Software Suite (MeV) for Cordyceps militaris-treated carboplatin-resistant SKOV-3 cells. Heat map revealed the genes that underwent more than 2-fold changes after cell death in response to Cordyceps militaris treatment. The red and blue colors represent more than 3-fold change in the expression of upregulated and downregulated genes, respectively. The ratios of gene profiles are presented as heat map (left panel) and gene expression pattern (right panel). (b) Combined screenshots from the STRING website, showing the results obtained after entering a set of 15 proteins thought to be involved in the apoptotic signaling pathway. The insets are shown as follows (from top to bottom): the accessory information available for a single protein, reported enrichment of functional connections among the set of proteins, and statistical enrichments detected in functional subsystems. (c) Pathway analysis comparing between the nontreated and Cordyceps militaris-treated resistant SKOV-3 cells using Gene Ontology and KEGG Pathway analysis. One enriched function was selected, and the corresponding protein nodes in the network were automatically highlighted.
Activating transcription factor 3 (ATF3), an ATF/cAMP-responsive element-binding protein family member, is an adaptive responsive gene that is induced by a wide range of cellular stresses, including DNA damage, cellular injury, and oxidative stress. 27 Upregulation of ATF3 by intra- and extracellular stresses induces activation of genes associated with alleviation of cellular stress. However, when stresses cannot be overcome, the expression of ATF3 must be enhanced to promote apoptosis. 28,29 Apoptosis can be initiated through the upregulation of the downstream target of ATF3, CHOP/GADD153, which upregulates pro-apoptotic proteins. 30 Overexpression of ATF3 reduces the invasive potential of ovarian cancer cells. 31 ATF3 contains a central leucine zipper domain (Zip) that is well characterized as a mediator of protein-protein interaction. 32 ATF3 binds to p53 via this domain and blocks p53 ubiquitination catalyzed by MDM2, leading to upregulation of the p53 tumor suppressor activity independent of the ATF3 transcriptional activity. 33 Binding of ATF3 to p53, which is enhanced by DNA damage, also stabilizes the latter protein, resulting in an increase in the transcriptional activity of the tumor suppressor. 33 Moreover, ATF3 can be induced by a range of antitumorigenic compounds, including curcumin, and nonsteroidal anti-inflammatory drugs. 34,35 All these findings strongly suggest that ATF3 may be a novel therapeutic target.
Although ATF3 induction occurs frequently in the early stage of the DNA damage response, the role of ATF3 in cancer development remains undecided. In addition to suppression of E6-mediated p53 degradation, many studies previously demonstrated that ATF3 stabilizes and activates p53. 33 Therefore, as ATF3 binds to TP53, its downstream CHOP and PUMA expressions are activated and then pro-apoptotic protein expression including Bax, caspase-3-8, and PARP-1 was upregulated. 36 Therefore, we analyzed the expression alternation of ATF and TP53, also confirmed the expression downstream proteins, including CHOP and PUMA, related with apoptotic signaling pathway. First, we identified that C. militaris induced apoptosis in carboplatin-resistant SKOV-3 by regulating apoptosis-associated proteins. In order to confirm the establishment of resistance to carboplatin, we performed immunoblotting to identify the alternation of apoptosis-related protein expression by carboplatin in carboplatin-resistant and original parental SKOV-3. Bax, Bcl-2, and pro-apoptotic members were analyzed as target proteins. First, we confirmed that the carboplatin-resistant SKOV-3 cells established significant resistance to carboplatin compared with the original parental SKOV-3 cells (Figure 6(a)). Bax and cleaved caspase-3 and -8 were upregulated by carboplatin in SKOV-3, whereas there was no change in carboplatin-resistant SKOV-3. Next, to study the mechanism by which C. militaris inhibits cell proliferation and induces cell apoptosis, the carboplatin-resistant SKOV-3 cells treated with different doses of C. militaris (0, 100, and 500 µg/mL) and cotreated with C. militaris and 50 µg/mL of carboplatin were used for protein expression analysis. The analysis demonstrated that the protein expression levels of the cleaved caspase-3 and -8 increased significantly after treatment with C. militaris (Figure 6(b)). In addition, the Bcl-2, antiapoptotic protein expression levels were found to be remarkably decreased. Interestingly, we found that cotreatment of C. militaris and carboplatin but not carboplatin alone decreased the expression of Bax and pro-apoptotic proteins in carboplatin-resistant SKOV-3 (Figure 6(b)). Taken together, these results imply that C. militaris induces cell apoptosis through Bak, Bcl-2, and caspase-dependent pathways (Figure 6(a)).

C ordyceps militaris extract induces alternation of apoptotic protein expression in carboplatin-resistant SKOV-3 cells but not changed by carboplatin. Carboplatin-resistant SKOV-3 and SKOV-3 cell lines were exposed to (a) 50 and 100 µg/mL carboplatin and (b) 100 and 500 µg/mL Cordyceps militaris alone or cotreated with Cordyceps militaris and carboplatin for 48 hours whole-cell protein lysates were harvested and prepared for immunoblotting analysis using B-cell lymphoma 2, Bax, cleaved caspase-3, and cleaved caspase-8. Data were normalized to controls and represent the mean ± standard deviation for 3 independent experiments (* means P < 0.05, ** means P < 0.01, *** means P < 0.001, and ns means nonsignificance).
Further, C. militaris may play a secondary role in carboplatin-based chemotherapy by increasing the activities of the apoptosis-related signaling pathways. As mentioned earlier, C. militaris was designed to target and trigger the activation of ATF3 and TP53. Thus, we decided to study the ATF3 and TP53 levels using immunoblotting in the cotreatment of C. militaris with carboplatin or C. militaris treatment alone. In addition, we also confirmed the expression of ATF4, which is the upstream of ATF3 and TP53. Cordyceps militaris alone or in combination with 50 µg/mL of carboplatin triggered the upregulation of the ATF3 and TP53 levels in carboplatin-resistant SKOV-3. However, carboplatin treatment alone had no effect on the ATF3 and TP53 levels in carboplatin-resistant SKOV-3 (Figure 7(a)). In addition, we investigated the downstream proteins of ATF3/TP53 signaling, including CHOP, PUMA, and cleaved PARP-1 in C. militaris alone or in combination with 50 µg/mL of carboplatin. Immunoblotting also demonstrated that C. militaris treatment alone or cotreatment with 50 µg/mL of carboplatin markedly upregulated the CHOP and PUMA expression, which is important in regulating cell apoptosis (Figure 7(b)). Again, it was found that activation of ATF3/TP53 signaling was more increased by cotreatment of C. militaris and carboplatin than C. militaris alone, and that the downstream proteins of ATF3 and TP53 including CHOP, PUMA, and cleaved PARP-1 were also more upregulated. These findings indicate that C. militaris can reverse carboplatin resistance and increase drug sensitivity in resistant SKOV-3 by activating the ATF3/TP53 signaling pathway. In summary, our results demonstrate that C. militaris triggers apoptosis by activating the ATF3/TP53/CHOP/PUMA signaling pathway. Taken together, our findings describe the molecular mechanisms of apoptosis induced by C. militaris; the discovery of this synergistic combination, which is effective even when apoptosis is blocked, has important implications for the development of new treatment strategies.

C ordyceps militaris extract upregulates sensitivity of carboplatin via activation of ATF3/TP53 signaling pathway in carboplatin-resistant SKOV-3. Effect of Cordyceps militaris on (a) upstream protein ATF4 and ATF3/TP53 signaling and (b) downstream proteins including CHOP, PUMA, and cleaved PARP-1 in carboplatin-resistant SKOV-3. Carboplatin-resistant SKOV-3 cells were treated with (a) 50 and 100 µg/mL carboplatin or 100 and 500 µg/mL C ordyceps militaris alone or cotreated with Cordyceps militaris and carboplatin for 48 hours whole-cell protein lysates were harvested and prepared for immunoblotting analysis using ATF4, ATF3, TP53, CHOP, PUMA, and cleaved PARP-1. Data were normalized to controls and represent the mean ± standard deviation for 3 independent experiments (* means P < 0.05, ** means P < 0.01, *** means P < 0.001, and ns means nonsignificance).
Finally, to confirm whether C. militaris or in combination with carboplatin strongly inhibits tumor growth in in vivo carboplatin-resistant SKOV-3 subcutaneous tumor models, we progressed tumor xenograft experiment of carboplatin-resistant SKOV-3. The carboplatin/C. militaris treatment combination was assessed in vivo in subcutaneous carboplatin-resistant SKOV-3 tumor xenografts in nude mice. Seven days after the tumors were established, the mice were treated intraperitoneally with 40 mg/kg of carboplatin once 2 days for 10 days and with/or 500 mg/kg of C. militaris. Carboplatin treatment did not affect SKOV-3 tumor growth, confirming the carboplatin-resistant status of these tumors. In contrast to the in vitro results, C. militaris alone was able to inhibit tumor growth, with a slowdown in tumor growth (Figure 8(a)). The combination of carboplatin and C. militaris triggered a strong tumor growth inhibition with slowing of tumor growth and an apparent decrease in tumor weight (Figure 8(a) and (b)). For reference, the change of body weight confirmed that C. militaris has no cytotoxicity (Figure 8(c)). Taken together, these results indicate that C. militaris plays an antitumoric role in chemoresistant ovarian adenocarcinoma.

In vivo antitumor efficacy with Cordyceps militaris single and combination with carboplatin in xenograft. (a) Changes in tumor volume as a function of time in carboplatin-resistant SKOV-3 ovarian adenocarcinoma xenograft-bearing male nu/nu mice after 1 dose of paclitaxel and tamoxifen administered per 2 days for 10 days. The xenograft tumor in different treatment groups (b) The final tumor weight at necropsy at 10 days postadministration (c) Effect of Cordyceps militaris and carboplatin on the body weight of mice during 10 days of treatment. Data were normalized to controls and represent the mean ± standard deviation for 3 independent experiments (* means P < 0.05, ** means P < 0.01, *** means P < 0.001, and ns means nonsignificance).
In conclusion, the systematic investigation in this study has revealed the detailed molecular mechanism of the C. militaris-induced apoptosis in carboplatin-resistant ovarian cancer cells. In addition, these results indicate that C. militaris plays an antitumor role and an assistance therapeutic anticancer medicine for carboplatin-based chemotherapy in carboplatin-resistant ovarian adenocarcinoma.
Experimental
Preparation of Cordyceps militaris
Cordyceps militaris was purchased from Wonkwang University Jeonju Korean Medicine Hospital (Jeollabuk-do, Republic of Korea). Fresh bodies or mycelia of C. militaris were extracted using 50% ethanol at 80°C for 3 hours (5 times). The CME was filtered using 1 µm pore-size filters, concentrated, and dried. The total extract (200 g, yield [w/w], 11%) was diluted with water. The quality evaluation of CME, as determined using a high-resolution mass spectrometer, a traditional medicine library, and the nucleotide and cordycepin quadrupole time-of-flight (Q-TOF) mass spectrometry analysis was shown in our previous study. 37
Reagents and Chemicals
Fetal bovine serum (FBS) was purchased from Hyclone (HyClone, Logan, UT, United States), and antibiotic-antimycotic (100×) was obtained from Gibco (Waltham, MA, United States). Phosphate-buffered saline (PBS) and McCoy’s 5A medium were purchased from WelGENE Inc. (Daegu, Republic of Korea). Carboplatin and Annexin V-fluorescein isothiocyanate (FITC) Apoptosis Detection Kit were obtained from Sigma-Aldrich (St. Louis, MO, United States). Whole cell lysis buffer was procured from iNtRON Biotechnology Inc. (Seoul, Republic of Korea). Antibodies against ATF4, Bcl-1, Bcl-2, LAMP3, and PUMA used for immunocytochemistry were purchased from Abcam (Cambridge, United Kingdom), while Bax, caspase-3, and CHOP were supplied by Cell Signaling (Beverly, MA, United States). Those against ATF3, cleaved PARP-1, caspase-8, TP53, and β-actin were obtained from Santa Cruz (Dallas, TX, United States).
Cell Lines
SKOV-3 (ovarian adenocarcinoma) cell lines were also purchased from the american type culture collection (ATCC). 38 SKOV-3 and carboplatin-resistant SKOV-3 were grown in McCoy’s 5A medium. For all cell lines, the media were supplemented with 10% heat-inactivated FBS, 1× antibiotic-antimycotic (100 U/mL of penicillin, 100 µg/mL of streptomycin, and 0.25 µg/mL of amphotericin B) (Gibco). The carboplatin-resistant SKOV-3 cells were incubated with 50 µg/mL of carboplatin in a fresh growth medium. All cells were grown as monolayer cultures and maintained in a humidified atmosphere of 5% CO2 in air at 37°C. Establishment of carboplatin resistance in human ovarian cancer cells was made. The carboplatin-resistant SKOV-3 cell lines were derived from the original parental cell line via continuous exposure to carboplatin (Sigma-Aldrich, United Kingdom) following initial dose-response studies of carboplatin (0.1-100 µM) over 72 hours from which IC50 values were obtained. This development period was carried out for approximately 6 months; thereafter, the IC50 concentrations were re-assessed in the resistant SKOV-3 cell lines. The cells were then maintained continuously in the presence of cisplatin at these new IC50 concentrations for another 6 months. 39
Cell Viability/Cytotoxicity Assay
The carboplatin-resistant SKOV-3 and original parental cells were allowed to adhere and grow for 24 hours prior to the exposure to CME and/or carboplatin. In brief, 2 cell lines were seeded in 96-well plates at a density of 5 × 103 cells/well. After 24 hours of incubation, the cells were treated with various concentrations of CME (10, 50, 100, 200, 500, and 1000 µg/mL) and carboplatin (5, 10, 20, 50, 100, and 200 µg/mL) for 24, 48, and 72 hours. In addition, 50 µg/mL of carboplatin (maximum concentration for resistance) was treated at various CME concentrations to determine whether synergistic effects on anticancer activity occurred in cotreatment of CME and carboplatin. The optimal dose (without cytotoxic effects) was determined using the CCK-8 assay (Dojindo). Briefly, 10 µL of CCK-8 solution was added to each well at the end of the treatment, and the plate was incubated for 2 hours at 37°C. The absorbance was measured at a 450 nm wavelength using a Sunrise microplate absorbance reader (Tecan, Männedorf, Switzerland) relative to that of untreated controls in triplicate experiments.
Apoptosis Analysis via PI/Annexin V Staining
To determine the apoptotic effects of C. militaris on human ovarian carcinoma cells, we used the Annexin V-FITC Apoptosis Detection Kit (Sigma-Aldrich, United States). Briefly, the cells were treated with C. militaris for 24 hours, dissociated using trypsin, and washed twice with PBS. The cell suspension in PBS was centrifuged at 1500 rpm for 5 minutes, and the supernatant was carefully removed by pipetting. The cell pellet was resuspended in 500 µL of Annexin V binding buffer and treated with 0.1 µg/mL of Annexin V-FITC conjugate and 2 µg/mL of PI for 10 minutes at 25°C room temperature in the dark. The fluorescence of the samples was immediately detected using the Guava system (Millipore) at an excitation wavelength of 488 nm with a 530/30 nm band-pass filter to detect Annexin V and 670 nm high-pass filter to detect PI.
Transmission Electron Microscopy
The CME-treated carboplatin-resistant SKOV-3 cells were sequentially fixed with 2.5% glutaraldehyde and 1% osmium tetroxide on ice for 2 hours and washed with PBS. The tissues were dehydrated in ethanol and propylene oxide series, embedded in Epon 812 mixture, and polymerized in an oven at 70°C for 24 hours. The sections acquired from polymerized blocks were collected on grids, counterstained with uranyl acetate and lead citrate, and examined using the Bio-HVEM system (JEM-1400Plus at 120 kV and JEM-1000BEF at 1000 kV, JEOL Ltd., Japan).
Microarray Analysis
Transcriptional profiling of the CME-treated carboplatin-resistant SKOV-3 was conducted using a human twin 44K cDNA chip. Total RNA was extracted from vehicle- or C. militaris (500 µg/mL)-treated carboplatin-resistant SKOV-3 cells, and 50 mg of RNA was subjected to cDNA synthesis in the presence of aminoallyl-dUTP by reverse transcription. The cDNA was coupled with Cy3 (vehicle) or Cy5 dye (CME-treated). The genes were thought to be differentially expressed when the global M and log2 (R/G) values exceeded |1.0| (2-fold) at P values of < 0.05. The Student’s t-test was applied to assess the statistical significance among the differentially expressed genes after C. militaris treatment. To analyze the biological significance of these changes, the array data were categorized into specific gene groups.
Gene Ontology-Based Network Analysis
To study the biological functions of the regulated genes through an interaction network, we used the STRING database (IPA, http://string-db.org/) and examined the biological functions of the differentially regulated genes and proteins according to the ontology-related interaction network, including apoptosis signaling. Network generation was optimized on the basis of the obtained expression profiles with an aim of producing highly connected networks.
Immunoblotting
Total cell lysates were prepared after the homogenization of the cells in 2 mL of Tris-HCl (20 mM) containing a protease inhibitor cocktail (Roche). The cell homogenate was placed on ice for 30 minutes before centrifugation (10 minutes, 12 000 rpm, 4°C). The protein content in the supernatant was quantified using the bicinchoninic acid method. Denatured proteins (30 µg) were resolved using 12% sodium dodecyl sulfate polyacrylamide gel electrophoresis, and the separated bands were transferred onto a 0.2 µm nitrocellulose membrane in a transfer buffer for 2 hours. The membrane was blocked for 1 hour with 5% (w/v) skimmed milk in Tris-buffered saline with Tween-20 (TTBS), followed by incubation with the appropriately diluted primary antibodies at room temperature for 2 hours or 4°C overnight. After washing the membrane thrice with TTBS, it was probed with horseradish peroxidase-conjugated goat anti-mouse or rabbit anti-goat IgG (1:2000 dilution) in TTBS containing 5% (w/v) skimmed milk at room temperature for 1 hour. The membrane was rinsed thrice with 0.1% (v/v) TTBS. An enhanced chemiluminescence system (Thermo Scientific) was used to visualize the bands on the ChemiDoc MP system (Bio-Rad, Hercules, CA, United States). Densitometric measurement of the bands was performed using the ImageJ software. Protein levels were quantitatively analyzed after normalization with the β-actin level.
Animals
Four- to 5-week-old male BALB/c nude mice (Orient Bio, Inc., Gyeonggi, Republic of Korea) were included after approval from the Institutional Animal Care and Use Committee of the Korea Basic Science Institute (KBSI-AEC 1913). The mice were housed in a facility approved by the Association for Assessment and Accreditation of Laboratory Animal Care on a 12 hours light/12 hours dark cycle with food and water ad libitum. Mice experimentations began after 1 week of quarantine.
Tumor Xenograft Experiment
The 107 carboplatin-resistant SKOV-3 and original parental cells were implanted subcutaneously into 1 flank of 7 mice per subgroup. Xenograft tumors were established for 5 to 8 weeks, and their sizes were monitored. Thereafter, they were treated with saline or 500 mg/kg CME and/or 40 mg/kg carboplatin via intraperitoneal administration once every 2 days. The well-being and body weight of the mice were checked every 2 to 3 days. Tumor lengths and widths were measured twice a week, and the tumor volume was extrapolated using the following formula: (length ×width2)/2. The mice were sacrificed via cervical dislocation 12 days after the injection of C. militaris and/or carboplatin. The tumors were sampled and then conditioned for histological analysis (formol fixation).
Statistical Analyses
GraphPad Prism (GraphPad, San Diego, CA, United States) was used to perform the statistical analyses. Data were analyzed using one-way analysis of variance (ANOVA), followed by the Tukey-Kramer multiple comparisons test. The IC50 values were determined via nonlinear curve fitting using 5 data points and expressed as means ± standard deviations.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2017R1D1A1B03034936, 2019R1F1A1058077).
