Abstract
Mori Cortex Radicis (MCR) is a well-known Korean and Chinese folk medicine with anti-obesity, anti-inflammatory, anti-asthmatic, and hypoglycemic activities. This study was aimed to evaluate the total phenolic and flavonoid contents, as well as intracellular antioxidant and anti-inflammatory effects of water and 70% (v/v) ethanol extracts of MCR. The antioxidant activities of MCR extracts were determined with diphenyl-2-picrylhydrazyl and 2,2′-azinobis[3-ethylbenzothiazoline-6-sulfonic] scavenging activity assays. The suppressive activities of MCR extracts on the production of nitric oxide (NO*) and the expression of cytokines, c-Fos, activated p38-Mitogen-activated protein kinase (MAPK), and Nuclear factor Kappa B (NF-κB) and splenocytes proliferation in lipopolysaccharide-treated macrophages were determined. Furthermore, this study demonstrated the effects of MCR on reactive oxygen species production in murine macrophages. Mori Cortex Radicis restored deoxyribonucleic acid damages at higher concentrations of the extracts and significantly suppressed free radicals and NO* production. In this study, MCR significantly restored inflammatory responses and intracellular antioxidant activities in murine macrophages (RAW 264.7), which anticipated that MCR could be used as a natural anti-inflammatory agent.
Mori Cortex Radicis (MCR) indicates the root bark of Morus alba L. which has been long used for the treatment of diseases such as jaundice, hematemesis, and edema. 1 It is also a well-known oriental herbal medicine known as “Sang bai pi” in China and “Sambak pi” in Korea. 2 Morus alba is widespread in Europe, Asia, Africa, and America (United States, Colombia, Mexico, Panama, Argentina, and Brazil). The origin of most cultivated Morus varieties is traced back to areas around east Asia and the Himalaya foothills. About hundred species of Morus had been described but Morus taxonomy may characterize as complicated. 3 Living organisms generate reactive oxygen species (ROS) and free radicals during physiological processes. Though ROS have many key roles in normal physiological function including metabolism and respiration, a high amount of free radicals may lead to oxidative damage to biomolecules, impair the function of protein, trigger cell death, and cause tissue damage leading to chronic and lethal diseases. 4,5 The tumorigenic characteristics of ROS have prompted the evaluation of dietary antioxidants as potential preventive and therapeutic effects on animals and humans. 6 Mori Cortex Radicis is an enormous source of flavonoids, and other compounds such as stilbenes, benzofurans, terpenes, and alkaloids have also been previously reported as the phytochemical constituents of this plant. 7 Oxidation process generates free radicals; they are not stable cause of their unpaired electrons, and hence, they attack other stable biomolecules of organ. However, artificial antioxidants, such as Butylated hydroxytoluene (BHT) and Butylated Hydroxyanisole (BHA), have strong activities against free radicals but also have adverse effects on health. Therefore, plants are gaining a lot of attention as source of bioactive compounds which are natural antioxidant agents. Inflammatory pathways adversely affect organs and systems by producing excessive nitric oxide (NO*) and other inflammatory cytokines. Macrophages play pivotal roles in inflammation, and they are sometimes incurred in the pathogenesis of inflammatory diseases. 8 The potent preventive potentialities of prenylated flavonoids and phytochemicals from MCR against NO* or sodium nitroprusside-induced cell death in human neuroblastoma SH-SY5Y cell have been reported. 7 Different prenylated flavonoids and other phenolic compounds isolated from Morus species have anti-inflammatory activities illustrated by the inhibition of the production of cyclooxygenase-1, 2 COX-1), (COX-2), and NO. 9,10 NO is a critical reactive radical which has potent roles as oxidative biological signaling components in physiological processes, including blood pressure regulation, neurotransmission, prohibiting the platelet aggregation, smooth muscle relaxation, defense mechanisms, and immune regulation. NO* is also responsible for aggravating of inflammatory joint disease and it plays pivotal roles in cartilage catabolism through the inflammatory process. NO* is produced through NO* synthases (NOSs) by converting l-arginine to l-citrulline, and iNOS is activated by inflammatory mediators such as bacteria and pyrogen/lipopolysaccharide (LPS). 11 On the other hand, LPS is a potent stimulator for pro-inflammatory cytokines. 12,13 The components of MCR including sanggenone C, sanggenone D, kuwanon C, kuwanon G, morin, and morusin showed strong to downregulating effects of pancreatic lipase. 14 Pancreatic lipase is a potent enzyme that hydrolyzes triacylglycerides in the gastrointestinal tract. Mori Cortex Radicis extract ameliorated corticosterone response and suppressed c-Fos activity in the dentate gyrus. In addition, MCR treatment modulated the downregulation of serine/threonine protein phosphatase 5 and pGR(S232) by which MCR extract promotes antidepressant-like effects. 15 To our knowledge, this is the first time study to evaluate the intracellular antioxidant and anti-inflammatory activities of 2 different extracts (water and 70% ethanol) of MCR. Furthermore, antiproliferative activity of primary lymphocytes was studied against LPS. We hypothesized the therapeutic and preventive potential effects of MCR against inflammatory diseases, which are mediated by the suppression of free radicals and inhibition of inflammatory responses.
The yields were found as 19.37% and 13.14% (w/w) for water and 70% ethanol MCR extracts, respectively. Phenols are biofunctional and secondary metabolites and mostly found in plants, and they have anti-inflammatory activities and have the ability to scavenge free radicals. Antioxidant activity is one of the prominent effects of phenolics in mammalian body. 16,17 Total polyphenols, flavonoids, and yield of water and 70% ethanol (v/v) extracts of MCR are presented in Table 1. Water extract contained higher level of bioactive content than ethanol extract which can be attributed to the higher yield found in the water extract. Total phenolic content and total flavonoid content (TFC) of water extract of MCR were 1060 and 256 mg per 100 g of dry mass, respectively, while those of 70% ethanol extract were 777 and 25 mg per 100 g of dry mass, respectively. Total phenolic content was calculated from the gallic acid (GA) standard curve and TFC from the catechin standard curve by using regression equation where r 2 values were 0.9954 and 0.9997, respectively.
Total Phenolic, Flavonoid Content, and Yield of Mori Cortex Radicis.

CE, catechin equivalents; GAE, gallic acid equivalents.
Values are mean ± standard deviation (n = 3).
Diphenyl-2-picrylhydrazyl (DPPH) assay is effective in determining the antioxidant activities of sample. Antioxidant activity was assessed in terms of electron (or hydrogen) donating abilities (EDA) of DPPH. 18 In this study, both extracts have high DPPH scavenging activity. On the other hand, the standard (AA) showed a complete DPPH scavenging activity at a concentration of 0.06 mg/mL. Diphenyl-2-picrylhydrazyl and 2,2′-azinobis[3-ethylbenzothiazoline-6-sulfonic] (ABTS) radical scavenging activities and cytotoxicity effects by 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay of the extracts were illustrated in Figure 1, and the results indicate that MCR extracts scavenged the free radicals in a dose-dependent manner with regard to DPPH and ABTS, and no cytotoxic effect up to 1 mg/mL has been observed. The high scavenging activity of the extracts may be attributed to the high level of phenolic compounds that are naturally occurring in plants. 19,20 The nitrite radical scavenging and ferric reducing activity of MCR extract was observed at higher concentration that is provided as supplemental Figure. The conversion of ferric to ferrous is used to measure the EDA of antioxidants and this ability is one of the characteristics of phenolics. 16,17 Both extracts demonstrated almost similar ferric reducing activity in a dose-dependent manner. At 2 mg/mL, the water extract showed less ferric reducing activity than ethanol extract. Fenton reaction, a key biological source of hydroxyl radicals, is generated near deoxyribonucleic acid (DNA) in the presence of transitional metal ions such as iron and copper. In the negative control (without sample), DNA strand was damaged by H2O2 and FeSO4, and that damage was restored by 2 mg/mL of both MCR water and 70% ethanol extracts as shown in Figure 1. Hydroxyl radicals have the ability to join nucleotides of DNA and break the strand that is responsible for mutagenesis, carcinogenesis, and cytotoxicity 19

(a) Diphenyl-2-picrylhydrazyl and (b) 2,2′-azinobis[3-ethylbenzothiazoline-6-sulfonic] radical scavenging activities of Mori Cortex Radicis extracts. (c) Mori Cortex Radicis extract deoxyribonucleic acid preventive activities against oxidative damages stained by EtBr subjected to 0.8% agarose gel. Both extracts of Mori Cortex Radicis restored the plasmid deoxyribonucleic acid damages. (d) 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay of Mori Cortex Radicis extracts and both extracts showed no cytotoxic effect up to 1 mg/mL. Data were presented as mean ± standard deviation, where n = 3.
Cell viability of MCR extracts was evaluated against LPS on RAW 264.7 cells using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) method. Lipopolysaccharide contains lipid and polysaccharide, and it is a key stimulator of pro-inflammatory cytokines, which suppressed cell proliferation, endotoxin, and pyrogen, thereby causing cell damage. Both extracts of MCR significantly suppressed LPS-induced cell death in RAW 264.7 cells. Cell viability increased notably with MCR treatment as shown in Figure 2(a). NO* has a crucial role in our physiological system. The excessive production of NO* possibly occurs as a mechanism to prevent the host from infection. Moreover, NO* affects lymphocyte on proliferation and causing damage to other normal host cells is the same protective/destructive duality inherent in other major biomolecules of the immune system. 21 NO production was determined from NaNO2 calibration curve where r 2 value was 0.9984 (Figure 2(b)). Mori Cortex Radicis extracts inhibited NO generation in murine macrophage in a dose-dependent manner, with ethanol extract showing a significantly higher inhibitory activity than water extract. The level of NO in the cells treated with only LPS was 78 µM which was reduced to 48 and 60 µM by ethanol and water extracts of MCR, respectively, at their final concentrations.
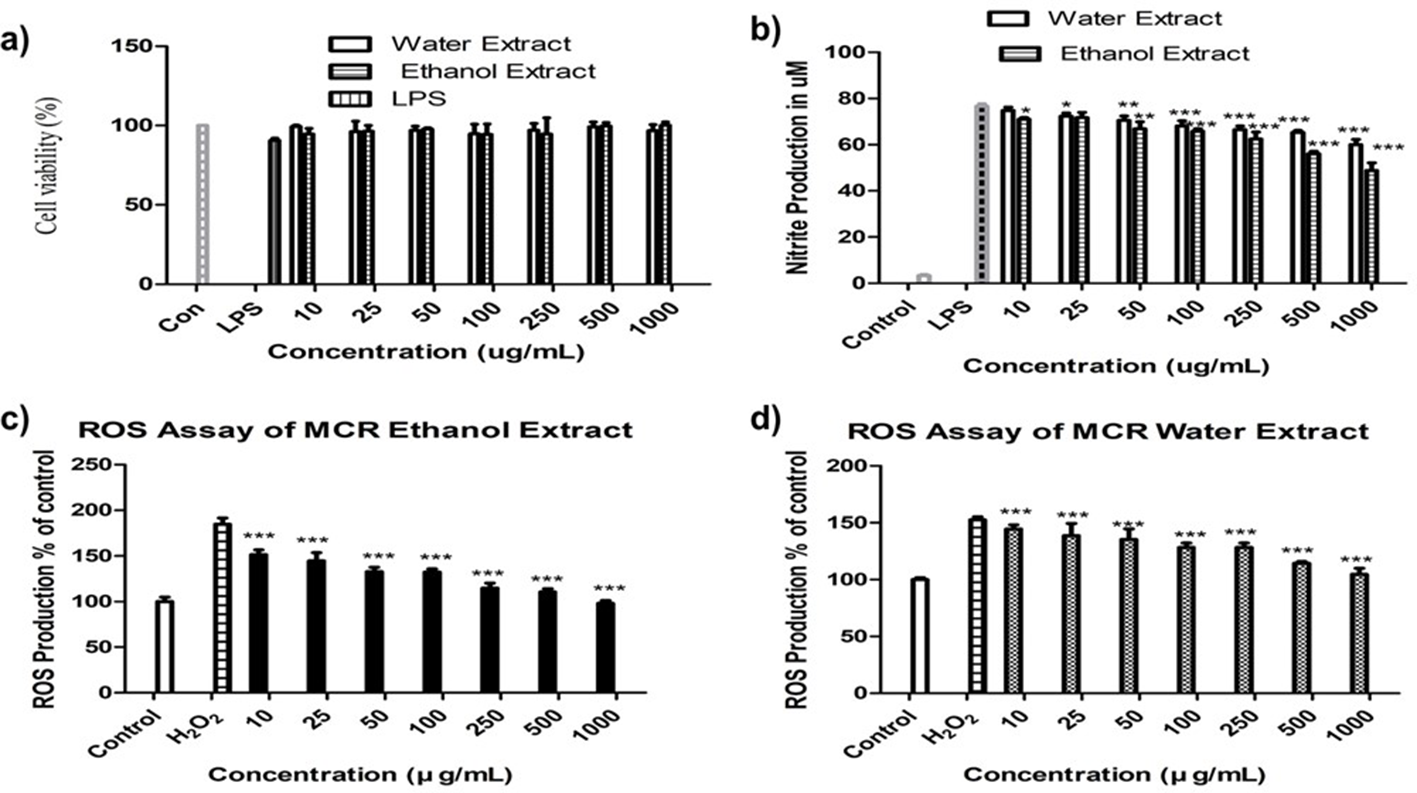
(a) The effect of Mori Cortex Radicis extracts on cell viability of inflammation mediator lipopolysaccharide-stimulated RAW 264.7 cells (b) and NO production (c). (d) Cellular antioxidant activities of Mori Cortex Radicis ethanol and water extracts, respectively. Data were presented as mean ± standard deviation, where n = 3, and *P < 0.05, **P < 0.01, and ***P < 0.001 compared to the lipopolysaccharide- or H2O2-treated group.
H2O2 is the key indicator of ROS accumulation in RAW 264.7 cell line. Intracellular bioactivity is more biologically relevant and effective; therefore, a cell-based experiment is an inevitable part of this study to evaluate antioxidant activity of MCR. A vast area of essential biomolecules such as proteins, enzymes, nucleic acid, lipids, and carbohydrates is damaged by the excessive production of ROS and the activation of MAPK/NF-kB. 22 Several studies have shown the protective effect of antioxidants against the action of ROS and other free radicals. In Figure 2(c) and (d), both extracts of MCR significantly suppressed the generation of ROS in a concentration-dependent manner. Ethanol and water extracts of MCR significantly suppressed H2O2-induced ROS production by 111%, 98% and 114%, 105% at the concentrations of 500 and 1000 µg/mL, respectively. Murine macrophage (RAW 264.7) cell line is widely used to assess NO* and ROS assays. 23,24 Macrophages are belonging to innate immune system, and hence, they play important roles in the pathogenesis of autoimmune diseases.
Western blot analysis showed the significant and potential downstream of inflammatory response and cytokine production induced by LPS in murine macrophages (Figure 3). Activated MAPK p-38 and NF-κB p65 play pivotal roles in inflammatory response, and their roles in body mechanisms are crucial. Activated MAPK p-38 and NF-κB may facilitate the production of excessive cytokines and chemokines which may perpetuate inflammatory diseases and disorders. The interaction among Tumor necrosis factor alpha (TNF-α), Interleukin (IL-6), and Phosphorylation of NF-kappaB (pNF-κB) initiating a virtuous circle in the cytokine network, IL-6 and TNF-α, can activate NF-κB, which again amplifies the inflammatory mediators and factors. Furthermore, pNF-κB may further activate or induce COX-2. 25 Mori Cortex Radicis notably suppressed the expression of TNF-α, c-Fos, p-p38, and pNF-κB p65. pNF-κB, phospo p-38 MAPk, and activated cells may result in tumor and cancers. 26 Moreover, MCR restored iNOS, COX-2, IL-10, and IL-1β expression in LPS-treated RAW 264.7 cells in a concentration-dependent manner, which possibly explains the therapeutic effect of MCR in several inflammatory diseases.
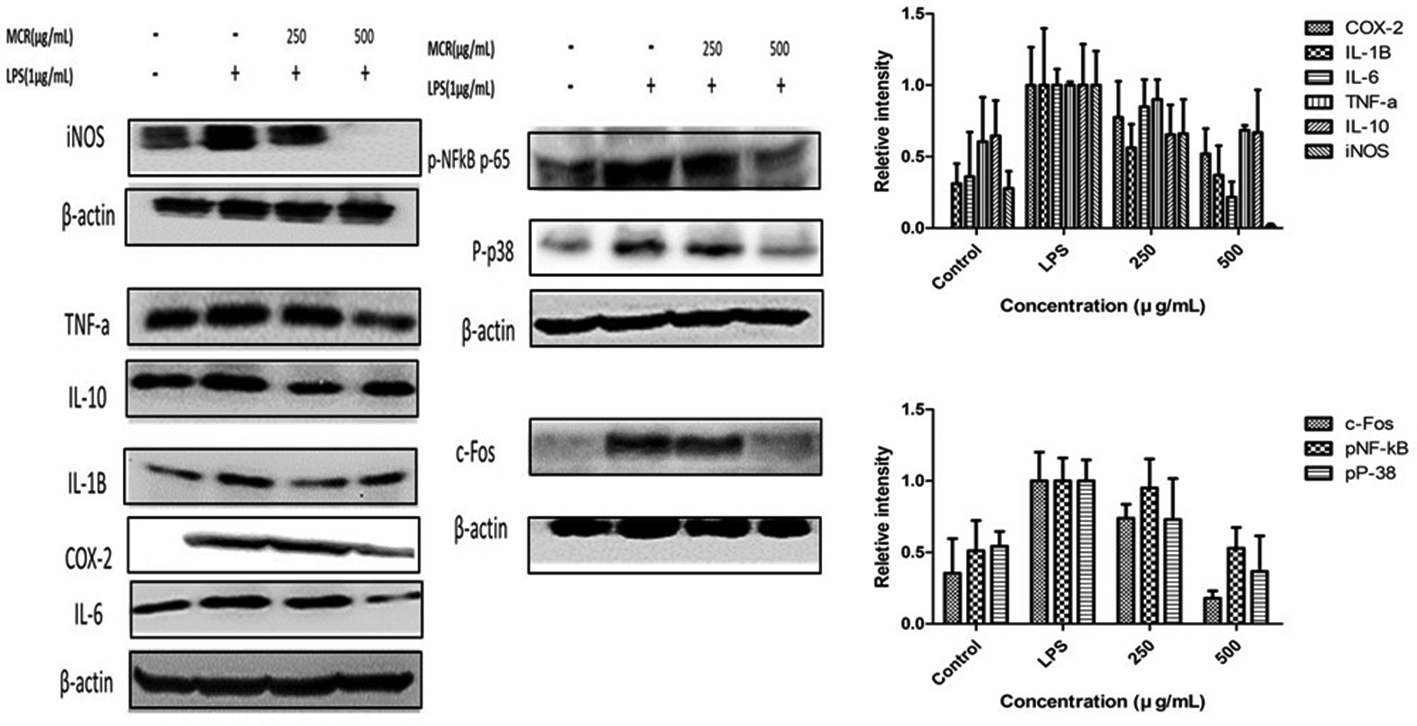
The effect of Mori Cortex Radicis water extract on inflammatory cytokines expression and activation of MAPK/NFkB and c-Fos against lipopolysaccharide-stimulated RAW 264.7 cells. The histogram of relative intensity was analyzed by lmageJ and normalized by B-actin. Data were presented as mean ± standard deviation, where n = 3.
Splenocytes consist of neutrophils, monocytes, macrophages, and T-lymphocytes which are vital parts of our adaptive and innate immune cells that protect the host of infection and also are important inflammatory mediators and phagocytic role in physiological process. Institute of Cancer Research (ICR-CrljOri:CD1) mouse splenocytes were isolated to investigated whether the MCR is effective against LPS-induced proliferation. Mori Cortex Radicis extract significantly reversed splenocytes proliferation. The proliferation of the splenocytes may cause phagocyte, other cells, or tissue damages that lead to malfunction in the physiological process. Whereas, the control group is considered as 100% that accelerated up to 170% by LPS stimulation which is attenuated by the MCR significantly shown in Figure 4.
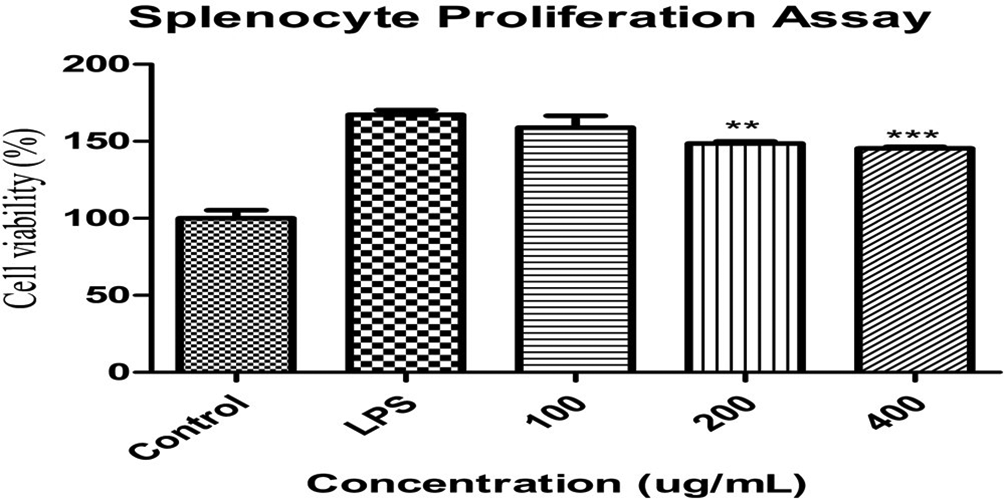
The effect of Mori Cortex Radicis on lipopolysaccharide-induced proliferation in splenocytes isolated from ICR mouse. Splenocytes were treated with 100 ng/mL lipopolysaccharide in the presence or absence of Mori Cortex Radicis for 24 hours. Cell proliferation (expressed as percentage of untreated control) was measured by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide assay. Values are mean ± standard deviation (n = 3). *P < 0.05, **P < 0.01, and ***P < 0.001 compared to the lipopolysaccharide-treated group and significant values are given by analysis of variance (unpaired t-test).
This study evaluated the biofunctional characteristics of MCR and its extracts showed significant attenuation of intracellular oxidants production and anti-inflammatory and antiproliferative activities of lymphocytes which may explain its preventive activity against aging and chronic disorders. Our results demonstrated the antioxidant properties of MCR extracts in macrophages, and this suggests that MCR is a potential biomedicinal component that may be used as a therapeutic or preventive agent of oxidative stress-mediated diseases. In addition, MCR extracts restored the expression of inflammatory mediators and cytokines. Previous studies have also reported the effects of MCR extracts against hyperglycemia and diseases caused by oxidative stress. 15,27,28 Further studies to determine the bioavailability and toxic effects (if have any) of MCR are necessary for proper elucidation of MCR bioactivities.
Experimental
Chemical and Reagents
Sodium nitrite, DPPH, ABTS, trichloroacetic acid (TCA), potassium persulfate, ammonium thiocyanate, Ethylene diamine tetra acetic acid (EDTA), anhydrous sodium phosphate (dibasic), anhydrous sodium phosphate (monobasic), ferrous sulfate, and pyrogallol were purchased from Sigma, Sigma Chemical (St. Louis, MO, United States). Sodium hydroxide and ferric chloride were obtained from Wako Pure Chemical Industries Ltd. (Osaka, Japan). Dulbecco’s Modified Eagle’s Medium (DMEM), fetal bovine serum (FBS), Dulbecco’s phosphate-buffered saline (DPBS), and trypsin EDTA were purchased from WEIGENE Inc. (Seoul, Korea). All other chemicals and reagents were of analytical grade and used without further purification.
Preparation of Water and Hydroethanolic Extracts of MCR
Mori Cortex Radicis had been dried and pulverized followed by the preparation of its extracts with distilled water (D.W.) and 70% ethanol (v/v). Mori Cortex Radicis (100 g) was soaked in D.W. (1000 mL) at 50°C for 24 hours and in 70% (v/v) ethanol in water (1000 mL) at room temperature (RT) for 24 hours, 29 separately. This process had been repeated 3 times and the samples were concentrated by vacuum rotary evaporator and lyophilizer. The yield (w/w) was calculated as [Yield (%) = (total extracted sample weight/total dry weight) × 100] and the extracts were stored at −20°C before use. The yield was expressed as the percentage (%) of dry mass.
Total Phenolic Content
Total phenolic contents were determined by Folin-ciocalteu (FC) method as previously mentioned 30 with some changes. The extracts (1 mg/mL) and various concentrations of GA (0-100 µg/mL) were prepared. 17,31 Furthermore, 40 µL of each sample was added to 20 µL of 1 N FC reagent and 60 µL Na2CO3 (20 %, w/v) and were incubated in the dark at normal temperature for 30 minutes. The absorbance was taken at 700 nm with a Versamax microplate reader (Molecular Device, CA, United States). The results were denoted as mg of GAE per 100 g of dry mass from the calibration curve of GA.
Total Flavonoids Content
Total flavonoids content was performed as previously described. 30,32 The concentrations of catechin (standard) solutions were 0.00 to 0.100 mg/mL and sample concentration was 1 mg/mL. Briefly, 25 µL of samples or standard were incubated with 125 µL of D.W. and 8 µL of 5% NaNO2 in a 96-well plate. After 5 minutes, 15 µL of AlCl3(10 %) was mixed with the mixture and kept at RT for 6 minutes. Furthermore, 50 µL of NaOH (1 M) and 27 µL of water were added into the mixture and the absorbance was taken at 510 nm. Total flavonoids content was determined from the catechin standard curve. Results were demonstrated as mg of catechin equivalent per 100 g of dry mass.
Diphenyl-2-picrylhydrazyl Free Radical Scavenging Activity
Diphenyl-2-picrylhydrazyl free radical-scavenging activities of water and 70% (v/v) ethanol extracts were determined by a previously described method. 4,5,33 Briefly, various concentrations (0.06-1.00 mg/mL) of sample and ascorbic acid (AA) as standard were prepared in water and ethanol. Afterward, 80 µL of every sample or standard solutions were added with 80 µL of DPPH (0.2 mM in methanol) solution. The mixture was placed on a shaker at 70 rpm in the dark for 30 minutes and the absorbance was measured at 517 nm. The controls were composed of the solvent of extraction. Diphenyl-2-picrylhydrazyl radical-scavenging activity was calculated using the following equation:

where C is the absorbance of DPPH + solvent, D is the absorbance of solvent + methanol, A is the absorbance of sample or standard + DPPH, and B is the absorbance of sample or standard + methanol
2,2′-Azinobis[3-Ethylbenzothiazoline-6-Sulfonic]+ Radical Scavenging Activity
The ABTS+ radical scavenging activity of MCR extracts was measured according to the previously described methods 31,34,35 with slight modifications. First, ABTS powder (purchased from Sigma Aldrich) was dissolved in D.W. and made up to 7 mM concentration, and ABTS+ was produced by the reaction of ABTS and potassium persulfate solution (found 2.45 mM final concentration) by incubating the mixture at RT for 14 hours before use. Next, prepared ABTS radical solution was diluted with 0.01 M phosphate-buffered saline (PBS, pH 7.4) to adjust the absorbance to 0.70 ± 0.02 at 734 nm. Different concentrations of sample (0.6 mL) and single concentration of AA (alone or in combination) were mixed with 0.8 mL of ABTS+ solution in cuvettes. The absorbance was taken at 734 nm after 5 minutes of reaction at RT. The controls were composed of the solvent. The scavenging activity of ABTS free radical was determined by the following equation:
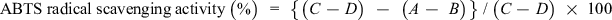
where C is the absorbance of ABTS+ solvent, D is the absorbance of solvent + potassium persulfate, A is the absorbance of sample/standard + ABTS+, and B is the absorbance of sample/standard + potassium persulfate.
Nitrite Radical Scavenging Activity
Nitrite radical scavenging activity was measured as previously mentioned in Hasnat et al, Debnat et al, Lee et al, and Choi et al. 4,17,33,36 Ethanol and water extracts or standard reagent (0.12 mL) at 0.5 to 10 mg/mL were added to 0.12 mL of 1 mM NaNO2. Next, 0.96 mL of 0.2 M citrate buffer was mixed to the mixture in e-tube and incubated at 37°C in the water bath for 1 hour. Then, 1 mL of the mixture was taken out and mixed with 2 mL mixture (2%, v/v) of acetic acid, 0.2 mL of Griess reagent A, and same amount of Griess reagent B. The combined solution was mixed by a vortex mixer and incubated at RT for 15 minutes and then the absorbance was taken at 520 nm. Ascorbic acid was used as positive control. The controls contained water and 70% ethanol (v/v). The scavenging activity of each sample and standard solution was calculated as follows:
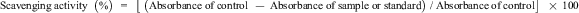
Reducing Power Activity
The ferric reducing power of the samples was measured according to a previously described method 37 with some changes. Furthermore, 500 µL of every sample or standard solution (where AA is considered as a standard solution) at various concentrations (0.5-10.00 mg/mL) was mixed with 500 µL of 0.2 M PBS and 500 µL of 10 mg/mL potassium ferricyanide solution. Then, the mixture was incubated at 50°C in water bath for 20 minutes, and 1 mL was taken out from the mixture into new tube followed by the addition of 0.5 mL TCA (100 mg/mL) and centrifugation at 3000 × g for 10 minutes at 4°C. Next, 0.5 mL of supernatant was added to 0.5 mL of D.W. and 100 µL of FeCl3 (0.1% [w/v]) was added to the mixture. The absorbance was measured at 700 nm.
Deoxyribonucleic Acid Protective Activity
Plasmid DNA (pBR322) and 6× loading had been purchased from Thermo Scientific. The ability of the extracts to prevent H2O2-induced DNA damage was investigated as mentioned in an earlier study. 19 pBR322 (1 µL, 0.5 µg/µL) was mixed with 2 µL of 0.08 mM FeSO4, 3 µL of 30% H2O2 (v/v), 5 µL of D.W., and 2 µL of both extracts at various concentrations (0.5, 1, and 2 mg/mL). Next, the mixture was incubated at 37°C for 1 hour and 2 µL of 6× loading dye was added to the mixture. The mixture was subjected to 0.8% agarose gel electrophoresis (Mupid-2plus; Advance Co., Ltd.) at 100 V at RT. Deoxyribonucleic acid bands (supercoiled, linear, and open circular) were stained with ethidium bromide (EtBr) and the gels were scanned on the gel documentation system (iBright CL1000, Invitrogen). Bands were detected using image analysis software.
Cell Culture
The murine macrophage (RAW 264.7) was purchased from Korean Cell Line Bank (Seoul, Korea) and maintained in the DMEM supplemented with 10% FBS and 1% penicillin-streptomycin (P.S.) in a controlled atmosphere of 5% CO2 at 37°C.
Cell Viability
Cell viability of the sample was carried out by following the treatment with LPS and MCR of RAW 264.7 cells. Cell viability was quantified by using MTT assay as previously described 11,38 with some modifications. In brief, the cells were seeded as 5 × 104 cells each well in 100 µL into a 96-well plate and maintained at 37°C with 5% CO2 in an incubator. After 24 hours, the cells were incubated with various concentrations of MCR extracts (10-1000 µg/mL) with or without LPS (1 µg/mL). The viability of treated cells was compared with that of the control (without MCR and LPS) and negative control (with LPS and without MCR). Cell viability was determined by measuring the absorbances at 570 nm and compared with that of control (100%).
NO Inhibitory Activity
The effect of MCR extracts on LPS-administrated nitric oxide (NO*) production by murine macrophage was investigated by Griess reagent method 38,39 with slight modification. In brief, RAW 264.7 cells were loaded in a 96-well plate as 5 × 104 cells per well and maintained at 37°C with 5% CO2 in an incubator. After 24 hours, the cells were treated with LPS with or without MCR and the control was treated with only media. After treatment for 24 hours, 80 µL of supernatant was collected from each well and the same amount of Griess reagent was mixed with the supernatant to quantify the amount of NO. The absorbance was taken at 540 nm. NO production was calculated from the calibration curve of NaNO2 which was also added in media to the Griess reagent as standard samples.
Cellular Antioxidant Activity
Intracellular antioxidant activity was investigated by the level of suppression of ROS. Reactive oxygen species suppressive activity was analyzed by the inhabitation of the oxidation of 2′,7′-dichlorodihydrofluorescein diacetate (DCF-DA) to fluorescent 2′,7′-dichlorofluorescein by hydroperoxide. 22 RAW 264.7 cells were loaded as 5 × 104 cells per well in two 96-well plates, one for water extract and another for 70% ethanol extract. After 24 hours incubation, the cells were incubated with various concentrations of both extracts (10-1000 μg/mL) for 24 hours in 2 different 96-well plates. Then, 10 µM DCF-DA in media was added into every well followed by incubation for 40 minutes. H2O2 (60 µM) was poured except control group and the fluorescence was taken after 1 hour. The fluorescence was measured by using a Soft MaxPro5 at the excitation and emission wavelengths of 485 and 530 nm, respectively.
Western Blot Analysis
Immunoblot expression of MCR in RAW264.7 cells was analyzed by Western blot analysis as previously described 25 with slight modifications. The cells were seeded in different 100-mmculture dishes at 2 × 106 cells per dish and incubated for 24 hours. They were treated with or without LPS (1 µg/mL) and MCR water extract (250 and 500 µg/mL). Proteins from treated cells were extracted with Radio immuno precipitation assay (RIPA) buffer (Thermo Scientific, United States) and the concentration of the protein extract was determined by Bio-Rad protein concentration dye (Bio-Rad, United States). Each sample was subjected to 10% polyacrylamide gel electrophoresis (30 µg/lane) and the gels were then transferred onto Poly vinylidene fluoride (PVDF) membrane. Afterward, membranes were treated with appropriate antibody at 4°C overnight. The immunosignal was captured by iBright (Invitrogen) with enhanced chemiluminescence kit.
Splenocytes Proliferation Assay
Primary spleen cells were isolated from the ICR 5-week-old male mouse (Orientbio Inc. Seongnam, Korea) and maintained in RPMI 1640 (WElGENE Inc., Seoul, Korea) media supplemented with 10% FBS and 1% P.S. The cells were seeded as 5 × 104 cells/well in 96-well plate for MTT assay. Twenty-four hours later, 100 ng/mL LPS was induced in cells with or without MCR (100, 200, and 400 µg/mL). Control group was treated only with media. After 24 hours of incubation, cell counting was performed by cytometer with trypan blue and MTT assay was carried out.
Statistical Analysis
All data are presented as the mean ± standard deviation for at least 3 independent experiments and statistical analyses were performed using Microsoft Excel 2016 edition and GraphPad Prism 5.0 software (GraphPad Software, Inc., San Diego, CA, United States). The significance of differences between groups was determined by using one-way analysis of variance.
Supplemental Material
Supplementary material - Supplemental material for Antioxidant, Anti-inflammatory, and Antiproliferative Activity of Mori Cortex Radicis Extracts
Supplemental material, Supplementary material, for Antioxidant, Anti-inflammatory, and Antiproliferative Activity of Mori Cortex Radicis Extracts by Al B. Bayazid, Jae G. Kim, Seo H. Park and Beong O. Lim in Natural Product Communications
Footnotes
Acknowledgment
This work was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (NRF-2016R1D1A1A09919529).
Author Contributors
A.B. Bayazid carried out the research and wrote the manuscript. A.B. Bayazid, J.G. Kim, and S.H. Park designed the research study. A.B. Bayazid, J.G. Kim, and B.O. Lim analyzed the data. B.O. Lim supervised the research and reviewed the manuscript. All authors reviewed and approved the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
