Abstract
Bone cancer pain is characterized by moderate to severe ongoing pain that commonly requires the use of opiates, which could produce tolerance or addiction. Baicalin is a flavonoid compound extracted from Huang Qin, possesses antioxidant properties, and has an analgesic effect on nitroglycerin-induced migraine in rats and neuropathic pain in spinal nerve ligation rats. However, the effect of baicalin on bone cancer pain is still unclear. Therefore, the aim of this study is to examine the analgesic effect of baicalin in a rat model of bone cancer pain. Bone cancer pain animal model was created by tumor cell implantation (TCI). Animal behaviors were measured using a set of mechanical or electronic von Frey apparatus and hot plate. mRNA expression and inflammation cytokine levels were examined by Quantitative polymerase chain reaction (qPCR) and enzyme linked immunosorbent assay (ELISA) methods. Baicalin suppressed the upregulation of transient receptor potential vanilloid 1 (TRPV1), but not transient receptor potential A1 in dorsal root ganglion (DRG) of TCI rats. In addition, the phosphorylation of extracellular regulated protein kinases (ERK) was also suppressed by baicalin injection in DRG of TCI rats. Our results revealed that baicalin might play a promising analgesic role by preventing the upregulation of TRPV1 in DRG of TCI rats. Baicalin administration prevented the progress of bone cancer pain and reduced mechanical allodynia and thermal hyperalgesia. Our study clearly established a novel role of baicalin as an analgesic agent for bone cancer pain. And the analgesic role of baicalin in bone cancer pain might involve a TRPV1.
The most common types of cancer, including breast, liver, and lung cancer, tend to metastasize to the bones. As bone remodeling progresses, severe spontaneous pain occurs in which the occurrence and severity of such pain may be acute and unpredictable. 1 An epidemiological study showed that 75% to 90% of patients with metastatic or advanced cancer experienced severe cancer-induced pain. 2 Bone cancer pain is a common symptom in patients with advanced cancer. The functional status and quality life of patients may be particularly debilitating due to bone cancer pain. 1,3 Generally, the treatment of pain from bone metastases involves the use of multiple complementary approaches including radiotherapy, surgery, chemotherapy, bisphosphonates, calcitonin, and analgesics. 1,4 However, bone cancer pain is a chronic pain that is very difficult to fully control since metastasis is usually not limited to a single site. 4 Moreover, the efficacy of commonly used analgesics, such as non-steroidal anti-inflammatory drugs (NSAIDS) 4 and opioids, 5,6 are limited, because of their significant side effects in the treatment of bone cancer pain. Accordingly, there is a need to develop new strategies for the treatment of bone cancer pain with acceptable side effects.
Several transient receptor potential (TRP) superfamily ion channels have been reported to be involved in nociception and thermosensation in the peripheral and central nervous systems. 7 Transient receptor potential vanilloid 1 (TRPV1) is activated by multiple mechanisms, such as noxious heat (>43°C), protons, and some pungent chemicals such as capsaicin. 7 Transient receptor potential A1 (TRPA1) functions as a sensor of environmental and endogenous chemical irritants, such as 4-hydroxynonena, allyl isothiocyanate, and icillin. 8 Transient receptor potential vanilloid 1 and TRPA1 are shown to have a crucial role in nociceptive transmission under pathological forms of pain. 9 -11 Moreover, it is clear that spinal synaptic plasticity is an important procedure for the pain transition from acute to chronic in which TRPV1 and TRPA1 play critical roles. 12 Transient receptor potential vanilloid 1 and TRPA1 expression in nociceptors are altered in different models of neuropathy under pathological conditions. 13,14 The cancer cells that metastasize to the bone marrow and grow rapidly could gradually destroy bone tissue, which was the initiation of a coordinated process of bone formation and absorption. 15 In squamous cell carcinoma rat and mouse models, upregulation of TRPV1 or TRPA1 was observed in the dorsal root ganglion (DRG) and spinal cord. 16 -18 In summary, targeted TRPV1 or TRPA1 could be a potential treatment in bone cancer pain.
Plant natural products are well-known as traditional Chinese medicine in the therapy of chronic comorbidities, such as diabetes, cancer, and pain 19 . Baicalin (7-glucuronic acid,5,6-dihydroxyflavone), a flavonoid compound extracted from Huang Qin, possesses antioxidant properties 20 and has an analgesic effect on nitroglycerin-induced migraine in rats, 21 neuropathic pain in spinal nerve ligation rats, 22 and diabetic neuropathic pain. 23 These studies suggested that the potential role of baicalin in the relief of pain originated from peripheral nervous system. However, the putative impact of baicalin on bone cancer pain and the underlying mechanisms remain unclear. Therefore, this study aims to investigate the analgesic effect of baicalin on bone cancer pain and its possible mechanisms.
In the present study, detailed experiments were carried out to investigate the analgesic role of baicalin on bone cancer pain. Our results demonstrated that baicalin administration reduced both neuropathic pain and TRPV1 expression in DRG neurons after tumor cell implantation (TCI) in rats. Our data highlighted the potential therapeutic role of baicalin in the treatment of bone cancer pain.
Materials and Methods
Animals
Male Sprague Dawley rats (200-220 g; Guangdong Laboratory Animal Center, Guangzhou, China) were maintained on a 12 hours/12 hours light/dark cycle. The animals were housed by 2 to 3 rats/cage at 22°C ± 1°C, with free access to water and rat chow. All experimental procedures were carried out in accordance with the guidelines of the International Association for the Study of Pain and approved by the Institutional Animal Care and Use Committee of Shenzhen Longgang People’s hospital. Animal treatments were performed according to the Guidelines of the International Association for the Study of Pain.
Induction of Bone Cancer Pain
Walker 256 cell line was purchased from China Center for Type Culture Collection (Wuhan, China). Bone cancer was induced by TCI as described. 24,25 The left leg was shaved and the skin was disinfected with 70% (v/v) ethanol, after rats were anesthetized by intraperitoneal administration of sodium pentobarbital (50 mg/kg, intraperitoneally [i.p.]). A rostrocaudal incision of 1 cm was made in the skin over the lower one-third of the tibia for clear exposure with minimal damage to nerves and muscles. The medullary canal was approached by inserting a 23-gage needle proximally through a hole drilled in the tibia. The needle was then replaced with a 20 µL microinjection syringe. A 10 µL volume of Walker 256 cells (2 × 105 cells) or boiled cells (sham group) was injected in the tibia straight after culturing them in vitro in plates. The syringe was removed after a 2-minute delay allowing cells to fill the cavity. The drill hole was sealed with bone wax and the wound was closed using 1-0 silk threads. The rats were allowed for unrestricted movement in the cages after recovery and the general condition of rats was monitored during the experiments.
Drug Administration and Experimental Design
Baicalin was purchased from Wako (Wako Pure Chemical Industries, Osaka, Japan) and dissolved in 2% Dimethyl Sulfoxide (DMSO) and saline. The stock solution was filtered through a 0.22-µm membrane filter before use. For behavioral experiments, rats were randomly divided into 4 groups (8 rats in each group): a vehicle group, a TCI group, a TCI group treated with baicalin (10 µg/kg), and a TCI group treated with baicalin (30 µg/kg). 23 Rat received injection (i.p.) of baicalin daily at 24-hour intervals, while vehicle and TCI animals received the same volume of saline.
Behavioral Tests
Mechanical Allodynia (von Frey Test)
The mechanical allodynia was assessed by von Frey test. Rats were placed in a clear plastic box (20*17*13 cm) with a wire mesh floor, 40 cm above the bench, and allowed to habituate to their environments for 15 minutes prior to testing. An electronic von Frey hair unit (IITC, United States) was used: the withdrawal threshold was evaluated by applying forces ranging from 0 to 50 g with a 0.2 g accuracy. Punctuate stimulus was delivered to the midplantar area of each hind paw from below the mesh floor through a plastic tip and the withdrawal threshold was automatically displayed on the screen. The paw sensitivity threshold was defined as the minimum force required to elicit a robust and immediate withdrawal reflex of the paw. Voluntary movements associated with locomotion were not considered as a withdrawal response. The hind paw of each rat was stimulated at 5-minute intervals. Measurements were repeated 3 times and the final value was obtained by average of 5 measurements. Mechanical threshold measurement was performed in a blinded manner.
Thermal Hyperalgesia
Thermal hyperalgesia was assessed using a hot plate analgesia meter (BIO-CHP, Bioseb, France). The animals were placed one at a time on a hot plate maintained at a temperature of 53°C ± 1°C. Latency of response either by jump or hind-paw-lick was recorded using an electronic timer. To prevent tissue damage, a cut-off time of 30 seconds was adopted. Each rat was measured 3 times, with a 10-minute interval between each measurement, and the average value was calculated. Lifting for normal locomotion was excluded. Behavioral testing was done with the experimenters blinded to treatment conditions.
Tissue Collection
Upon completion of behavioral testing, rats were deeply anesthetized with sodium pentobarbital (50 mg/kg, i.p., Sigma-Aldrich, MO, United States). The lumbar (L) 4-L5 DRGs, which corresponded to the innervation of nerves in the hind paw that is in anatomical proximity to the tibia, were quickly removed and flash frozen in liquid nitrogen. Samples were stored at −80°C until assay.
Isolation of Total RNA and Quantitative Real-Time PCR (qRT-PCR)
The L4-L5 DRGs were collected from each rat and placed in tubes with Trizol reagent (Sigma, United States). RNA isolation was followed by chloroform extraction and isopropanol precipitation, frozen and stored at −80°C. For cDNA synthesis, 2 µg of total RNA was reverse transcribed using the PrimeScript Master Mix kit (Takara, Japan) following the manufacturer’s instructions. Real-time Polymerase chain reaction (PCR) was carried out in a Roche Light Cycler 96 System (Roche, Switzerland) according to the manufacturer’s instructions using TransStart Tip Green qPCR SuperMix (TransGen Biotech, China). The results were standardized for comparison by measuring levels of glyceraldehyde-3-phosphate dehydrogenase. IL-1β: Interleukin 1β (GAPDH) mRNA in each sample. The primers were used according to the previous report. 26,27
Western Blot
Upon completion of behavioral test, rats in the experiment were deeply anesthetized with sodium pentobarbital (50 mg/kg, i.p. Sigma, United States) and perfused intracardially with warm 0.9% saline. The L4-L5 DRGs were removed, dissected, and homogenized in 300 µL ice-cold (4°C) Radio Immunoprecipitation Assay (RIPA) buffer and lysed as previously described. 28 The supernatant was carefully collected, and the protein concentration was measured using a protein assay kit (Bio-Rad, United States). Equal amounts of protein (20 µg) were separated by 10% Tris-Tricine Sodium dodecylsulphate polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto polyvinylidene difluoride (PVDF) membranes. Polyvinylidene difluoride membranes were then blocked in 10% nonfat milk for 1 hour at room temperature followed by overnight incubation at 4°C with rabbit anti-TRPV1 (1:1000; Abcam, MA, United States), ERK (1:500; Abcam, MA, United States), phosphorylation of ERK (pERK, 1:500; Abcam, MA, United States), and GAPDH (1:5000; Abcam, MA, United States) primary antibody. Polyvinylidene difluoride membranes were then incubated for 30 minutes at room temperature with goat antirabbit polyclonal IgG (1:2000, Abcam, MA, United States). Among these steps, PVDF membranes were rinsed with Tris buffer system-Triton (TBS-T) 3 times for 10 minutes. Protein bands were visualized with the ECL kit (Amersham Pharmacia Biotech, Piscataway, United States) and then detected by using the ChemiDoc Imaging System (Bio-Rad, Berkeley, United States). The density of the bands was quantified after background subtraction and then normalized with respect to the β-actin or ERK levels using NIH Image J software.
Statistical Analysis
The results are presented as mean ± SEM; statistical significance between groups was determined using repeated measures Analysis of Variance (ANOVA) followed by multiple t-tests with Bonferroni correction or one-way ANOVA followed by 2-tailed t-test with Bonferroni correction. Values of *P < 0.05, **P < 0.01 or # P < 0.05, ## P < 0.01 were considered significant and the number of repetitions (n) is indicated in the figure legends.
Results
Bone Cancer Induced Thermal Hyperalgesia and Mechanical Allodynia in Rat
First of all, Walker 256 cells (2 × 105 cells) or boiled cells (vehicle group) were injected into the bone cavity of rats to create bone cancer pain model in rats. After tumor inoculation, we tested the mechanical thresholds and paw withdrawal latencies upon heat stimulation in rats from vehicle and TCI groups. On the third day after receiving Walker 256 cells injection, the mechanical thresholds in the left hand paw (ipsilateral side) of rats were significantly decreased, compared with vehicle group rats. On the other hand, the mechanical thresholds in the right hand paw (contralateral side) of rats were not changed. The mechanical sensitivity is kept at a stable level until 21 days in rats after TCI (P < 0.01, n = 6, Figure 1(a)). In addition, a significant thermal hyperalgesia in response to noxious heat stimulation was observed in TCI model rats as well (P < 0.01, n = 6, Figure 1(b)). These results suggested that the bone cancer pain model was successfully established in rats after TCI surgery.
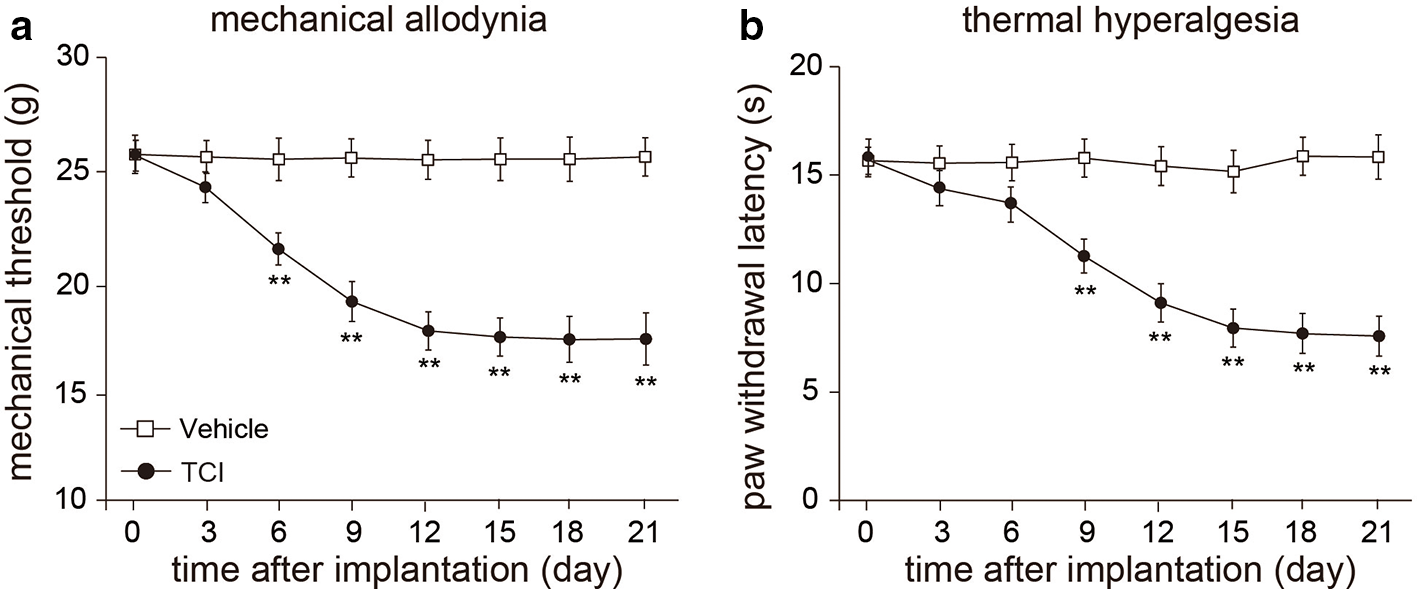
Tumor cell implantation induced mechanical allodynia and thermal hyperalgesia in rats. (a) Mechanical allodynia in tumor cell implantation induced bone cancer pain rats. (b) Thermal hyperalgesia in tumor cell implantation induced bone cancer pain rats. Mean ± SEM, n = 8; **P < 0.01 vs vehicle group. Repeated measures ANOVA followed by multiple t-tests with Bonferroni correction.
Baicalin Ameliorated Mechanical Allodynia and Thermal Hyperalgesia in Bone Cancer Pain Rats
We next explored the analgesic effect of baicalin administration i.p. in rat model with bone cancer pain. Baicalin (10 or 30 µg/kg) was injected (i.p.) every day. Our data showed that the mechanical threshold (ipsilateral) and paw withdrawal threshold upon heat stimulation were significantly decreased in TCI group (Figure 2(a) and (b), P < 0.01, n = 8, repeated ANOVA). Moreover, the decreases of the mechanical threshold and paw withdrawal latency in response to thermal stimulation were significantly prevented in either TCI + baicalin 10 µg/kg or TCI + baicalin 30 µg/kg group, compared to TCI + saline group (Figure 2(a) and (b), P < 0.05 or P < 0.01, n = 8, repeated ANOVA). Our results suggested that baicalin may prevent the progress of tumor-induced neuropathic pain in rat after TCI.

Reduction of mechanical allodynia and thermal hyperalgesia by consecutive injection of baicalin in tumor cell implantation rats. Prevention of mechanical allodynia (a) and thermal hyperalgesia (b) by repeated injection of baicalin. Baicalin 10 or 30 µg/kg was repeatedly injected (intraperitoneally, every day). Paw withdrawal threshold in the von Frey tests and paw licking latency in the hot plate tests were measured in rats before and after surgery. Mean ± SEM, n = 8, **P < 0.01 vs vehicle group; # P < 0.05, ## P < 0.01 vs tumor cell implantation group. Repeated measures ANOVA followed by multiple t-tests with Bonferroni correction.
Baicalin Suppressed the Upregulation of Trpv1 mRNA Expression in DRG of TCI Rats
To explore the potential mechanisms which baicalin involved in the prevention of neuropathic pain, we examined Trpv1 and Trpa1 mRNA expression in DRG of TCI rats using quantitative Reverse Transcription-Polymerase Chain Reaction (RT-PCR) method. Our results demonstrated that Trpv1 and Trpa1 mRNA expression levels were dramatically increased in DRG of TCI rats 12 and 21 days after implantation, but only Trpv1 mRNA expression was significantly prevented by repeated baicalin administration (Figure 3(a) and (b), P < 0.01, n = 8, one-way ANOVA) in DRG of TCI rats 12 and 21 days after surgery. These results revealed that baicalin suppresses Trpv1 mRNA expression upregulation in DRG of rat after tumor cell implantation.

mRNA expression levels of Trpv1 and Trpa1 in dorsal root ganglion of tumor cell implantation rats after baicalin treatment. Trpv1 (a) and Trpa1 (b) mRNA expression levels in DRG of CCI model rats before and after saline or baicalin treatment for 0, 3, 12, and 21 days. Mean ± SEM, n = 8; **P < 0.01 vs o day. # P < 0.05, ## P < 0.01 vs tumor cell implantation group. One-way ANOVA followed by 2-tailed t-test with Bonferroni correction.
Baicalin May Suppress the Upregulation of TRPV1 via ERK Phosphorylation
We next examined the expression of TRPV1 protein expression in DRG of rats. Western blot results demonstrated that the protein expression of TRPV1 was significantly increased in DRG of rats 21 days after TCI surgery. But this increase was prevented by repeated baicalin (30 µg/kg) administration in TCI rats (Figure 4(a) and (b), P < 0.01, n = 8, one-way ANOVA). We further asked the signaling pathway involved in the analgesic effect of baicalin. Western blot results demonstrated that the pERK was significantly increased in DRG of TCI rats, but this increase did not happen in the DRG of TCI rats repeatedly treated with baicalin (30 µg/kg, Figure 4(a) and (c), P < 0.01, n = 8, one-way ANOVA). These results further suggested that baicalin relieves neuropathic pain by preventing TRPV1 upregulation and ERK phosphorylation in DRG of TCI rats.

Protein expression of transient receptor potential vanilloid 1 and ERK in dorsal root ganglion of tumor cell implantation rats after consecutively baicalin treatment. (a) Representative images of Western blot result of transient receptor potential vanilloid 1, phosphorylation of ERK, ERK, and GAPDH from dorsal root ganglion of tumor cell implantation rats consecutively treated with baicalin (30 µg/kg) for 21 days. Comparison of transient receptor potential vanilloid 1 (b) and phosphorylation of ERK (c) protein levels in DRG of tumor cell implantation rats consecutively treated with baicalin (intraperitoneally) for 21 days. Mean ± SEM, n = 8, **P < 0.01 vs sham group, ## P < 0.01 vs saline group. One-way ANOVA followed by 2-tailed t-test with Bonferroni correction.
Discussion
Bone cancer pain is considered to be one of the most difficult chronic pain to fully control, which seriously affects the quality of life in patients. 29 Therefore, we still need to further seek new analgesics and treatment options. The injection of Walker 256 rat breast cancer cells can successfully prepare a rat bone cancer pain model similar to human bone cancer pain. 24,25 The mechanical thresholds and paw withdrawal thresholds in response to noxious heat stimulation in rats were significantly decreased from day 3 after the injection of Walker 256 cells into the upper tibia, suggesting that the bone cancer pain model was established successfully (Figure 1). In this study, the analgesic effect of baicalin on bone cancer pain models was investigated. Baicalin dose-dependently prevented the progression of bone cancer pain upon TCI. Moreover, TRPV1 and TRPA1 were significantly upregulated in DRG of TCI rats. Baicalin suppressed the upregulation of TRPV1, but not TRPA1 in DRG of TCI rats. In addition, the pERK was also suppressed by baicalin injection in DRG of TCI rats. Our results revealed that baicalin might play a promising analgesic role by preventing the upregulation of TRPV1 in DRG of TCI rats.
The mechanism of bone cancer pain includes inflammatory pain and neuropathic pain components, but it is not a simple addition of these 2 types of pain. The bone cancer pain includes not only the tumor itself but also the inflammatory reaction, peripheral sensitization, and central sensitization. Transient receptor potential channels are reported to act as thermal, chemical, and mechanical sensors. 30 Accumulating evidences indicated that some TRP channels are responsible for cancer pain. Transient receptor potential vanilloid 1 is a well-known channel that is involved in nociception and thermosensation in peripheral and central nervous systems. 7 Transient receptor potential vanilloid 1 is shown to have a crucial role in nociceptive transmission under pathological forms of pain. 31 Transient receptor potential vanilloid 1 receptors are expressed primarily by primary afferents/nociceptors, and most TRPV1 is localized presynaptically. 32 Transient receptor potential vanilloid 1 expression in nociceptors is altered in different models of neuropathy under pathological conditions. 13,14 Our data showed that cumulative injection of baicalin (i.p., every day) when given during the induction of bone cancer pain prevented the establishment of TCI-induced bone cancer pain in rats (Figure 2). These results suggested that baicalin might be a promising agent for amelioration of TCI-induced bone cancer pain. Repeated baicalin given during the development of bone cancer pain significantly suppressed TRPV1 upregulation in DRG (Figure 4), indicating that the analgesic effect of baicalin might be associated with the suppression of the TCI-induced TRPV1 upregulation in DRG of rats. These data suggested that TRPV1 might be involved in the analgesic effect of baicalin on bone cancer pain. The analgesic effects of baicalin on bone cancer pain might also involve other possible mechanisms, such as anti-inflammation, 33 regulating P2X3, 34 and we could not exclude these possibilities.
How does baicalin regulate the expression of TRPV1 in DRG of TCI-induced bone cancer pain rats? It has been reported that bone cancer pain involves a strong inflammatory component and TRPV1 is a key molecule in the process of inflammatory sensitization of afferent sensory nerve fibers. Several pro-inflammatory cytokines such as tumor necrosis factor alpha and IL-1β activate signal transduction pathways in sensory neurons leading to downstream activation/sensitization of TRP channels. 35 Transient receptor potential vanilloid 1 is upregulated on sensory neurons by inflammatory condition. 36,37 On the other hand, baicalin has a strong anti-inflammation effect. 33 Moreover, mitogen-activated protein kinases are the main protein phospho-regulating effectors that mediate nociceptive sensitization. 38 It has been reported that TRPV1 involves in endometriosis pain via ERK signaling pathway. 39 Our results also demonstrated that baicalin prevents TRPV1 upregulation and ERK phosphorylation in DRG of Paclitaxel-induced peripheral neuropathic pain (PIPNP) rats. Our data suggested that TRPV1 may play an analgesic role in neuropathic pain signal transduction, which may be mediated by ERK signaling pathway. Moreover, it has been reported that baicalin modulates the expression of Protein kinase C (PKC), 40,41 which is upstream of TRPV1. Therefore, we could not exclude the other possible mechanisms by which baicalin may be involved in TCI-induced bone cancer pain.
In conclusion, our study clearly established a novel role of baicalin as an analgesic agent for bone cancer pain. And the analgesic role of baicalin in bone cancer pain might involve a TRPV1. Our data suggested that baicalin plays an important analgesic role in bone cancer pain and might serve as a potential compound in clinical treatment and prevention of bone cancer pain.
Footnotes
Author note
D. Xiong and A. Li were responsible for the concept and design of the study; P. Li, Y. Bi, and Y. Deng were involved in experimental and analytical aspects of the manuscript; D. Xiong, A. Li, and P. Li performed data interpretation, presentation, and writing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Grants from Health and Family Planning Commission of Shenzhen Municipality (No. SZFZ2018036) and Shenzhen Longgang District Scientific Research Program of China (No. LGKCYLWS2018000095).
