Abstract
In our preliminary screening study on the protein tyrosine phosphatase 1B (PTP1B) inhibitory and cytotoxic activities, an ethyl acetate soluble fraction of the aerial part of Orthosiphon stamineus Benth. was found to inhibit PTP1B activity. Thus, based on assay-guided isolation of this active fraction, ten compounds (
Protein tyrosine phosphatase 1B (PTP1B) is a member of nontransmembrane protein tyrosine phosphatase that plays a key role in metabolic signaling and is a promising drug target for type 2 diabetes and obesity. 1,2 Originally, this enzyme is located on the cytoplasmic face of endoplasmic reticulum, and is firstly identified from human placenta and executed as a critical regulator of multiple signaling pathways involved in many kinds of human cancers. 3,4 Accumulating evidences indicated that PTP1B is involved in leptin signal of cancer as a key negative regulator. 3,4 However, several contrasting findings suggested that it could exert both tumor-suppressing and tumor-promoting effects depending on the substrate involved and the cellular context. 5 Among human cancers, breast cancer is the most common malignancy and accounts for the second-highest number of deaths in women cancer worldwide. Protein tyrosine phosphatase 1B had recently drawn attention as an attractive target for the treatment of breast cancer. 5,6 Protein tyrosine phosphatase 1B promoted breast cancer metastasis by regulating phosphatase and tensin homolog but not epithelial-mesenchymal transition. 7 In addition, the level of PTP1B was higher in breast cancer tissues than the corresponding adjacent normal tissues in several patients. It was positively associated with lymph node metastasis and estrogen receptor status, and acted as an independent prognostic factor and predicted a poor prognosis in estrogen receptor-positive breast cancer patients. 7,8 These findings consequently raised the hypothesis that inhibitors of PTP1B may offer new avenues for the treatment of breast cancer. Therefore, finding new PTP1B inhibitors from natural plants was investigated. A few PTP1B inhibitors from natural products have been identified. The results showed that natural compounds found to inhibit PTP1B were diverse including pterocarpan derivatives, 9,10 diterpenoids and triterpenoids, 11 flavonoids, 12 sesquiterpenoids, 12 coumarins, 12 alkaloids, 12 and phenolic compounds. 13,14 These results encourage us to continue our studies in the discovery of PTP1B inhibitors from natural sources.
In our screening program for PTP1B inhibitors from medicinal plants, the ethyl acetate (EtOAc) fraction of the aerial parts of Orthosiphon stamineus Benth. (Lamiaceae) exhibited 67.8% inhibition on PTP1B at a concentration of 30 µg/mL. Orthosiphon stamineus is found mainly in Southeast Asia, Australia, and Africa. 15 In Vietnam, O. stamineus is called “Rau Meo” and the aerial parts are used for the treatment of urinary lithiasis, edema, eruptive fever, influenza, hepatitis, and jaundice. 16 In Myanmar, this plant is known as “Secho” or “Myit-shwe” and the leaves are reputed to be an antidiabetic drug and decoctions of the air-dried leaves are used to cure urinary tract and renal diseases. 17 This plant is also known as “Neko no hige” in Japan and consumed as a healthy Java tea to facilitate body detoxification. In Indonesia, O. stamineus is known as an effective folk medicine for the treatment of diabetes, hypertension, rheumatism, tonsillitis, and menstrual disorder. 18 Orthosiphon stamineus had been reported to possess various pharmacological activities such as anti-inflammatory, 19,20 antioxidant, 21 hepatoprotective, 22,23 anticancer, 24,25 antihypertensive, 19,26 gastroprotective, 22 antisebum, 27 hyperlipidemic, 28 nephroprotective, 21 antipyretic, 29 anti-angiogenic, 30 antibacterial, 31 antiviral, 32 diuretic, 33 and antidiabetic. 34,35 Previous studies on chemical constituents of O. stamineus showed the presence of essential oils, 36 diterpenoids, triterpenoids, flavonoids, and phenolics. 19,21,37 Some studies have shown PTP1B inhibitory 34,35 and cytotoxic activities 38 of extracts and fractions from O. stamineus. However, there has been no investigation of chemical constituents and inhibitory activities of PTP1B and cytotoxicity of O. stamineus. Therefore, this paper described the isolation and structural elucidation of these compounds as well as the evaluation of their PTP1B inhibitory and cytotoxic activities on human breast cancer cells.
The methanol extract of the aerial parts of O. stamineus was partitioned into chloroform, ethyl acetate, and water-soluble fractions. In a preliminary experiment, the inhibitory effect of these fractions was tested in vitro on PTP1B enzyme assay. The result showed that the ethylacetate fraction was the most active fraction against the enzyme activity (see supplemental figures S2–S4 for details). Therefore, the ethyl acetate fraction was selected for further isolation of active components. Chromatographic purification of this fraction (see supplemental figure S3) led to the isolation of 10 compounds (

Chemical structure of isolated compounds
The 1H-nuclear magnetic resonance (NMR) spectrum of
Compound
Compound
Compound
Similar to
All isolates
Protein tyrosine phosphatase 1B Inhibitory Activity of Isolated Compounds (1-10) From Orthosiphon stamineus Benth.

aResults are expressed as IC50 values (µM), determined by regression analysis and expressed as the means ± SD of 3 replicates.
bData not determined.
cPositive control.
The Lineweaver-Burk plot and Dixon plot experiments were performed with the presence and absence of the inhibitors with various concentrations of the substrate para-Nitrophenylphosphate (p-NPP) to determine the inhibition type of the active compounds (

Lineweaver-Burk plots for the inhibition of compounds (5, 6, 9, and 10) on the protein tyrosine phosphatase 1B-catalyzed hydrolysis of p-NPP. Data are expressed as the mean reciprocal of initial velocity for n = 3 replicates at each substrate concentration.
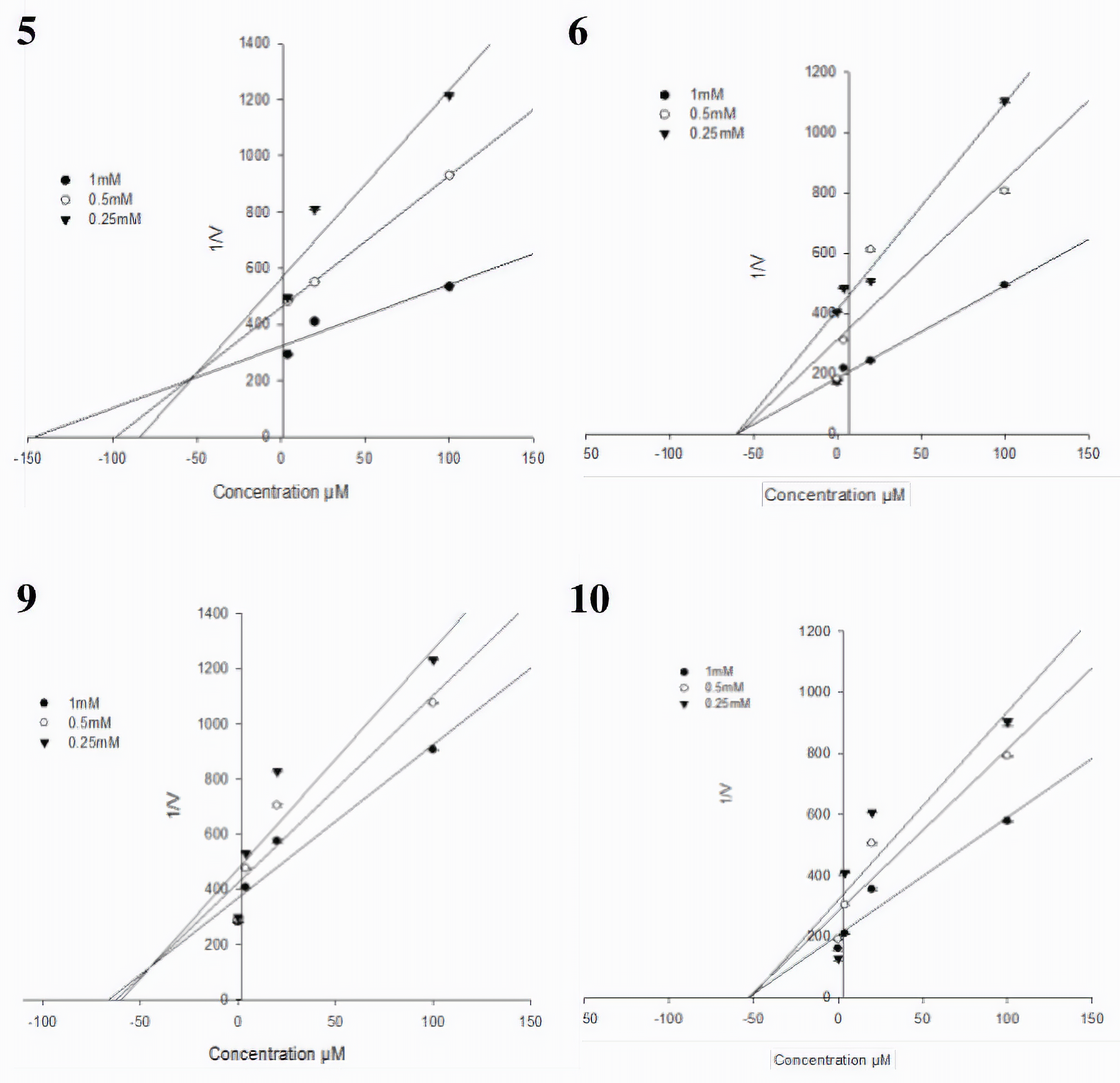
Dixon plots for isolated compounds (5, 6, 9, and 10) used for determining the inhibition constant K i. K i values were determined from the negative x-axis value at the point of the intersection of 3 lines. Data are expressed as the mean reciprocal of initial velocity for n = 3 replicates at each substrate concentration.
For diabetic and cancer objects, the PTP1B enzyme displayed an important role as an attractive target in the treatment, especially for breast cancer. Many studies have shown that in human breast cancer PTP1B was overexpressed, and that the inhibition of PTP1B delayed erbB2-induced mammary tumorigenesis and protected from lung metastasis.
6,50
Thus, the isolated flavonoids were further investigated for their growth inhibition on MCF7, MCF7/TAMR, and MDA-MB-231 breast cancer cell lines (Table 2). Among the isolates, compounds
Cytotoxic Activities of Isolated Compounds (1-10) Against Human Breast Cancer Cell Lines.
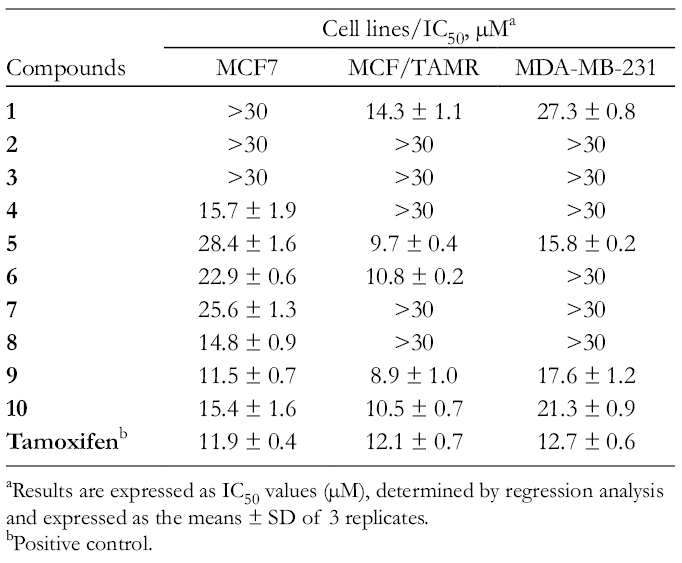
aResults are expressed as IC50 values (µM), determined by regression analysis and expressed as the means ± SD of 3 replicates.
bPositive control.
Experimental
General Experimental Procedures
1H-NMR (400 MHz) and 13C-NMR (100 MHz) were measured on a Varian Unity Inova 400 MHz spectrometer. Electron spray ionization mass spectroscopy (ESI-MS) was obtained from a Varian FT-MS spectrometer and MicroQ-TOF III (Bruker Daltonics, Ettlingen Germany). Infrared (IR) spectroscopy was fulfilled on Nicolet Impact 410 spectrometer. UV was performed in spectroscopic V-630 UV-VIS instrument. Column chromatography was carried out on silica gel (Si 60 F254, 40-63 mesh, Merck, St. Louis, MO, United States). All solvents were redistilled before use. Precoated thin layer chromatography (TLC) plates (Si 60 F254) were used for analytical purposes. Compounds were visualized under UV radiation (254-365 nm) and by spraying plates with 10% H2SO4 followed by heating with a heat gun. The high performance liquid chromatography (HPLC) runs were carried out using a Gilson System LC-321 pump with an UV/Vis-155 UV detectors, and an Optima Pak C18 column (10 × 250 mm, 10 µm particle size), J. sphere ODS-H80 (20 × 150 mm, 4 µm particle size) RS Tech, Korea, and Shodex-C18M-10E (10 × 250 mm, 10 µm particle size), and/or YMC-Pak ODS-AM (6.0 × 150 mm, 5 µm particle size) for semipreparative runs.
Plant Materials
The aerial parts of O. stamineus Benth. were collected in January 2017 at Ngu Hiep, Thanh Tri, Ha Noi. The sample was identified by Dr Nguyen Quoc Binh (Viet Nam National Museum of Nature, VAST). A voucher specimen (SH-164) was deposited at the Institute of Natural Products Chemistry (INPC), VAST (see supplemental figure S1 and supplemental references S1–S2 for details).
Extraction and Isolation
The aerial parts of O. stamineus (1.1 kg) were extracted with methanol (5 L × 3 times) at room temperature using sonication for 5 hours. After removing the solvent under reduced pressure, the methanol extract (121 g) was suspended in hot water (500 mL) and partitioned with n-hexane (1 L × 3), chloroform (CHCl3, 1 L × 3), and ethylacetate (EtOAc, 1 L × 3), successively. The resulting fractions were concentrated in vacuo to give the hexane, CHCl3, and EtOAc-soluble fractions, respectively. By the guided-fractionation activity, the EtOAc-soluble fraction (43.2 g) was directly chromatographed on an open silica gel column (15 × 60 cm; 63-200 mm particle size, Merck) using a stepwise gradient of n-hexane/EtOAc (from 20:1 to 1:0) to give 5 fractions (OA-1-OA-5) according to their TLC profiles. Fraction OA-1 (3.2 g) was then subjected onto an open silica gel column chromatography (4.0 × 60 cm; 40-63 mm particle size), eluted with a stepwise gradient solvent system of n-hexane/acetone (from 20:1 to 1:1), resulted in the isolation of compound
Fraction 3 (OA-3) (650 mg) was also subjected onto an open silica gel column chromatography (4.0 × 60 cm; SiO2, 40-63 mm particle size), eluted with a stepwise gradient solvent system of n-hexane/acetone (from 10:1 to 1:0) to afford 6 subfractions (OA-3.1-OA-3.6). Compounds
Protein Tyrosine Phosphatase 1B Inhibition Assay
Protein tyrosine phosphatase 1B (human recombinant) was purchased from Biomol International LP, Plymouth Meeting, PA, United States, and the inhibitory activities of the tested samples were evaluated using the method as described. 51 Briefly, 0.05 to 0.1 µg of PTP1B (BIOMOL International L.P., Plymouth Meeting, PA, United States) and 4 mM p-NPP in a buffer containing 1 mM dithiothreitol, 0.1 M NaCl, 1 mM EDTA, and 50 mM citrate (pH 6.0), with or without test compounds, were added as 100 µL of a final volume to each of the 96 well. After the reaction mixture was incubated at 37°C for 30 minutes, 10 M NaOH was added to quench the reaction. Protein tyrosine phosphatase 1B enzyme activity was determined by the amount of produced p-nitrophenol at 405 nm. The nonenzymatic hydrolysis of the substrate was corrected by measuring the control, which did not contain PTP1B enzyme.
Determination of the Inhibition Mode of Active Compounds
The Lineweaver-Burk plot and Dixon plot experiments were carried out in the presence and absence of the inhibitors with various concentrations of p-NPP as the substrate. The inhibition modes of the tested compounds were assessed on the basis of their inhibitory effects on K m (dissociation constant) and V max (maximum reaction velocity) of the enzyme, which were determined by Lineweaver-Burk plot experiment. The Lineweaver-Burk plot is the double reciprocal plot of the enzyme reaction velocity (V) vs the substrate (p-NPP) concentration (1/V vs 1/[p-NPP]).
Cytotoxic Activity Assay
The cancer cell lines were maintained in Roswell Park Memorial Institute (RPMI) 1640, which included l-glutamine with 10% fetal bovine serum (FBS) and 2% penicillin-streptomycin. Cells were cultured at 37°C in a 5% CO2 incubator. Cytotoxic activity was measured using a modified 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. 52 Viable cells were seeded in the growth medium (100 µL) into 96-well microtiter plates (1 × 104 cells per well) and incubated at 37°C in a 5% CO2 incubator. The test sample was dissolved in dimethyl sulfoxide (DMSO) and adjusted to final sample concentrations ranging from 5.0 to 150 µM by diluting with the growth medium. Each sample was prepared in triplicate. The final DMSO concentration was adjusted to <0.1%. After standing for 24 hours, 10 µL of the test sample was added to each well. The same volume of DMSO was added to the control wells. On removing medium after 48 hours of the test sample treatment, MTT (5 mg/mL, 10 µL) was also added to the each well. After 4 hours incubation, the plates were removed and the resulting formazan crystals were dissolved in DMSO (150 µL). The OD was measured at 570 nm. The IC50 value was defined as the concentration of sample that reduced absorbance by 50% relative to the vehicle-treated control.
Statistical Analysis
Data are represented as means ± SD of at least 3 independent experiments performed in triplicated assays and determined by regression analysis. For statistical analysis of the data for single comparison, the significance between means was determined by Student’s t test. Sigma Plot program version 11.0 was used for analysis of the kinetic data.
Supplemental Material
Supplementary material - Supplemental material for PTP1B Inhibitory Flavonoids From Orthosiphon stamineus Benth. and Their Growth Inhibition on Human Breast Cancer Cells
Supplemental material, Supplementary material, for PTP1B Inhibitory Flavonoids From Orthosiphon stamineus Benth. and Their Growth Inhibition on Human Breast Cancer Cells by Dao-Cuong To, Duc-Thuan Hoang, Manh-Hung Tran, Minh-Quan Pham, Nhu-Tuan Huynh and Phi-Hung Nguyen in Natural Product Communications
Footnotes
Acknowledgment
The authors wish to thank the Center for Applied Spectroscopy, Institute of Chemistry, VAST, for the spectroscopic measurements.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a Grant from Vietnam Academy of Science and Technology (VAST) (Project code number: VAST.ĐLT.06/17-18).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
