Abstract
A new aurone, (2Z)-2-[(4′-hydroxyphenyl) methylene]-6-methoxy-7-prenyl-3(2H)-benzofurane (
Psoralea corylifolia L. (Leguminosae; Substance SID of PubChem:135313809) is an annual herbaceous plant which is predominantly distributed in Southeast Asia. The seeds of this species are known as “Bu-Gu-Zhi” in China, and they are used as an important folk medicine to treat asthma,
1
bacterial infections,
2
osteoporosis,
2
spermatorrhea,
2
and gynecological bleeding.
3
Previous studies have showed that the major constituents of P. corylifolia are coumarins,
4
flavonoids,
5
and meroterpenoids of the monoterpene phenol class.
6
Several of these compounds have displayed potential biological activities such as anti-inflammatory,
7
antioxidant,
8
antibacterial,
9
and anticancer properties.
10
-12
During the course of our search for natural compounds with anticolon cancer activity from P. corylifolia, a new aurone (
Results and Discussion
Compound
1H and 13C Nuclear Magnetic Resonance Spectral Data of Compound 1 in Acetone-d 6 (1H: 500 MHz and 13C: 125 MHz).


The structures of compounds
Moreover, 6 known compounds (
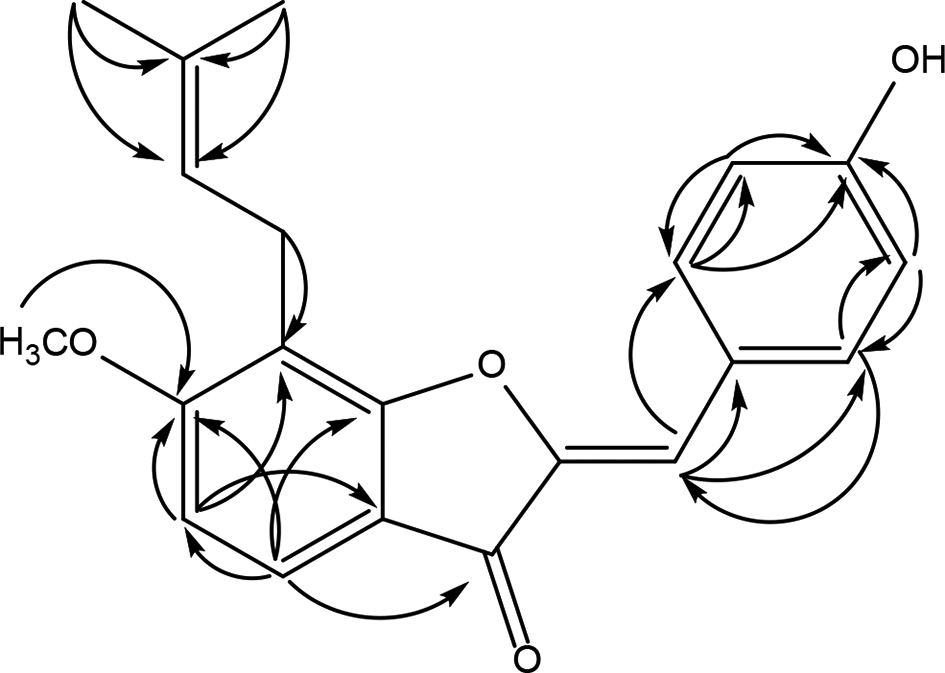
The key HMBC correlations of compound
All the isolates were evaluated for their cytotoxicity against the SW620 cell line; 5-fluorouracil was used as the positive control. Compounds
The Cytotoxicity of Compounds 1 to
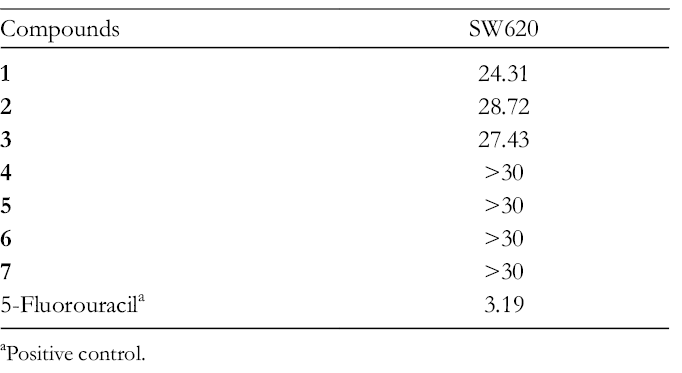
aPositive control.
Experimental
General Experimental Procedures
Optical rotations were determined on a Jasco P-2000 polarimeter (Daklia Trading Corporation, Mandaluyong, Philippines). UV spectra were recorded in MeOH using a Shimadzu UV-2401PC spectrophotometer (Shimadzu, Tokyo, Japan). IR (Infrared Radiation) spectra were recorded with a JASCO FT-IR 620 spectrophotometer. Nuclear magnetic resonance spectra were obtained from a Varian Unity Inova 500 MHz spectrometer (Varian Unity Inova, Phoenix, United States) using TMS (Tetramethylsilane) as the internal standard. Mass spectra were obtained on a QTOF2 (Quadrupole Time of Fight) high-resolution mass spectrometer (Micromass, Wythenshawe, United Kingdom). Column chromatography was conducted using silica gel 60 (200 μm particle size, Yantai Xinde Chemical Co., Ltd, Yantai, China) and RP-18 (150-163 μm particle size, Merck, Darmstadt, Germany). For thin-layer chromatography (TLC), precoated TLC silica gel 60 F254 plates from Merck were used. HPLC (High Performance Liquid Chromatography) was carried out using a Shimadzu System LC-10AD pump equipped with a model SPD-10Avp UV detector (Shimadzu, Tokyo, Japan) and an Optima Pak C18 column (10 × 250 mm, 10 µm particle size, Shiseido Fine Chemicals, Tokyo, Japan).
Plant Material
The seeds of P. corylifolia were collected in Xishuangbanna, Yunnan province, Republic of China, and authenticated by Professor Jia Zhao (College of Pharmacy, Shenyang Pharmaceutical University). A voucher specimen of the plant (No. 20180515) was deposited at Liaoning University of Traditional Chinese Medicine, Shenyang, China.
Extraction and Isolation
The dried seeds of P. corylifolia (5.0 kg) were extracted 3 times with 95% aqueous EtOH at room temperature. The combined solutions were concentrated to obtain a crude extract (748.3 g), which was suspended in H2O and partitioned successively with light petroleum, EtOAc, and n-BuOH to afford petroleum, EtOAc, and n-BuOH soluble fractions, respectively. Part of the EtOAc-soluble fraction (100.0 g) was chromatographed over a silica gel column using a gradient of CH2Cl2-MeOH (from 0:1 to 0:1), and separated into 12 fractions (Fr.1-Fr.12). Fr.4 (7.3 g) was subjected to a silica gel column and eluted with CH2Cl2-MeOH (from 0:1 to 0:1) to obtain 10 fractions (Fr.4.1-Fr.4.10). Fr.4.4 (2.43 g) was chromatographed on a RP-18 column eluted with MeOH/H2O (2:8 to 10:0) to afford 16 fractions. Fr.4.4.5 (182.1 mg) was then separated by HPLC, using a gradient solvent system of 40% to 65% MeOH in H2O over 70 minutes, yielding compounds
(2Z)-2-[(4′-Hydroxyphenyl)methylene]-6-Methoxy-7-Prenyl-3(2H)-Benzofurane (1)
Light yellow amorphous powder; UV (MeOH) λ max (nm) (log ε): 260 (3.92), 402 (4.34); IR (KBr) ν max3424, 1768, 1654, 1611 cm−1; 1H (500 MHz) and 13C NMR (125 MHz) data in acetone-d 6, see Table 1; HREIMS m/z 336.1358 [M]+ (calcd for C21H20O5, 336.13621).
Cytotoxicity Screening Assays
The MTT (Methyl Thiazolyl Tetrazolium) assay was modified from that previously described. 15 An adherent SW620 cell line was cultured in RPMI (Roswell Park Memorial Institute) medium 1640 as adherent monolayers in flasks supplemented with 10% fetal bovine serum, 2 mM l-glutamine, 100 U/mL penicillin, and 100 µg/mL streptomycin in a humidified 37°C incubator supplied with 5% CO2. Briefly, cells were harvested with trypsin and dispensed into 96-well microtiter assay plates at 2000 cells/well for the SW620 cell line and then incubated for 18 hours at 37°C with 5% CO2. The test compounds were dissolved in 1% DMSO (Dimethyl Sulfoxide) in PBS (Phosphate Buffer Saline) (v/v) and aliquots (10 µL) tested over a series of final concentrations ranging from 10 nM to 30 µM. Control wells were treated with 1% aqueous DMSO. After 48 hours incubation at 37°C with 5% CO2, an aliquot (20 µL) of MTT in PBS (5 mg/mL) was added to each well (final concentration of 0.5 mg/mL), and the microtiter plates incubated for further 4 hours at 37°C with 5% CO2. After this final incubation, the medium was aspirated and the precipitated formazan crystals dissolved in DMSO (100 µL/well). The absorbance of each well was measured at 580 nm with a liquid scintillation counter (ALOKA LSC-8000, Aloka, Inc., Tokyo, Japan). All experiments were performed in duplicate and 5-fluorouracil was used as a positive control.
Supplemental Material
Supplementary material - Supplemental material for Anticolon Cancer Potential of (2Z)-2-[(4′-Hydroxyphenyl) Methylene]-6-Methoxy-7-Prenyl-3(2H)-Benzofurane, a New Aurone From Psoralea corylifolia
Supplemental material, Supplementary material, for Anticolon Cancer Potential of (2Z)-2-[(4′-Hydroxyphenyl) Methylene]-6-Methoxy-7-Prenyl-3(2H)-Benzofurane, a New Aurone From Psoralea corylifolia by Ai-Hua Liu, Li-Hua Liu and Chun-Ying Liu in Natural Product Communications
Footnotes
Acknowledgment
We thank Dr Huangxiu Wang in the Research Center for Chemistry, Shenyang Pharmaceutical University.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
