Abstract
Mallotus japonicus has been evaluated for the treatment of dermatitis, inflammatory conditions, and cancer. Diterpenes, one of the major constituents of M. japonicus, possess various pharmacological effects. In this study, 2 known diterpenes, anomaluone (
Mallotus japonicus, which belongs to the Euphorbiaceae family, has been used for a long time as a traditional medicine for the treatment of various diseases, including gastropathy, scald, and cancer. 1 Several species of the genus Mallotus are rich in biologically active compounds, such as phloroglucinols, tannins, terpenoids, coumarins, benzopyrans, and chalcones. 2 -8 Phytochemical studies of this genus have shown that kauranes, which are produced through the mevalonate pathway and consist of a perhydrophenanthrene unit fused with a cyclopentane unit, 9 are an important group of tetracyclic diterpenes. 10 Ent-kaurane diterpene is one of 2 enantiomeric series that resulted from triterpene cyclization. Kaurane and ent-kaurane diterpenes differ in their inverted configurations of carbons C-5, C-9, and C-10. 9 They possess many different biological activities, including plant growth regulatory, antimicrobial, antiviral, anti-inflammatory, and antitumoral activities. 11,12 The broad spectrum of biological activities of kaurane diterpenes have motivated countless studies of structural modifications of the kaurane skeleton, aiming to obtain new bioactive substances, especially those with antitumor activities. 13,14 Evasion of apoptosis is one of the most common cellular malfunctions that can facilitate the development of various cancers. Cancer cells easily evade apoptosis because of defects in the programmed cell death signaling pathway. Therefore, induction of apoptosis in cancer cells is an effective cancer prevention strategy. 15 -18 Antitumoral activities have been reported for some ent-kaurane diterpenes; oridonin induced apoptosis in Jurkat cells via activation of caspase 3, 19 glaucocalyxin A induced apoptosis in HL-60 cells through mitochondrial membrane potential loss, 20 and ent-11α-hydroxy-16-kaurene-15-one induced apoptosis in HL-60 cells through caspase 8. 21 Ent-Kaurane diterpenes with strong apoptosis-inducing activity are expected to have potential as anti-cancer drugs.
In this study, 2 ent-kaurane diterpenes, anomaluone (
The EtOAc fraction from the roots of M. japonicus was subjected to repeated column chromatography (CC), RP C18-MPLC (medium-pressure liquid chromatography), and high-performance liquid chromatography (HPLC) to yield 7n known compounds. The structures of the isolated compounds were elucidated using 1-dimensional (1D) and 2D nuclear magnetic resonance (NMR) spectroscopy (1H, 13C, heteronuclear single quantum coherence, heteronuclear multiple bond correlation, correlation spectroscopy, and rotating frame overhauser effect spectroscopy [ROESY]). Comparison of the obtained spectroscopic data with those reported in the literature showed that compounds

(a) Strnctures of compounds 1 to 8 isolated from Mallotus japonicus roots. (b) Key COSY, HMBC, and ROESY correlations for compounds 6 and 7. COSY, correlation spectroscopy; HMBC, heteronuclear multiple bond correlation; ROESY, rotating frame Overhauser effect spectroscopy.
Compound
1H and 13C Nuclear Magnetic Resonance Data for Compounds 6 and 7 (Dimethyl Sulfoxide-D 6).

Compound
The antiproliferative potential of M. japonicus compounds and fractions was tested in a cell proliferation assay (CellTiter Glo) using a blood cancer-derived cell line (please see supplemental material S7). Of the isolated compounds, compound

Antiproliferative effects of Mallotus japonicus compounds 1 to 8 on Jurkat cells. Cell viability was assessed by the MTS assay at 48 and 72 hours post treatment. DMSO, dimethyl sulfoxide, ***p < 0.001 vs control group.
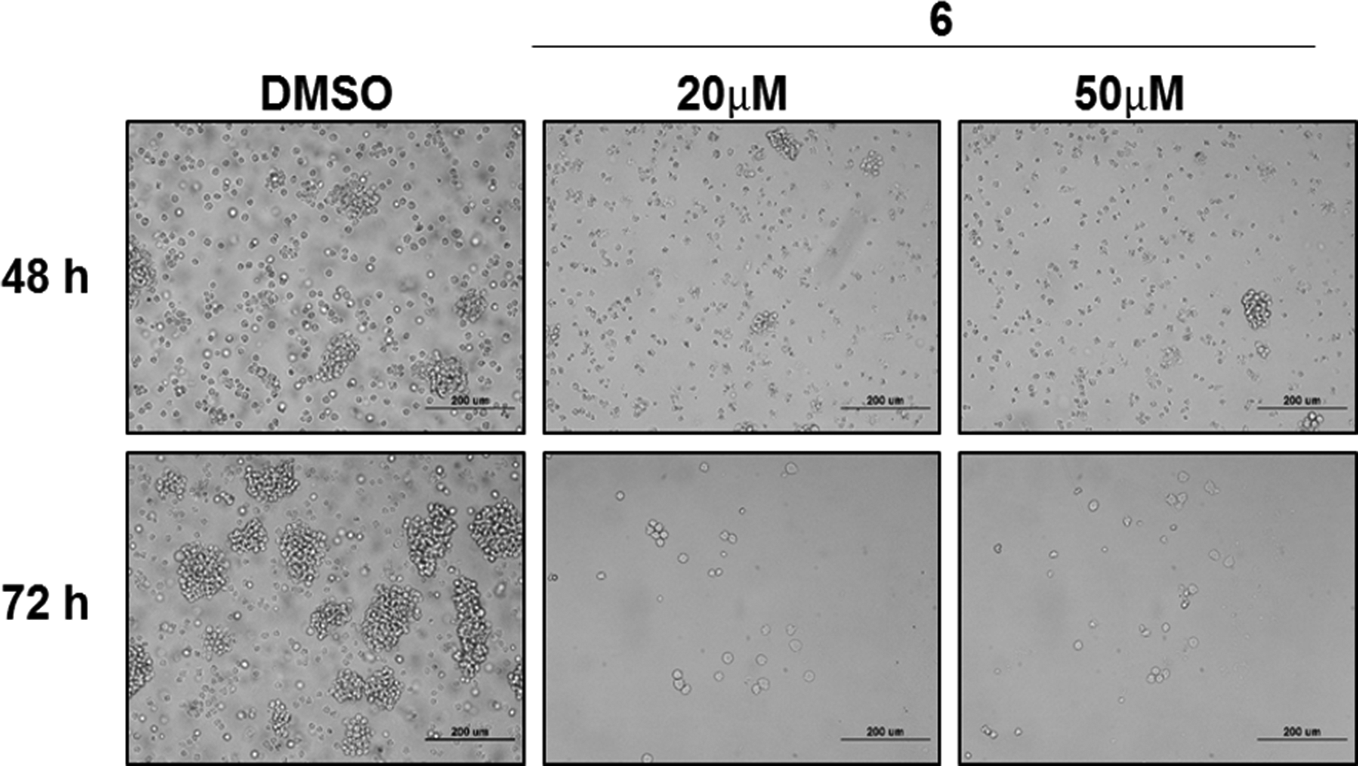
Morphological changes in Jurkat cells treated with compound 6. Cells treated with compound 6 (20 and 50 μM) show morphological changes that are characteristic of apoptosis at 48 and 72 hours post treatment. Phase contrast microscopy (400× magnification).
Compound
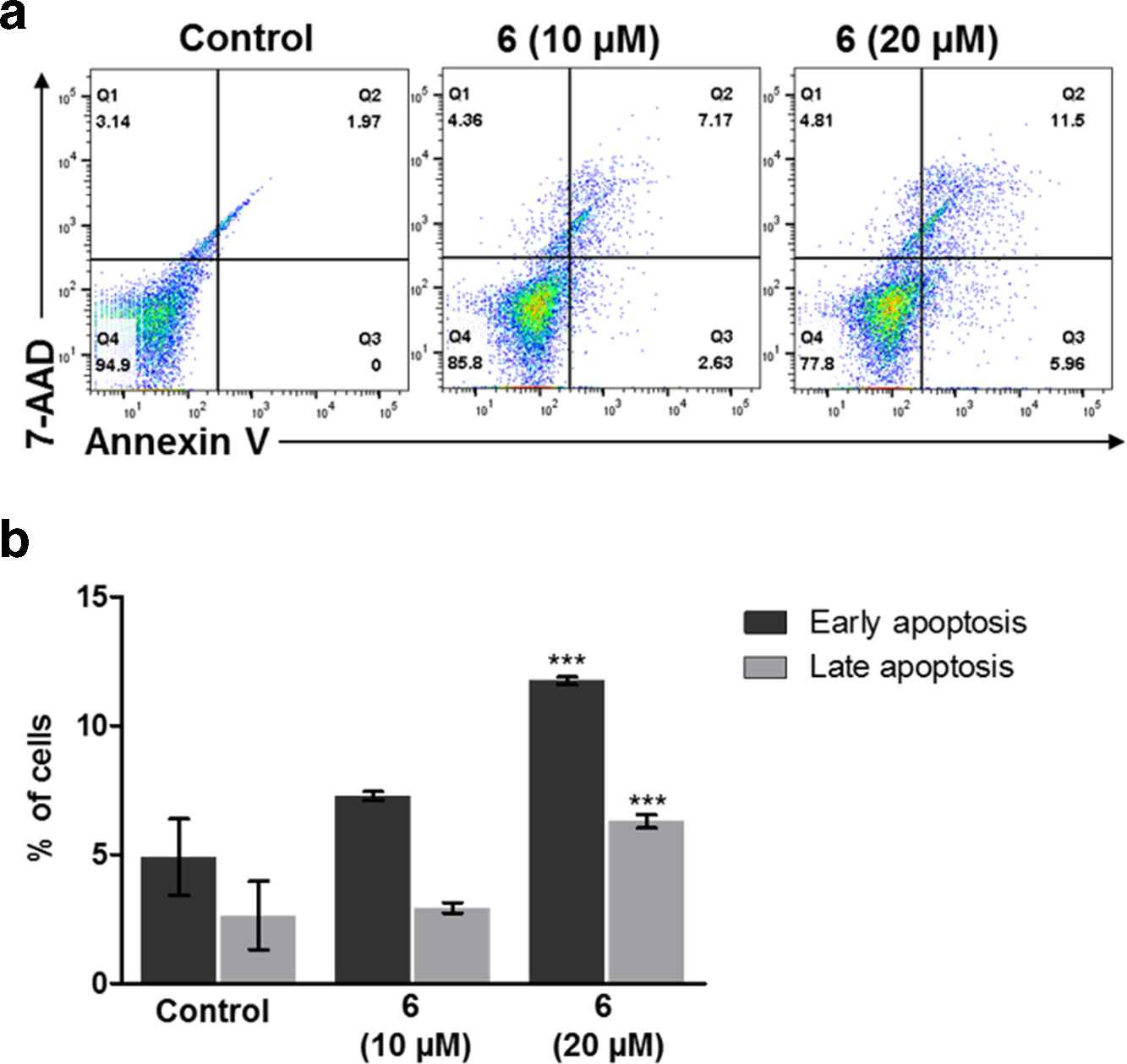
Compound
To determine whether the reduction in cell viability in Jurkat cells induced by compound
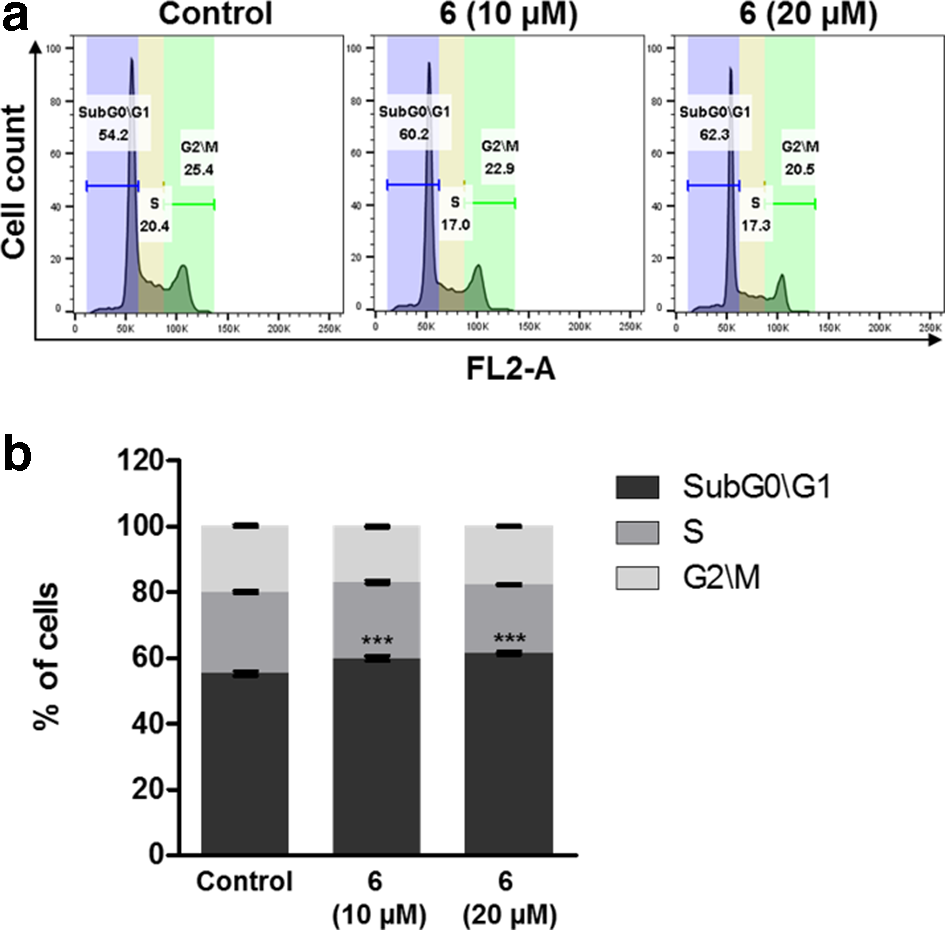
Compound
Next, the effects of compound

Compound
Cancer cells maintain ROS production by suppressing antioxidant generation.
33,34
Cancer cells exhibit a redox imbalance due to increased ROS levels compared to those in normal cells. ROS production induced by anticancer agents has been shown to be associated with promotion of apoptosis and cell-cycle arrest.
35,36
To examine how compound
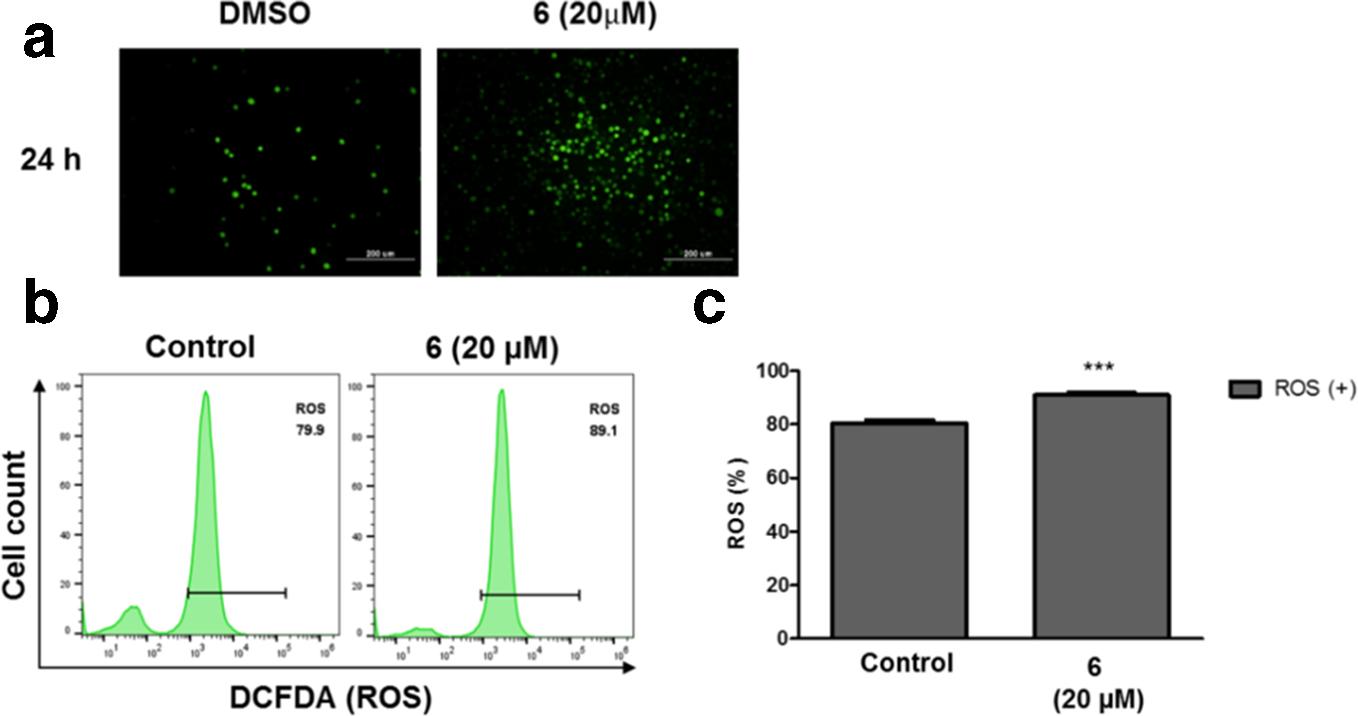
Compound
Experimental
General Experimental Procedures
Optical rotations were measured using a Perkin-Elmer model 343 plus polarimeter (JASCO Corp., Japan). UV spectra were recorded on a JASCO V-530 UV/VIS spectrometer (JASCO Corp., Japan). IR spectra were obtained using a JASCO FTIR 300-E spectrometer. NMR spectra were recorded by an AVANCE-400 NMR spectrometer (Bruker, Billerica, MA, USA). CC was performed using Kieselgel 60 silica gel (<0.063 mm; Merck, Germany) and octadecyl (C18) silica gel (Yamazen, Japan). Thin-layer chromatography (TLC) spots were visualized on Kieselgel 60 F254-coated normal-phase silica gel TLC plates and TLC silica gel 60 RP-18 F254 (Merck, Germany). All solvents were obtained from Daejung Chemicals & Metals Co., Ltd. (Siheung, Korea).
Plant Specimens and Reagents
Mallotus japonicus roots were collected in June 2018 from the Medicinal Plant Garden of Boseong, Jeollanam-do, Korea and were identified by H.S. Park (Medicinal Plant Garden of Boseong). A voucher specimen (CNU-0602) was deposited at the College of Pharmacy, Chonnam National University, South Korea.
Sample Extraction
The dried roots of M. japonicus (2.0 kg) were percolated with 100% MeOH at room temperature. Evaporating in vacuo yielded a residue (120 g) that was suspended in H2O and fractionated with EtOAc and n-butanol consecutively. The EtOAc extract (30 g) was subjected to CC using silica gel and eluted with a gradient of CH2Cl2:MeOH (100:1-1:100, v/v), which yielded 6 fractions (E1-E6). Fraction E2 (2.1 g) was subjected to RP C18-MPLC (10% MeOH to 100% MeOH), which yielded 6 subfractions (M1-M6). Compound
Cell Culture
Jurkat cells were obtained from the American Type Culture Collection (ATCC). Cell-culture medium was prepared as recommended by ATCC. The cells were cultured in a humidified atmosphere of 5% CO2 at 37°C and subcultured (1:5) when the cell density reached 80%-90%, which was every 3 or 4 days.
Antiproliferation Assay
The antiproliferative effect of the M. japonicus compounds on the cells was measured using the CellTiter 96 AQueous One solution Cell Proliferation Assay Kit (Promega, Madison, WI, USA). Cells were seeded at a density of 5 × 105 cells/well in RPMI 1640 (100 µL) and cultured for 24 hours. Then, the cells were treated with different concentrations of each compound (10, 25, and 50 µM). DMSO-treated cells were included as a control. To estimate cell viability (%) at 48 hours post treatment, the cells were incubated with 20 µL of CellTiter 96 AQueous One solution Cell Proliferation Assay reagent for 2 hours at 37°C. Then, the optical density at 490 nm was measured using a microplate reader (Synergy HTX, BIO-TEK Instruments, Inc.).
Microscopy
To detect morphological changes, compound-treated Jurkat cells were examined under a phase contrast microscope (Olympus). Photomicrographs of the cells (400× magnification) were taken at 48 and 72 hours post compound treatment and analyzed for changes in shape, size, and number.
Cytotoxicity Assay
To explore the mechanism underlying compound-induced cytotoxicity in Jurkat cells, we investigated the changes in cell-cycle progression and apoptosis using flow cytometry.
Apoptosis Analysis
Compound-treated Jurkat cells were visualized by flow cytometry using the PE Annexin V Apoptosis Detection Kit with 7-AAD (BioLegend). Annexin V binds to phosphatidylserine (PS) when PS flips from the inner leaflet to the outer leaflet of the plasma membrane during early apoptosis. In contrast, PI stains DNA when the cell membrane is disrupted during late apoptosis. To distinguish PE fluorescence from PI fluorescence and thus discriminate between early (Annexin V+) and late (7-AAD+) apoptotic cell populations, the FL-2 and FL-3 channels were selected to measure the PE and 7-AAD fluorescence, respectively. Compound-treated and untreated (control) Jurkat cells were seeded in 6-well plates and incubated at 37°C for 48 hours. The cells were harvested, washed with phosphate-buffered saline (PBS), and resuspended in binding buffer (1×). The cells were stained with annexin V-PE and 7-AAD, according to the manufacturer’s instructions. After staining, the cells were categorized as early (Annexin V+/7-AAD-) or late (Annexin V+/7-AAD+) apoptotic cells using a flow cytometer (BD FACSVerse, BD Biosciences), and the cell percentages were determined using FlowJo software (BD Biosciences).
Tetramethylrhodamine Methyl Ester Perchlorate (TMRM) Assay
Compound-treated Jurkat cells were harvested at 48 hours post-treatment, washed with PBS, and incubated with the cell-permeable fluorescent indicator TMRM (100 nM; Thermo Fisher Scientific), which was prepared by diluting the 10 mM stock in PBS, for 30 minutes at 37°C. To assess mitochondrial membrane potential, the cells were washed, resuspended in PBS, and analyzed with a flow cytometer (BD FACSVerse) and FlowJo v10 software.
Cell-Cycle Analysis
Univariate analysis of the cellular DNA content of compound-treated cells enables the detection of cells in different cell-cycle phases (Sub-G1/G1 vs. S vs. G2/M) and the detection of apoptotic cells with fractional DNA content. Compound-treated Jurkat cells were seeded in 6-well plates and incubated for 48 hours. Then, the cells were harvested, washed with PBS, and fixed with ice-cold 70% ethanol for 3 hours at 4°C. For the cell-cycle analysis, the cells were resuspended in PBS containing RNase A (5 µg/mL) and PI (50 µg/mL) and then evaluated using a flow cytometer (BD FACSVerse,). The percentages of cells in G0/G1, S, and G2/M phases were analyzed using FlowJo software.
Measuring ROS
Intracellular ROS levels were detected using 2′,7′-dichlorodihydrofluorescein diacetate acetyl ester (H2DCFDA) (Thermo Fisher Scientific). Cells were continuously perfused with PBS buffer (37°C) and imaged using an inverted fluorescent microscope (Olympus). For this analysis, the cells were incubated with DCFDA (1 µM) for 30 minutes at room temperature, washed with cold PBS, and then resuspended in PBS supplemented with 1% fetal bovine serum. Intracellular fluorescence accumulation was analyzed using a flow cytometer (BD FACSVerse).
Statistical Analyses
Statistical analyses were performed using GraphPad Prism 5 (GraphPad Software, Inc.), and the data are presented as the mean ± SD. The results were analyzed by 1-way analysis of variance and t-test. P values less than 0.05 were considered statistically significant.
Anomaluone (6)
White powder;
16-epiabbeokutone (7)
White powder;
Supplemental Material
Supplementary Material - Supplemental material for An Antiproliferative ent-Kaurane Diterpene Isolated from the Roots of Mallotus japonicus Induced Apoptosis in Leukemic Cells
Supplemental material, Supplementary Material, for An Antiproliferative ent-Kaurane Diterpene Isolated from the Roots of Mallotus japonicus Induced Apoptosis in Leukemic Cells by Joo-Eun Lee, Nguyen Thi Thanh Thuy, Youngju Lee, Namki Cho and Hee Min Yoo in Natural Product Communications
Footnotes
Acknowledgment
The authors are grateful to the Center for Research Facilities at the Chonnam National University for their assistance in the analysis of the organic structure (FT-IR, FT-NMR, HRTOFMS).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Korea Research Institute of Standards and Science for the “Development of New Chemical Medical Measurement Standard Technology” [grant number KRISS-2019-GP2019-0009]; the National Research Foundation of Korea (NRF) [grant number NRF-2018R1C1B5083127]; and Chonnam National University [grant number 2018-3487].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
