Abstract
Oil-soluble organosulfur compounds in garlic are known for the anticancer effect. However, there are limited experimental studies to describe the effect of S-allylcysteine (SAC), a main water-soluble derivative of garlic, in carcinogenesis. This study investigates the prevention function of SAC on carcinogen benzo(a)pyrene (B(a)P)-induced precancerous activity in human lung cells (A549). A549 cells were either pretreated (PreTM) or concurrently treated (CoTM) with 1 μM B(a)P and either 10 or 50 μM SAC. The 50 μM CoTM group inhibited B(a)P-induced cell proliferation by approximately 100%. The 50 μM SAC CoTM and PreTM inhibited the B(a)P-induced G2/M phase shift by 119% and 100%, respectively. Furthermore, the SAC PreTM exhibited the potential to reduce the generation of reactive oxygen species (ROS) in cells relative to the B(a)P group by approximately 100%. The CoTM and PreTM elevated superoxide dismutase (SOD) by at least 70% compared with B(a)P group. In this study, we demonstrated that the mechanisms involved in the inhibitory role of SAC in B(a)P-induced carcinogenesis, including suppression of cell proliferation and DNA damage, cell cycle regulation, attenuation of ROS formation, increase of SOD activity, and inhibition of nuclear factor-kappa B (NF-κB) activity, which indicated that SAC is potentially a novel therapeutic candidate for the prevention and treatment of B(a)P-induced human lung cancer.
Cigarette smoke (CS) is a mixture of more than 4000 chemicals that include significant amounts of free radicals, particles, reactive chemicals, and gases, producing an overwhelming oxidant burden on the lung 1 and second-hand smoke as a predictor of smoking cessation among lung cancer survivors. 2 Benzo(a)pyrene (B(a)P) is well known as a polycyclic aromatic hydrocarbon and potent carcinogen in CS. 3 Emerging evidences indicate that exposure to B(a)P could promote invasive and metastatic behaviors of human lung cells, 4 which are resulting from its carcinogenic potential, including oxidative damage from increasing reactive oxygen species (ROS). 5
Garlic (Allium sativum L.) has been employed as a medical herb for centuries. 6,7 and widely studied regarding its antiviral, 8 antioxidant, 9 anti-inflammatory, 10 antifungal, 11 cardiovascular, 12 and anticancer effects, 13,14 which can result from oxidative stress. 15 Oxidative stress is defined as an imbalance between the production of ROS and their elimination by antioxidants. Individual organosulfur compounds (OSCs) in garlic have been studied to identify the mechanisms of their anticarcinogenic activity, especially for oil-soluble compounds such as diallyl sulfide, diallyl disulfide, and diallyl trisulfide . 16,17
In vitro studies have shown that the antioxidant effect of aged garlic extract may be due to its modulation of the glutathione (GSH) redox cycle and superoxide dismutase (SOD) activity in vascular endothelial cells. 18 Moreover, aged black garlic extract has been found with strong antioxidant capacity compared with fresh raw garlic. 19 Studies also showed that the major water-soluble fraction, S-allylcysteine (SAC), can suppress cancer risk and alter the biological behaviors of various types of human tumors. 20 In vivo studies have demonstrated the protective effect of aqueous garlic extract against naphthalene-induced oxidative stress in mice. 21 Thus, these findings imply that SAC may also have chemopreventive potential in inhibiting carcinogenesis initiation, but the mechanism is still unclear in human lung cancer cells.
In this study, we choose 2 potentially physiological doses of SAC to evaluate the efficacy in B(a)P-induced precancerous carcinogenesis in A549 cells. A549 cells are a continuous tumor-cell line derived from a human lung carcinoma with the properties of human type II alveolar epithelial cells. 22,23 We choose B(a)P-induced precancerous carcinogenesis in A549 cells as an in vitro model. 9,24,25 Cell proliferation, cell cycle, and signaling pathway was examined to explore the mechanism of SAC in carcinogenesis.
Materials
S-Allylcysteine (purity of 99%) was synthesized and purified by recrystallization in our laboratory with a modified procedure as previously reported. 26 Benzo(a)pyrene was purchased from Tokyo Chemical Industry (Tokyo, Japan). Propidium iodide, 4,6-diamidino-2-phenylindole (DAPI), and sulforhodamine B (SRB) were purchased from Sigma-Aldrich Chemical Co. (St. Louis, MO, United States). The alpha inhibitor of NF-κB (IκB-α) polyclonal antibodies and the RELA polyclonal antibodies (p65) were purchased from Proteintech Biotechnology (Rocky Hill, United States). Fluorescein (FITC)-conjugated AffiniPure Goat Anti-Rabbit IgG (H + L) was provided by Proteintech Biotechnology (Rocky Hill, United States). Dichlorofluorescein diacetate (DCFH-DA) was purchased from Invitrogen Biotechnology (California, United States). Fetal bovine serums (FBS) was provided by Sijiqing Biotechnology Corp. (Hangzhou, China). Roswell Park Memorial Institute (RPMI)-1640 was obtained from Gibco (Grand Island, NY, United States). Penicillin and streptomycin were provided by Solarbio Biotechnology Corp. (Beijing, China). Radioimmunoprecipitation assay (RIPA) lysis buffer were provided by the Beyotime Institute of Biotechnology (Beijing, China).
Cell Culture and Cell Viability Assay
A549 cells (provided by the Committee of Type Culture Collection of Chinese Academy of Sciences) were cultured in RPMI-1640 with 10% FBS, penicillin (100 U/mL), and streptomycin (100 µg/mL) at 37°C in 5%-CO2 incubator. A stock solution of SAC (5 mM) was prepared fresh in phosphate buffer saline (PBS). Benzo(a)pyrene was dissolved in dimethyl sulfoxide (DMSO) as a 4 mM stock solution and dilutions were made in RPMI-1640. The total DMSO concentrations were kept below 0.05% (v/v), which showed no influence on cell growth. Cells were seeded in 96-well plates at a concentration of 1 × 104 cells/well. After 24 hours, cells were categorized into 3 groups: B(a)P only, SAC pretreatment (PreTM), and SAC concurrent treatment (CoTM). The PreTM group was treated with 10 and 50 µM of SAC for 4 hours, followed by 1 µM of B(a)P. The CoTM group was treated with 1 µM of B(a)P in combination with 10 or 50 µM of SAC. The cell viability is determined at 3, 6, 12, and 24 hours after SAC and B(a)P treatment by using SRB assay. 27
DNA Ladder Assay
Cells were categorized into 4 groups: B(a)P only, SAC only, PreTM, and CoTM. The PreTM group was treated with 50 µM of SAC for 4 hours, followed by 1 µM of B(a)P. The CoTM group was treated with 1 µM of B(a)P in combination with 50 µM of SAC. At 6 and 24 hours after SAC and B(a)P treatment, isolation of cellular DNA was performed following the protocol provided by the manufacturer, using the DNA Isolation Kit (Tiangen Biotech Co. Ltd, Beijing, China). The isolated DNAs were subjected to electrophoresis in 1% agarose gel with the logic DNA Marker (Tiangen Biotech Co. Ltd, Beijing, China).
Cell Cycle Distribution Analysis
Cells were seeded in 6-well plates at a concentration of 1 × 105/mL per well and cultured for 24 hours. After treatment by chemicals for 24 hours, as described above, cells were collected and washed with PBS. Analysis of cell cycle distribution was performed using a modification of a previously described method. 28 The cells were analyzed by a Beckman Coulter model FC500 flow cytometer (Brea, CA, United States). Data were analyzed by using MODFIT and CELLQUEST software (Verity Software House, Topsham, Maine, United States).
Reactive Oxygen Species Determination
After treatment as described above, the cells were incubated with 10 µM DCFH-DA at 37°C for 20 minutes in the dark. After harvested and suspended in PBS, cells were immediately analyzed and illuminated with 488 and 518 nm laser.
The Determination of SOD and GSH-Px Activity
A549 cells were seeded in 6-well plates at a density of 10 × 104/mL and cultured overnight to achieve 80% confluence. After B(a)P only, SAC pretreatment, and concurrent treatment, cells were harvested resuspended in PBS without Mg2+ or Ca2+. After frozen-thaw 1 to 2 times, the cell suspension was ultrasonicated. Superoxide dismutase and glutathione peroxidase (GSH-Px) activity were measured using commercial kits (Jiancheng Bioengineering Co. Ltd, Nanjing, China). Total protein contents in PBS were measured using a BCA protein reagent (Beyotime Institute of Biotechnology, China) according to the manufacturer's instructions.
Western Blot Analysis
After B(a)P and SAC only, SAC pretreatment, and concurrent treatment, the cells were harvested and washed twice in PBS at 4°C. Total cell lysates were homogenized in cell lysis buffer, containing 50 mM Tris (pH 7.4), 1 mM ethylenediamine tetraacetic acid (EDTA), 150 mM NaCl, 1 µM phenymethylsulfonyl fluoride, 5 µg/mL leupeptin, 5 µg/mL aprotinin, 1% sodium deoxycholate, 1% Triton X-100, and 0.1% sodium dodecyl sulfate (SDS). Equal amounts of the resulting supernatant (30-50 μg) were separated electrophoretically using SDS-polyacrylamide gel electrophoresis. The gel was then transferred to 0.45 µm polyvinylidene fluoride membranes (Millipore, Bedford, MA, United States). Membranes were soaked in blocking buffer (5% nonfat milk) and probed using the primary antibodies, with subsequent horseradish peroxidase conjugated antibodies. Immune complexes were then determined.
Immunofluorescence Analysis
The treated cells were washed twice in PBS, fixed with acetone and methyl alcohol (1:1, v/v), and blocked with 5% bull serum albumin (BSA). Cells were then sequentially incubated with anti-NF-κB p65 antibody, FITC-conjugated sheep anti-rabbit IgG, and DAPI and examined for NF-κB translocation using a confocal laser scanning microscope (LSM 700, Carl Zeiss, Germany).
Statistical Analysis
Data were done by triplicate independently. P < 0.05 was considered statistically significant. Results are expressed as the mean ± SD. The Prism’s t-test between experimental groups was used to determine significance.
S-Allylcysteine Suppresses B(a)P-Stimulated Cell Proliferation
The SRB assay was used to evaluate cell viability after treatment with B(a)P and SAC. Optimal treatment conditions were first established. Benzo(a)pyrene was shown to significantly (P < 0.05) increase A549 cell proliferation at 3 and 6 hours, and sustained a slight increase in cell viability at 12 and 24 hours, compared with control. The 50 µM SAC CoTM inhibited B(a)P-induced cell proliferation at 6, 12, and 24 hours by an average of 89%, 106%, and 176%, respectively. The 50 µM PreTM concentration of SAC inhibited B(a)P-induced increases in cell proliferation at 3, 6, and 24 hours by an average of 70%, 104%, and 266%, respectively (Figure 1(b-e)). Thus, SAC can have the potential to inhibit cell proliferation.
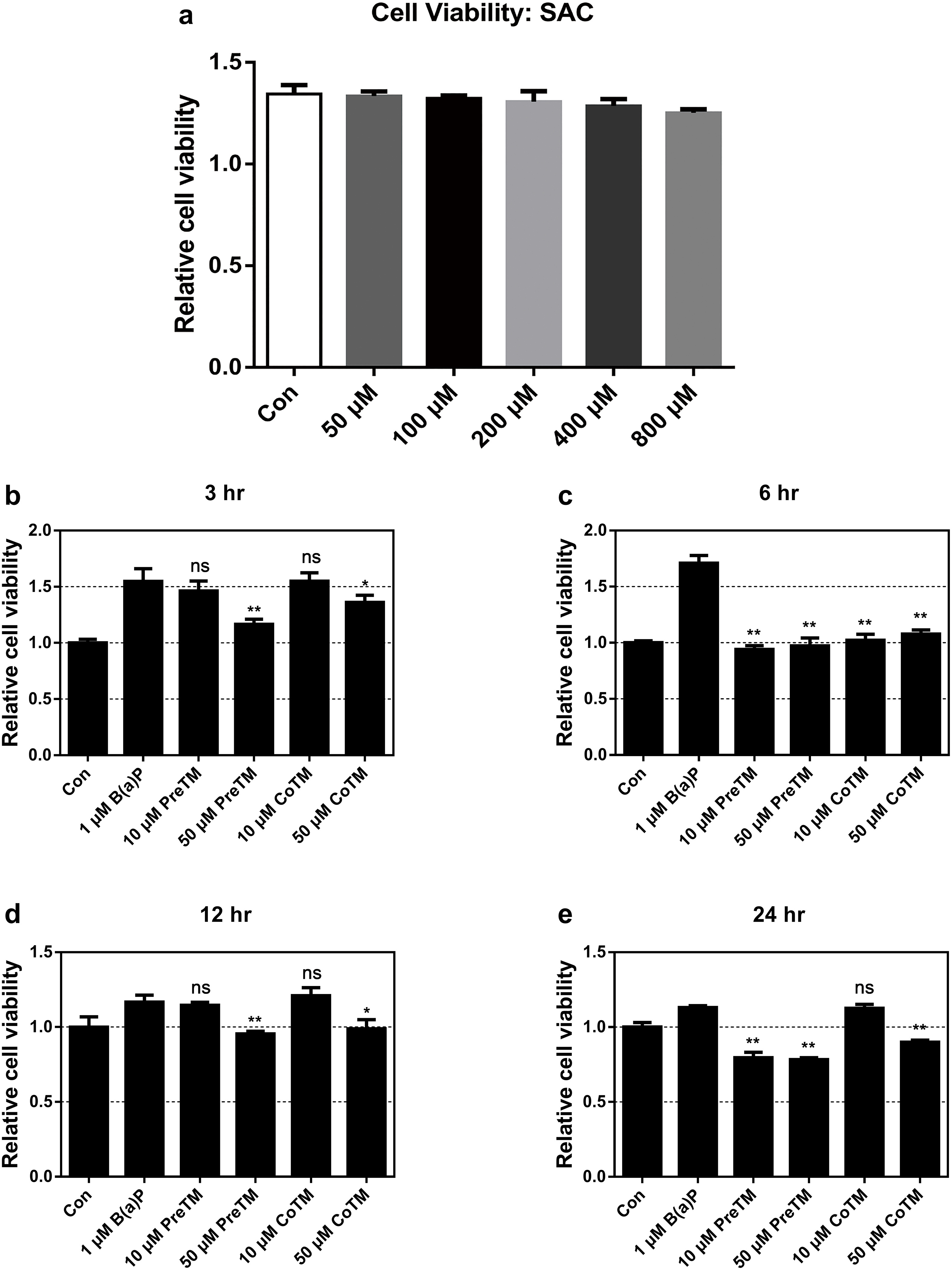
Effects of S-allylcysteine on cell viability in A549 cells (a). Cell viability and proliferation resulting from concurrently treated and pretreated with S-allylcysteine and benzo(a)pyrene. A549 cells were treated with S-allylcysteine and 1 µM benzo(a)pyrene at 3 (b), 6 (c), 12 (d), and 24 hours (e). The results were expressed as a percentage of the control (untreated) culture. The data are expressed as mean ± SD (n = 3) from experiments of 4 replicates (*P < 0.05 and **P < 0.01 when compared with the benzo(a)pyrene only treated group).
S-Allylcysteine Suppresses B(a)P-Stimulated DNA Damage
The results revealed that B(a)P causes significant DNA damage compared with control in A549 cells at 6 hours and the damage was recovered at 24 hours. 29 However, the 50 µM SAC treatment reduced B(a)P-induced DNA damage at 6 hours (Figure 2). As a result, SAC showed the potential to protect the DNA damage from B(a)P in A549 cells.

S-Allylcysteine inhibition of B(a)P-induced DNA damage. A549 cells were either cotreated with S-allylcysteine and benzo(a)pyrene (1 µM) or treated with S-allylcysteine 4 hours before benzo(a)pyrene treatment for 6 and 24 hours.
Effects of SAC on Cell Cycle
Benzo(a)pyrene induces G2/M and S phase arrest in breast cancer cells. 30 To examine the mechanism, cell cycle distribution was evaluated using flow cytometry. Benzo(a)pyrene increased G2/M phase shift of the cells from 4% to 10%. S-Allylcysteine (10 and 50 µM) inhibited the B(a)P-induced G2/M phase shift by 129% and 119% (CoTM), and 113% and 100% (PreTM), respectively (Figure 3). As a result, SAC showed the potential to counteract B(a)P-induced G2/M shifts.

Cell cycle distribution of A549 cells treated with S-allylcysteine and benzo(a)pyrene. A549 cells were either cotreated with S-allylcysteine and benzo(a)pyrene (1 µM) for 24 hours (a) or treated with S-allylcysteine 4 hours before benzo(a)pyrene treatment for 24 hours (b). The results are represented as mean ± SD.
S-Allylcysteine Suppresses B(a)P-Stimulated Production of ROS
Figure 4 shows the ability of the samples to generate ROS assessed by the DCFH-DA assay in A549 cells. Treatment with 1 µM B(a)P significantly increased ROS levels by 1.6-fold over the control (P < 0.05). Pretreated group treated with 10 or 50 µM SAC attenuated ROS levels by 89% and 108%, respectively, compared with the B(a)P group. As a result, SAC may inhibit B(a)P-induced production of ROS.

Reactive oxygen species determination resulting from concurrently treated and pretreated at 24 hours after S-allylcysteine and benzo(a)pyrene treatment. The graphs represent the average relative cell viability for 3 replicates. **P < 0.01, indicating a significant difference from the benzo(a)pyrene only treated group.
S-Allylcysteine Suppresses B(a)P-Stimulated SOD Activities
Superoxide dismutase concentrations (U/mg protein) in cell lysates are presented in Figure 5. Statistically significant variations in SOD activities were found in cells exposed to B(a)P vs control. Benzo(a)pyrene decreased SOD activities by 29% relative to control. While the decrease was significantly inhibited by using 50 µM SAC by 94% (PreTM) and 111% (CoTM), respectively.

Effects of S-allylcysteine on benzo(a)pyrene-induced superoxide dismutase activities from pretreated and concurrently treated at 24 hours after S-allylcysteine and benzo(a)pyrene treatment. Values are the means ± SD for n = 3 (*P < 0.05 and **P < 0.01 when compared with the benzo(a)pyrene only treated group).
Effects of Chemicals on GSH-Px Activity
The activities of GSH-Px could not be detected in all groups, suggesting that GSH-Px activities were under the detection limit (0.1 U/mL) in A549 cells. Our results were consistent with the previous research. 31
S-Allylcysteine Suppresses B(a)P-Stimulated NF-κB Activation
The effect of SAC on NF-κB activation was examined using Western blot. As shown in Figure 6, Western blot assay results demonstrate that stimulation of cells with B(a)P decreased the expression of IκBα. At all concentrations, SAC treatment significantly inhibited the degradation of IκBα. Similar results were found using immunofluorescence microscopy, showing that NF-κB p65 translocated from the cytosol to the nucleus after B(a)P treatment. S-Allylcysteine inhibited the translocation to some extent (Figure 7). These results suggest that the PreTM and CoTM groups of SAC inhibited NF-κB activation in B(a)P-induced A549 cells by the suppression of IκB degradation and nuclear translocation of NF-κB.
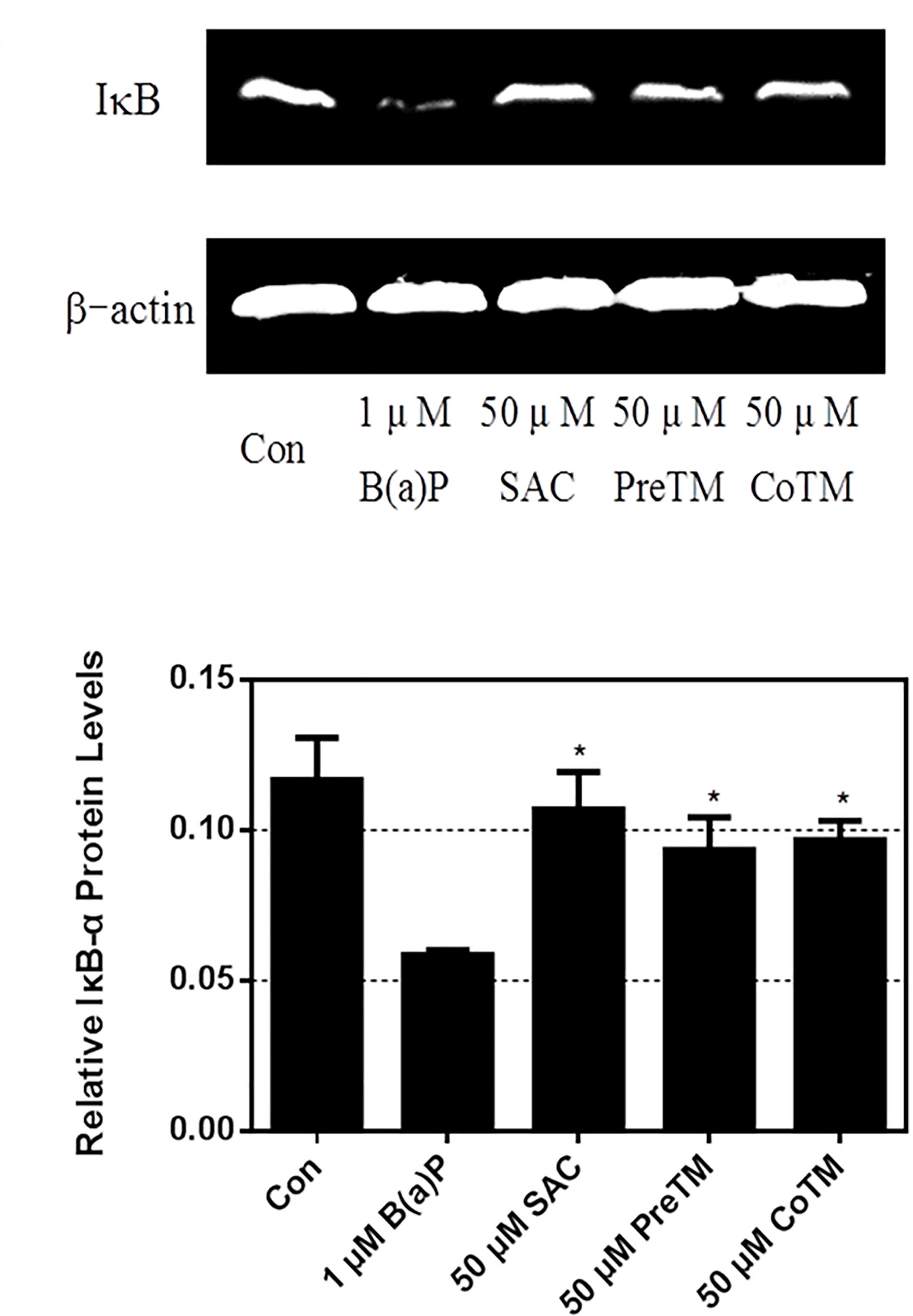
Effects of S-allylcysteine on nuclear factor-kappa B activity in benzo(a)pyrene-induced A549 cells. A549 cells were pretreated with 50 µM S-allylcysteine for 4 hours before benzo(a)pyrene treatment and treated with 50 µM S-allylcysteine concurrent with 1 µM benzo(a)pyrene. After 24 hours, cells were harvested for 24 hours and the IκB was investigated with the β-actin antibody serving as a loading control.

Effects of S-allylcysteine on nuclear factor-kappa B activity in benzo(a)pyrene-induced A549 cells. A549 cells were pretreated with 50 µM S-allylcysteine for 4 hours before 1 µM benzo(a)pyrene treatment and harvested after 24 hours. After immunofluorescence staining with nuclear factor-kappa B p65 antibody (green), cells were stained with 4,6-diamidino-2-phenylindole for visualization of nuclei (blue). Results are represented by triplicate independently. Bar, 15 µm.
In this study, we firstly reported that SAC Pre- and CoTM inhibited B(a)P-induced cellular proliferation, G2/M transition, and production of ROS in A549 cells. These effects were accompanied by the increase of SOD activities, possibly by suppression of NF-κB nuclear translocation.
Cell proliferation may not significantly increased following a single exposure of B(a)P for 24 hours in normal and cancerous cell lines. 30 Our study showed similar results, but promotion of cell proliferation by B(a)P was significant at 3 and 6 hours, which were not evaluated in other studies. Garlic OSCs such as SAC have antiproliferative effects in cancer cells 32 and suppress the growth of a human androgen-independent prostate cancer xenograft, CWR22R. 33 Cell viability was not significantly decreased by the concentrations of SAC evaluated. However, SAC inhibited B(a)P-induced A549 cell proliferation at 6 hours both in PreTM and CoTM group compared with control. Interestingly, B(a)P induced DNA damage at 6 hours, which was reversed by SAC Pre- and CoTM. This result may indicate SAC has an inhibitory role in B(a)P-induced carcinogenesis.
The cell cycle change was investigated further. The 50 µM doses of SAC Pre- and CoTM were the most effective counteract B(a)P-induced G2/M shifts. A549 cells exhibited decreased cell numbers in the G1 phase and a corresponding accumulation of cells in S phase after treated by SAC Pre- and CoTM, which could result from the protective mechanisms employed by SAC against B(a)P-induced shift to G2/M and S phases.
Reactive oxygen species are radicals, ions, or molecules that can initiate the cellular injury, which may lead to aging and disease. 34 The harmful activity of a small percentage of ROS is inhibited by the natural antioxidants occurring in the cell, which may be divided into enzymatic (SOD and GSH-Px) and nonenzymatic (vitamins E, C, and A) groups. 35 When, however, the amount of free radical increases due to smoking, the pool of antioxidants is saturated and the excess of free radicals damages biological structures. High levels of ROS have been detected in almost all cancer cells. Benzo(a)pyrene and its cellular metabolite anti-benzo(a)pyrene-7,8-diol-9,10-epoxide can induce DNA damage through increasing ROS levels, initiating oxidative damage to nucleic acids and proteins. In this work, we found that B(a)P significantly increased ROS levels compared to the control. Meanwhile, cellular levels of SOD were also decreased. Fortunately, SAC can utilize both enzymatic and nonenzymatic endogenous antioxidant defenses to minimize cell injury. 36 As shown in our experiments, SAC treatment diminished ROS production and repressed the decrease of SOD, which were induced by B(a)P. Thus, regulation of ROS and SOD may have great potential for the prevention and treatment of B(a)P-associated diseases.
While NF-κB has been shown to involve in suppressing the accumulation of ROS and regulating Mn-SOD expression, 37,38 the role of NF-κB was further discussed in our study. S-Allylcysteine Pre- and CoTM reduced the degradation of IκBα in the cytoplasm with B(a)P treatment. The NF-κB factor p65 translocated from the cytosol to the nucleus after the B(a)P treatment, which was markedly inhibited by SAC. These results suggested that the inhibitory effects of SAC on production of free radicals induced by B(a)P were at least partially mediated by suppression of NF-κB signaling.
Taken together, our data firstly provided the evidence that SAC had a beneficial effect in the inhibition of B(a)P-induced carcinogenesis in A549 cells. The mechanisms of SAC inhibition of precancerous activities of B(a)P were found to include inhibition of cell proliferation and DNA damage, attenuation of G2/M shifts, suppression of ROS production, and increase of SOD activities. These effects were related at least part of NF-κB signaling pathway, but additional mechanisms remain to be elucidated (Figure 8). These studies suggested that SAC or related compounds 39 could be an effective approach to chemoprevention and therapy of smoke-induced carcinogenesis.
The mechanisms of S-allylcysteine inhibition of benzo(a)pyrene-induced precancerous carcinogenesis
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for this work was provided by funds from Opening Project of Engineering Laboratory of Chemical Resources Utilization in South Xinjiang of Xinjiang Production and Construction Corps (CRUYB1803), Key Research and Development Program of Shandong Province (2018CXGC1411), Shandong Key Development Project (Grant # 2018GSF118205), and Programs Foundation of University of Jinan (160100204).
