Abstract
A new manzamenone derivative (
The marine sponges of the genus Plakortis have been recognized as a rich source of oxygenated fatty acids such as plakortin, 1 cyclic peroxides, 2,3 untenone, 4 plakoridine A, 5 untenolide A, 6 and manzamenones. 7 -11 Manzamenones constitute a family of natural dimeric 7 -10 and trimeric 11 fatty acid derivatives. Especially, it has been proposed that 3,6-dioxo-4-docosenoic acid might be a universal key intermediate of either the manzamenones or untenone A, and furthermore, all of these molecules are likely to be biosynthetically related to one another. 8 As a consequence, the manzamenones feature saturated long alkyl chains and ester groups around a cyclopentenone core. From the biological activity viewpoint, several Plakortis-derived manzamenones exhibit a broad spectrum of bioactivity. 10 -12
As part of our ongoing efforts to identify bioactive metabolites from marine sponges,
13
we collected the sponge Plakortis from Weno Island, Chuuk State, Federated States of Micronesia. The crude organic extract of this marine sponge exhibited considerable antioxidant activity. Stimulated by the fact, we initiated an investigation on this organism. Fractions of interest were selected on the basis of the 1H nuclear magnetic resonance (NMR) analysis of the crude extracts obtained from solvent-partitioning. Subsequently, an integrated separation process for isolation and purification of compounds was executed by different chromatographic methods including vacuum flash chromatography on silica and normal-phase high-performance liquid chromatography (HPLC). In consequence, the separation process yielded 5 compounds, which were later identified by combined spectroscopic analyses to be manzamenone A (
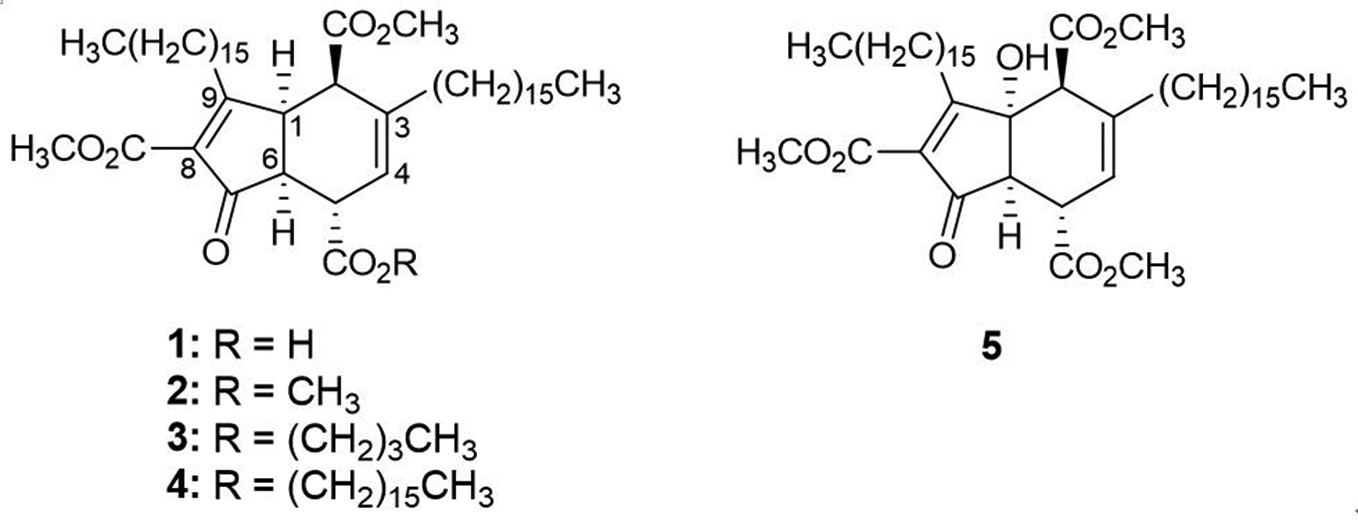
Structures of compounds
Compound
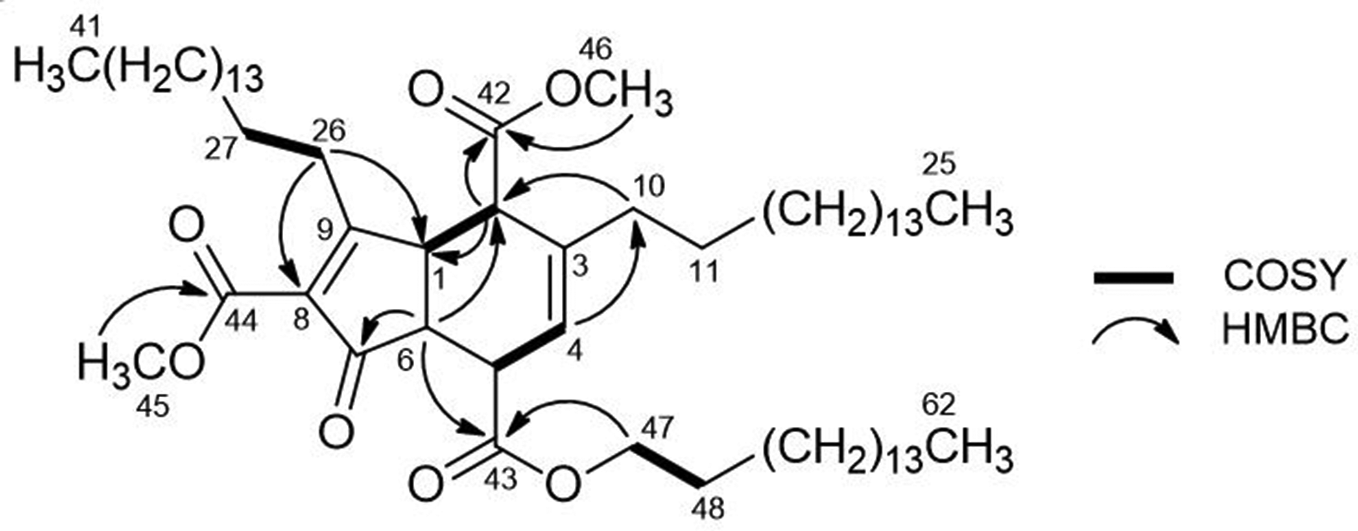
Selected 2-dimensional correlations of compound 4.
The isolation of natural products from the sponge Plakortis yielded a considerable quantity of 43-O-methylmanzamenone A (
As shown in Scheme 1, the reaction of compound

Oxidations of compound 2. (a) H2O2, methanol, 40°C, 53%; (b) mCPBA, CH2Cl2, rt, 34%; (c) RNH2, THF, 50°C, 35% for

Key nuclear Overhauser effect spectroscopy correlation for compound 6.
Interestingly, oxidation of
The treatment of compound
Based on our preliminary data on antioxidant activity of the crude MeOH extract, natural manzamenones

Inhibitory effects of manzamenone derivatives
In conclusion, a series of manzamenones have been prepared by the isolation of secondary metabolites from the marine sponge Plakortis sp. and the subsequent chemical modifications of the manzamenone
Experimental
NMR spectra were recorded on a Varian Unity 500 instrument at 500 MHz for 1H and 125 MHz for 13C. All chemical shifts were recorded with respect to the residual solvent signals as an internal standard (CDCl3 δ H 7.26 ppm, δ C 77.0 ppm). The infrared spectra were recorded using a JASCO FT/IR-4100 spectrophotometer as thin film. Optical rotations were measured from a JASCO DIP-370 automatic polarimeter. Mass spectral data were obtained at the Korean Basic Science Institute, Seoul, Korea.
Collection and Identification
Specimens (Registry No. 102-CH-231) of Plakortis sp. were collected by hand with scuba equipment at a depth of 20-25 m off the shore of Weno Island, Chuuk State, Federated States of Micronesia, in February 2010. The sample was identified by Dr Y. A. Kim, and a voucher specimen has been deposited at the Marine Biotechnology Research Center, Korea Institute of Ocean Science & Technology.
Extraction and Isolation
The specimens were lyophilized (wet wt. 153 g) and repeatedly extracted with MeOH (500 mL × 2) and CH2Cl2 (500 mL × 1). The extract was filtered and concentrated under reduced pressure to afford 13.4 g of crude extract. The residue was partitioned between H2O and n-BuOH to yield 12.4 g of organic-soluble material. The n-BuOH layer was repartitioned between 15% aqueous MeOH and n-hexane (9.3 g). The residue of the n-hexane layer was subjected to silica vacuum flash chromatography using gradient mixtures of n-hexane and EtOAc as eluents (elution order: 10%, 20%, 30%, 40%, 50% EtOAc in n-hexane, and 100% EtOAc). The fraction eluted with 20% EtOAc in n-hexane (2.71 g) was dried and separated by silica HPLC (YMC silica column, 250 × 10 mm; 10% EtOAc in n-hexane) to afford in order of elution, 53.0 and 2.1 mg of
Compound
1H and 13C nuclear magnetic resonance data of compound 4.

Reaction of 2 with H2O2
43-O-Methylmanzamenone A (
Reaction of 2 with mCPBA
43-O-Methylmanzamenone A (
Reaction of 2 with Alkylamines
Compound
Inhibition of LDL Oxidation
The inhibitory effects against copper-induced LDL oxidation were measured by the reported method.
15
LDL (100 µg/mL) was incubated in a phosphate-buffered saline (pH 7.4, 1 mL) containing 25 µM CuSO4, in the absence (control) or presence of manzamenone derivatives (5 µM) at 37°C for 6 hours. The reaction mixture (1 mL) (15% trichloroacetic acid, 0.375% thiobarbituric acid, and 0.25 N hydrochloric acid) was added to the LDL mixture prepared as mentioned earlier. The resultant mixture was heated at 95°C for 30 minutes. After cooling, the absorbance of the pink chromophore was measured at 515 nm. A similar amount of 2,6-di-tertbutyl-4-methylphenol (BHT) was used as a reference compound. When comparing with the control, the inhibition rates against LDL oxidation for the compounds
Supplemental Material
Supplementary material - Supplemental material for Antioxidant Properties of the Manzamenones from the Tropical Marine Sponge Plakortis sp.
Supplemental material, Supplementary material, for Antioxidant Properties of the Manzamenones from the Tropical Marine Sponge Plakortis sp. by MyoungLae Cho, Yeon-Ju Lee, Jong Seok Lee, Hee-Jae Shin and Hyi-Seung Lee in Natural Product Communications
Footnotes
Acknowledgments
We are grateful to the Department of Marine Resources, State of Chuuk, Federated States of Micronesia, for supporting marine organism research. This research was partially supported by the KIOST (PE99721), and the Ministry of Oceans and Fisheries, Republic of Korea (PM61620). We would like to thank the Basic Science Research Institute in Seoul, Korea, for providing mass spectrometric data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
