Abstract
A key pathologic event in neurodegenerative diseases, such as Alzheimer’s disease and Parkinson’s disease, is endoplasmic reticulum (ER) stress-induced neuronal cell death. ER stress-induced generation of reactive oxygen species (ROS) has been implicated in neurological disease processes. Betulin is one of the major triterpenoids found in Betula platyphylla that possesses several biological properties, including cytoprotective and antioxidative effects. Therefore, we investigated whether betulin could prevent ER stress-induced neurotoxicity in HT-22 hippocampal neuronal cells. We observed that betulin reduced the thapsigargin (TG, an ER stress inducer)-induced apoptosis of HT-22 cells. Moreover, the cytoprotective effects of betulin were comparable to those of tauroursodeoxycholic acid, a potent ER stress-reducing agent. In our study, we confirmed that the ER stress-induced accumulation of ROS plays an important role in HT-22 cell death. Betulin also displayed cytoprotective effects in TG-injured HT-22 cells by reducing ROS generation; these results were comparable to those for N-acetyl-L-cysteine, a known ROS inhibitor. In addition, SnPP, a heme oxygenase-1 (HO-1) inhibitor significantly blocked the cytoprotective effects and ROS scavenging activity of betulin. Based on these results, we believe that betulin-mediated induction of HO-1 may contribute to the neuroprotective effects against ER stress in HT-22 hippocampal cells. We also found that betulin significantly inhibited the TG-induced expression of CHOP and caspase-12. These results demonstrated that betulin could serve as a potential therapeutic agent against ER stress-induced neurodegenerative diseases.
HT-22 hippocampal neuronal cell death has been implicated in several neurodegenerative disorders, such as Alzheimer’s disease and Parkinson’s disease. 1 The immortalized mouse hippocampal HT-22 cell line has been used in several studies to determine the specific effects of oxidative stress on cell death. 1 -3 The endoplasmic reticulum (ER) is an organelle that synthesizes secretory or membrane proteins, and ER stress-induced apoptotic cell death has been reported as a key pathologic event in the development of neurological diseases. 4 -6 ER stress is the major excitatory neurotransmitter in the brain and can induce neuronal cell damage through continuous oxidative stress. 4 -6 When ER functions are severely impaired, ROS levels get elevated, triggering apoptosis. 6 -8 These results suggest the importance of finding a potent therapeutic modality to regulate ER stress-induced apoptotic cell death in neurodegenerative disorders. 6 -9
Heme oxygenase (HO) is a cytoprotective protein with an ability to confer a beneficial effect on normal cells. 10 It mainly functions as a rate-limiting enzyme in heme degradation, yielding carbon monoxide (CO), iron, and biliverdin as the end products. 11 HO-1 and HO-2 are isoforms of HO in mammals. In particular, the role of HO-1 as a protective enzyme is well known. 10 -12 For example, its antioxidant, antiapoptotic, and anti-inflammatory effects have been extensively studied. 12,13 The cytoprotective effect of HO-1 could have several distinct underlying mechanisms, including degradation of heme to the antioxidant bilirubin; coordinated induction of ferritin, which chelates the pro-oxidant free iron; and release of CO, which exerts significant antioxidant effects. 12,13
Our previous study showed the ameliorative effects of betulin, isolated from Betula platyphylla bark, on scopolamine-induced amnesic mice. 14 Betulin, a naturally occurring pentacyclic triterpene, is commonly isolated from the bark of birch trees. Betulin and its derivatives have been widely used in traditional herbal medicine against various inflammatory diseases and ischemia. 14,15 Betulin is cytoprotective, and its anti-inflammatory actions are based on radical quenching and antioxidative effects. 14,15 Thus, betulin could serve as a potential therapeutic candidate for brain disorders. However, no study has yet reported the biological mechanism of betulin in ER stress-injured HT-22 in vitro models. In the present study, we aimed to uncover the mechanism on the neuroprotective effects of betulin on ER stress-induced apoptosis in HT-22 murine hippocampal neuronal cells. To further study the mechanism underlying the neuroprotection induced by betulin, we monitored its effects on the production of HO-1 and ROS in cells incubated with thapsigargin (TG), a known ER stress inducer. In addition, we investigated the role of apoptosis-related proteins, including CHOP and caspase-12.
Results and Discussion
Several studies have been conducted to find naturally occurring small molecules with an ability to reduce cellular damage caused by ER stress. The use of such molecules could be a potential therapeutic strategy for pathologies associated with oxidative stress. 16 -18 TG has been known as a non-competitive inhibitor of the sarco/endoplasmic reticulum Ca2+ ATPase (SERCA), which induces ER stress, ultimately causing cellular death. 19 In our screening system using TG-injured HT-22 hippocampal cells, we found betulin isolated from B. platyphylla bark to exert significant neuroprotective activity. The structure of betulin is shown in Figure 1a; betulin is a pentacyclic lupane-type triterpenoid with a double bond at position 20 (29) and three beta-hydroxy and 28 hydroxymethyl substituents. Since betulin can be easily converted to betulinic acid, several previous reports have compared the effects of betulin with those of betulinic acid in a broad spectrum of biological and pharmacological activities. 20,21 In particular, the antioxidant effect of betulin is more outstanding than betulin acid in hepatic stellate cell. 22 In our results, betulinic acid, a pentacyclic lupane-type triterpene derivative of betulin, exhibited mild cytotoxicity towards HT-22 hippocampal cells owing only to the structural difference of 28 carboxy substituents (Figure 1b).
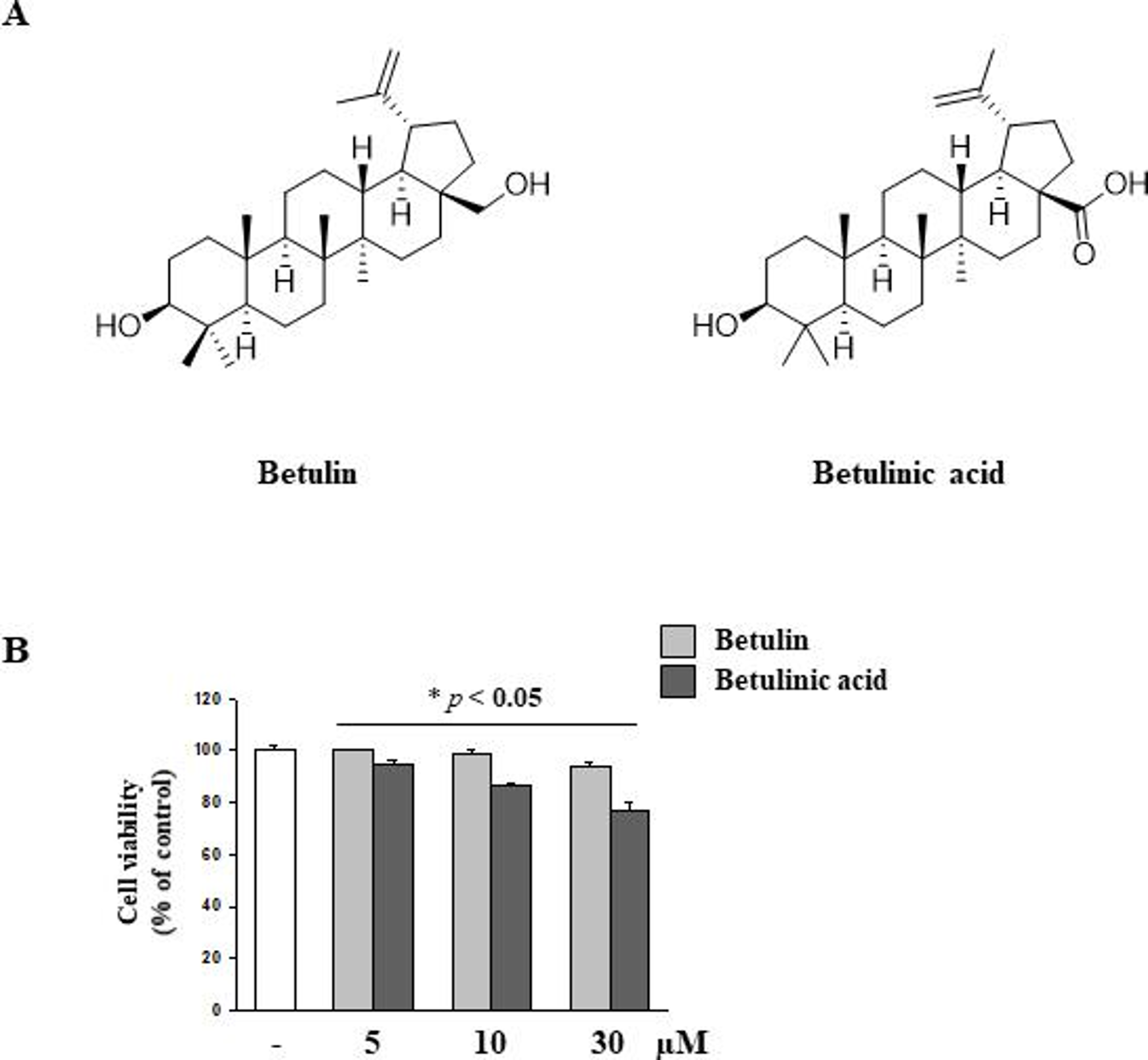
Chemical structure of betulin and betulinic acid, and measurement of cytotoxicity in HT-22. (a) Chemical structures of betulin and betulinic acid. (b) HT-22 cells were incubated for 24 hours with various concentrations of betulin and betulinic acid (10 µM and 30 µM). Data are representative of three independent experiments and expressed as mean ± SD (n = 3), *P < 0.05 compared with the group.
We used betulin at a concentration of up to 10 µM to gain further clarity regarding its mechanism.
To identify phytochemicals capable of exerting neuronal protection against ER stress, we investigated the effects of betulin in HT-22 cells using TG, a known ER stress inducer. Treatment with 10 µM TG for 24 hours increased HT-22 cell death by up to 50% compared with its effect on untreated cells (Figure 2a). On the contrary, treatment with betulin attenuated the TG-induced ER stress-mediated apoptosis of HT-22 cells. In our study, 100 µM of tauroursodeoxycholic acid (TUDCA), a potent ER-stress reducing agent, was used as a positive control. 23 Recent reports have suggested that ROS accumulation caused by ER stress plays an important role in inducing HT-22 cell death. 24 -27 ER stress could induce mitochondrial damage through the accumulation of ROS. 24 -27 Moreover, neurons are particularly susceptible to damage by ROS owing to low levels of endogenous antioxidant enzymes and weak antioxidant defense systems. 24 -27 Thus, the oxidative stress induced by ROS contributes to neuronal cell death in the brain. 24 -27 Similar to these findings, we detected significantly high ROS levels in HT-22 cells after treatment with TG. On the contrary, inhibition of ROS generation by betulin was comparable with that caused by TUDCA, after treatment with TG (Figure 2b). To investigate the cytoprotective effects of betulin through ROS reduction, we examined its effects on the reduced cell viability caused by TG and hydrogen peroxide (H2O2), two well-known oxidative stress inducers. As shown in Figure 2c, betulin increased the cell viability decreased by TG and H2O2 treatment, an effect comparable with that of N-acetyl-L-cysteine (NAC), a known ROS inhibitor. These results indicated that betulin protects HT-22 hippocampal cells by acting as a ROS scavenger under ER stress.
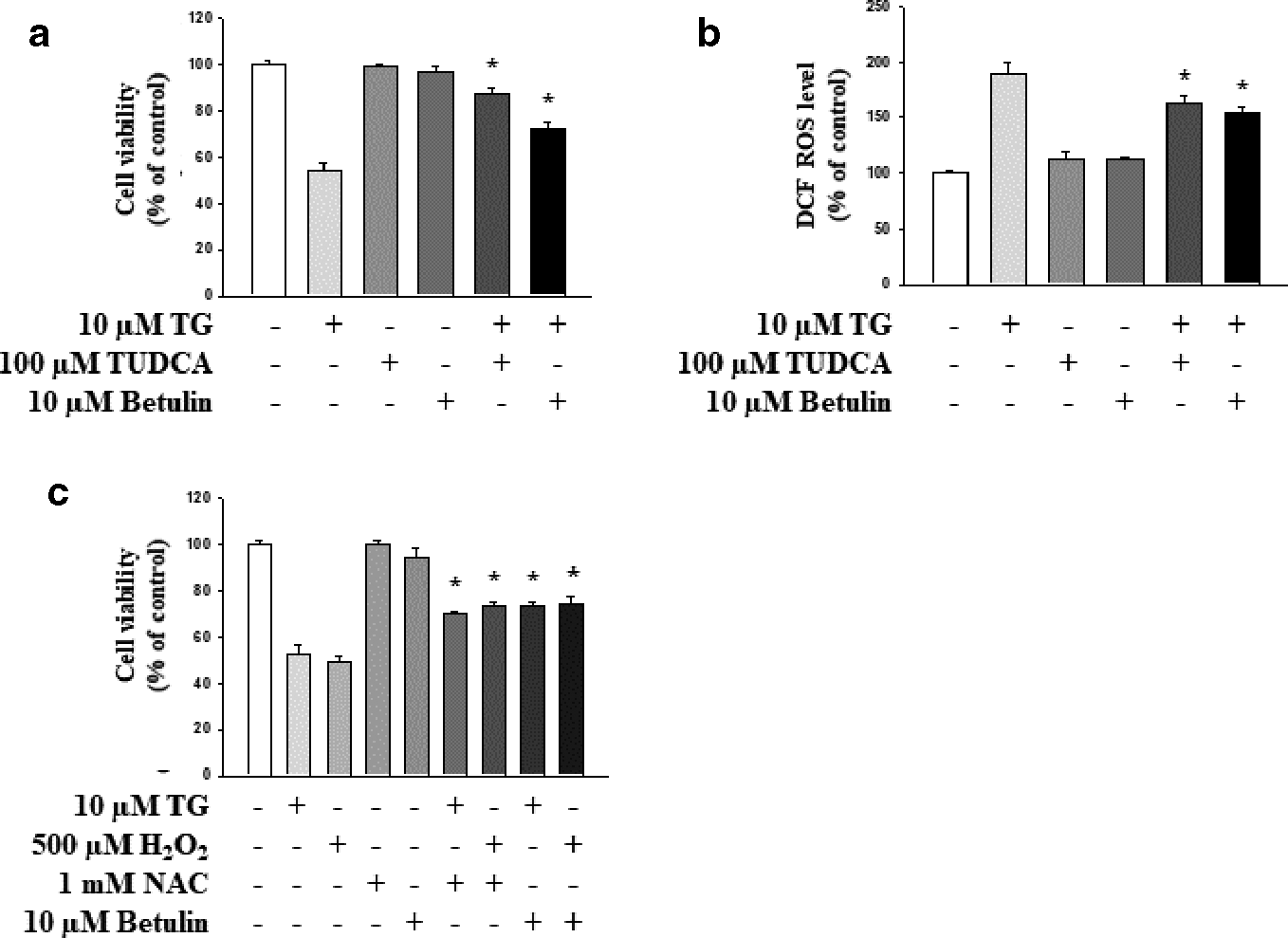
Antioxidant effects of betulin on ER stress-induced reactive oxygen species. (a) Cells were co-treated with 10 µM betulin and 10 µM TG. TUDCA (100 µM) was used as a positive control. (b, c) Incubation of cells with TG (10 µM) for 24 hours increased the production of reactive oxygen species. Betulin effectively inhibited the TG-induced generation of reactive oxygen species. NAC (1 mM) was used as the positive control. Data are representative of three independent experiments and expressed as mean ± SD (n = 3), *P < 0.05 compared with the group treated with TG only or H2O2 only. ER, endoplasmic reticulum; H2O2, hydrogen peroxide; NAC, N-acetyl-L-cysteine; SD, standard deviation; TUDCA, tauroursodeoxycholic acid; TG, thapsigargin.
Heme oxygenase-1 (HO-1) is an enzyme that catalyzes the breakdown of heme into the antioxidant biliverdin, CO, and iron. 26 HO-1 has received considerable attention for the potential neuroprotective effects in cell death models. 27,28 We used hemin, an HO-1 inducer, and SnPP, an HO-1 inhibitor, to investigate whether HO-1 is involved in ER stress-induced cytotoxicity in HT-22 cells by assessing the cell viability by measuring the ROS levels (Figure 3a and b). As shown in Figure 3b and c, the viability of HT-22 cells after treatment with TG and hemin increased as compared with that after a single treatment of TG; however, co-treatment with TG and SnPP decreased the viability of HT-22 cells as compared with a single treatment of TG. We also confirmed that hemin blocks TG-induced ROS production (Figure 3c).

Intracellular role of HO-1 in the TG-induced death of HT-22 cells. (a) HT-22 cells were treated with 10 µM TG and incubated with 0.5 µM hemin or 0.2 µM SnPP, followed by the measurement of HO-1 expression. (b, c) To confirm whether HO-1 induction inhibited TG-induced ROS production and cell death, an MTT assay was performed and ROS levels using dichlorofluorescin diacetate were measured. Data are representative of three independent experiments and expressed as mean ± SD (n = 3), *P < 0.05 compared with the group treated with TG only. #P < 0.05 compared with the group treated with TG and hemin. GAPDH, glyceraldehyde 3-phosphate dehydrogenase; HO, heme oxygenase; MTT, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide; ROS, reactive oxygen species; SD, standard deviation; TG, thapsigargin.
To determine the effects of betulin-induced HO-1 expression, HT-22 hippocampal cells were seeded and treated with betulin at various concentrations for 24 hours. Treatment with betulin gradually increased the expression of HO-1 in HT-22 cells (Figure 4a). In addition, we evaluated the effects of betulin on HO-1 induction under the ER stress. As shown in Figure 4b, treatment with both TG and betulin (lane 3) elevated the levels of HO-1 as compared with treatment with TG only (lane 4). Furthermore, as shown in lane 5 in Figure 4c, the HO-1 inhibitor SnPP inhibited the protective effects of betulin after treatment with TG, implying the involvement of the HO-1 pathway in betulin-mediated cytoprotective effects against ER stress. We also confirmed that SnPP blocked the ROS scavenging effects of betulin after treatment with TG (Figure 4d). Figures 4 and 3, thus, suggest that the betulin-mediated induction of HO-1 could contribute to neuroprotective effects against ER stress in HT-22 hippocampal cells.

Effect of betulin on HO-1 expression in HT-22 cells. (a) HT-22 cells were incubated with various concentrations of betulin. (b) SnPP, an HO-1 inhibitor, decreased HO-1 expression in a betulin-mediated manner. (c, d) SnPP inhibited the antioxidant and cytoprotective effects of betulin. GAPDH was used as a loading control. Data are representative of three independent experiments and expressed as mean ± SD (n = 3), *P < 0.05 compared with the group treated with TG only; #P < 0.05 compared with the group treated with TG and betulin; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; HO, heme oxygenase; SD, standard deviation; TG, thapsigargin.
Studies have reported CHOP to be an important mediator of ER stress-related apoptosis, such that its inhibition is considered as a potential therapy for neurodegenerative diseases, such as Alzheimer’s disease. 29,30 Therefore, we investigated whether betulin affects the expression of CHOP, a pro-apoptotic ER protein, using western blotting. In addition, we evaluated the activation of caspase-12, which is closely related to ER stress-induced cell death. Previous studies have reported that caspase-12 cleavage and activation results in pro-apoptotic actions of ER stress. 30,31 In our study, the levels of CHOP and cleaved caspase-12 significantly increased upon treatment with increasing concentrations of TG (Figure 5a and b). Next, we examined whether betulin affects the expression of CHOP and caspase-12 during ER-stress induced cytotoxicity in HT-22 hippocampal cells (Figure 5c and d). We found that betulin significantly inhibited TG-induced CHOP and caspase-12 expression, suggesting that the cytoprotective effects of betulin against ER stress were mediated through the CHOP pathway. In addition, we observed that SnPP attenuated the antiapoptotic effects of betulin in Figure 5c.

Inhibitory effect of betulin on the TG-activated apoptotic pathway. (a) HT-22 cells were incubated with various concentrations of TG for 24 H. (a, b) The expression of CHOP and caspase-12 was detected in HT-22 cells after treatment with TG and co-treatment of TG with betulin. (c) Bcl2 and cleaved caspase-3 were detected in the cell with SnPP. Data are representative of three independent experiments and expressed as mean ± SD (n = 3), *P < 0.05 compared with the control group; #P < 0.05 compared with the group treated with TG only. GAPDH, glyceraldehyde 3-phosphate dehydrogenase; SD, standard deviation; TG, thapsigargin.
To conclude, the findings of our study provide insights into the mechanisms underlying betulin-induced neuroprotection in TG-injured HT-22 hippocampal cells. We demonstrated that betulin inhibited ER stress-induced cell death by reducing ROS accumulation. Moreover, the cytoprotective effects of betulin could be attributed to the betulin-mediated induction of HO-1 expression after TG-induced neurotoxicity in HT-22 hippocampal cells. Since ER stress has been increasingly gaining importance as a factor contributing to the development of neuronal diseases, we suggest that betulin could serve as a promising natural agent for treating neurodegenerative diseases.
Experimental
Cell
The neuronal hippocampal HT-22 cell line was obtained from Professor S.H. Sung (Seoul National University, Seoul, Korea). HT-22 cells were cultured according to the directions of the Korean Cell Line Bank in RPMI medium, supplemented with 10% fetal bovine serum and 1% streptomycin (pen-strep) in a humidified atmosphere containing 5% CO2 at 37°C.
Sample Preparation
The bark of B. platyphylla (500 g) was grounded and extracted with chloroform (CHCl3) in an ultrasonic apparatus at room temperature. The CHCl3 extract was concentrated in vacuo to form a crude extract (52.5 g). Subsequently, the CHCl3 extract was suspended in water (H2O) and partitioned successively with n-hexane. After the removal of the non-polar organic constituents, residues were loaded onto a silica gel chromatography column and sequentially eluted with 150 mL of hexane-EtOAc (10:1). Next, the column was continuously eluted with 500 mL of hexane-EtOAc (4:1) with 500 drop fractions retained in each collection tube. Betulin (10.5 g) was isolated from each tube by recrystallization. Its purity was calculated to be >97% using high-performance liquid chromatography (HPLC) on the basis of its UV absorption at 200 nm. 15
Chemicals and Reagents
The chemicals, TUDCA, TG, hemin, and SnPP were purchased from Sigma-Aldrich Corporation (St. Louis, MO, USA).
Cell Viability Assay
The cell viability of activated HT-22 cells was determined using the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay. For the assay, 100 µL of cells were plated in a growth medium in a 96-well plate (105 cells/well) and incubated for 24 hours. Betulin was added to all wells at varying concentrations. Next, the MTT solution (5 mg/mL) was added to each well and the formazan precipitate formed upon incubation of cells for 2 hours was dissolved in 100 µL of dimethyl sulfoxide. Absorbance was measured using an automated microplate reader (Bio Tek; Winooski, VT, USA) at a wavelength of 562 nm. The relative cell survival (%) was calculated as a ratio between the absorbances of the treated and control (untreated) cells. The assay was performed at least thrice with a triplicate set for each condition.
Western Blotting and Antibodies
Cells were lysed in the radioimmunoprecipitation assay buffer containing 1× phosphate-buffered saline ( 1% [v/v]), Nonidet P-40, 0.5% (w/v) sodium deoxycholate, 0.1% (w/v) sodium dodecyl sulfate (SDS), 0.1 mg/mL phenylmethylsulfonyl fluoride, 30.0 µL/mL aprotinin, and 1.0 mM sodium orthovanadate. Cell lysates were centrifuged, and the resulting supernatants were collected. Proteins were separated using 8% to 15% SDS polyacrylamide gel electrophoresis gels and transferred onto a polyvinylidene difluoride membrane. Each membrane was blocked in Tris-buffered saline containing 0.1% Tween-20 (TBST) and 5% non-fat dry milk for 1 hour at room temperature, followed by overnight incubation with a primary antibody in TBST containing 1% non-fat dry milk at 4°C. Anti-HO-1, anti-CHOP, anti-caspase-12, and anti-glyceraldehyde phosphate dehydrogenase antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Membranes were washed with TBST and incubated with goat anti-rabbit or anti-mouse horseradish peroxidase-conjugated immunoglobulin G secondary antibody for 2 hours. The antibody signal was measured using the chemiluminescence system (GE Healthcare; Piscataway, NJ, USA).
Statistical Analysis
All experiments were conducted at least in triplicate, and data are expressed as mean ± standard deviation. Statistical significance was determined using the t test or one-way analysis of variance with Tukey’s test at indicated P-values. All statistical analyses were performed using the SPSS 18.0 software (SPSS Inc., Chicago, IL, USA).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclose the receipt of the following financial support for the research, authorship, and/or publication of this manuscript: This work was supported by the Chonnam National University (grant number: 2018-3487); and the Development of New Chemical Medical Measurement Standard Technology, funded by Korea Research Institute of Standards and Science (grant number: KRISS – 2019 – GP2019-0009).
