Abstract
A new glycoside, glutacoside (
The genus Gluta (Anacardiaceae) comprises approximately 42 species and is widely distributed throughout the tropical forests of Southeast Asia. Gluta usitata (wall) Ding Hou (previously known as Melanorrhoea usitata), locally known as Namkliang or Thai lac tree, is a flowering plant, medium to large tree with straight clean cylindrical trunks and dark green leaves. 1 Gluta usitata is well known as a major source of lacquer. The lacquer contains various compounds including urushiol, laccol, thitsiol, resorsinol, and phenol derivatives. 2 -4 Some urushiol derivatives have been evaluated as histone deacetylase 8 (HDAC8) inhibitors by molecular docking study. 5 Recently, we have reported the chemical constituents from the twigs and the flowers of this plant. 6,7 To the best of our knowledge, no compounds from the root of this plant have been reported.
Acetylation and deacetylation of histone protiens play an important role in the epigenetic regulation of transcription in cell. Acetylation is carried out by histone acetyl transferases (HATs) and deacetylation is controlled by histone deacetylases (HDACs). The balance between the action of HDACs and HATs defines the level of acetylation of the histones and the effect involved in the control of the cell cycle progression, differentiation, apoptosis, and tumorigenesis. Therefore, HDACs have been validated as prominent therapeutic target for a broad range of human disorders such as cancers, diabetes, and brain disorders. 8 -11 In humans, HDACs are grouped into 4 classes based on their homology to yeast models, subcellular localization, and enzymatic activities. Eighteen mammalian isoforms of HDACs have been reported. 12 Class I (HDAC1, 2, 3, and 8), class IIa (HDAC4, 5, 7, and 9), class IIb (HDAC6 and 10), and class IV (HDAC11) HDACs are zinc-dependent metalloproteins. Class III (SIRT1-7) HDACs are NAD+-dependent proteins. Different HDAC isoforms have been associated with different types of diseases and cancers. For example, class I, HDAC1, 2, and 3, has been associated with ovarian, pancreatic, lung, and gastric cancers, whereas HDAC2 has been detected in neurodegenerative diseases as well. 13 -18 Class IIa, HDAC4, has been involved in colon, ovarian, and gastric cancers, however, HDAC7 plays an important role in cardiovascular developments and diseases. 19 -22
In this article, we report the bioactive compounds: a new glycoside, glutacoside (
Experimental
General Experimental Procedures
Column chromatography (CC) was carried out on a silica gel 60 (Merk, Germany). Melting points were measured on a Gallenkamp SANYO melting point apparatus. 1H-NMR and 13C-NMR were recored on a Varian Mercury Plus spectrometer operated at 400 MHz (1H) or 100 MHz (13C). Mass spectrometer was operated using a Micromass Q-TOF 2 hybrid quadrupole time-of-flight. The fluorescence was measured using spectra Max Gemini XPS microplate spectrofluorometer. The UV spectra were obtained using a model Agilent 8453 UV-Vis Spectrometer. Optical rotation was measured on a JASCO DIP-1000 digital polarimeter.
Plant Material
The fresh roots of G. usitata were collected at Ubolratanaphitthayakhom School, Khon Kaen, in November 2014. The plant was independently verified by Dr Boonchuang Boonsuk. A voucher specimen (P. Kumboonma 01) has been deposited in the Herbarium of Khon Kaen University (KKU).
Extraction and Isolation
Air-dried and powdered root of G. usitata (2 kg) were extracted successively with methanol (3 × 10 L, 3 days each time) at room temperature. The combined extracts were concentrated to dryness under vacuum to obtain a methanol extract (105 g). The methanol extract (105 g) was isolated with a normal phase CC using a gradient of dichloromethane-methanol (10:0 to 5:5), to yield 4 fractions (F1-F4) based on Thin layer chromatography (TLC) profiles. Fraction F1 was purified on silica gel column, eluting with an isocratic of dichloromethane-methanol (9:1) to yield compound
Glutacoside (1)
White powder; mp 142°C to 143°C,
3,4,5-Trimethoxyphenol-1-O-β-d-Glucopyranoside (2)
White amorphous solid; mp 199°C to 202°C,
Acid Hydrolysis of 1 and 2
Acid hydrolysis was performed by heating a solution of compounds
β-Sitosterol-3-O-β-d-Glucoside (3)
Light-yellow amorphous solid; IR (neat) υ max 3369, 2957, 2930, 2867, 1461, 1367, 1068, 1020 cm−1 (supplemental figure S16); 1H-NMR (400 Hz, CD3OD): δ (ppm) = 5.36 (bs, 1H, H-6), 4.85 (dd, J = 11.7, 2.5 Hz, 1H, H-6ʹ), 4.84 (dd, J = 11.7, 5.2 Hz, 1H, H-6ʹ), 4.40 (t, J = 4.5 Hz, OH, H-5ʹ), 4.20 (d, J = 7.9 Hz, 1H, H-1ʹ), 3.58 (d, J = 4.5 Hz, OH, H-2′), 3.56 (d, J = 4.5 Hz, OH, H-3′), 3.40 (d, J = 4.5 Hz, OH, H-4′), 3.10 (m, 1H, H-3′), 3.06 (m, 1H, H-5′), 3.00 (m, 1H, H-4′), 2.98 (m, 1H, H-3), 2.89 (m, 1H, H-2′), 2.26 to 2.21 (m, 4H, H-2 and H-4), 0.99 (s, 3H, H-19), 0.89 (d, J = 6.3 Hz, 3H, H-21), 0.84 (t, J = 7.6 Hz, 3H, H-29), 0.81 (d, J = 6.9 Hz, 6H, H-26 and 27), 0.68 (s, 3H, H-18) (supplemental figure S14). 13C-NMR (100 Hz, CD3OD): δ (ppm) = 140.9 (C-5), 121.6 (C-6), 101.3 (C-1′), 77.5 (C-3′), 77.2 (C-3), 77.2 (C-5′), 73.9 (C-2′), 70.5 (C-4′), 61.5 (C-6′) 56.7 (C-14), 55.9 (C-17), 50.1 (C-9), 45.7 (C-24), 42.3 (C-13), 38.8 (C-4), 37.3 (C-12), 36.7 (C-1), 36.7 (C-10), 36.0 (C-20), 33.8 (C-22), 31.9 (C-7), 31.9 (C-8), 29.7 (C-2), 29.2 (C-25), 28.3 (C-16), 26.0 (C-23), 24.3 (C-15), 23.1 (C-28), 21.1 (C-11), 20.1 (C-26), 19.5 (C-19), 19.4 (C-27), 19.1 (C-21), 12.2 (C-29), 12.1 (C-18) (supplemental figure S15).
Fisetin (7)
White-yellow powder; IR (neat) υ max 3358, 3235, 1727, 1600, 1459, 1378, 1252, 1163, 1105, 1012 cm−1 (supplemental figure S19); 1H-NMR (400 Hz, CD3OD): δ (ppm) = 7.97 (d, J = 8.8 Hz, 1H, H-5), 7.77 (d, J = 2.0 Hz, 1H, H-8), 7.65 (dd, J = 8.8, 2.0 Hz, 1H, H-6), 6.92 (m, 1H, H-2′), 6.90 (m, 1H, H-6′), 6.86 (m, 1H, H-5′) (supplemental figure S17). 13C-NMR (100 Hz, CD3OD): δ (ppm) = 173.0 (C-4), 162.8 (C-7), 157.1 (C-8a), 147.3 (C-2), 146.1 (C-4′), 144.8 (C-3′), 137.1 (C-3), 126.1 (C-5), 123.0 (C-1′), 120.2 (C-6′), 114.8 (C-5′), 114.6 (C-2′), 114.5 (C-6), 114.1 (C-4a), 101.6 (C-8) (supplemental figure S18).
The structure elucidation of (−)-eriodictyol (
Histone Deacetylase Activity Assay
The isolated compounds were evaluated for their ability to inhibit HDAC enzymes. Inhibition of HDAC activity in vitro was measured using the Fluor-de-Lys HDAC activity assay kit. Trichostatin A was used as the positive control. A 5 µL of each sample (in Dimethyl sulfoxide), 1 µL of the Hela nuclear extract, and 19 µL of buffer were added into the 96-well plate and incubated at 37°C for 5 minutes. Next, the substrate (25 µL) was added and incubated again at 37°C for 15 minutes and then 50 µL of the developer was added to generate a fluorophore. Finally, the fluorescence signals of the fluorophores with excitation at 360 nm and emission at 460 nm were detected. A decrease of fluorescence signal indicated an inhibition of HDAC activity.
Antioxidant Assay
The antioxidant activities of all compounds were measured using 2,2-diphenylpicrylhydrazyl (DPPH) free radical. Gallic acid was used as a positive control. Five different concentrations of each antioxidant were tested. The DPPH solution in methanol (1 × 10–4 M) 3 mL was added to each sample in methanol. The final concentration of DPPH solution was 0.6 × 10–4 M. Incubation of the reaction mixtures was done in the dark at room temperature for 30 minutes. The absorbances of the solutions were measured at 517 nm. From the absorbance values monitored, the percentage inhibition was calculated and the result is expressed in terms of IC50. The percentage of DPPH inhibition was calculated as
Molecular Docking Studies
The crystal structures of HDACs were obtained from the Protein Data Bank [PDB entry code: 3MAX (HDAC2), 2VQW (HDAC4), 3C0Z (HDAC7), and 1T64 (HDAC8)]. All water and noninteracting ions as well as ligands were removed. Then, all missing hydrogens and sidechain atoms were added using the ADT program. Gasteiger charges were calculated for the system. For ligand setup, the ligands were drawn in Gauss View 03 program. Energy of the molecules was minimized with the AM1 level using Gaussian program. Docking software AutoDock 4.2 Program supplied with AutoGrid 4.0 and AutoDock 4.2 was used to produce grid maps. The spacing between grid points was 0.375 Å. The Lamarckian Genetic Algorithm was chosen to search for the best conformers. Molecular docking studies were operated for 50 runs.
Results and Discussion
Structure Elucidation
Chromatographic separation of the methanol extract of the dried G. usitata root yielded a new glycoside, glutacoside (
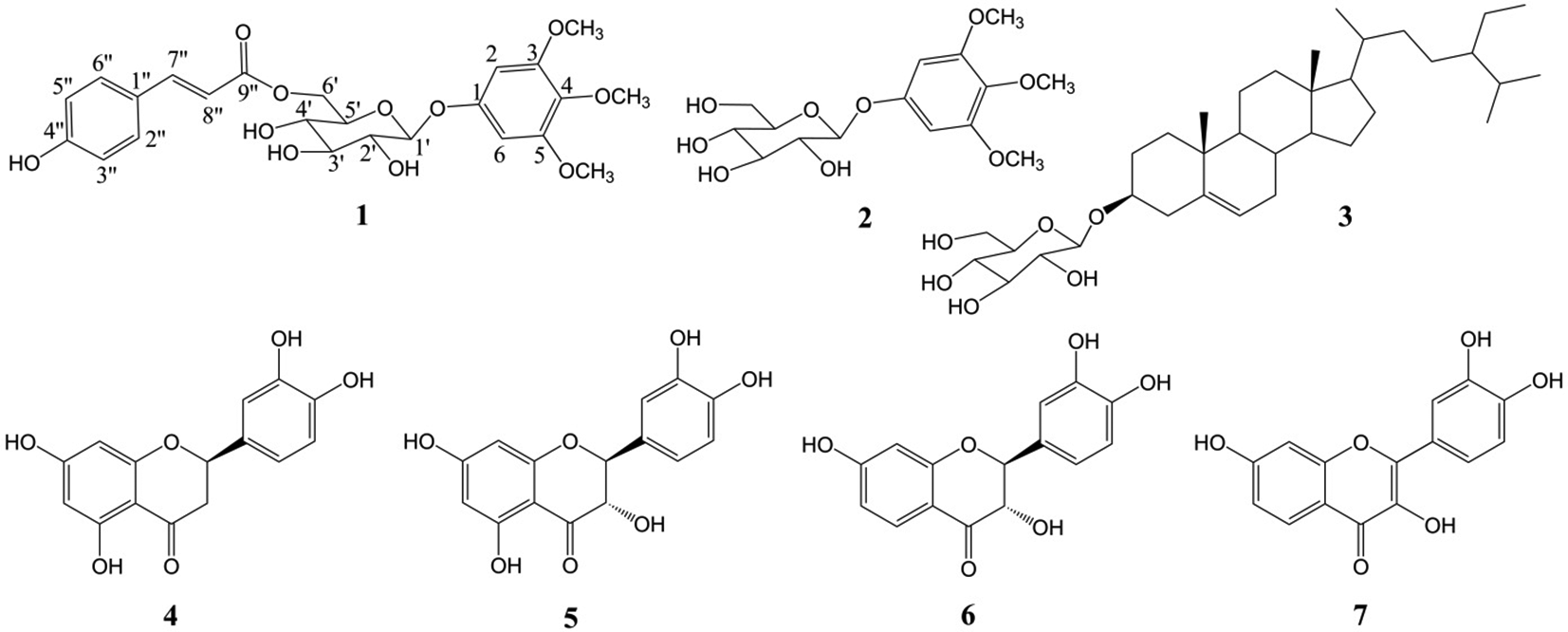
Structures of isolated compounds from the root of Gluta usitata.
Glutacoside (

Key correlations observed in the 2D NMR spectra of compound 1.
NMR Data of Glutacoside (
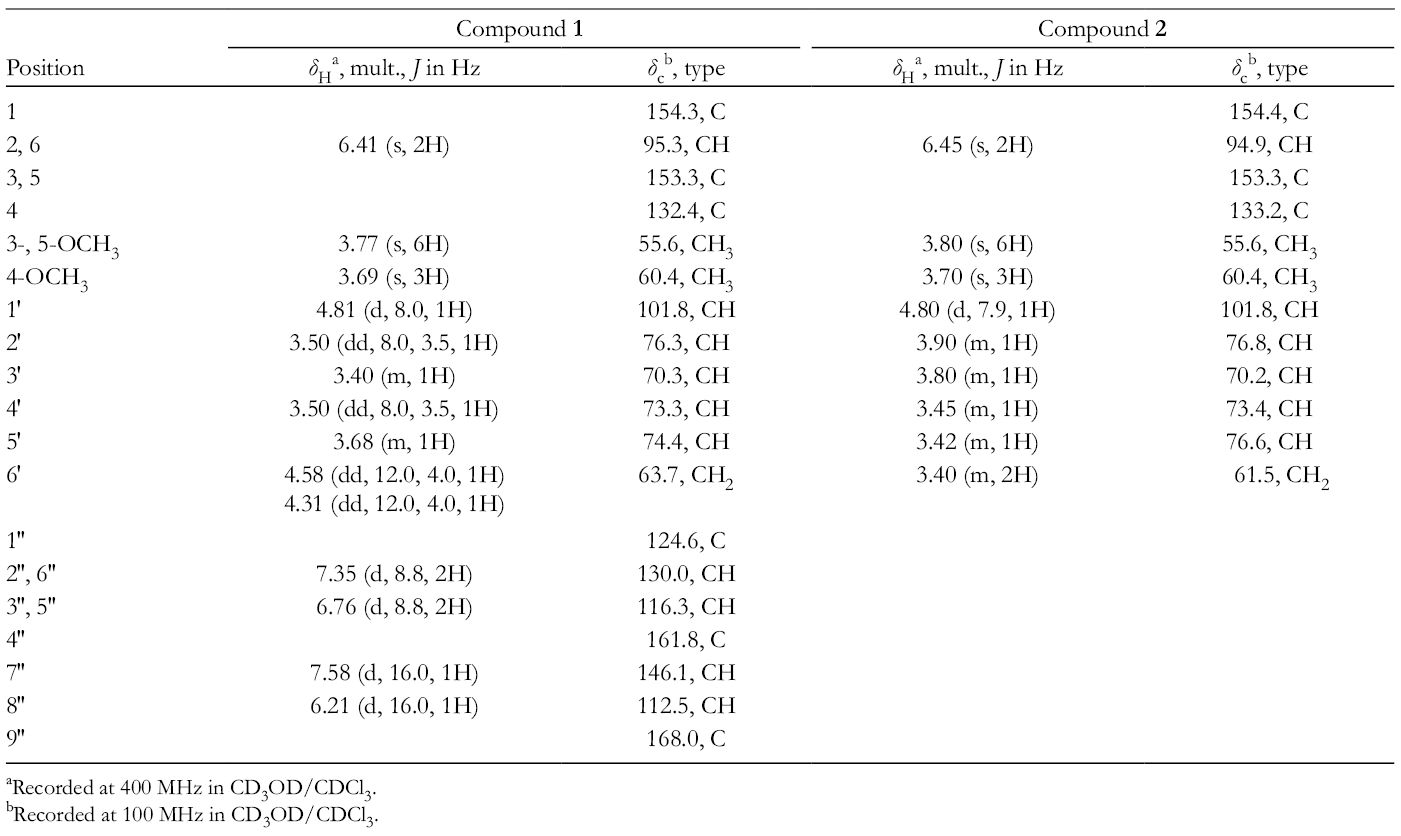
aRecorded at 400 MHz in CD3OD/CDCl3.
bRecorded at 100 MHz in CD3OD/CDCl3.
Histone Deacetylase Inhibitory Activity
Histone deacetylase inhibitory activities of all compounds were initially tested at 1 and 100 µM by a fluorimetric assay (Fluor de Lyse) (see supplemental tables S1 and S2). The results are shown in Table 2. All compounds demonstrated weak HDAC inhibitory activities at 1 µM. However, most compounds showed high percentage inhibitions against HDAC at 100 µM. Glycosides (
Histone Deacetylase Inhibitory Activity of the Compounds.

HDAC, histone deacetylase; TSA, trichostatin A.
aKumboonma et al. 6
bPositive control at 25 µM.
Molecular Docking Studies
To explore a possibility of being HDAC isoform-selective inhibitor, the most active in vitro compound

The interaction mode of 7 in the active site of histone deacetylase 8.
The I n Silico Histone Deacetylase Inhibitory Activity of Compound 7.

HDAC, histone deacetylase.
Antioxidant Studies
The antioxidant activities of the isolated compounds were evaluated by DPPH method. The DPPH assay is expressed in terms of IC50 values and the results are demonstrated in Table 4. Glycosides (
The I n Vitro Antioxidant Activity of the Compounds.

Conclusion
A new glycoside, glutacoside (
Supplemental Material
Supplementary material - Supplemental material for Histone Deacetylase Inhibitors and Antioxidants From the Root of Gluta usitata
Supplemental material, Supplementary material, for Histone Deacetylase Inhibitors and Antioxidants From the Root of Gluta usitata by Pakit Kumboonma, Somprasong Saenglee, Thanaset Senawong and Chanokbhorn Phaosiri in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by Rajamangala University of Technology Isan (RMUTI). We also would like to thank Mr. Kittisak Poopasith for the excellent NMR data.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is funded by Rajamangala University of Technology Isan (RMUTI).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
