Abstract
Reports on β-secretase inhibitors of natural origin are listed in order to reveal their chemical diversity. Various types of compounds were found to inhibit β-secretase, and natural resources included a wide spectrum of biological species. Among them, some triterpenes and moracin derivatives, which are nonpeptidic compounds, were determined to be competitive inhibitors. In addition, no peptide compounds were reported from natural resources. These points will be clarified in future studies.
There are now around 50 million dementia patients worldwide and this number has been increasing sharply. In 2050, there will be 130 million dementia patients. This sharp increase in the number of patients is expected to occur mainly in developing countries. One of the major problems caused by this increase will be national medical costs. The high medical cost will pressurize the national budget. Therefore, strategies to prevent dementia are urgently required.
There are various types of dementia, the most common of which is Alzheimer’s disease (AD), accounting for 60% to 70% of cases. The pathogenesis of AD is the cleavage of amyloid precursor protein by secretases that then liberate amyloid β peptide. Amyloid β peptide aggregates to form pigmentation. The pigmentation of amyloid β peptide is known as “senile plaque” and it destroys the nerve cells in the brain. The destroyed cells cannot be regenerated and neurotransmission is impaired. The pathogenesis of AD is thought to begin in younger people. Therefore, if an effective agent or compound could be administered or consumed over a long time period from a young age, AD could be prevented.
To develop an “effective” agent, inhibition of β-secretase is a promising molecular target. β-Secretase or BACE (beta-site amyloid precursor protein cleaving enzyme) is an arginine protease that cleaves the β-site of amyloid precursor protein. The inhibition of β-secretase directly inhibits the production of amyloid β peptide.
To discover novel β-secretase inhibitors and their chemical structures, screening of extracts from natural resources or naturally occurring compounds is a promising strategy. Many researchers have done extensive research in this field. In this review, the chemical diversity of β-secretase inhibitors from natural resources is presented by listing their structures. Reports on β-secretase inhibitors after 2011 are presented and discussed since Williams et al have already listed the structures of β-secretase inhibitors reported before 2011. 1
Reports on β-Secretase Inhibitors of Natural Origin
Paudel et al reported that bromophenols isolated from a red algae, Symphyocladia latiuscula, are potent β-secretase inhibitors (Figure 1). 2 Among the compounds tested, bis(2,3,6-tribromo-4,5dihydroxybenzyl)ether showed the most potent activity with an IC50 value of 2.3 μM. The compound also showed potent inhibition of acetylcholinesterase and butyrylcholinesterase. These data suggest that bromophenols are promising candidates for both therapeutic treatment and prevention of AD. Yatsu et al isolated meroterpenoids from the fruiting body of the mushroom Boletinus asiaticus. 3 Among them, asiaticusinol C, asiachromenic acid, and asiaticusin A showed inhibitory activities with IC50 values of 14.7, 11.4, and 2.0 μM, respectively (Figure 2). Ali et al reported that 3 flavanone glycosides—didymin, poncirin, and prunin—possessed inhibitory activities against β-secretase with IC50 values of 2.3, 4.0, and 13.4 μM, respectively (Figure 3). 4 Their potencies were higher than that of a flavone, quercetin (IC50 = 21.7 μM). They also revealed that the modes of inhibition of these flavanones were mixed type based on kinetic analyses. Choi et al isolated pterosin glycoside, (-)-pteroside N, and seco-illudoid sesquiterpene, pterosinone, as β-secretase inhibitors (Figure 4). 5 The IC50 values were deduced to be 30.6 μM for (-)-pteroside N and 19.4 μM for pterosinone. Furthermore, pterosinone was revealed to be a mixed-type inhibitor from kinetic analysis. Qi et al isolated meroterpenoids as β-secretase inhibitors from the fungal strain Aspergillus terreus. 6 Among the compounds isolated, asperterpene E, F, and J showed promising inhibitory effects on β-secretase with IC50 values of 3.3, 5.9, and 31.7 μM (Figure 5). Interestingly, other meroterpenoids showed no obvious inhibitory activities. The authors concluded that a cis-fused A/B ring may be important for the inhibitory activity since other compounds have a trans-fused A/B ring or a Δ 5 A/B ring system. Furthermore, an open D ring may reduce the potency based on the activity data of asperterpene E and J. Qi et al also isolated novel 3,5-dimethylorsellinic acid-based meroterpenoids from the same organism (Figure 6). 7 Among the compounds isolated, terreusterpene A and B showed inhibitory effects on β-secretase with IC50 values of 6.0 and 11.4 μM, respectively. Interestingly, terreusterpene D, a synthetic derivative of terreusterpene C, possessed the most potent inhibitory activity with an IC50 value of 1.9 μM, whereas terreusterpene C showed no significant inhibitory activity. Terreusterpene D also showed a potent inhibitory effect on acetylcholinesterase with an IC50 value of 8.9 μM. Terreusterpene D is therefore a good potential compound for both prevention and therapy of AD. Wagle et al found that glycyrrhizin, and its metabolites 18α- and 18β-glycyrrhetinic acid, isolated from the seaweed Hizikia fusiformis showed inhibitory activity against β-secretase (Figure 7). 8 In their report, glycyrrhizin and 18α- and 18β-glycyrrhetinic acid showed an inhibitory effect on β-secretase with IC50 values of 20.1, 104.4 and 8.9 μM, respectively. In addition, modes of inhibition for glycyrrhizin and 18β-glycyrrhetinic acid were determined to be noncompetitive and competitive, respectively. Son et al investigated moracin derivatives isolated from the root bark of the white mulberry, Morus alba. 9 Moracin M, O, P, and S showed inhibitory effects on β-secretase with IC50 values of 18.7, 36.8, 18.2, and 3.1 μM, respectively (Figure 8). In addition, moracin M and S were revealed to be competitive inhibitors from kinetic analyses. Interestingly, the nonpeptidic compounds moracin M and S are competitive inhibitors against β-secretase. Kuk et al also isolated mulberrofuran G, albanol B, and kuwanon G from the same plant material (Figure 9). 10 Mulberrofuran G, albanol B, and kuwanon G showed inhibitory effects on β-secretase with IC50 values of 0.3, 0.5, and 1.0 μM, respectively. In addition, these 3 compounds showed mixed-type inhibition against β-secretase. Xu et al isolated 4 prenylated flavonoids, bavachin, bavachinin, bavachalcone, and isobavachalcone, from the fruit of Cullen corylifolium, which is known as Psoralea Fructus (Figure 10). 11 All compounds showed moderate inhibitory activity against β-secretase. Among them, bavachalcone was the most potent with approximately 63% of inhibition at 100 μM, as shown in the graph presented in the report. Bavachalcone also showed inhibitory activity against Aβ42 aggregation. Therefore, bavachalcone could be developed as a multifunctional compound to prevent AD. Shrestha et al isolated rubrofusarin and its glycosides from the seeds of Cassia obtusifolia (Figure 11). 12 Among the compounds isolated, nor-rubrofusarin 6-O-β-d-glucoside showed the most potent inhibitory effect on β-secretase with an IC50 value of 14.4 μM, while the IC50 values of rubrofusarin and rubrofusarin 6-O-β-d-gentiobioside were 90.0 and 85.7 μM, respectively. In addition, rubrofusarin 6-O-β-d-glucopyranoside showed a moderate inhibitory effect with an IC50 value of 190.6 μM. The mode of inhibition of nor-rubrofusarin 6-O-β-d-glucoside against β-secretase was determined to be mixed type. Xie et al isolated sesquiterpenes from the fungus Phomopsis sp. TJ507A. 13 Among the compounds isolated, phomophyllin A, B, C, D, E, F, G, and I, radulone B, onitin, and 7-hydroxy-10-oxodehydrodihydrobotrydial showed inhibitory effects on β-secretase ranging from 19.4% to 43.8% inhibition at a concentration of 40 μM (Figure 12). Zhu et al isolated 4 steroidal compounds from marine Urechis unicinctus. 14 Among them, hecogenin and cholest-4-en-3-one showed inhibitory effects on β-secretase with IC50 values of 116.3 and 390.6 μM, respectively (Figure 13). The authors concluded that the potencies were comparable to other natural product-based inhibitors previously reported. Wang et al isolated 7 xanthones from the pericarps of mangosteen, Garcinia mangostana, and demonstrated their inhibitory activities against β-secretase (Figure 14). 15 α-Mangostin, 8-deoxygartanin, gartanin, garciniafuran, garcinone C, garcinone D, and γ-mangostin showed 60.3%, 16.9%, 39.8%, 36.3% 18.7%, 62.7%, and 42.1% of inhibition at 100 μM, respectively. α-Mangostin and garcinone D showed potent inhibitory activities against β-secretase among the compounds tested and the former compound also showed inhibitory activities against Aβ42 self-aggregation, both in bacteria and in vitro, while the latter showed its inhibitory activity only in vitro. Therefore, mangosteen containing these xanthones has the potential to prevent AD. In addition, α-mangostin is a promising candidate agent to prevent AD due to its potent enzyme inhibition and inhibitory activity against Aβ42 self-aggregation. Zhao et al also determined that α-mangostin, a polyphenolic xanthone, possessed β-secretase inhibitory activity with an IC50 value of 13.2 nM (Figure 15). 16 α-Mangostin also decreased the production of Aβ40 and Aβ42 in mouse primary neuronal cells (cortical neurons) without affecting the expression of enzymes related to nonamyloidogenic and amyloidogenic pathways or amyloid precursor protein maturation. These data indicate that α-mangostin is a highly potent β-secretase inhibitor and could be a multifunctional compound to prevent AD. Seong et al isolated meroterpenoids from a brown algae, Sargassum serratifolium. 17 Sargahydroquinoic acid, sargachromenol, and sargaquinoic acid showed inhibitory activity against β-secretase with IC50 values of 4.4, 7.0, and 12.1 μM, respectively (Figure 16). Inhibition of sargahydroquinoic acid and sargaquinoic acid was mixed type while that of sargachromenol was noncompetitive. Lee et al isolated 8 anthraquinones from Morinda officinalis using a bioassay-guided approach. 18 Among the compounds isolated, alizarin-1-methyl ether, 1,2-dimethoxy-3-hydroxy anthraquinone, 2-methoxy anthraquinone, rubiadin-1-methyl ether, 1-hydroxy-3-hydroxymethyl anthraquinone, and rubiadin showed potent inhibitory activities against β-secretase with IC50 values of 192.4, 114.6, 9.3, 25.9, 178.4, and 19.8 μM, respectively (Figure 17). In addition, alizarin-1-methyl ether, 1,2-dimethoxy-3-hydroxy anthraquinone, and 2-methoxy anthraquinone possessed moderate inhibitory activity against acetylcholinesterase and butyrylcholinesterase. Therefore, the consumption of M. officinalis containing these 3 compounds over a long time period may contribute to the prevention and treatment of AD. Koirala et al found that the crude extract of Pueraria lobata possessed an inhibitory effect on β-secretase with an IC50 value of 80.4 μg/mL. 19 The active principles were determined to be lupeol and lupenone by activity-guided fractionation (Figure 18). The IC50 values of lupeol and lupenone for β-secretase inhibition were calculated as 5.1 and 63.0 μM, respectively, and their modes of inhibition were competitive. Ali et al isolated umbelliferones from Angelica decursiva and evaluated their inhibitory activities against β-secretase as an indicator of anti-AD activity. 20 Among the compounds tested, 6-formyl umbelliferone showed the most potent inhibitory activity against β-secretase with an IC50 value of 1.3 μM, whereas umbelliferone and 8-formyl umbelliferone showed moderate inhibitory activities with IC50 values of 168.5 and 39.8 μM, respectively (Figure 19). The modes of inhibition of 6-formyl umbelliferone and 8-formyl umbelliferone were determined to be noncompetitive. Youn et al demonstrated inhibitory effects of polymethoxyflavones, nobiletin, tangeretin, and sinensetin, on β-secretase and their IC50 values were 59, 49, and 63 μM, respectively (Figure 20). 21 Bhakta et al demonstrated the inhibitory activities of cornuside and gallotannins contained in fruits of Cornus officinalis. 22 Among them, 1,2,3,6-tetra-O-galloyl-β-d-glucose showed a potent inhibitory effect on β-secretase with an IC50 value of 20.2 μM in a noncompetitive manner (Figure 21). In addition, isoterchebin, 1,2,3-tri-O-galloyl-β-d-glucose, and tellimagrandin II showed weaker inhibitory activity with IC50 values of 33.3, 41.2, and 45.6 μM, respectively. Tellimagrandin II and isoterchebin were mixed-type inhibitors against β-secretase. Cornuside and tellimagrandin I possessed moderate inhibitory activity with IC50 values of 55.8 and 52.8 μM, respectively. Dai et al isolated 4 novel pregnane-10,2-carbolactones and 1 known compound from a marine sponge of the genus Myrmekioderma, and tested their inhibitory effects on β-secretase (Figure 22). 23 The novel chlorinated compound (6β-chloro-3β-hydroxy-17-methyl-17α-pregna-4,13-diene-10,2-carbolactone) showed weak inhibitory activity against β-secretase with an IC50 value of 82 μM, while the other compounds showed no activity at the test concentration of 80 μM. Youn et al demonstrated the inhibitory effect of isoflavone biochanin A, which can be found in legumes and red clover, on β-secretase (Figure 23). 24 Biochanin A inhibited β-secretase in a dose-dependent manner with 30.45%, 56.11%, 69.69%, and 74.69% inhibition at 12.5, 25, 50, and 100 μM, respectively. From these data, the IC50 value appears to be around 20 μM. Biochanin A was found to be a noncompetitive inhibitor by kinetic analyses. Jung et al isolated compounds from the heartwood of Juniperus chinensis and revealed their inhibitory effects on β-secretase. 25 Among the compounds tested, 9′-methoxycalocedrin, α-methyl artoflavanocoumarin, and 5,7,4′-trihydroxy-2-styrylchromone showed significant inhibitory activities against β-secretase with IC50 values of 16.1, 11.9, and 6.3 μM, respectively (Figure 24). In addition, savinin, calocedrin, 10-oxowiddrol, (+)-taxifolin, (7S,8R)-dihydro-3′-hydroxy-8-hydroxymethyl-7-(4-hydroxy-3-methoxyphenyl)-1′-benzofuranpropanol, styraxlignolide C, (7R,8S)-dihydro-3′-methoxy-8-hydroxymethyl-7-(4-hydroxy-3-methoxyphenyl)-1′-benzofuranpropanol 4-O-β-d-glucopyranoside, and (+)-catechin exhibited moderate inhibitory activities against β-secretase with IC50 values of 185.0, 87.7, 93.8, 142.8, 86.3, 62.0, 107.1, and 23.2 μM, respectively. In this report, lignans and flavonoids were potent inhibitors of β-secretase. Moreover, most of the β-secretase inhibitors also possessed an inhibitory effect on acetyl- and butyrylcholinesterase, suggesting that the plant J. chinensis has good potential for both prevention and therapy of AD. Jung et al revealed that the extracts from various parts of Nelumbo nucifera possessed an inhibitory effect on β-secretase. 26 Among them, they found that an embryo extract showed potent inhibitory activity with an IC50 value of 7.47 μg/mL. Activity-guided fractionation led them to isolate 2 alkaloids (neferine and liensinine), 2 flavonoids (vitexin and quercetin 3-O-glucoside), and an isoquinoline alkaloid (northalifoline) (Figure 25). Liensinine showed the most potent inhibitory activity with an IC50 value of 6.4 μM. Neferine, vitexin, quercetin 3-O-glucoside, and northalifoline showed IC50 values of 28.5, 19.3, 41.2, and 33.2 μM, respectively. In addition, all compounds possessed inhibitory activities against acetyl- and butyrylcholinesterase at IC50 values ranging from 0.3 to 102.6 μM. Therefore, the N. nucifera embryo could be a promising candidate for prevention and therapy of AD if the acute toxicity of its alkaloids is low. Lam et al isolated bioactive steroidal alkaloids from Buxus macowanii and tested for inhibitory activities against β-secretase. 27 Among them, 31-hydroxybuxatrienone and N b-demethylpapillotrienine showed inhibitory activities against β-secretase with IC50 values of 15.0 and 25.0 μM, respectively (Figure 26). Nguyen et al isolated 4 novel compounds and 9 known compounds from Lycopodiella cernua and tested for anti-β-secretase activities. 28 Among the compounds isolated, 4 novel compounds, 3β,21β,29-trihydroxyserrat-14-en-24-oic acid-3β-yl-(7′-hydroxycinnamate), 21β-hydroxyserrat-14-en-3,16-dione, 3β,14α,15α,21β-tetrahydroxyserratan-24-oic acid-3β-yl-(4′-methoxy-5′-hydroxybenzoate), and (2E,4E,6R)-6-hydroxydeca-2,4-dienoic acid and a known compound, 3β,21β,29-trihydroxyserrat-14-en-24-oic acid-3β-yl-(4′-hydroxybenzoate), showed inhibitory effects on β-secretase with IC50 values of 1.1, 0.2, 1.0, 6.9, and 0.3 μM, respectively (Figure 27). Their modes of inhibition will be elucidated in further experiments. Choi et al isolated polyphenols from a brown algae, Ecklonia cava, and their inhibitory activities against β-secretase were noted. 29 Dieckol, 6,6′-bieckol, 8-8′-bieckol, eckol, and phlorofurofukoeckol-A showed inhibitory effects against β-secretase with 1 μM as 36.8%, 18.6%, 35.0%, 25.6%, and 58.3% inhibition, respectively (Figure 28). Among the compounds tested, phlorofurofukoeckol-A showed the most potent activity. In addition, phlorofurofukoeckol-A also possessed inhibitory activity against acetylcholinesterase, butyrylcholinesterase, and biosynthesis of amyloid precursor protein. These data show that phlorofurofukoeckol-A is a promising candidate for inhibition of AD pathogenesis. Brown algae may be a good source for lead compounds with anti-AD effects. Rauf et al isolated a steroidal compound, pistagremic acid, from Pistacia integerrima and tested for its inhibitory activity against β-secretase (Figure 29). 30 Pistagremic acid showed potent inhibitory activity against β-secretase with an IC50 value of 350 nM. Youn et al found that an ethyl acetate fraction obtained from a 95% ethanolic extract of Geranium thunbergii possessed the most potent inhibitory activity against β-secretase with 69.39% of inhibition at 50 μg/mL. 31 Geraniin and corilagin were isolated by bioactivity-guided fractionation as the active principles of the ethyl acetate fraction’s inhibitory activity (Figure 30). Geraniin and corilagin showed inhibitory activities against β-secretase with IC50 values of 4.0 and 34.0 μM, respectively. These data suggest that a crude extract of G. thunbergii has potential to prevent AD. In addition, some tannins could also be lead compounds for potent β-secretase inhibition. Zhang et al isolated metabolites including pyrrolidone, ianthellidones, and lamellarin derivatives from a southern Australian marine sponge, Ianthella sp. 32 Among the various compounds isolated, lamellarin O1 showed the most potent inhibitory activity against β-secretase with 60% of inhibition at 10 μM while ianthellidone F, lamellarin O2, and lamellarin O showed 40% inhibition at the same concentration (Figure 31). Tao et al isolated flavanones and dihydrochalcones from Balanophora involucrata and tested for the inhibitory activities against β-secretase. 33 Among the compounds tested, 3,4,2′,6′-tetrahydroxydihydroflavone-4′-O-β-d-glucopyranose showed the most potent inhibitory activity of 21.4% at 100 μM (Figure 32). The other compounds listed in Figure 32 showed weak activities with 5.3% to 18% inhibition at the same concentration. Youn et al found that an ethyl acetate fraction obtained from an ethanolic extract of Corni fructus possessed potent inhibitory activity against β-secretase. 34 They isolated ursolic acid, p-coumaric acid, and gallic acid as candidates for inhibitory activity against β-secretase. Among the isolated compounds, p-coumaric acid showed the most potent activity with an IC50 value of 90 μM (Figure 33). In addition, the mode of the inhibition was deduced to be noncompetitive by kinetic analyses. These data suggest that a C. fructus extract has potential to prevent AD, while p-coumaric acid could be a lead compound for developing a new treatment. Dai et al revealed that an ethyl acetate extract of the yellow fruit of Ficus benjamina possessed inhibitory activity against β-secretase with 50% inhibition at 30 μg/mL. 35 In addition, they isolated 2 coumarano-chroman-4-ones and 6 novel isoflavonoids from the ethyl acetate extract. Among the novel compounds isolated, 5,7,2′,4′-tetrahydroxy-8-(3,7-dimethyl-2,6-octadienyl) isoflavone showed the most potent inhibitory activity with an IC50 value of 45 μM, while the 2 coumarano-chroman-4-ones, 3,5,7-trihydroxy-4′-methoxycoumarano-chroman-4-one and 6-(3-methyl-2-buten-1-yl)-3,5,7-trihydroxy-4′-methoxycoumarano-chroman-4-one, showed inhibitory activities with IC50 values of 139 and 91 μM, respectively (Figure 34). The most potent compound tested in this report was lupiwighteone and its IC50 value was 27 μM. Marumoto et al revealed the structure-activity relationships of coumarins against β-secretase inhibition. 36 Among the various coumarin derivatives tested, 5-geranyloxy-8-methoxypsoralen showed the most potent inhibitory activity with an IC50 value of 9.9 μM (Figure 35). Bergamottin, 8-geranyloxypsoralen, and 8-geranyloxy-5-methoxypsoralen exhibited IC50 values of 32.2, 20.4, and 11.1 μM, respectively. In addition, phellopterin and kinidilin showed moderate inhibitory activities, with IC50 values of 143 and 344 μM, respectively. Bergamottin, 5-geranyloxy-8-methoxypsoralen, and 8-geranyloxy-5-methoxypsoralen were proven to be mixed-type inhibitors by kinetic analyses, while 8-geranyloxypsoralen was a noncompetitive inhibitor. Sorribas et al isolated 3 novel lanostane-type triterpenes from the fruiting body of a fungi Daedalea sp. by activity-guided purification. 37 Among the compounds tested for anti-β-secretase activity, daedalol C showed the most potent activity with an IC50 value of 14.2 μM (Figure 36). The epoxide moiety in the molecule may play an important role in expressing the activity based on structure-activity relationship studies. Cho et al found that a methanolic extract of the stem bark of Morus lhou possessed an inhibitory effect on β-secretase with an IC50 value of 78.4 μg/mL. 38 Activity-guided fractionation led them to isolate 8 prenylated flavones (Figure 37). All isolated compounds showed dose-dependent inhibitory activity against β-secretase and their IC50 values were as follows: norartocarpetin, 60.6 μM; kuwanon C, 3.4 μM; morusin, 59.4 μM; kuwanon A, 5.3 μM; cyclomorusin, 101.2 μM; morusinol, 135.9 μM; neocyclomorusin, 146.1 μM; mormin, 103.5 μM. These data suggest that M. lhou extract could be a promising candidate for prevention of AD and that prenyl flavones, especially kuwanon C, could be good lead compounds to develop β-secretase inhibitors. Sasaki et al screened biflavonoids isolated from various plants for inhibitory activity against β-secretase. 39 Among them, 2,3-dihydroamentoflavone and 2,3-dihydro-6-methylginkgetin showed potent inhibitory activities against β-secretase with IC50 values of 0.75 and 0.35 μM, respectively (Figure 38). Amentoflavone, sequoiaflavone, bilobetin, sotetsuflavone, podocarpusflavone A, ginkgetin, amentoflavone-7,7″-dimethyl ether, podocarpusflavone B, and isoginkgetin also showed moderate inhibitory effects on β-secretase with IC50 values of 1.5, 1.4, 20.2, 1.6, 0.99, 4.2, 6.3, 4.2, and 3.0 μM, respectively. Biflavonoids are also promising lead compounds for development of AD prevention agents due to their high potencies. Marumoto et al found that a chloroform extract of Angelica dahurica possessed potent inhibitory activity against β-secretase with 46% of inhibition at 100 μg/mL. 40 Activity-guided fractionation led them to isolate 5 furanocoumarins (Figure 39). All the isolated furanocoumarins exhibited an inhibitory effect on β-secretase and their IC50 values were as follows: isoimperatorin, 244.2 μM; imperatorin, 91.8 μM; (+)-oxypeaudanin, 359.2 μM; (+)-byakangelicol, 104.9 μM; (+)-byakangelicin, 219.7 μM. Tian et al found that an ethanolic extract of Homalomena occulta possessed a moderate inhibitory effect on β-secretase. 41 Four phenolic acids and a sesquiterpene were isolated by activity-guided fractionation. Of these, the 4 phenolic acids, 2-[(Z)-heptadec-11-enyl]-6-hydroxybenzoic acid, 2-[(6Z,9Z,12Z)-heptadeca-6,9,12-trienyl]-6-hydroxybenzoic acid, 2-[(9Z,12Z)-heptadeca-9,12-dienyl]-6-hydroxybenzoic acid, and 2-hydroxy-6-(12-phenyldodecyl)benzoic acid showed inhibitory activities against β-secretase with IC50 values of 6.2, 6.3, 7.9, and 7.7 μM, respectively (Figure 40). In addition, 2-[(Z)-heptadec-11-enyl]-6-hydroxybenzoic acid was deduced as a noncompetitive inhibitor of β-secretase by kinetic analysis.

Structures of bromophenols isolated from Symphyocladia latiuscula. Inhibitory activities against β-secretase are hereinafter shown as IC50 values in figures.

Structures of meroterpenoids isolated from fruiting body of Boletinus asiaticus.

Structures of flavanone derivatives.

Structures of (-)-pteroside N and pterosinone isolated from Pteridium aquilinum.

Structures of asperterpene E, F, and J isolated from Aspergillus terreus.

Structures of terreusterpene A, B, C, and D isolated from Aspergillus terreus.
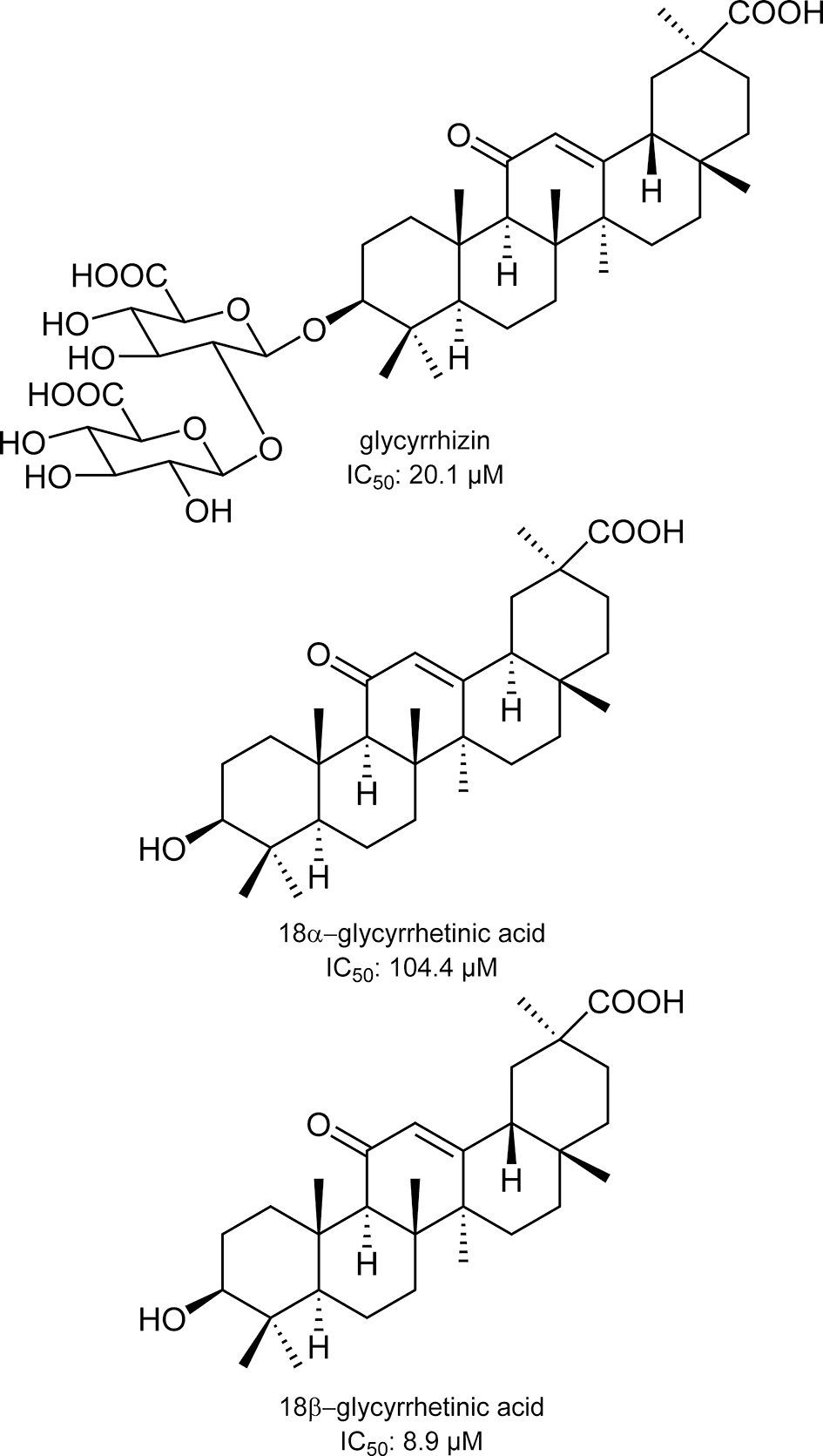
Structures of glycyrrhizin and 18α- and 18β-glycyrrhetinic acid isolated from Hizikia fusiformis.

Structures of moracin M, O, P, and S isolated from Morus alba.
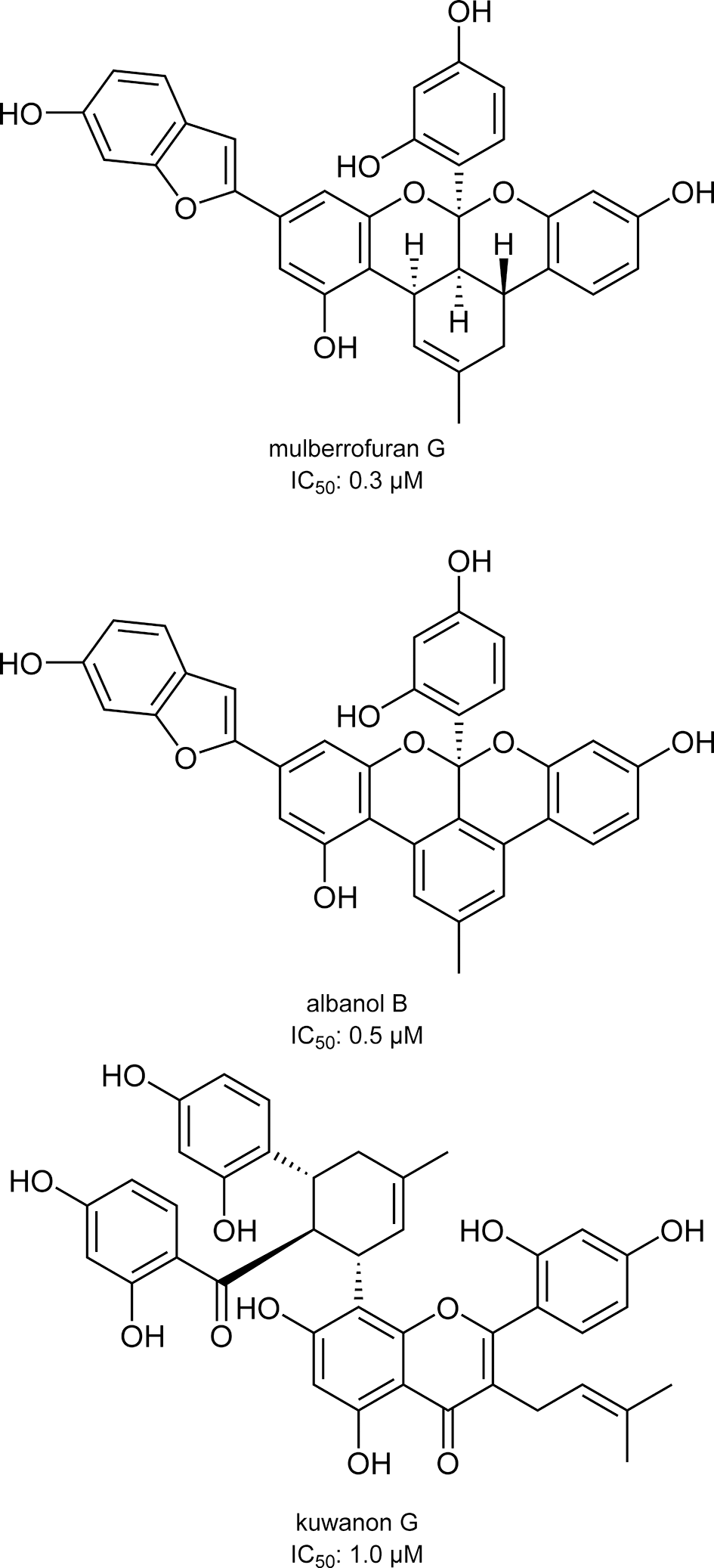
Structures of mulberrofuran G, albanol B, and kuwanon G isolated from Morus alba.

Structures of bavachin, bavachinin, bavachalcone, and isobavachalcone isolated from Cullen c orylifolium.

Structures of rubrofusarin and its derivatives isolated from Cassia obtusifolia.
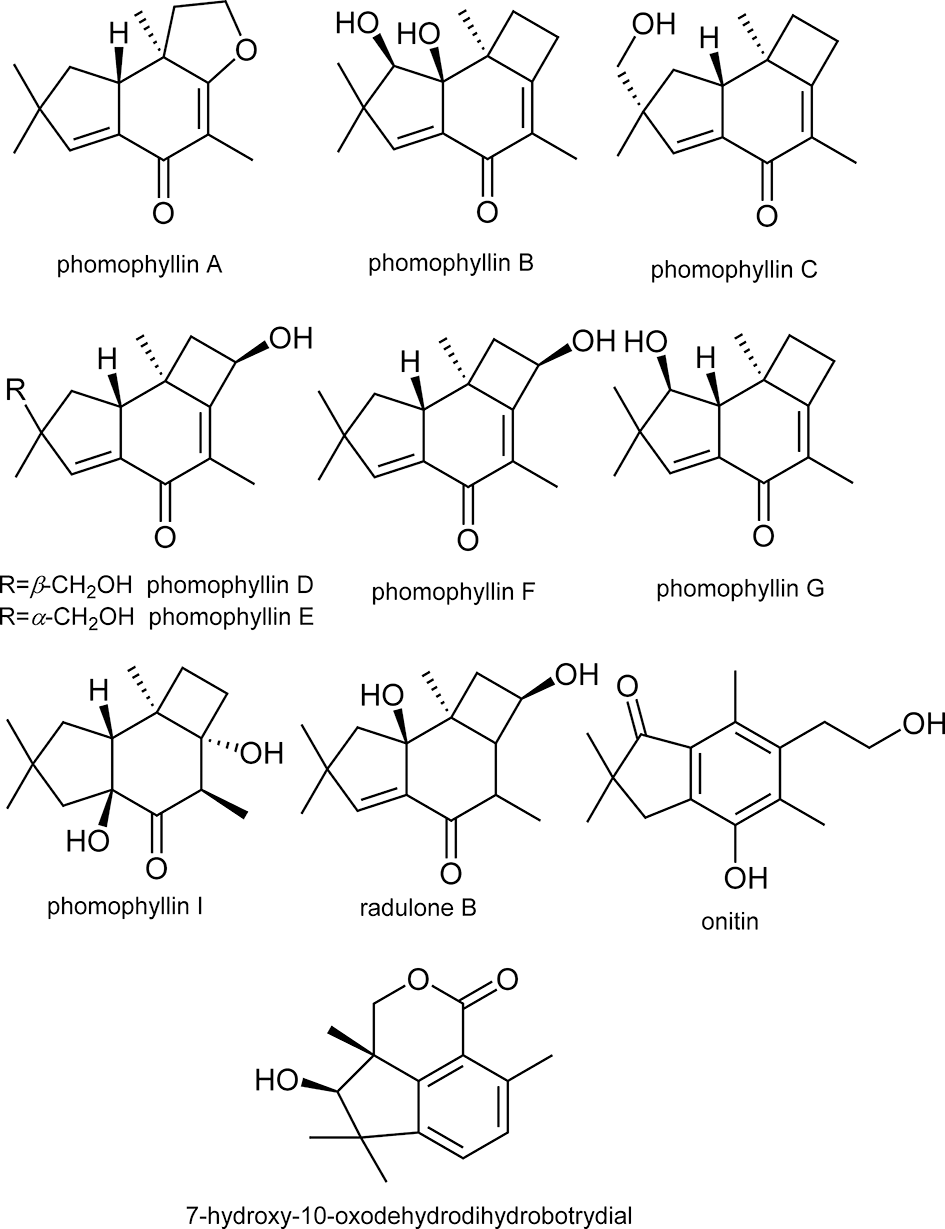
Structures of sesquiterpenes isolated from Phomopsis sp. TJ507A.

Structures of steroidal compounds isolated from Urechis unicinctus.

Structures of xanthones isolated from Garcinia mangostana. Numbers in parenthesis show the inhibitory activities at 100 μM.

Structure of α-mangostin.

Structures of sargahydroquinoic acid, sargachromenol, and sargaquinoic acid isolated from Sargassum serratifolium.

Structures of anthraquinones isolated from Morinda officinalis.

Structures of lupeol and lupenone.

Structures of umbelliferones isolated from Angelica decursiva.

Structures of nobiletin, tangeretin, and sinensetin isolated from citrus peel.

Structures of cornuside and gallotannins isolated from Cornus officinalis.
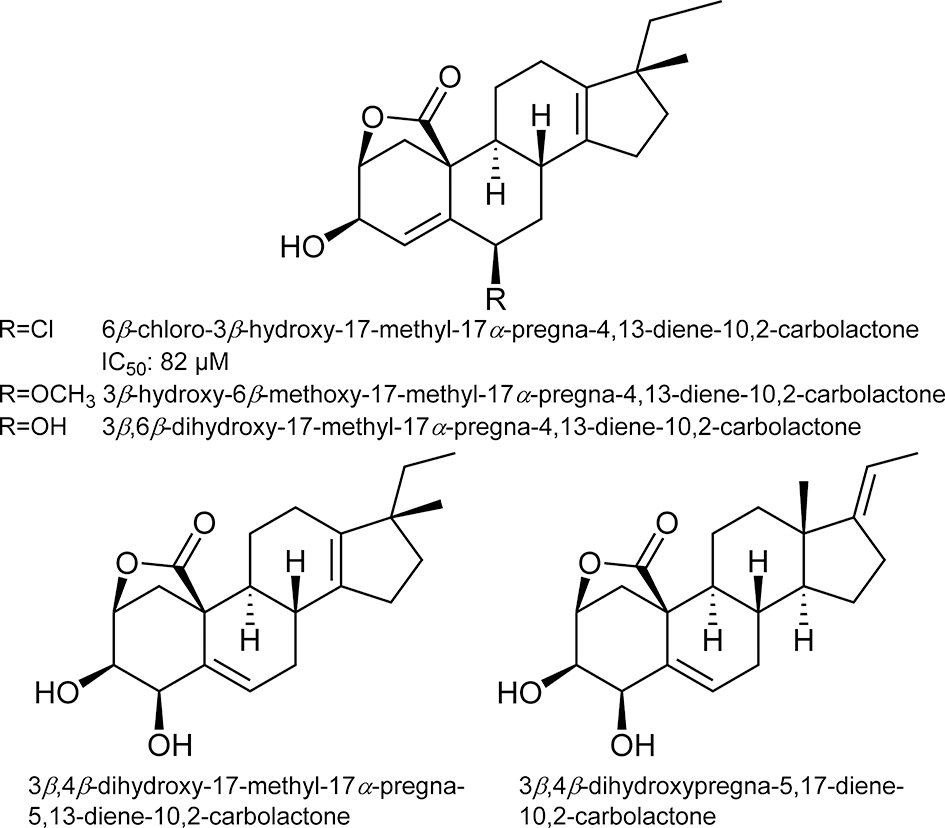
Structures of pregnane-10,2-carbolactones isolated from genus Myrmekioderma.
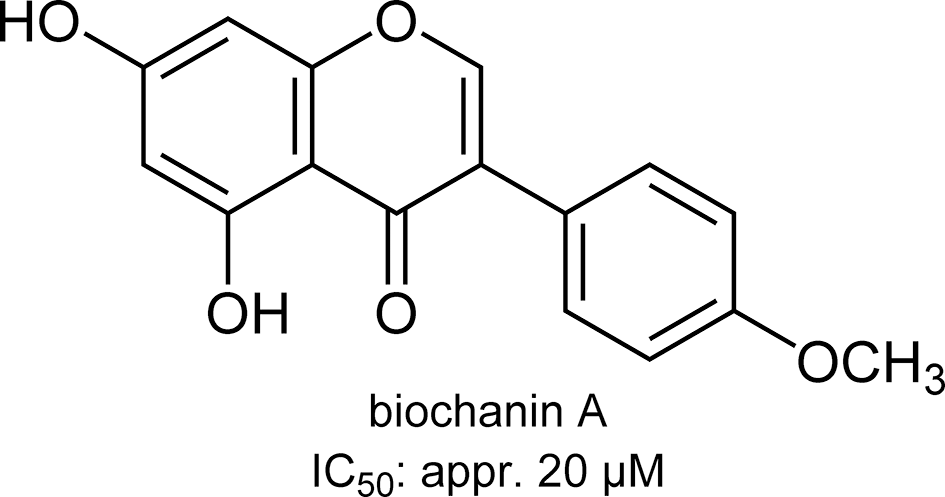
Structure of biochanin A.

Structures of compounds isolated from Juniperus chinensis
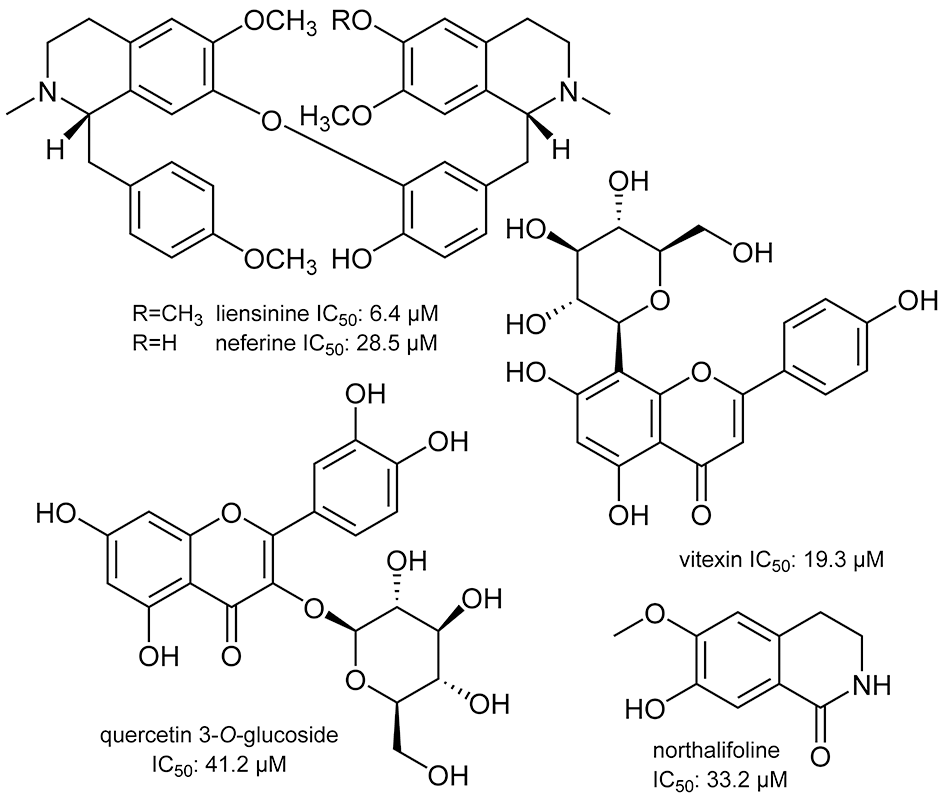
Structures of compounds isolated from Nelumbo nucifera.

Structures of compounds isolated from Buxus macowanii.

Structures of compounds isolated from Lycopodiella cernua.

Structures of polyphenols isolated from Ecklonia cava.

Structure of pistagremic acid isolated from Pistacia integerrima.

Structure of geraniin and corilagin.
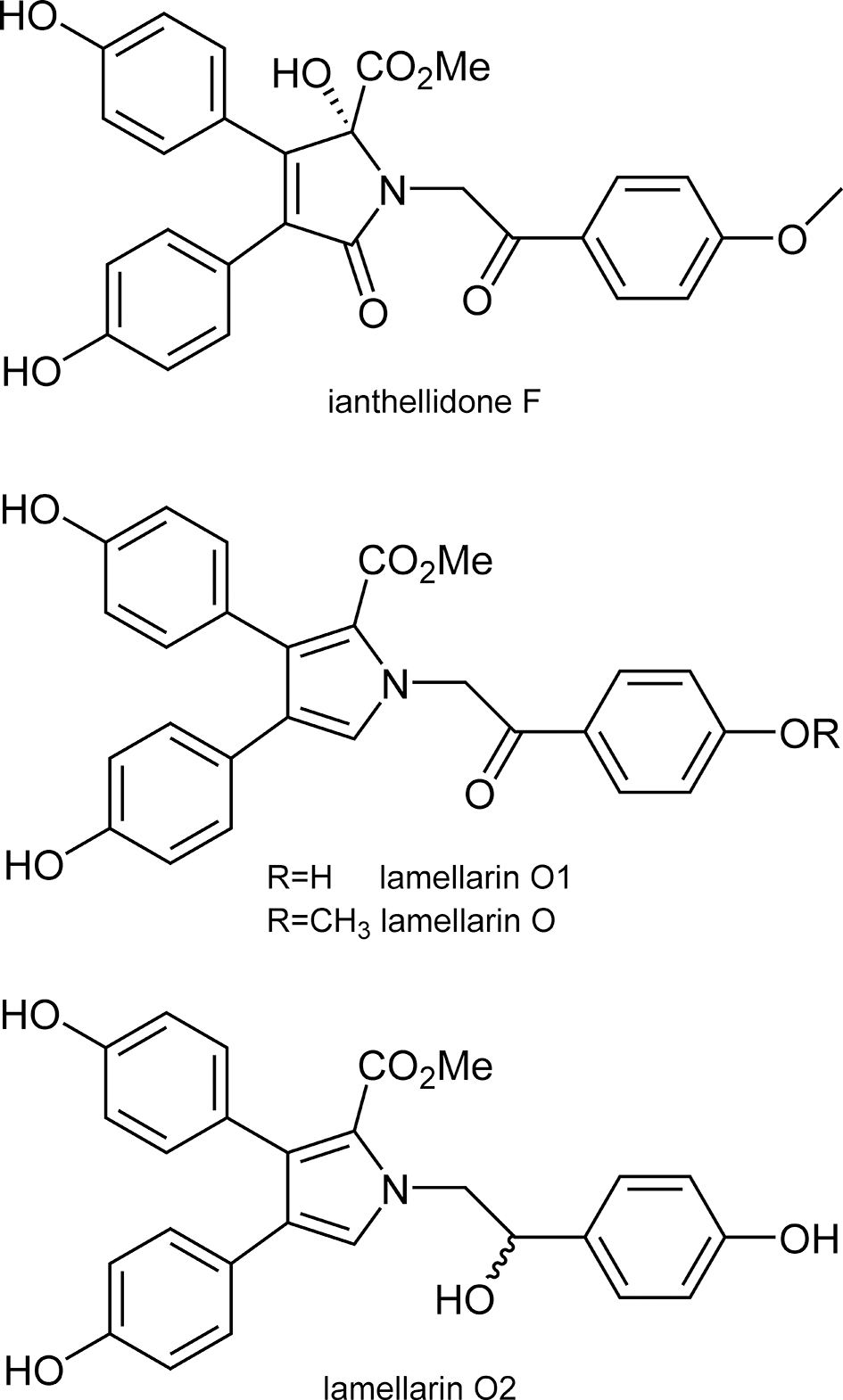
Structures of ianthellidone and lamellarin derivatives isolated from Ianthella sp.

Structures of compounds isolated from Balanophora involucrate.

Structure of p-coumaric acid.

Structures of coumarano-chroman-4-ones and isoflavonoids isolated from Ficus benjamina.

Structures of coumarins as β-secretase inhibitors.

Structure of daedalol C isolated from Daedalea sp.
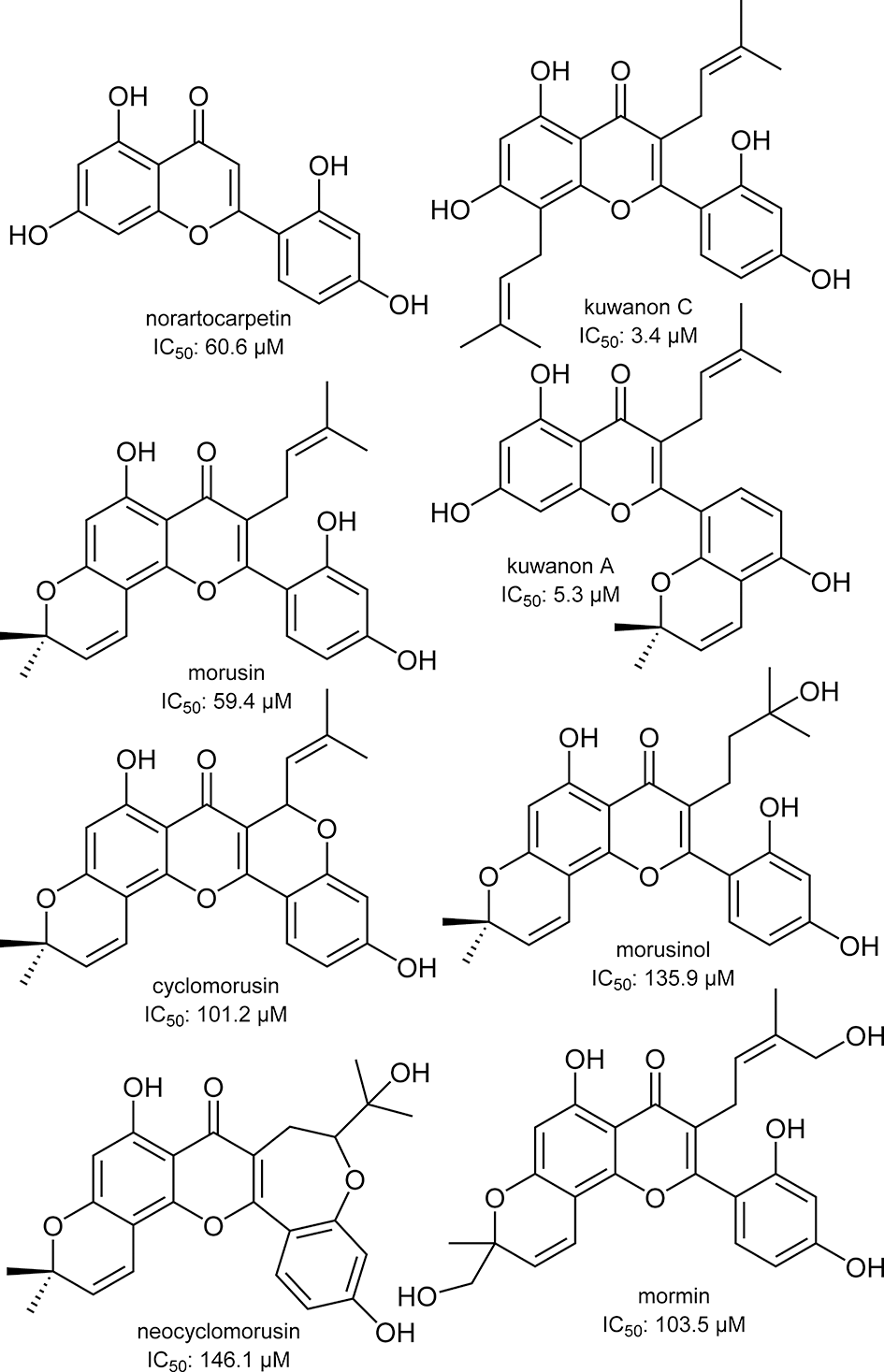
Structure of flavones isolated from Morus lhou.
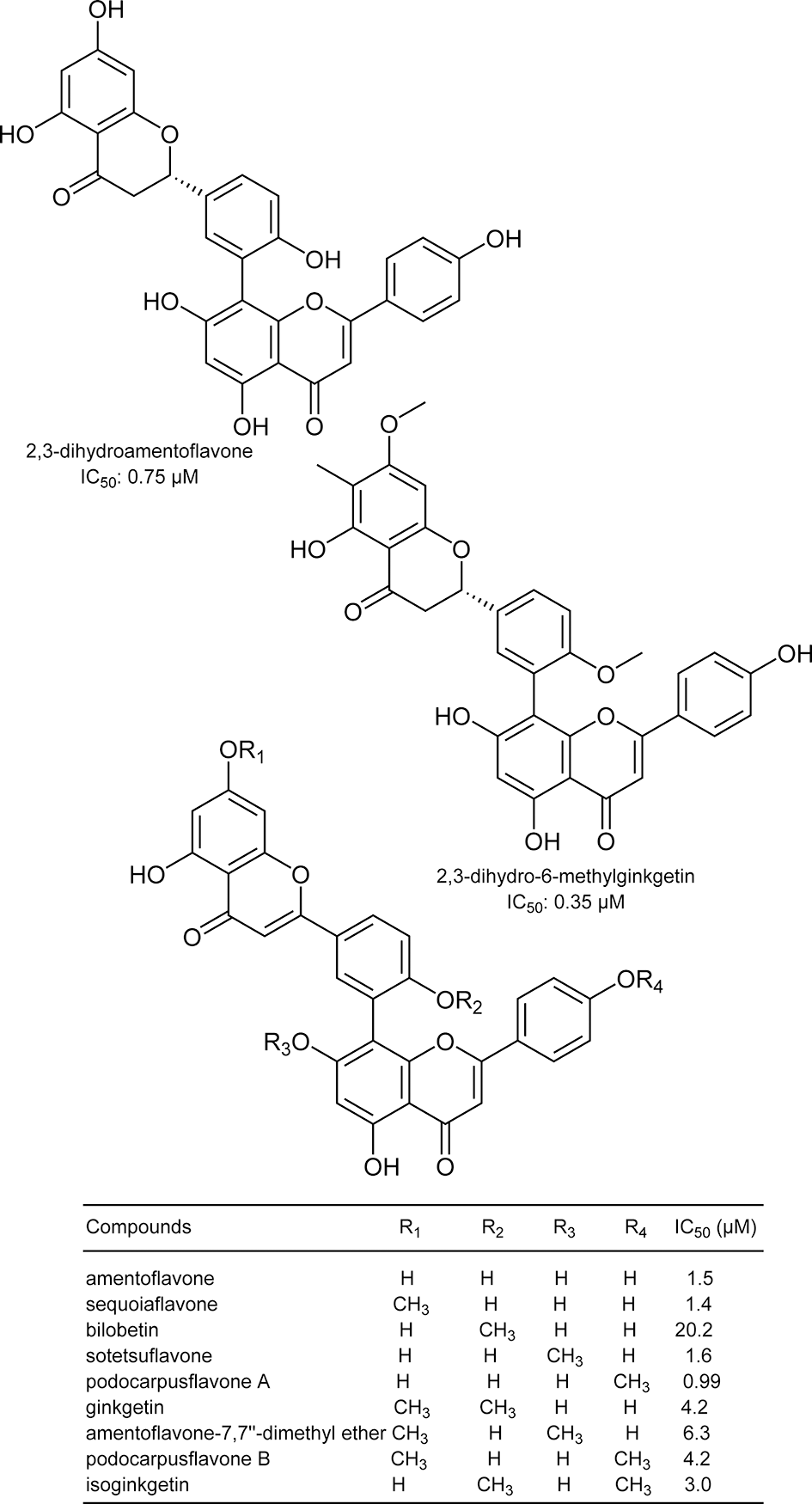
Structures of biflavonoids used in the study. 39

Structures of furanocoumarins isolated from Angelica dahurica.
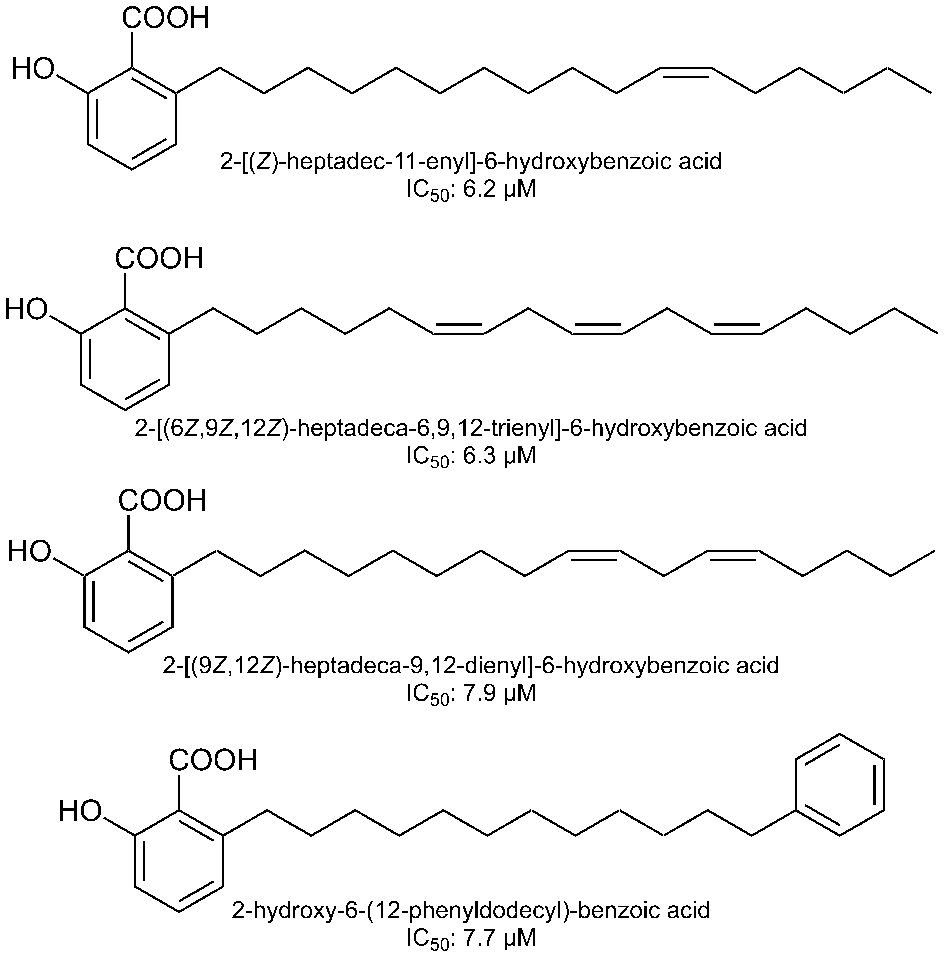
Structures of phenolic acids isolated from Homalomena occulta.
Discussion
The number of reports on β-secretase inhibitors significantly increased after 2015, probably due to the increasing interest in β-secretase as an anti-AD agent. However, the inhibitory activities of inhibitors reported were low, in the order of micromolar, so these inhibitors were not recognized as lead compounds for therapeutic development. However, these low activities can still be advantageous if used as functional foods or materials. Therefore, more reports on the activities of crude extracts are expected.
The reason for the limited number of reports on crude extracts may be the lack of an efficient analysis method for β-secretase inhibition in vitro. Almost all reports used β-secretase assay kits based on the use of a fluorescent resonance energy transfer substrate and fluorescent microplate reader. We have reported that this assay kit may not detect the inhibition activities of crude extracts accurately as the fluorescence from the test sample itself causes strong interference. 42 In this report, we proposed that fluorescent high-performance liquid chromatography for detecting the degraded fluorescent fragment is an ideal method to evaluate the inhibitory activity accurately. Using this strategy, we have already reported on its application to various crude extracts from plant resources to reveal their inhibitory activities and investigate their active constituents. 43 -45 This method is expected to be recognized as an efficient strategy for natural product studies.
Among the β-secretase inhibitors mentioned above, triterpenes, 18β-glycyrrhetinic acid, lupeol and lupenone, and moracin M and S, were determined to be competitive inhibitors. These compounds likely bind to the same site for peptic substrate despite their nonpeptidic structures. In addition, there are no reports on novel peptide compounds that act as β-secretase inhibitors. These points will be clarified in future studies and discovery of novel peptides is another promising strategy to identify β-secretase inhibitors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
