Abstract
Naringin (NRG) is a common dietary flavonoid in citrus fruits and has been documented to possess multiple pharmacological activities, including anti-oxidant, anti-inflammatory, and neuroprotective effects. Naringin is frequently consumed in combination with common clinical drugs. To date, the effects of NRG on cytochrome P450 enzymes have not been fully investigated yet. In this study, the activities of hepatic CYP1A2, CYP2D2, CYP2C9, CYP2C19, and CYP2E1 in rats after the continuous oral administration of NRG (50 and 500 mg/kg) were evaluated using cocktail probe-drug method. The concentrations of 5 probe drugs (phenacetin, dextromethorphan, diclofenac sodium, omeprazole, and chlorzoxazone) in rat plasma were simultaneously determined with a validated HPLC-MS/MS (high performance liquid chromatography-tandem mass spectrometry) method and then used to calculate corresponding pharmacokinetic parameters. Compared with the control group, the AUC(0-t), AUC(0-∞), t 1/2, and C max of each probe drug in treatment groups showed no significant differences. Meanwhile, fluorescence quantitative polymerase chain reaction (FQ-PCR) analysis revealed that NRG did not significantly affect the mRNA expressions of genes CYP1a2, CYP2d2, CYP2c6, CYP2c11, and CYP2e1 in rat liver. Based on these results, it could be concluded that NRG showed no significant effects on the activities and mRNA expressions of tested CYP450 in rats.
Cytochrome P450 enzymes (CYPs) are a series of heme monooxygenases and widely located on the endoplasmic reticulum in hepatocytes. 1 More than 50 kinds of CYP450 isoenzymes have been identified in human body among which CYP3A4, CYP2D6, CYP2C9, CYP1A2, CYP2C19, and CYP2E1 are responsible for biotransformation of 80% clinical drugs. 2 Drug metabolism can be affected by other co-administered drug or food that have potential inhibitory or inducible effects on CYP450 isoenzymes, thus causing drug-drug or drug-food interactions. 3 The occurrence of CYP-mediated drug interactions were up to 80% among elder population in Canada and more than 73% among adults in United Kingdom. 4,5 Accordingly, it is crucial to fully investigate a compound’s potential inhibitory or inducible effect on the activity of CYP450 enzymes in the process of new drug development.
Naringin (NRG), chemically 5,7,4′-trihydroxyflavanone-7-O-rhamnoglucoside, is a predominant flavanone glycoside in citrus fruits. 6,7 Previous pharmacological studies have demonstrated that NRG possessed multiple bioactivities, including anti-oxidant, anti-inflammatory, and neuroprotective effects. 8 -11 In vivo and in vitro studies revealed that NRG had excellent effects on relieving cough and eliminating phlegm, suggesting that NRG was a promising potential drug in the treatment of respiratory diseases. 12,13
Flavonoids were considered to activate multiple nuclear receptors, including aromatic hydrocarbon receptor (AhR), 14 pregnane X receptor, 15 and constitutive androstane receptor. 16 The activation generally shows inductive effects on CYPs and thereby change the catalytic capacity of these enzymes, which play an important role in drug metabolism. 17 As a dietary flavonoids, NRG was frequently consumed in combination with common clinical drugs. 18 Grapefruit juice, which contained abundant NRG, was experimentally documented to alter the pharmacokinetic profiles of multiple drugs. 19 Naringin was considered as a potential modulator in grapefruit juice. 20 In vitro microsomal incubation experiments suggested that NRG was an inhibitor of CYP3A and CYP2C9. 21 Moreover, the activity of CYP1A2 in mice was found to be inhibited after successive oral administration of NRG for 7 days. 22 Based on the reported results, it is rational to believe that NRG could alter the activities of CYP450 to some extent. To date, many studies have focused on the effect of NRG on CYP3A4, 23,24 while the effects of NRG on other major CYPs (CYP1A2, CYP2D6, CYP2C9, CYP2C19, and CYP2E1) have not been evaluated. Given these CYPs also play a predominant role in drug metabolism, it is of great importance to assess the potential inhibition or induction effects of NRG on them.
Rodent CYP1A2, CYP2D2, CYP2C6, CYP2C11, and CYP2E1 are homologous with human CYP1A2, CYP2D6, CYP2C9, CYP2C19, and CYP2E1, respectively. 25 The purpose of this study was to investigate the effects of NRG on the activities and mRNA expressions of hepatic CYP1A2, CYP2D2, CYP2C6, CYP2C11, and CYP2E1 in rats, with the method of cocktail probe drugs and FQ-PCR. Phenacetin, dextromethorphan, diclofenac sodium, omeprazole, and chlorzoxazone were adopted as probe drug for CYP1A2, CYP2D2, CYP2C6, CYP2C11, and CYP2E1, respectively. After continuous oral administration of NRG for 7 days and subsequent injection of cocktail probe drugs, plasma samples were collected from rats and analyzed with a developed HPLC-MS/MS method. To evaluate the effects of NRG on the mRNA expressions of hepatic CYPs, liver samples were obtained from rats with continuous oral administration of NRG for 7 days, and then detected using the FQ-PCR technique. Obtained results would be useful in clarifying the effects of NRG on CYP450 enzymes.
Materials and Methods
Chemicals and Reagents
The reference standards NRG (purity: 94.7%), phenacetin (purity: 98.7%), dextromethorphan hydrobromide (purity: 94.8%), diclofenac sodium (purity: 99.0%), chlorzoxazone (purity: 99.9%), antipyrine (purity: 100.0%), and glipizide (purity: 99.7%) were obtained from the National Institute for the Control of Pharmaceutical and Biological Products (Beijing, China). Omeprazole (purity: 99.0%) was purchased from Shanghai Yuanmu Biotechnology Co., Ltd (Shanghai, China). Chloral hydrate was purchased from Damao Chemical Reagent Factory (Tianjin, China). Heparin sodium was obtained from Aladdin Reagent Co., Ltd (Shanghai, China).
HPLC grade methanol and acetonitrile were purchased from Honeywell B&J (NJ, United States). Mass spectrometry (MS) grade formic acid were acquired from Sigma-Aldrich (St. Louis, MO, United States). Deionized water was prepared using a Milli-Q purification system (Millipore, Bedford, MA, United States). Naringin powder (purity: 98.4%), for oral administration, was produced in Guangdong Medi-World Pharmaceutical Co., Ltd (Foshan, China).
Animals
Male Sprague-Dawley rats (230-270 g) were acquired from Guangdong Medical Laboratory Animal Center (Guangzhou, China, animal certificate number: SCXK(Yue) 2013-002). Animals were maintained with feed and water available ad libitum in a breeding room with an ambient temperature at 20 ℃ to 25 ℃, a relative humidity of 55% ± 15%, and 12-hour light-dark cycles. All experimental procedures and protocols were approved by the Animal Ethics Committee of the School of Life Sciences in Sun Yat-sen University.
Pharmacokinetic Study
A total of 21 rats were randomly assigned into 3 groups, namely control (CON) group, low-dose naringin (NRG-L) group, and high-dose naringin (NRG-H) group. The CON group was intragastrically treated with deionized water for 7 days. The NRG-L and NRG-H group were orally administrated with low-dose (50 mg/(kg·d)) and high-dose (500 mg/(kg·d)) NRG for 7 days in succession, 26 -28 respectively.
After the last dose, animals were fasted for 24 hours with water available ad libitum. Then, a cocktail solution, containing phenacetin (4 mg/kg), dextromethorphan (6 mg/kg), diclofenac sodium (0.5 mg/kg), omeprazole (3 mg/kg), and chlorzoxazone (1 mg/kg), was injected into the caudal vein of all rats. 29,30 Blood samples (300 µL) were collected from retro orbital plexus into heparinized tubes at predose (0 minute), 5, 10, 20, 30, 45, 60, 90, 120, 180, 270, 360, and 480 minutes after the injection. Plasma samples were obtained by immediate centrifugation (5000 rpm for 30 minutes) and stored at −80 ℃ until analyzed.
Sample Preparation
An aliquot of 100 µL plasma sample was mixed with 10 µL internal standard (IS) spiking solution (containing antipyrine and glipizide with the concentration of 500 ng/mL) and 290 µL acetonitrile. Then, samples were vortex-mixed for 1 minute and centrifuged at 15 000 rpm for 10 minutes at 4 ℃. Finally, an aliquot of 10 µL supernatant was injected into the HPLC-MS/MS system for analysis.
Preparation of Calibration Standards and Quality Control (QC) Samples
The stock solutions of phenacetin, dextromethorphan, diclofenac sodium, omeprazole, chlorzoxazone, antipyrine (IS 1), and glipizide (IS 2) were individually prepared as 1.0 mg/mL in methanol. All stock solutions were stored at 4 ℃ and brought to room temperature before use. Working standard and IS spiking solutions of each compound were prepared from the stock solutions by serial dilution with 70% acetonitrile. Obtained solutions were vortex-mixed with blank plasma to yield calibration standards of 2, 5, 10, 25, 50, 100, 200, 375, and 500 ng/mL for each analyte. Another separate stock of each analyte was prepared and used for preparation of quality controls (QC). QC samples were prepared at final concentrations of 5, 100, and 375 ng/mL in the same manner as the calibration standards. Calibration standards, QC samples, and IS spiking solution were all prepared accompanying each analytical batch.
LC-MS/MS Condition
Samples were analyzed with Agilent 1200 RRLC tandem 6410 triple quadrupole mass spectrometers with an electrospray ionization source (ESI; Agilent Technology, Santa Clara, CA, United States). Chromatographic separation was carried out on an Agilent Zorbax Eclipse XDB-C18 column (4.6 mm × 50 mm, 1.8 µm, Agilent, United States) at 40 ℃. Isocratic elution was performed for 4.5 minutes using acetonitrile and water with 0.1% formic acid (v / v) at the ratio of 70:30, with the flow rate of 0.3 mL/min. The HPLC effluent was directly introduced to mass spectrometer without splitting.
Mass spectrometry detector was operated in multiple reaction monitoring mode at unit mass resolution with a dwell-time of 200 ms for all analytes. To obtain optimal response, phenacetin, dextromethorphan, diclofenac sodium, omeprazole, and corresponding IS antipyrine (IS 1) were determined in positive ionization mode, while chlorzoxazone and its IS glipizide (IS 2) were detected in negative ionization mode. The MS/MS transitions and corresponding fragmentors, collision energies (CEs) were optimized as follows: m/z 180.1→110.0 for phenacetin (fragmentor: 110 V, CE: 20 eV), m/z 272.2→171.1 for dextromethorphan (fragmentor: 150 V, CE: 44 eV), m/z 296.2→250.0 for diclofenac sodium (fragmentor: 100 V, CE: 9 eV), m/z 346.1→198.1 for omeprazole (fragmentor: 90 V, CE: 9 eV), m/z 189.3→56.1 for antipyrine (fragmentor: 130 V, CE: 38 eV), m/z 167.9→132.1 for chlorzoxazone (fragmentor: 120 V, CE: 19 eV), and m/z 444.2→319.0 for glipizide (fragmentor: 175 V, CE: 20 eV). The ESI ion source parameters were set as follows: Capillary 4000 V, gas flow 10 L/min, nebulizer 25 psi, and gas temperature 350 ℃. And other instrument parameters were adopted based on the recommended values. Agilent Masshunter software (version 1.0; Agilent Technology, Santa Clara, CA, United States) was used for data acquisition and processing.
Method Validation
The method validation was conducted according to the Guidance for Bioanalytical Method Validation issued by Chinese Pharmacopoeia Commission in 2015. 31
The selectivity of this method was evaluated by comparing responses of target analytes in blank plasma (from 6 different sources), standard-spiked plasma, and postdosing plasma samples. Calibration curves of the above-mentioned analytes were constructed and fitted by linear least-squares regression analysis to plot the IS-normalized responses against corresponding concentrations. Precision and accuracy were determined by repeated analyses of lower limit of quantification (LLOQ) and QC samples (n = 6) at concentrations of 2, 5, 100, and 375 ng/mL on 3 separate days. Intra- and interday precisions were expressed by the relative standard deviation (RSD, %), while the accuracy was expressed as relative error (RE, %). The extraction recoveries of analytes were evaluated by comparing the peak area of extracted 3 levels QC samples to that of the analytes spiked to the blank sample extracts at the corresponding concentration. The matrix effect was calculated by comparing the IS-normalized responses of analytes spiked into blank plasma extracts with that acquired in the neat solution at an equivalent concentration.
Effects of NRG on mRNA Expression of CYP Isozymes in Rats
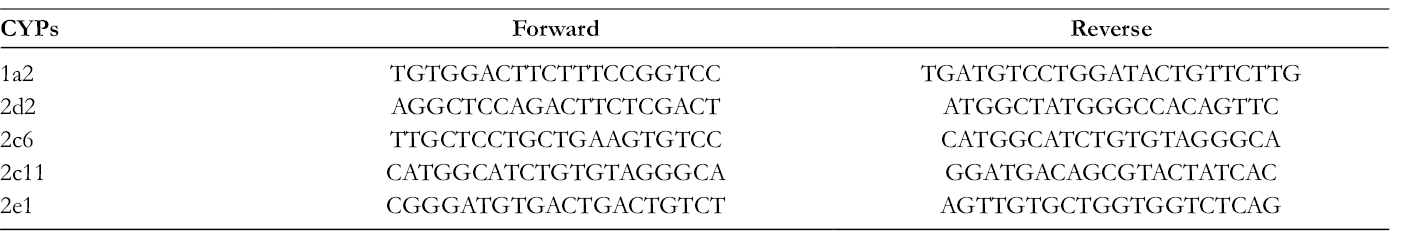
The other 21 rats were randomly divided into 3 groups (i e, CON, NRG-L, and NRG-H) and treated same as the animals in the above-mentioned pharmacokinetic study. After the administration for 7 consecutive days, the rats were fasted for 24 hours and then sacrificed. Liver samples were removed from rats. Total RNA was isolated from rat liver samples according to the manufacturer’s instruction. The quality of the isolated RNA was evaluated using the 260/280 nm absorbance ratio (1.8-2.0 indicates a highly pure sample). Subsequently, 1 µg of RNA from each sample was reverse transcribed to cDNA using a GoScript RNA RT Reagent Kit (Promega, United States) in accordance with the recommended protocol. The total RNA concentration of each reaction was 200 µg/mL. FQ-PCR reactions were carried out in a final volume of 10 µL containing SYBR Green mix 5 µL, cDNA 2 µL, F-primer 0.5 µL, R-primer 0.5 µL, and distilled water 2 µL. The amplification conditions were as follows: initial denaturation at 95 ℃ for 10 minutes, followed by 45 cycles of denaturation at 95 ℃ for 10 seconds, annealing at 60 ℃ for 20 seconds, extension at 72 ℃ for 20 seconds. The sequences of the forward and reverse primers used in this experiment are shown in Table 1. 32
Sequences of Primers for FQ-PCR Analyses.
Data Analysis
The pharmacokinetic parameters were calculated with DAS (Drug and statistics) software (Version 3.0, Shanghai University of Traditional Chinese Medicine, China). The mean concentration-time curves were plotted using GraphPad Prism (Version 7.0; GraphPad Software Inc., La Jolla, CA, United States). Obtained parameters of drug-treated groups were compared with those of CON group using one-way ANOVA and Dunnett’s test in GraphPad Prism 7.0. P-value less than 0.05 was considered statistically significant.
Results and Discussion
Method Development and Instrumental Optimization
In this study, we compared liquid-liquid extraction and protein precipitation to obtain the optimum extraction method. As a result, the processed method of protein precipitation with acetonitrile showed simple operation, stable recovery and good repeatability. To obtain good separation and abundant signal responses, different solutions were investigated to optimize the chromatographic conditions. Finally, acetonitrile and water with 0.1% formic acid were selected as mobile phases. Moreover, the MS/MS parameters were optimized to acquire abundant responses for target analytes. The following MS/MS transitions were selected for quantification: m/z 180.1→110.0 for phenacetin, m/z 272.2→171.1 for dextromethorphan, m/z 296.2→250.0 for diclofenac sodium, m/z 346.1→198.1 for omeprazole, m/z 189.3→56.1 for antipyrine (IS 1), m/z 167.9→132.1 for chlorzoxazone, and m/z 444.2→319.0 for glipizide (IS 2), respectively.
Method Validation
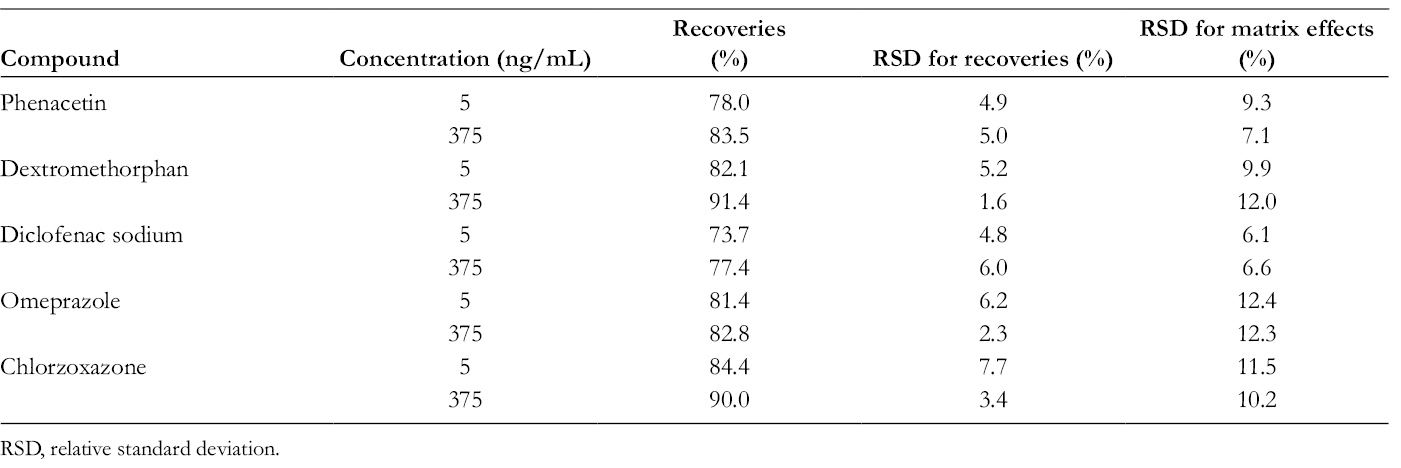
In this study, sharp and fine peaks were obtained for all analytes with optimized chromatographic conditions (shown in Figure 1). No significant interference was observed. Calibration curves showed good linearity over the range of 2 to 500 ng/mL for phenacetin (r = 0.998), dextromethorphan (r = 0.996), diclofenac (r = 0.997), omeprazole (r = 0.999), and chlorzoxazone (r = 0.996). As presented in Table 2, the intraday and interday precision of LLOQ and QC samples were all within 11.4%, and the accuracy ranged from 91.5% to 113.2%. The extraction recoveries for these analytes were stable and greater than 73.7% (shown in Table 3). Relative standard deviation for matrix effects of each analyte at assigned concentration level is within 15% (shown in Table 3). These results revealed that the developed methods were reliable and reproducible for the quantitative analysis or mentioned probe drugs.

Extracted ion chromatograms of 6 probe drugs and internal standards in rat plasma: (a) blank plasma, (b) blank plasma spiked with cocktail drugs and internal standard, and (c) plasma sample collected 1 hour after the injection of cocktail drugs.
Intra- and Interday Accuracies and Precisions of Phenacetin, Dextromethorphan, Diclofenac Sodium, Omeprazole, and Chlorzoxazone in Rat Plasma.

RE, relative error; RSD, relative standard deviation.
Recoveries and Matrix Effects for Phenacetin, Dextromethorphan, Diclofenac Sodium, Omeprazole, and Chlorzoxazone in Rat Plasma.
RSD, relative standard deviation.
Effect of NRG on the Activities of CYPs
With the developed method, the concentration of target analytes in plasma samples collected after the injection of cocktail drugs were determined. Mean concentration-time curves of CON group, NRG-L group, and NRG-H group for 5 probe drugs were drawn with GraphPad Prism 7.0 (shown in Figure 2). Pharmacokinetic parameters were calculated and analyzed with DAS 3.0 and SPSS 18.0 software, respectively (presented in Table 4).

Mean concentration-time curves of phenacetin (a), dextromethorphan (b), diclofenac sodium (c), omeprazole (d), and chlorzoxazone (e) (mean ± SD, n = 7).
Pharmacokinetic Parameters of Phenacetin, Dextromethorphan, Diclofenac Sodium, Omeprazole, and Chlorzoxazone in Rat Plasma After the Injection of Cocktail Drugs (n = 7).
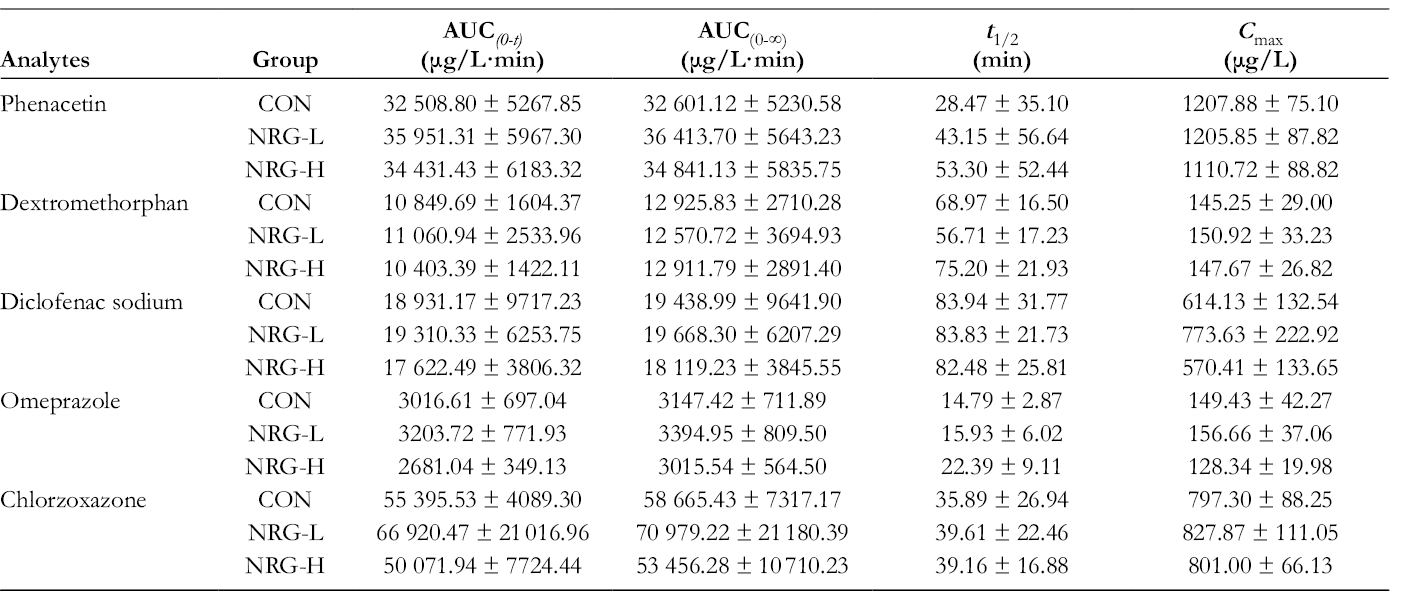
As shown in Figure 2, after the continuous gavage with NRG for 7 days, the mean concentration-time curves of phenacetin, dextromethorphan, diclofenac sodium, omeprazole, and chlorzoxazone in the treated group were similar to those in CON group. Meanwhile, the AUC(0-t), AUC(0-∞), t 1/2, and C max shown no significant differences (P > 0.05) compared with the CON group, suggesting that NRG exerted no significant effects on the metabolic reaction mediated by CYP1A2, CYP2D2, CYP2C9, CYP2C19, and CYP2E1.
Naringin and its aglycone are primary flavanone glycosides in citrus fruits, especially in grapefruit. 24 Fuhr et al 33 evaluated the effects of grapefruit juice, NRG and naringenin on the activity of CYP1A2, using caffeine as a probe substrate. In vitro experiments revealed that NRG was a potent competitive inhibitor of caffeine 3-demethylation catalyzed by CYP1A2. However, grapefruit juice (containing 0.5 g/L NRG) and naringenin just showed slight effects on the in vivo clearance of caffeine. That is to say, NRG and naringenin barely affected the in vivo activity of CYP1A2, aligned with the results obtained in this work. As to CYP2C9, in vitro assay indicated that naringenin was an important inhibitor of CYP2C9-catalyzed diclofenac 4′-hydroxylation. 21 Nevertheless, no remarkable changes were observed in the pharmacokinetic profile of diclofenac in rats after the successive oral administration of NRG for 7 days. Thus, there existed a gap between the in vitro and in vivo results, warning that extrapolation from in vitro results to in vivo studies should be cautious.
Effects of NRG on Rat Liver CYP mRNA Expression Levels
In this study, liver tissue were collected as samples for the analysis of CYP mRNA expression levels. It should be noted that there exist remarkable individual differences in the contents and activities of CYPs, aligned with the reported results. 34
Flavonoids are an important class of polyphenols and widely distributed in the diets. 35 The reported results suggested that flavonoids could activate multiple nuclear receptors, leading to the mRNA expression changes of CYPs. 14 -16 These changes would alter drug metabolism catalyzed by CYPs 17 and ultimately resulted in drug interactions. Therefore, it is important to investigate the effects of dietary flavonoids on CYPs. Given multiple healthy benefits, NRG is increasingly used as dietary supplements and frequently consumed in combination with drugs. However, the effects of NRG on most CYPs are still unknown, except for CYP3A4. In this work, FQ-PCR was used to detect the expression changes of the rat liver genes CYP1a2, CYP2d2, CYP2c6, CYP2c11, and CYP2e1 after the continuous administration of NRG (shown in Figure 3). Compared with the CON group, no significant differences (P > 0.05) were observed in the mRNA expression level of the above-mentioned CYPs in NRG-treated groups, which were consistent with the pharmacokinetic results.
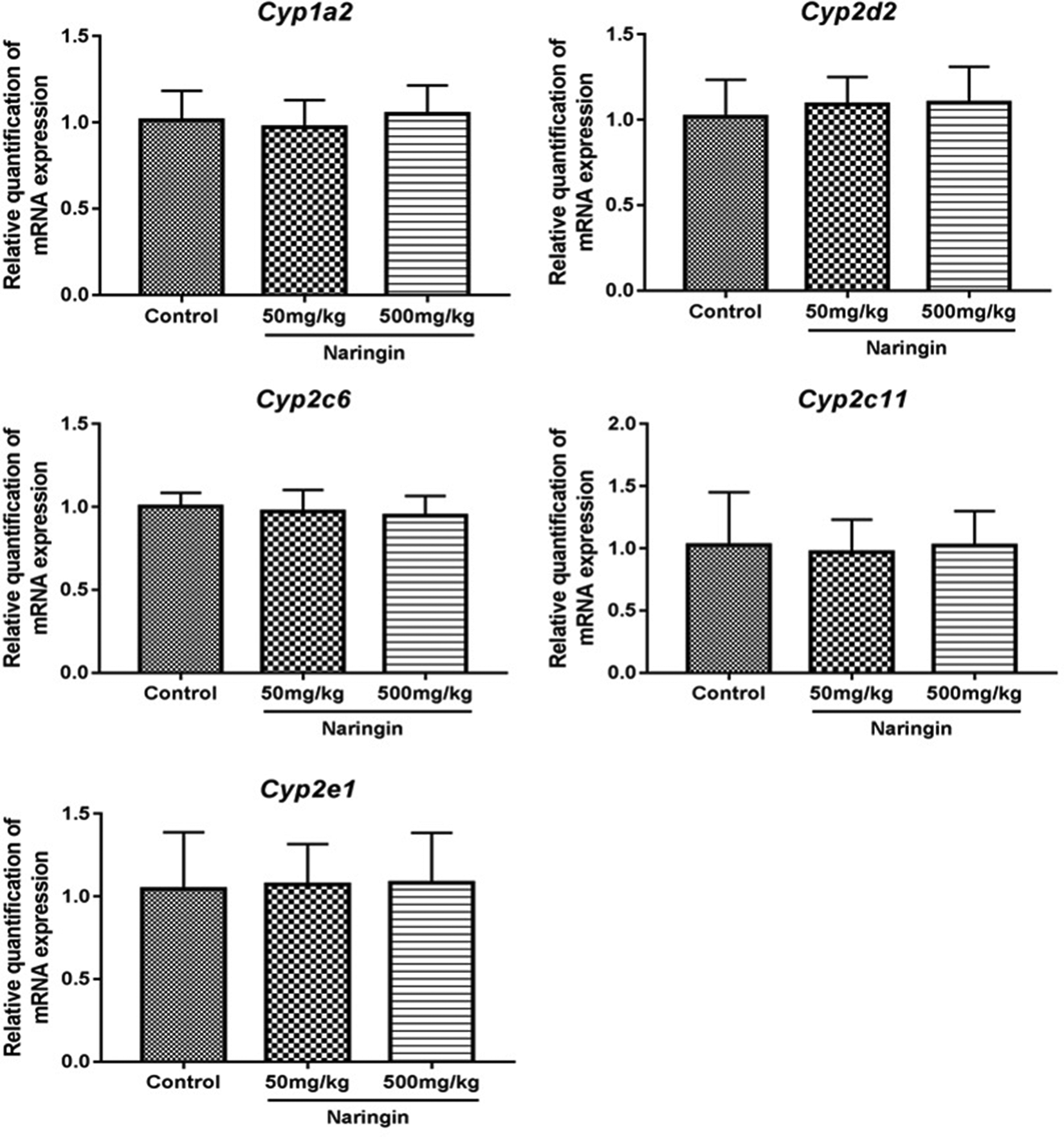
Effect of naringin on mRNA expression of CYP genes in rat liver (mean ± SD, n = 7).
Aromatic hydrocarbon receptor is a ligand-activated transcription factor and plays an important role in the induction of CYPs. 36 In an in vitro assay, marked induction of AhR-dependent transcriptional activity was observed with the treatment of naringenin. 37 In addition, an increase in CYP 1a2 transcription in pigs was found to be associated with the activation of AhR. 38 However, in this study, the mRNA expression of CYP1a2 in rat liver barely changed after the long-term intervention with NRG (a glycoside of naringenin), which was inconsistent with the reported studies. A possible explanation is that species variation may lead to the difference in the regulation of CYP1a2 transcription. More work is still required to explain this phenomenon.
Conclusion
In this study, the effects of NRG on the activity and mRNA expression of 5 CYP isozymes in rats were evaluated with cocktail drugs and FQ-PCR, respectively. Compared with the CON group, pharmacokinetic parameters and mRNA expression level obtained in NRG-treated groups showed no significant differences (P > 0.05) after the continuous administration of NRG for 7 days. Obtained results indicated that NRG had no significant effects on the activity and mRNA expression of CYP1A2, CYP2D2, CYP2C9, CYP2C19, and CYP2E1 in rats. These results would be helpful for further clinical use of NRG.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the National Natural Science Foundation of China (No. 31571830) and Applied Science and Technology R&D Special Fund Project of Guangdong Province (No. 2016B020239003).
