Abstract
Continuing our exploration for dual functions antidiabetic and antioxidant agents from Myanmar medicinal plant, a new carbazole-pyranocoumarin conjugate, carbazomarin-C (
Clausena excavata Burm. f. is commonly found in the tropical and subtropical regions such as India, China, and Southeast Asia countries. The plants are a member of Rutaceae family and they are in a form of wild shrubs. They are known to have medicinal properties since its leaves, twigs, and roots are widely used for the traditional treatment of cold, fever, wound, abdominal pain, snake-bite, a preliminary stage of AIDS, and skin diseases. 1 Previous phytochemical analyses found that C. excavata possesses an abundant amount of coumarins, 2 -4 carbazole alkaloids, 5 and a few limonoids. 6 The coumarins isolated from this plant raised the writers’ attention due to its bioactive properties. For instance, clauslactones A to J which were isolated from the leaves exhibited tumor promotion inhibitory effects. Nordentatin showed antibacterial and antioxidant properties, while pyranocoumarin and clausenidin which were isolated from roots displayed an anti-HIV-1 activity. 4,7
Diabetes mellitus (DM) is one of the complex chronic illness which demands constant medical checkup. As a consequence, many strategies are already developed in order to reduce the multifactorial risk through glycemic control.
8
Elevated plasma glucose causes overproduction of free radicals and other reactive oxygen species that destroy cells through oxidative stress, which supports the goal of developing antidiabetic drugs with radical scavenging. Dual function agents which have both antidiabetic (α-glucosidase inhibitor) and radical scavenging capacities are particularly relevant for the treatment of T2DM (Type 2 Diabetes Mellitus) and its complications. In this study, we searched for an antidiabetic and antioxidant agent having a dual mechanism from a medicinal plant.
9
Here, we have been isolated 1 new carbazomarin-C (
1H (600 MHz), 13C (151 MHz) NMR and HMBC Spectral Data of 1 in CDCl3.
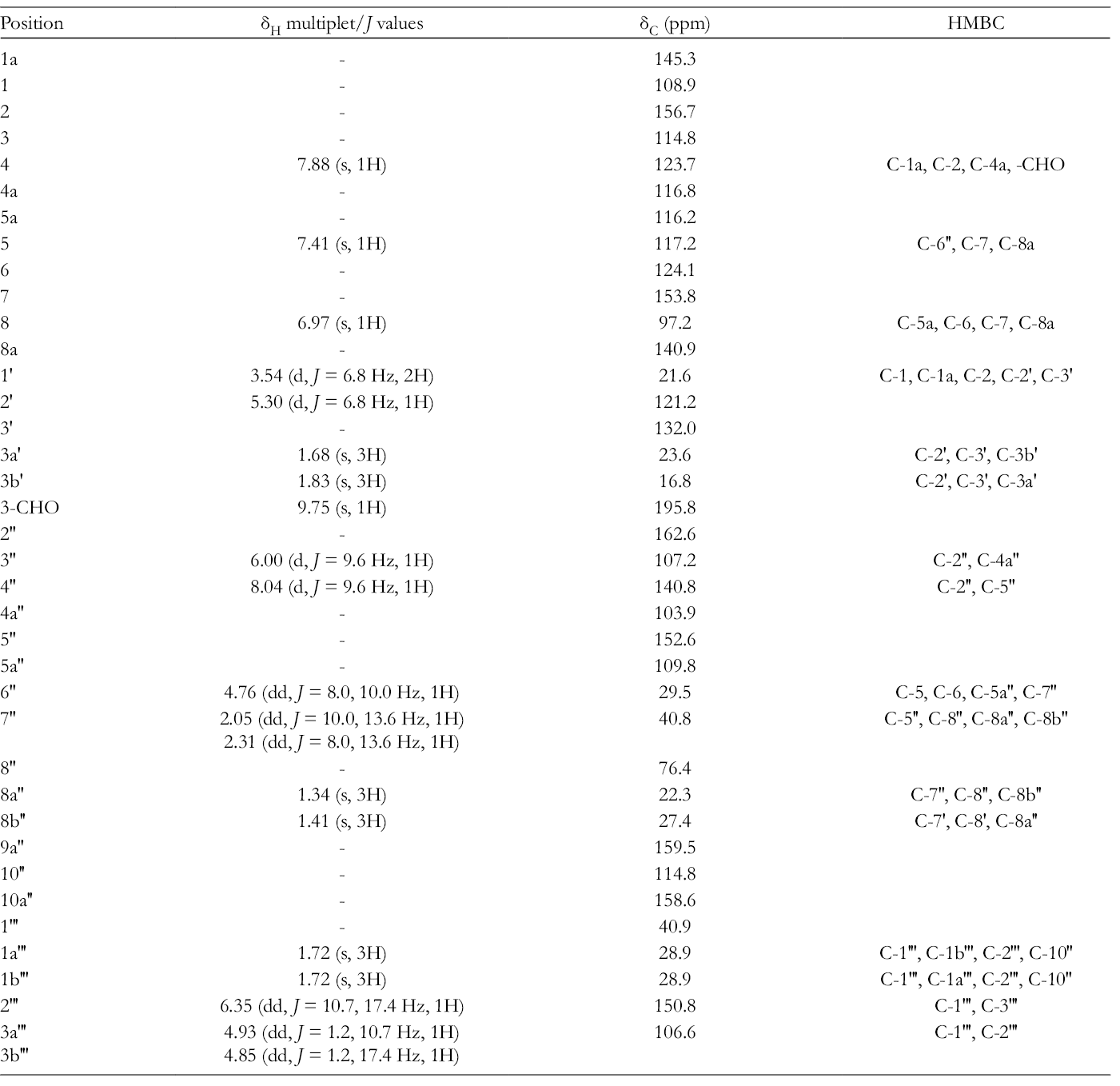

COSY (black bold), key HMBC (blue), and NOESY (red) correlation of 1.
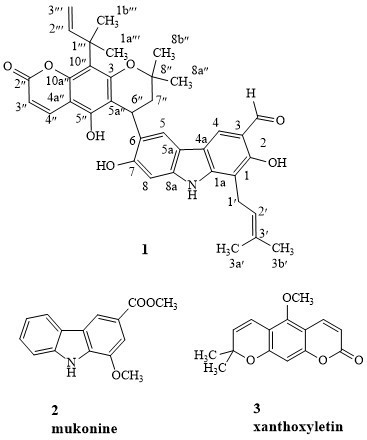
The structure of Clausena excavata’s isolated compounds.
Careful analyses were carried out to determine the physicochemical properties and spectroscopic data. Later on, a comparison step was carried out with the previously reported literatures where 2 known compounds were identified as mukonine
2,6
and xanthoxyletin
3,12
as shown in Figure 2. All of the isolated compounds were then undergone examination to measure antidiabetic activity. The examination was done by using yeast α-glucosidase inhibitory assay. Meanwhile, in order to measure the antioxidant activity, DPPH assay (Table 2) was carried out. There was a potent inhibition demonstrated by the isolated compounds
α-Glucosidase Inhibitory and the Isolated Compounds’ (1-3) Radical Scavenging Activities.

NI, no inhibition.
Experimental
Plant Material
The collection of C. excavata was done in Myanmar, precisely on Pyin Ma Nar Township. The plants’ substantial (voucher specimen MU-22032018) were collected on October 2016 under the authentication of Prof. Soe Myint Aye. Prof. Aye works as a botanist in the Botany Department of Mandalay University in Myanmar. 4
Extraction and Isolation
The roots sample of C. excavata (3.6 kg) was air-dried before finally being extracted with 95% EtOH (12.0 L) for 14 days under ambient temperature. After removing the solvent, 156 g of extract was obtained. Afterward, as much as 100 g of the extract was partitioned by liquid-liquid extraction. It was successfully done 3 times by using the solvent of n-hexane and MeOH with the ratio of 1:1 in volumes. Furthermore, a vacuum liquid chromatography (VLC) was exposed to as much as 80.4 g of methanol portion over silica gel which was eluted with different mixtures of n-hexane and ethylacetate. It was done by slowly intensifying the gradient polarity until 7 combined fractions (MF-1 to -7) were acquired. Fraction MF-3.2 was exposed to silica gel column chromatography. There were 3 different solvent systems precisely the mixture between n-hexane, CHCl3, and EtOAc with the ratio 100:5:20. As a result, there were 300 fractions gained and produced 6.8 mg of compound (
Carbazomarin-C, yellowish solid, mp. 251°C-252°C; UV (MeOH), λmax (log ε) 335 (1.23), 279 (2.08), 228 (1.53). 1H NMR (Methanol-d 4, 600 MHz), δ 9.75 (1H, s), 8.04 (1H, d, J = 9.6 Hz), 7.88 (1H, s), 7.41 (1H, s), 6.97 (1H, s), 6.35 (1H, dd, J = 17.4, 10.6 Hz), 6.00 (1H, d, J = 9.6 Hz), 5.41-5.24 (2H, m), 4.93 (2H, dd, J = 17.4, 1.2 Hz), 4.85 (2H, dd, J = 10.6, 1.2 Hz), 4.78-4.73 (3H, m), 3.54 (3H, d, J = 6.8 Hz), 3.35 (1H, s), 2.31 (2H, dd, J = 13.8, 7.8 Hz), 2.04 (3H, dd, J = 13.8, 10.1 Hz), 1.90 (0H, s), 1.83 (4H, s), 1.72 (6H, s), 1.68 (3H, s), 1.68 (3H, s), 1.41 (3H, s), 1.34 (3H, s) and 13C NMR (151 MHz, Methanol-d 4) δ 195.78, 162.58, 159.54, 158.62, 156.75, 153.87, 150.76, 145.26, 144.77, 140.83, 132.02, 124.08, 123.68, 121.22, 117.73, 116.80, 114.78, 110.12, 109.76, 108.93, 107.65, 106.63, 103.99, 97.20, 76.47, 42.78, 41.66, 40.88, 40.52, 29.41, 28.91, 28.15, 24.49, 24.49, 23.75, 23.03, 22.33, 16.66.
Supplemental Material
Supplementary material - Supplemental material for Carbazomarin: A New Potential of α-Glucosidase Inhibitor From Clausena excavata Roots
Supplemental material, Supplementary material, for Carbazomarin: A New Potential of α-Glucosidase Inhibitor From Clausena excavata Roots by Nanik S. Aminah, Tin M. Thant, Alfinda N. Kristanti, Rico Ramadhan, Hnin T. Aung and Yoshiaki Takaya in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors would like to thank Universitas Airlangga for its Airlangga Development Scholarship (ADS) and Riset Mandat Grand which have funded this research.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
