Abstract
A series of N-tosyl α-amino acids
Quinazoline alkaloids are isolated from natural plants or obtained synthetically in view of their well-established pharmacological and biological activities. 1 Peganum harmala was classified among the best-known species to contain quinazoline alkaloids. Its seeds have been widely investigated and were reported to contain several quinazoline alkaloids, including vasicinone which has been isolated as bioactive natural product. 2 From long time, this alkaloid has been used in folk medicine as a remedy for cold, asthma, and bronchitis. It also received great attention due to its diverse pharmacological and biological activities, such as anti-inflammatory, antimicrobial, cytotoxic, and antiproliferative. 3,4
Recent studies motivated by various pharmacological activities of α-amino acids encouraged their use as building blocks for pharmaceutical peptides. 5 Moreover, several studies demonstrated that α-amino acids have been implicated in the regulation of gene expression and control of some physiological functions. 6
On the other hand, organophosphorus compounds have, for a long time, attracted the interests of both chemists and biologists because of their diverse chemical and biological properties. Many organophosphorus derivatives exhibited a broad range of biological activities such as antidepressant, antibiotic, and cytotoxic. 7,8
In view of the above observations and as a continuation of our previous work on the synthesis of new structural analogs of alkaloids isolated from P. harmala seeds, 9 we report here the synthesis of new series of α-amino acid ester and phosphonate derivatives of (±)-vasicinone. Furthermore, the new synthesized compounds were tested in vitro for their acetylcholinesterase (AChE) and 5-lipoxygenase (5-LOX) inhibitor potentials and cytotoxic activity against MCF-7 (Human breast carcinoma), OVCAR-3 (human ovarian carcinoma), and HCT-116 (Human colon carcinoma).
(±)-Vasicinone
As illustrated in Scheme 1 and in order to prepare new ester derivatives consisting of both (±)-vasicinone and α-amino acids, we started by the protection of the amino group in the α-amino acids using tosyl chloride to generate N-tosyl α-amino acids

Synthetic pathway to compounds 2a-e, 3a-e, and 3b′-e′.
Then, (±)-vasicinone
Yield (%) and HPLC Retention Time t
r (min) of Derivatives 3a-
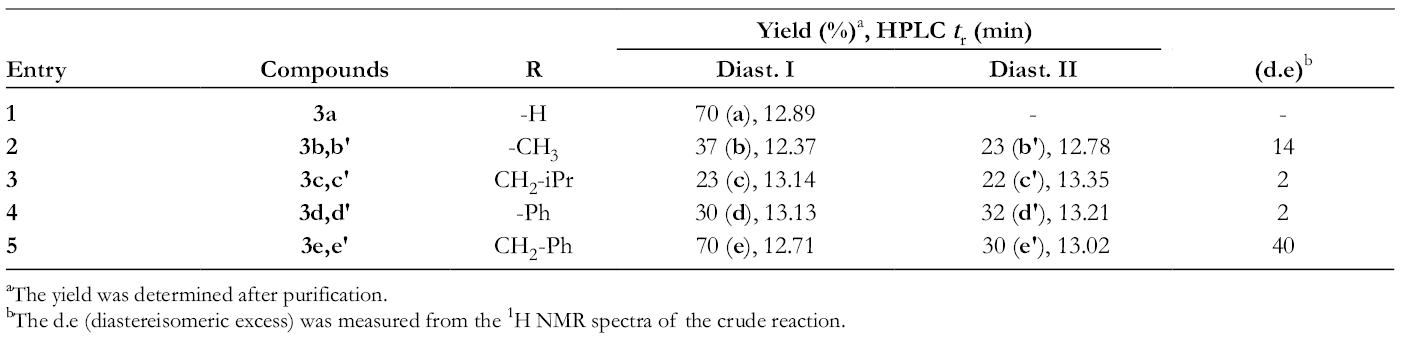
aThe yield was determined after purification.
bThe d.e (diastereisomeric excess) was measured from the 1H NMR spectra of the crude reaction.
On the other hand, encouraged by our results and in conjunction with the pharmaceutical importance known for fused heterocyclic incorporating a phosphonate moiety, we decided to continue to explore the secondary alcohol function of (±)-vasicinone
Our approach to the target fused (±)-vasicinone-phosphonate derivatives firstly started by the preparation of the chlorinated derivative
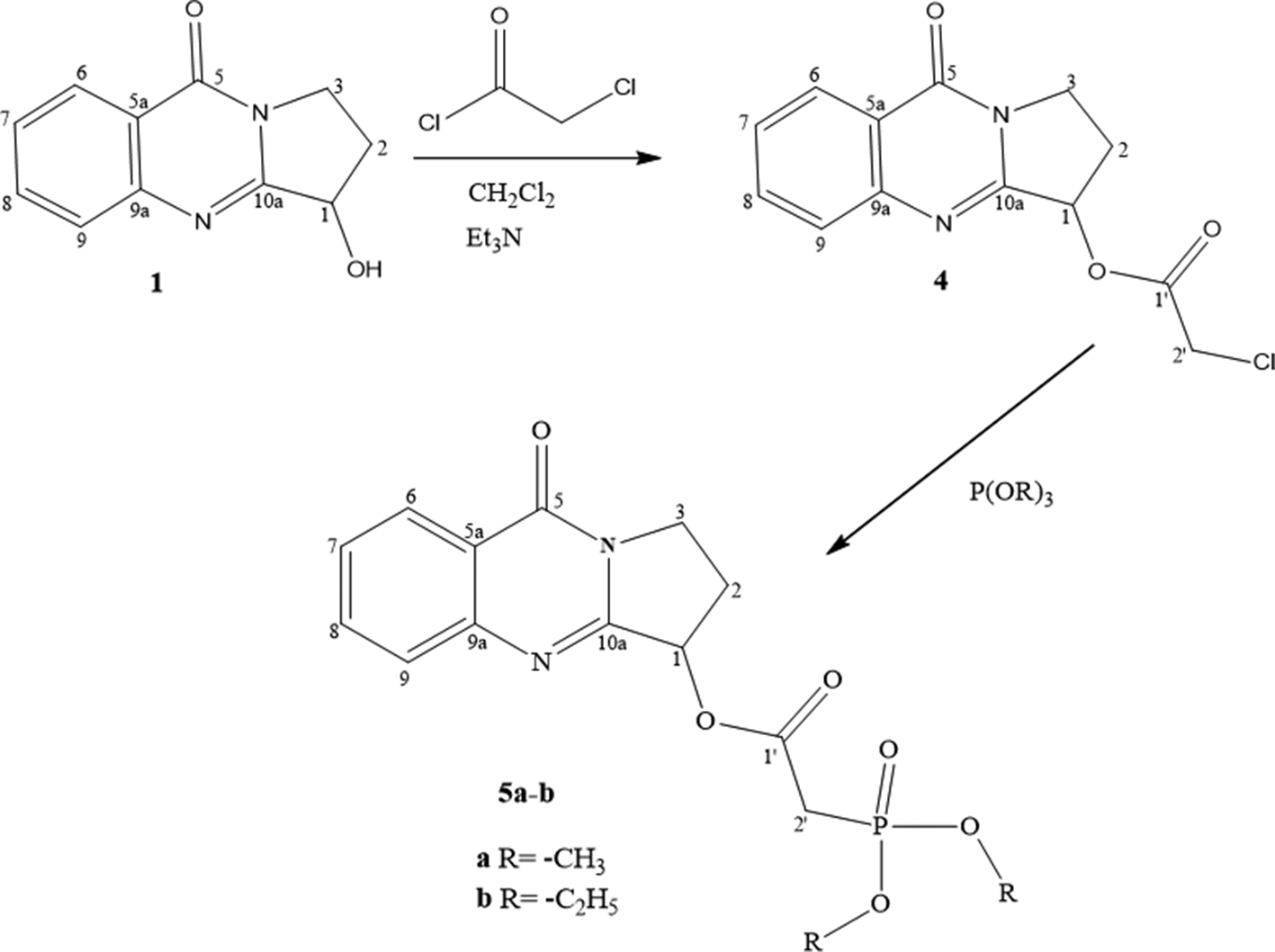
Synthetic pathway to compounds 4 and 5a,b.
Yields (%) of Derivatives 4 and 5a,b.
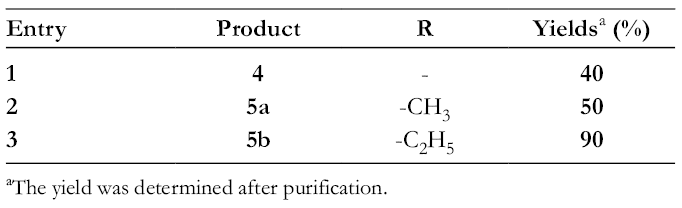
aThe yield was determined after purification.
Finally, the ES-HRMS mass spectra of all the synthesized compounds (
In order to survey the possible biological activities of (±)-vasicinone
The cytotoxic assay (Table 3) showed that (±)-vasicinone
Cytotoxic Activity (MCF-7, OVCAR-3, and HCT-116) of (±)-Vasicinone 1 and its Derivatives 3a-
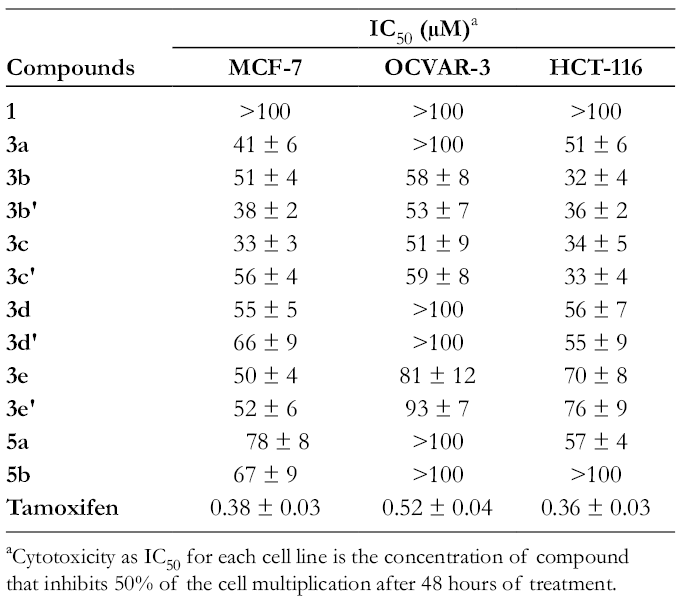
aCytotoxicity as IC50 for each cell line is the concentration of compound that inhibits 50% of the cell multiplication after 48 hours of treatment.
However, the ester derivatives
The relatively high activity of compounds
The results given in Table 3 show that phosphonate derivatives
On the other hand, the derivative
(±)-Vasicinone
Acetylcholinesterase and 5-Lipoxygenase Inhibition Capacity of (±)-Vasicinone 1 and its Derivatives 3a-e, 3b′-e

NDGA, nordihydroguaiaretic acid.
aIC50 values represent the concentration of inhibitor required to decrease enzyme activity by 50% and are the mean of 3 independent measurements, each performed in triplicate.
For the phosphonate derivatives, only
(±)-Vasicinone
In summary, in this paper, we designed and synthesized a new series of α-amino esters
Experimental
Chemistry
Melting points were taken on a Buchi-510 capillary melting point apparatus. 1H (300 MHz) and 13C (75 MHz) NMR spectra were recorded on a Bruker AM-300 spectrometer, using CDCl3 and DMSO-d 6 as solvent and nondeuterated residual solvent as internal standard. Chemicals shifts (δ) are given in parts per million (ppm) and coupling constants (J) in Hertz (Hz). High-resolution mass spectra were acquired with a Q TOF (Quadripole Time-of-Flight) 1er (Waters, ESI technique, positive mode). Preparative HPLC analysis was performed on a Ascentis Express C18, HPLC Column. 5 µm particle size, L × I.D. 25 cm × 4.6 mm. Solvents (H2O, A and acetonitrile, B) were pumped by a Prostar two-way binary high-pressure gradient pump (Varian). UV detection was carried out at 240 and 280 nm by a Prostar 345 detector (Varian). The mobile phase was composed of 2 solvents: A, water (20%) and B, acetonitrile (80%). pH = 2.65 acetic acid. Degasser: ERC-3114; Ferryman sample: AS300, spectra Series thermoseparation products. Samples were dissolved in MeOH. Flow rate: 1.2 mL/min.
Extraction of (±)-Vasicinone 1
A total of 2.9 kg of P. harmala seeds-dried powder were extracted 3 times with methanol by stirring at 50°C for 1 hour. The extracts were combined and evaporated to dryness. The residue was dissolved in HCl solution (2%) and then filtered. The filtrate was extracted 2 times with pentane. The aqueous acid layer was basified (pH = 10) with NH4OH and extracted 3 times with chloroform. The filtered extract was concentrated under reduced pressure and then the residue (35 g) was purified by chromatography on silica gel eluting successively with ethyl acetate-methanol (90:10 to 0:100) to afford 9 fractions; 1 g of (±)-vasicinone as a white solid was obtained after precipitation of fraction 2 in ethyl acetate-methanol (60:40). 9
(±)-Vasicinone 1: 1-Hydroxy-2,3-Dihydropyrrolo[2,1-b]quinazolin-5-one
White solid. mp 200°C to 202°C.
N-Amino Acid Protection: Preparation of Compounds 2a-e
A total of 1 g of amino acid was dissolved in 15 mL of a solution of NaOH (2 M) followed by the addition of tosyl chloride (1 equiv.), trimethylamine (1 equiv.), and acetone (2 mL). After 10 minutes, the mixture was stirred at room temperature for 6 hours. The mixture was then extracted 3 times with diethyl ether after which the aqueous layer was acidified by the addition of concentrated HCl (pH = 1) and this aqueous phase was extracted 3 times with ethyl acetate. After evaporating the solvent, the residue was washed with ethyl acetate-diethyl ether (1:1) and the product was obtained after filtration. 11
Compound 2a: Tosylglycine
White solid. Yield 50%. 1H NMR (300 MHz, CDCl3) δH: 7.58 (2H, d, J = 8.1 Hz , H-2′ and H-6′), 7.33 (2H, d, J = 8.1 Hz, H-3′ and H-5′), 3.79 (2H, s, H-2), 2.43 (3H, s, CH3). 13C NMR (75 MHz, CDCl3) δC: 168.7 (C=O), 143.2 (C-1′′), 136.0 (C-4′), 129,1 (C-3′ and C-5′), 126.7 (C-2′ and C-6′), 43.5 (C-2), 20.8 (CH3).
Compound 2b: Tosyl-l-Alanine
White solid. Yield 90%. 1H NMR (300 MHz, CDCl3) δH: 7.76 (2H, d, J = 7.8 Hz , H-2′ and H-6′), 7.35 (2H, d, J = 8.1 Hz, H-3′ and H-5′), 3.63 (2H, q, J = 6.9 Hz, H-2), 2.41 (3H, s, CH3), 1.31 (3H, d, J = 6.9 Hz, CH3(3)) . 13C NMR (75 MHz, CDCl3) δC: 179.1 (C=O), 143.5 (C-1′′), 139.1 (C-4′), 129.7 (C-3′′ and C-5′′), 127.0 (C-2′′ and C-6′′), 54.6 (C-2), 21.2 (CH3), 17.2 (C-3).
Compound 2c: Tosyl-l-Leucine
White solid. Yield 85%. 1H NMR (300 MHz, CDCl3) δH: 7.32 (2H, d, J = 8.4 Hz , H-2′ and H-6′), 7.80 (2H, d, J = 8.4 Hz, H-3′ and H-5′), 5.09 (1H, d, J = 3.6 Hz, NH), 3.96 (1H, dd, J = 6.3 Hz and J = 5.1 Hz, H-2), 2.44 (3H, s, CH3), 1.81 (2H, m, H-3), 1.54 (1H, dd, J = 6.9 Hz and J = 6.3 Hz, H-4), 0.92 (3H, d, J = 6.6 Hz, CH3(5)), 0.85 (1H, d, J = 6.6 Hz, CH3(5′)).
Compound 2d: (S)-2-((4-Methylphenyl)sulfonamido)-2-Phenylacetic Acid
White solid. Yield 70%. 1H NMR (300 MHz, CDCl3) δH: 7.58 (2H, d, J = 8.1 Hz , H-2′′ and H-6′′), 7.26 (2H, d, J = 7.8 Hz, H-3′′ and H-5′), 7.15 (5H, m, H-2′, H-3′, H-4′, H-5′, and H-6′), 4.03 (1H, dd, J = 8.4 Hz and J = 5.7 Hz, H-2), 3.04 (1H, dd, J = 13.8 Hz and J = 5.7 Hz, H-3a), 2.87 (1H, J = 13.8 Hz and J = 8.1 Hz, H-3b), 2.41 (3H, s, CH3). 13C NMR (75 MHz, CDCl3) δC: 173.1 (C=O), 143.0 (C-1′′), 137.7 (C-1′), 136.4 (C-4′), 129.1 (C-3′′ and C-5′′), 129.0 (C-2′′ and C-6′′), 127.9 (C-2′ and C-6′), 126.6 (C-3′ and C-5′), 126.3 (C-4′), 57.4 (CH), 38.5 (CH2), 20.3 (CH3), 17.8 (2CH3).
Compound 2e: Tosyl-l-Phenylalanine
White solid. Yield 40%. 1H NMR (300 MHz, CDCl3) δH: 7.58 (2H, d, J = 8.1 Hz , H-2′′ and H-6′′), 7.26 (2H, d, J = 7.8 Hz, H-3′′ and H-5′′), 7.15 (5H, m, H-2′, H-3′, H-4′, H-5′, and H-6′), 4.03 (1H, dd, J = 8.4 Hz and J = 5.7 Hz, H-2), 3.04 (1H, dd, J = 13.8 Hz and J = 5.7 Hz, H-3a), 2.87 (1H, dd, J = 13.8 Hz and J = 8.1 Hz, H-3b), 2.41 (3H, s, CH3). 13C NMR (75 MHz, CDCl3) δC: 173.1 (C=O), 143.0 (C-1′′), 137.7 (C-1′), 136.4 (C-4′), 129.1 (C-3′′ and C-5′′), 129.0 (C-2′′ and C-6′′), 127.9 (C-2′ and C-6′), 126.6 (C-3′ and C-5′), 126.3 (C-4′), 57.4 (C-2), 38.5 (C-3), 20.3 (CH3).
Preparation of Esters 3a-e and 3b′-e′
A mixture of (±)-vasicinone
Compound 3a: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosylglycinate
White solid. Yield 70%. ES-HRMS m/z [M+H]+ 414.1401. 1H NMR (300 MHz, CDCl3) δH: 8.32 (1H, d, J = 8.1 Hz , H-6), 7.75 (2H, d, J = 8.4 Hz, H-2′′ and H-6′′), 7.52 (1H, m, H-8), 7.70 (1H, d, J = 4.8 Hz, H-9), 7.50 (1H, t, J = 6.9 Hz, H-7), 7.26 (2H, d, J = 6.3 Hz, H-3′′ and H-5′′), 6.01 (1H, dd, J = 7.8 Hz and J = 5.4 Hz, H-1), 5.40 (1H, d, J = 10.5 Hz, NH), 4.24 (1H, m, H-3a), 4.11 (1H, m, H-3b), 3.95 (2H, d, J = 5.4 Hz, H-2′), 2.69 (1H, m, H-2a), 2.41 (3H, s, CH3), 2.11 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 167.6 (C=O), 159.8 (C-5), 154.0 (C-10a), 148.3 (C-9a), 143.2 (C-1′′), 136.1 (C-4′′), 133.8 (C-8), 129.1 (C-3′′ and C-5′′), 127.1 (C-7), 126.8 (C-9), 126.7 (C-6), 125.9 (C-2′′ and C-6′′), 120.6 (C-5a), 74.1 (C-1), 43.8 (C-3), 43.1 (C-2′), 26.6 (CH3), 20.8 (C-2).
Compound 3b: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Alaninate
White solid. Yield 37%. ES-HRMS m/z [M+H]+ 428.1272. 1H NMR (300 MHz, CDCl3) δH: 8.32 (1H, d, J = 8.1 Hz , H-6), 7.73 (4H, m, H-8, H-9, H-2′′, and H-6′′), 7.54 (1H, td, J = 7.8 Hz and J = 1.5 Hz, H-7), 7.22 (2H, d, J = 8.7 Hz, H-3′′ and H-5′′), 5.91 (1H, dd, J = 7.5 Hz and J = 5.7 Hz, H-1), 5.47 (1H, d, J = 8.1 Hz, NH), 4.15 (3H, m, H-3a, H-3b and H-2′), 2.59 (1H, m, H-2a), 2.37 (3H, s, CH3), 2.05 (1H, m, H-2b), 1.46 (3H, d, J = 6.9 Hz, CH3(3′)). 13C NMR (75 MHz, CDCl3) δH: 170.5 (C=O), 159.7 (C-5), 154.2 (C-10a), 148.0 (C-9a), 143.0 (C-1′′), 136.9 (C-4′′), 133.9 (C-8), 129.0 (C-3′′ and C-5′′), 126.9 (C-7), 126.8 (C-2′′ and C-6′′), 126.6 (C-9), 126.0 (C-6), 120.6 (C-5a), 73.8 (C-1), 51.1 (C-2′), 43.1 (C-3), 26.6 (CH3), 20.8 (C-2), 19.0 (CH3).
Compound 3b′: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Alaninate
White solid. Yield 23%. ES-HRMS m/z [M+H]+ 428.1272. 1H NMR (300 MHz, CDCl3) δH: 8.32 (1H, d, J = 8.1 Hz , H-6), 7.76 (2H, d, J = 8.7 Hz, H-2′′ and H-6′′), 7.74 (2H, m, H-8 and H-9), 7.52 (1H, td, J = 7.8 Hz and J = 1.5 Hz, H-7), 7.24 (2H, d, J = 7.8 Hz, H-3′′ and H-5′′), 5.94 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.34 (1H, d, J = 8.7 Hz, NH), 4.19 (1H, m, H-3a), 4.10 (2H, m, H-3b and H-2′), 2.59 (1H, m, H-2a), 2.38 (3H, s, CH3), 2.05 (1H, m, H-2b), 1.45 (3H, d, J = 6.9 Hz CH3(3′)). 13C NMR (75 MHz, CDCl3) δH: 171.0 (C=O), 159.8 (C-5), 154.2 (C-10a), 148.2 (C-9a), 143.1 (C-1′′), 136.7 (C-4′′), 133.8 (C-8), 129.0 (C-3′′ and C-5′′), 127.1 (C-7), 126.9 (C-2′′ and C-6′′), 126.8 (C-9), 126.0 (C-6), 120.6 (C-5a), 73.8 (C-1), 51.9 (O=C-CH-N), 43.1 (C-3), 26.5 (CH3), 20.8 (C-2), 18.9 (CH3).
Compound 3c: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Leucinate
White solid. Yield 23%. ES-HRMS m/z [M+H]+ 470.1749. 1H NMR (300 MHz, CDCl3) δH: 8.36 (1H, d, J = 8.1 Hz , H-6), 7.76 (4H, m, H-8, H-9, H-2′′, and H-6′′), 7.55 (1H, td, J = 7.8 Hz and J = 1.5 Hz, H-7), 7.26 (2H, d, J = 7.8 Hz, H-3′′ and H-5′′), 5.83 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.01 (1H, d, J = 9.6 Hz, NH), 4.14 (3H, m, H-3a, H-3b and H-2′), 2.59 (1H, m, H-2a), 2.41 (3H, s, CH3), 2.03 (1H, m, H-2b), 1.83 (1H, m, H-4′), 1.59 (2 H, m, H-3′), 0.93 (3H, d, J = 6.6 Hz, CH3(6′)), 0.90 (3H, d, J = 6.6 Hz, CH3(5′)). 13C NMR (75 MHz, CDCl3) δC: 171.1 (C=O), 160.4 (C-5), 154.5 (C-10a), 149.1 (C-9a), 143.6 (C-1′′), 137.0 (C-4′′), 134.2 (C-8), 129.5 (C-3′′ and C-5′′) , 127.7 (C-7), 127.4 (C-2′′ and C-6′′), 127.3 (C-9), 126.5 (C-6), 121.2 (C-5a), 74.3 (C-1), 54.3 (C-1′), 43.5 (C-3), 42.2 (C-3′), 29.6 (C-4′), 27.0 (C-2), 24.3 (CH3), 22.6 (CH3), 21.4 (CH3).
Compound 3c′: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Leucinate
White solid. Yield 22%. ES-HRMS m/z [M+H]+ 470.1749. 1H NMR (300 MHz, CDCl3) δH: 8.35 (1H, dd, J = 8.1 Hz and J = 1.2 Hz, H-6), 7.75 (2H, d, J = 8.4 Hz, H-2′′ and H-6′′), 7.70 (2H, m, H-8 and H-9), 7.55 (1H, td, J = 6.9 Hz and J = 1.5 Hz, H-7), 7.30 (2H, d, J = 8.7 Hz, H-3′′ and H-5′′), 5.88 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.06 (1H, d, J = 10.2 Hz, NH), 4.19 (2H, m, H-3a and H-3b), 4.00 (1H,m, H-2′), 2.54 (1H, m, H-2a), 2.42 (3H, s, CH3), 2.05 (1H, m, H-2b), 1.85 (1H, m, H-4′), 1.60 (2 H, m, H-3′), 0.87 (3H, d, J = 6.6 Hz, CH3(5′)), 0.85 (3H, d, J = 6.6 Hz, CH3(6′)). 13C NMR (75 MHz, CDCl3) δC: 171.6 (C=O), 160.4 (C-5), 154.4 (C-10a), 149.1 (C-9a), 143.6 (C-1′′), 137.1 (C-4′′), 134.3 (C-8), 129.5 (C-3′′ and C-5′′), 127.7 (C-7), 127.5 (C-2′′ and C-6′′), 127.2 (C-9), 126.4 (C-6), 121.3 (C-5a), 74.3 (C-1), 54.3 (C-2′), 43.5 (C-1), 42.2 (CH2), 29.6 (CH), 27.0 (C-2), 24.3 (CH3), 22.6 (CH3), 21.4 (CH3).
Compound 3d: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl (2S)-2-((4 Methylphenyl)sulfonamido)-2-Phenylacetate
White solid. Yield 30%. ES-HRMS m/z [M+H]+ 490.1439. 1H NMR (300 MHz, CDCl3) δH: 8.34 (1H, dd, J = 8.1 Hz and J = 1.2 Hz , H-6), 7.79 (2H, m, H-8 and H-9), 7.66 (2H, d, J = 8.4 Hz, H-2′′′ and H-6′′′), 7.53 (1H, td, J = 6.9 Hz and J = 1.5 Hz, H-7), 7.24 (5H, m, H-2′′, H-3′′, H-4′′, H-5′′, and H-6′′), 7.24 (2H, d, J = 8.1 Hz, H-3′′′ and H-5′′′), 5.96 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.64 (1H, d, J = 7.8 Hz, NH), 5.21 (1H, d, J = 7.5 Hz, H-2′), 4.05 (2H, m, H-3a and H-3b), 2.51 (1H, m, H-2a), 2.40 (3H, s, CH3), 1.95 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 169.2 (C=O), 160.4 (C-5), 154.2 (C-10a), 148.0 (C-9a), 143.5 (C-1′′′), 137.2 (C-4′′′), 134.9 (C-1′′), 134.3 (C-8), 129.5 (C-2′′ and C-6′′), 128.9 (C-3′′′ and C-5′′′), 128.8 (C-3′′ and C-5′′), 127.8 (C-7), 127.3 (C-2′′′ and C-6′′′), 127.2 (C-4′′), 127.1 (C-9), 126.4 (C-6), 121.2 (C-5a), 75.0 (C-1), 59.5 (C-2′), 43.5 (C-3), 26.9 (C-2), 21.3 (CH3).
Compound 3d′: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl (2S)-2-((4-Methylphenyl)sulfonamido)-2-Phenylacetate
White solid. Yield 32%. ES-HRMS m/z [M+H]+ 490.1439. 1H NMR (300 MHz, CDCl3) δH: 8.30 (1H, dd, J = 8.1 Hz and J = 1.2 Hz , H-6), 7.74 (3H, m, H-8, H-2′′′ and H-6′′′), 7.64 (1H, d, J = 8.1 Hz, H-9), 7.50 (1H, td, J = 8.1 Hz and J = 1.2 Hz, H-7), 7.30 (7H, m, H-2′′, H-3′′, H-4′′, H-5′′, H-6′′, H-3′′′, and H-5′′′), 6.00 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.63 (1H, d, J = 8.4 Hz, NH), 5.14 (1H, d, J = 8.4 Hz, H-2′), 4.16 (2H, m, H-3a and H-3b), 2.60 (1H, m, H-2a), 2.40 (3H, s, CH3), 2.17 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 170.0 (C=O), 160.4 (C-5), 154.4 (C-10a), 148.9 (C-9a), 143.5 (C-1′′′), 137.2 (C-4′′′), 134.9 (C-1′′), 134.3 (C-8), 129.5 (C-2′′ and C-6′′), 128.9 (C-3′′′ and C-5′′′), 128.8 (C-3′′ and C-5′′), 127.8 (C-7), 127.3 (C-2′′′ and C-6′′′), 127.2 (C-4′′), 127.1 (C-9), 126.4 (C-6), 121.2 (C-5a), 74.7 (C-1), 59.5 (C-2′), 43.5 (C-3), 26.9 (C-2), 21.3 (CH3).
Compound 3e: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Phenylalaninate
White solid. Yield 70%. ES-HRMS m/z [M+H]+ 504.1589. 1H NMR (300 MHz, CDCl3) δH: 8.36 (1H, d, J = 8.1 Hz , H-6), 7.80 (2H, d, J = 7.8 Hz, H-9 and H-8), 7.68 (2H, d, J = 8.1 Hz, H-2′′′ and H-6′′′), 7.54 (1H, td, J = 7.8 Hz and J = 0.9 Hz, H-7), 7.16 (7H, m, H-2′′, H-3′′, H-4′′, H-5′′, H-6′′, H-3′′′, and H-5′′′), 5.83 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.09 (1H, d, J = 9 Hz, NH), 4.34 (1H, m, H-3a), 4.06 (2H, m, H-3b and H-2′), 3.13 (2H, d, J = 6.6 Hz, H-3′), 2.47 (1H, m, H-2a), 2.39 (3H, s, CH3), 1.90 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 170.0 (C=O), 165.4 (C-5), 154.4 (C-10a), 148.9 (C-9a), 143.5 (C-1′′′), 134.8 (C-1′′ and C-4′′′), 134.2 (C-8), 129.6 (C-3′′′ and C-5′′′), 129.4 (C-3′′, C-5′′, C-2′′′, and C-6′′′), 128.6 (C-2′′ and C-6′′), 127.7 (C-7), 127.4 (C-9), 127.3 (C-4′′), 126.4 (C-6), 121.2 (C-5a), 74.7 (C-1), 56.7 (C-2′), 43.6 (C-3), 39.3 (C-3′), 26.9 (C-2), 21.3 (CH3).
Compound 3e′: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl Tosyl-l-Phenylalaninate
White solid. Yield 30%. ES-HRMS m/z [M+H]+ 504.1589. 1H NMR (300 MHz, CDCl3) δH: 8.36 (1H, d, J = 8.1 Hz , H-6), 7.78 (2H, d, J = 7.8 Hz, H-9 and H-8), 7.68 (2H, d, J = 8.1 Hz, H-2′′′ and H-6′′′), 7.54 (1H, td, J = 7.8 Hz and J = 0.9 Hz, H-7), 7.24 (2H, d, J = 8.1 Hz, H-3′′′ and H-5′′′), 7.16 (5 H, m, H-2′′, H-3′′, H-4′′, H-5′′, and H-6′′), 5.95 (1H, dd, J = 7.5 Hz and J = 5.1 Hz, H-1), 5.11 (1H, d, J = 9 Hz, NH), 4.31 (1H, m, H-3a), 4.14 (2H, m, H-3b and CH-N), 3.12 (2H, d, J = 6.6 Hz, CH2), 2.55 (1H, m, H-2a), 2.41 (3H, s, CH3), 2.05 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 170.0 (C=O), 160.4 (C-5), 154.4 (C-10a), 148.9 (C-9a), 143.5 (C-1′′′), 136.9–134.6 (C-1′′ and C-4′′′), 134.2 (C-8), 129.6 (C-3′′′ and C-5′′′), 129.5 (C-3′′, C-5′′, C-2′′′, and C-6′′′), 128.5 (C-2′′ and C-6′′), 127.8 (C-7), 127.2 (C-9), 126.4 (C-6), 121.2 (C-5a), 74.4 (C-1), 56.4 (C-2′), 43.6 (C-3), 39.1 (C-3′), 27.1 (C-2), 21.3 (CH3).
Preparation of Compound 4
Under argon atmosphere, (±)-vasicinone
Compound 4: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl 2-Chloroacetate
Yellow oil. Yield 40%. ES-HRMS m/z [M+H]+ 279.0815. 1H NMR (300 MHz, CDCl3) δH: 8.32 (1H, d, J = 8.1 Hz , H-6), 7.75 (2H, m, H-8 and H-9), 7.52 (1H, m, H-7), 6.15 (1H, dd, J = 7.2 Hz and J = 5.1 Hz, H-1), 4.31 (1H, m, H-3a), 4.17 (2H, s, H-2′), 4.12 (1H, m, H-3b), 2.77 (1H, m, H-2a), 2.33 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 165.8 (C=O), 159.9 (C-5), 154.0 (C-10a), 148.3 (C-9a), 133.8 (C-8), 127.2 (C-7), 126.8 (C-9), 125.9 (C-6), 120.6 (C-5a), 74.9 (C-1), 43.6 (C-3), 40.5 (C-2′), 27.3 (C-2).
Preparation of Compounds 5a,b
A mixture of compound
Compound 5a: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl 2 (Dimethoxyphosphoryl)acetate
Yellow oil. Yield 50%. ES-HRMS m/z [M+H]+ 353.0897. 1H NMR (300 MHz, CDCl3) δH: 8.33 (1H, d, J = 8.1 Hz , H-6), 7.73 (2H, m, H-8 and H-9), 7.52 (1H, m, H-7), 6.09 (1H, t, J = 5.1 Hz, H-1), 4.29 (1H, m, H-3a), 4.12 (1H, m, H-3b), 3.81 (6H, d, J = 11.1 Hz, 2CH3), 3.11 (2H, d, J = 21.6 Hz, H-2′), 2.72 (1H, m, H-2a), 2.37 (1H, m, H-2b). 13C NMR (75 MHz, CDCl3) δC: 166.3 (C-1′), 160.5 (C-5), 159.8 (C-10a), 149.0 (C-9a), 134.2 (C-8), 127.7 (C-7), 127.2 (C-9), 126.5 (C-6), 120.5 (C-5a), 74.7 (C-1), 54.5 (d, JP-C
= 98.7 Hz, O
Compound 5b: 5-Oxo-2,3-Dihydropyrrolo[2,1-b]quinazolin-1-yl 2 (Diethoxyphosphoryl)acetate
Yellow oil. Yield 90%. ES-HRMS m/z [M+H]+ 381.1212. 1H NMR (300 MHz, CDCl3) δH: 8.33 (1H, d, J = 8.1 Hz , H-6), 7.75 (2H, m, H-8 and H-9), 7.51 (1H, m, H-7), 6.09 (1H, dd, J = 7.5 Hz and J = 4.8 Hz, H-1), 4.25 (6H, m, H-3a, H-3b and 2(C
Biological Evaluation
Cytotoxic assay
(±)-Vasicinone
Acetylcholinesterase Inhibitor Activity Assay
First, 25 µL of each compound was dissolved in DMSO and then diluted in the buffer (sodium phosphate buffer, pH = 8.0); 25 µL of enzyme solution, 50 µL of 0.1 M sodium phosphate, already prepared, and 125 µL of 5,5′-dithiobis[2-nitrobenzoic acid] were added in a 96-well microplate and then incubated for a period of 15 minutes at a temperature of 25°C.
After the incubation step, 25 µL of a solution of acetylthiocholine iodide was added. The mixture was then incubated for 10 minutes at 25°C. Finally, at 412 nm, the absorbance was determined and the inhibitory power of the derivatives was given as a percentage of inhibition (I%) at different concentrations as follows:

where A control is the absorbance of the control reaction containing all reagents except the tested derivative and A sample is the absorbance of the test derivative.
For each compound, the IC50 was determined, which shows the concentration of each derivative through which it is capable of inhibiting 50% of the enzyme. Galanthamine was employed as positive control and the test was conducted in triplicate. 16
5-Lipoxygenase Inhibitor Activity Assay
A volume of 20 µL of different concentrations of each compound was dissolved in DMSO and mixed individually with 150 µL of sodium phosphate buffer (pH 7.4) containing 20 µL of 5-LOX enzyme and 60 µL of linoleic acid (3.5 mM), yielding a final volume of 250 µL. All compounds were resuspended in DMSO followed by dilution in the buffer so that DMSO does not exceed 1%. The mixture was incubated at 25°C for 10 minutes and the absorbance was determined at 234 nm. Nordihydroguaiaretic acid was used as positive control. All measurements were performed in triplicate. 17
Footnotes
Acknowledgment
The authors are grateful to Prof. Fethia Harzallah-Skhiri, High Institute of Biotechnology of Monastir, Tunisia, for botanical identification
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support to our Laboratory (LR11ES39) was given by the Ministry of Higher Education and Scientific Research of Tunisia, General Directorate for Scientific Research.
