Abstract
Dahuang Zhechong Pill (DHZCP) has been widely used in the treatment of hepatocarcinoma in China. The aim of our study was to identify the characteristic components of DHZCP. First, HPLC fingerprint of DHZCP was established to analyze the common components of 14 batches of DHZCP samples, which were purchased from different manufacturers. The results of HPLC fingerprint detected 164 peaks in these 14 batches of DHZCP. Through similarity analysis, cluster analysis, and principal component analysis, we identified 20 common components upon which to conduct quantitative analysis conducted by an HPLC method. After that, a cytotoxicity test was carried out to screen the active components in DHZCP. The results showed that hypoxanthine, rhein, emodin, aloe emodin, and wogonin are the active components of DHZCP for the treatment of hepatocarcinoma, as they have significant inhibitory effect against the activity of drug-resistant hepatocarcinoma cells (SMMC-7721/DOX) than others.
Dahuang Zhechong Pill (DHZCP) consists of 12 traditional Chinese medicines, including rhubarb, soil locust, leech, locust, grub, peach kernel, true lacquertree dried lacquer, S cutellaria baicalensis, radix paeoniae alba, amygdala amara, rehmannia, and licorice (Figure 1). It is widely used in the treatment of liver diseases, such as liver fibrosis, cirrhosis, and hepatocarcinoma, in China . 1 There are many manufacturers of DHZCP on the market, and the quality control is based on the Chinese Pharmacopoeia 2015. Our previous studies have shown that DHZCP plays a pharmacological role in reversing drug resistance in hepatocarcinoma by inducing apoptosis and inhibiting energy metabolism. 2,3 However, the pharmacodynamic substances in DHZCP remain undefined, requiring further research on its pharmacological mechanisms.

Prescription composition of DNZCP. DNZCP, Dahuang Zhechong Pill.
Fingerprint technology has been widely used in analyzing components in traditional Chinese medicines (TCM). This technology uses certain analytical methods to obtain maps of common peaks of different samples, 4,5 which indicate the characteristic components of TCM. Through similarity analysis, cluster analysis, and principal component analysis, the information of these chemical components can be fully extracted. 6 In comparison to thin layer chromatography, fingerprint analysis provides higher specificity and emphasizes the relatively stable proportion of multiple components that have common peaks. In addition, not only can the known components be identified, but also the unknown components. Therefore, the analytical model of fingerprint technology combined with multi-component quantification has become the main development direction of the chemical research of TCM. 7,8
In this study, the component database of DHZCP was established by searching the Chinese Pharmacopoeia and periodical literature. The fingerprints of the chemical components in DHZCP were obtained from a high-performance liquid chromatography (HPLC) system. On the basis of characterizing the overall chemical information of DHZCP, we got the common peaks of these components through similarity analysis. Then, qualitative and quantitative analyses of the characteristic components of DHZCP were obtained referring to the standard substances. Cytotoxicity test of SMMC-7721/DOX cell lines was used to analyze the anti-tumor components in DHZCP.
Analysis of the 14 batches of DHZCP as described in Table 1 was performed using an HPLC system. Sample S10 was used as reference to establish the fingerprint of DHZCP shown in Figure 2(a) by the method of multi-point correction and generate the reference fingerprint shown in Figure 2(b) by the average method using the Similarity Evaluation System for Chromatographic Fingerprint of TCM software. Common peaks in the reference fingerprint among the 14 batches of DHZCP were marked in Figure 2(b) according to the matched data generated by the software. The results showed that there were 57 common peaks in 14 batches of DHZCP. Among them, 20 main peaks were identified by standard substances as characteristic peaks, including peak 5 (hypoxanthine), peak 6 (allantoin), peak 8 (gallic acid), peak 14 (amygdalin), peak 15 (gentisic acid), peak 18 (paeoniflorin), peak 20 (verbascoside), peak 23 (p-methoxyphenylacetic acid), peak 27 (liquiritigenin), peak 28 (baicalin), peak 29 (naringenin), peak 31 (paeonol), peak 36 (baicalein), peak 39 (wogonin), peak 41 (aloe emodin), peak 45 (glycyrrhizinate), peak 46 (rhein), peak 49 (emodin), peak 53 (chrysophanol), and peak 55 (physcion).
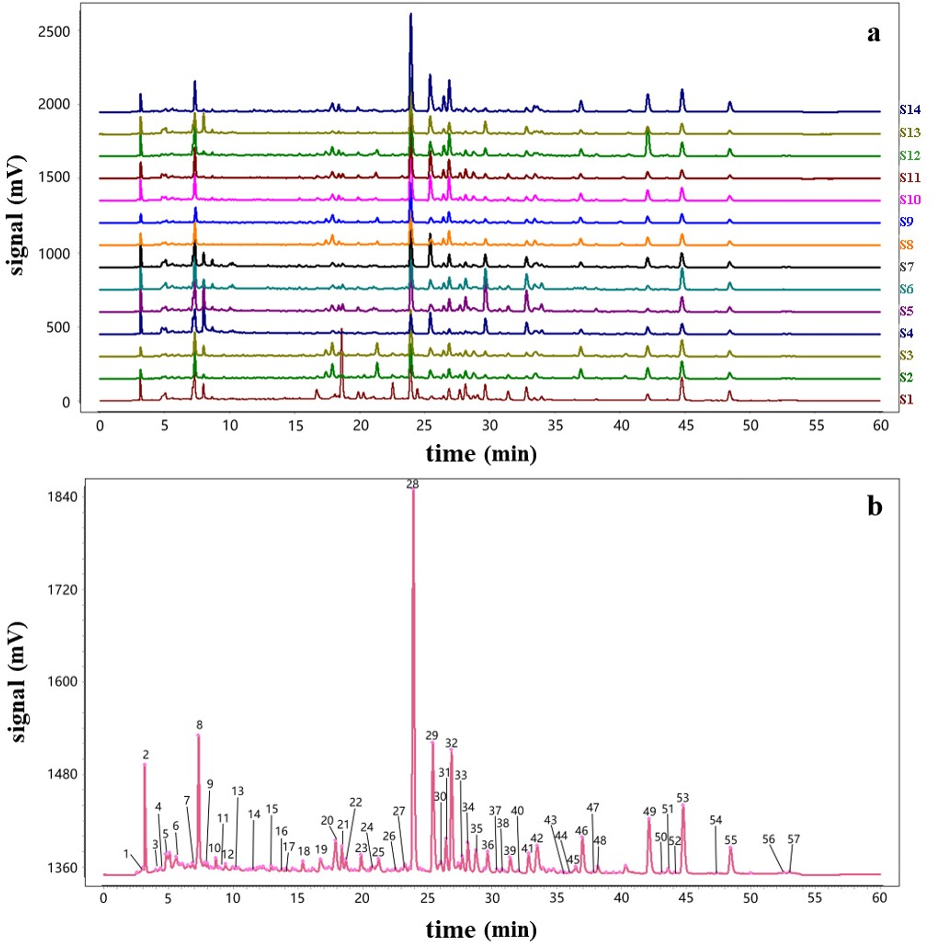
The high-performance liquid chromatography (HPLC) fingerprint of Dahuang Zhechong Pill (DHZCP). (a) The chemical fingerprints of 14 batches of DHZCP. (b) Common peaks of the reference fingerprint.
DHZCP Information for Analyzed Batches From Various Manufacturers.
DHZCP, Dahuang Zhechong Pill.
The area of peak 28 in the DHZCP HPLC fingerprint was stable with good resolution. Therefore, it was selected as the reference peak to calculate the ratio of retention time and area of each common peak of the 14 batches of DHZCP. The results of the relative retention time (RRT) and relative peak area (RPA) analysis showed that the relative standard deviations (RSDs) of the RRT of each common peak in the 14 batches were less than 0.47%, indicating that the retention times of the common peaks were relatively stable.
The peak with an area of more than 0.5% of the whole peak area was selected. Next, the similarities between the reference fingerprint and the sample maps of each batch as well as the similarities between batches were calculated using the Similarity Evaluation System for Chromatographic Fingerprint of TCM (Table 2). The similarity values between these 14 batches and the reference fingerprint, from S1 to S14, were 0.757, 0.909, 0.947, 0.763, 0.886, 0.926, 0.941, 0.958, 0.949, 0.958, 0.957, 0.957, 0.965, and 0.935, respectively. The result indicated that the 14 DHZCP samples obtained from different locations exhibited considerable variation. The similarity value among (S8, S9, S10, S11) and (S2, S3) samples was higher, prompting that the same brand has good chemical stability.
Similarity Evaluation of 14 Batches of DHZCP.

DHZCP, Dahuang Zhechong Pill.
The Pearson correlation coefficient was used to perform the analysis of these 14 batches of DHZCP, and the results are shown in Figure 3(a). When the class distance was set to 10, the samples of DHZCP could be classified into 4 types: (i) S5, S6, S7, S13; (ii) S1; (iii) S4; (iv) S8, S9, S10, S11, S12, S14, S2, S3. Next, we performed PCA on these samples to identify the main components in DHZCP. The results showed that the cumulative contribution rate of the first 2 components was up to 89.49%, the characteristic value of principal component 1 was 11.458, and the variance contribution rate was 81.843%. The characteristic value of principal component 2 was 1.071, and the variance contribution rate was 7.647%. The scatter plot was constructed by plotting the principal component 1 (X-axis) against the principal component 2 (Y-axis) of the 14 samples. The results of the PCA are largely consistent with those of the similarity and clustering analysis, as shown in Figure 3(b) and (c). Therefore, according to the result shown in Figure 3(a), we selected the samples with the higher similarity (S2, S3, S8, S9, S10, S11, S12, S14) to carry on with further analysis.
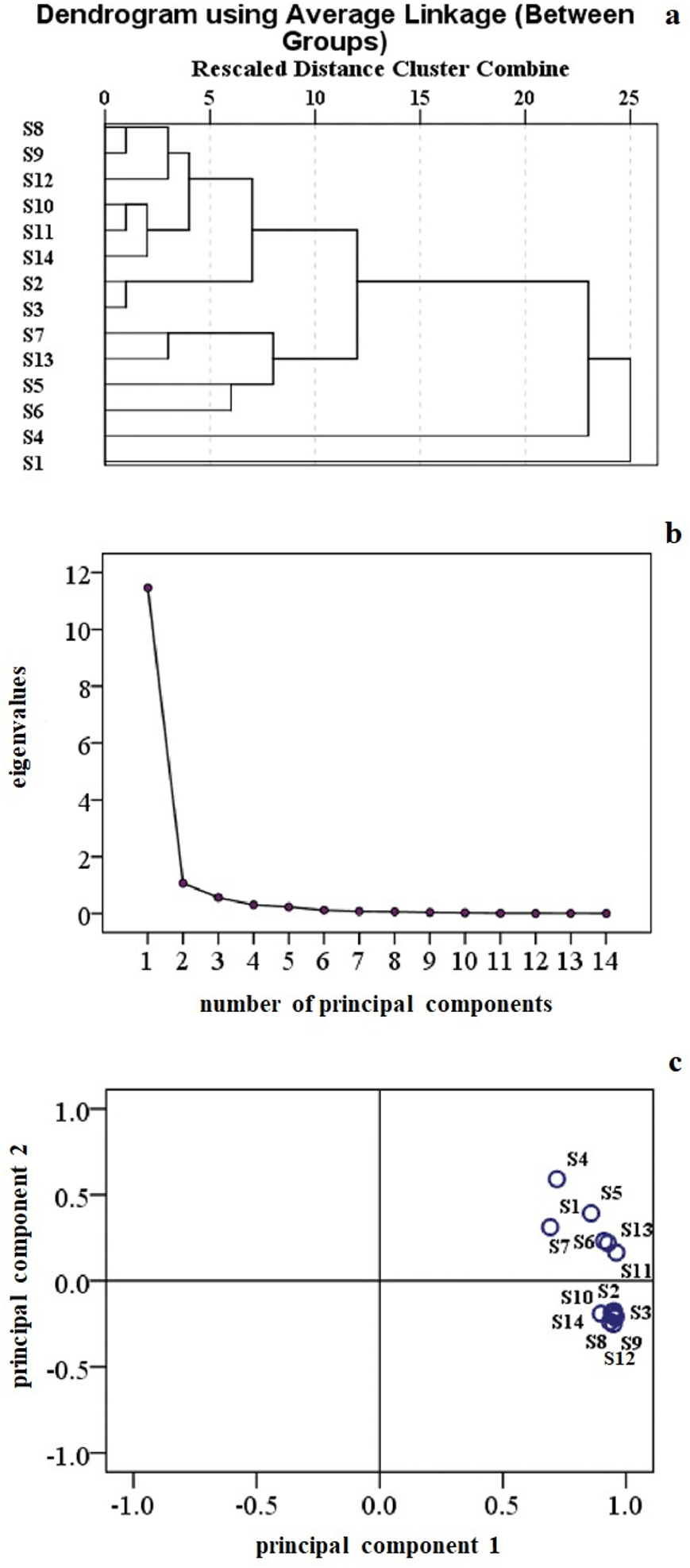
Principal components and cluster analysis of 14 Dahuang Zhechong Pill (DHZCP) batches. (a) Gravel chart. (b) Score chart. (c) Cluster analysis chart.
There were 164 peaks in the 14 batches of DHZCP, and each peak had a large discrete value, complicating the following steps of the analysis. Therefore, we selected the samples S2, S3, S8, S9, S10, S11, S12, S14 to proceed with PCA to reduce the number of variables and data dimensionality. The analysis steps were as follows: (i) the coefficient of variation (CV) of each peak was calculated according to the formula CV = SD/ mean × 100%. The results are shown in Figure 4(a) and (b). The peaks with CVs under 0.6 were selected. (ii) Based on the reference substance in Figure 4(c), the peaks with high content and low CV were selected for chemical identification in order to determine the characteristic components. (iii) The literature was reviewed to verify the results. Finally, 20 peaks were identified (Figure 4(d)), including peak 5 (hypoxanthine), peak 6 (allantoin), peak 8 (gallic acid), peak 14 (amygdalin), peak 15 (gentisic acid), peak 18 (paeoniflorin), peak 20 (verbascoside), peak 23 (p-methoxyphenylacetic acid), peak 27 (liquiritigenin), peak 28 (baicalin), peak 29 (naringenin), peak 31 (paeonol), peak 36 (baicalein), peak 39 (wogonin), peak 41 (aloe emodin), peak 45 (glycyrrhizinate), peak 46 (rhein), peak 49 (emodin), peak 53 (chrysophanol), and peak 55 (physcion).

Characteristic chemical constituent analysis of Dahuang Zhechong Pill (DHZCP). (a) Variable coefficient. (b) Ratio of single peak area to total area. (c) Single standard chromatograms. (d) Full spectral distribution of characteristic components (5, hypoxanthine; 6, allantoin; 8, gallic acid; 14, amygdalin; 15, gentisic acid; 18, paeoniflorin; 20, verbascoside; 23, p-methoxyphenylacetic acid; 27, liquiritigenin; 28, baicalin; 29, naringenin; 31, paeonol; 36, baicalein; 39, wogonin; 41, aloe emodin; 45, glycyrrhizinate; 46, rhein; 49, emodin; 53, chrysophanol; and 55, physcion).
Based on the results described above, we carried out a quantitative analysis by HPLC method of the characteristic components of DHZCP. The data in Figure 5 showed that gallic acid was found with the highest content (2783.5 ± 412.5 µg/g), followed by baicalin (2252.9 ± 547.0 µg/g), amygdalin (1987.6 ± 279.7 µg/g), and verbascoside (1987.6 ± 279.7 µg/g). The content of anthraquinones in rhubarb was slightly different. Among them, chrysophanol had the highest content, followed by aloe emodin, rhein, emodin, and physcion (Figure 5(b)). In the ingredients of animal medicine, hypoxanthine had the highest content. The contents of baicalein and gentisic acid were less than 100 µg/g, which were lower than others.

Quantitative analysis of Dahuang Zhechong Pill (DHZCP). (a) Chemical structural formula of the characteristic components. (b) Quantitative analysis of the 20 main components.
Finally, we studied the effect of the characteristic chemical ingredients of DHZCP on the activity of SMMC-7721/DOX cells. The results in Figure 6 showed that the components’ effect on inhibiting the activity of SMMC-7721/DOX cells differed from every chemical compound and can be divided into approximately 4 types: (i) hypoxanthine, rhein, emodin, aloe emodin, and wogonin. These 5 components exhibited a significant inhibitory effect on the activity of SMMC-7721/DOX cells, and their IC50s were 3.807, 24.99, 62.26, 64.20, and 133.7 µM, respectively. (ii) Chrysophanol, physcion, gentisic acid, p-methoxyphenylacetic acid, baicalein, naringenin, liquiritigenin, and glycyrrhizinate could inhibit the activity of SMCC-7721/DOX cells within their maximum soluble concentration; although, their inhibition rates were less than 50% and there were no significant dose-dependent relationships. (iii) Allantoin, amygdalin, paeoniflorin, verbascoside, and Baicalin, whose maximum inhibition rates were less than 20%, had no significant effect on cell viability. (iv) Gallic acid and paeonol apparently promoted cell growth at higher concentrations.

Effect of the characteristic components of Dahuang Zhechong Pill (DHZCP) on the activity of SMMC-7721/DOX cells.
The fingerprint analysis method, which has become an effective and simple method to study the components of TCM, can comprehensively detect the composition of various chemical components.
In this study, 14 batches of DHZCP were collected to analyze their chemical components by using fingerprint analysis. From the DHZCP HPLC fingerprint, we identified 57 common peaks. After the analysis of peak proportions and CVs, we selected 20 characteristic components. The results of the cell viability test showed that hypoxanthine, rhein, emodin, aloe emodin, and wogonin were the major medicinal substances, which exhibited a significant inhibitory effect on the activity of SMMC-7721/DOX cells, in these components. These characteristic components belong to the purine, anthraquinone, and flavonoid classes.
Purine and its derivatives are a class of organic heterocyclic aromatic compounds that are involved in a multitude of cellular processes, including energy storage, synthesis of nucleic acids and coenzymes, translation, and signaling. 9 At the same time, increasing evidence suggests that purine compounds are involved in regulating the purine nucleotide homeostasis, playing unexpected roles in tumorigenesis, which may provide a novel treatment for cancer. 10,11
Hypoxanthine is a type of alkaloid and is considered as an important pharmacodynamic substance in TCM. 12 The generation of free radicals through oxidative metabolism is considered as one of the mechanisms of its pharmacological action. Recent studies have shown that hypoxanthine as a DNA lesion generated by deamination of adenine is a major factor of cell toxicity. 13 In this study, hypoxanthine showed a strong cytotoxic effect on SMMC-7721/DOX cells, and its specific mechanism of action requires further study.
The anti-cancer activity of rhein in hepatocarcinoma cells is primarily attributed to the inhibition of energy metabolism and induction of the opening of the mitochondrial permeability transition pore. 14 Emodin has been found to be effective for cancer prevention and treatment due to its pleiotropic properties to scavenge free radicals, inhibit cell growth, and induce apoptosis. 15 Similarly, aloe emodin showed a strong anti-tumor effect by inducing tumor cell apoptosis. 16,17 In our study, similar experimental results were obtained, as the anthraquinone components in DHZCP showed a strong inhibitory effect in SMMC-7721/DOX cells.
Wogonin is a flavonoid isolated from Scutellaria baicalensis, which has shown anti-cancer potential by inhibiting proliferation, inducing apoptosis, inhibiting angiogenesis, and reversing drug resistance, hence attracting considerable attention. 18 Our results corroborate the anti-cancer properties of Wogonin.
It is noteworthy that certain chemical components in this prescription have exhibited toxicity for SMMC-7721/DOX cells. Although their drug potency was similar, their molecular structure is quite different. We speculate that they have different targets when exerting their pesticide effect. The synergistic effect of these components imbues the drug with anti-cancer properties. In addition, whether the chemical components with no direct effect on SMMC-7721/DOX cells can play an indirect anti-cancer role in the liver through other mechanisms and the possible modalities remain unknown. We will conduct further study on these components to find a more comprehensive method to analyze the material basis of DHZCP for its anti-cancer effect.
In conclusion, hypoxanthine, rhein, emodin, aloe emodin, and wogonin in DHZCP had a significant effect on inhibiting the activity of drug-resistant hepatocarcinoma cells, and their single peaks occupied more area in the total peak area compared to the other components. Therefore, they may be the characteristic components in fighting against hepatocarcinoma cells directly.
Experimental
Chemicals and Reagents
Fourteen batches of DHZCP were purchased from 9 different companies in China (listed in Table 1). Reference standards of hypoxanthine, allantoin, gallic acid, amygdalin, gentisic acid, paeoniflorin, verbascoside, p-methoxyphenylacetic acid, liquiritigenin, baicalin, naringenin, paeonol, baicalein, wogonin, aloe emodin, glycyrrhizinate, rhein, emodin, chrysophanol, and physcion were purchased from Chengdu Herbpurify Co., Ltd, China. Chromatographic pure methanol (CNW Technologies GmbH, Germany), chromatographic pure formic acid (ACS, USA), and CellTiter-LumiTM Plus kit (Biyuntian Biotechnology Co. Ltd, China) were also purchased.
Instruments
Liquid chromatography was performed on a Waters E2695 series LC system (Waters Corp., USA) equipped with a 2998 UV detector and controlled using Empower chromatography data software. Other apparatuses used were KQ-500 Ultrasonic cleaning instruments, Heidolph vortex mixer, and Heraeus centrifuge with a Sepatech rotor (centrifuge: A1301026, Shanghai Aige Electronic Technology Co. Ltd, China).
HPLC Analysis
The chromatographic separation was performed on Thermo Hypersil GOLD column (4.6 mm × 250 mm, 5 µm). The mobile phase consisted of an aqueous solution of formic acid 0.1% (v/v) (solvent A) and methanol (solvent B) at a flow rate of 1 mL/min. The gradient elution program was set as follows: 0 to 5 minutes, 5% to 25% B; 5 to 25 minutes, 25% to 65% B; 25 to 35 minutes, 65% to 80% B; 50 minutes, 95% B; 60 minutes, 5% B. The chromatograms were monitored at a wavelength of 270 nm during the experiment, and the column temperature was maintained at 30℃. The injection volume was 10 µL each time.
Sample Preparation
In addition to the DHZCP samples purchased from different companies, a DHZCP sample was prepared in lab according to the Chinese Pharmacopoeia 2015. Rhubarb 10 g, soil locust 1.0 g, leech 2.0 g, locust 1.5 g, grub 1.5 g, peach kernel 4.0 g, true lacquertree dried lacquer 1.0 g, S cutellaria baicalensis 2.0 g, radix paeoniae alba 4.0 g, amygdala amara 4.0 g, rehmannia 10 g, and licorice 3.0 g were combined and crushed into powder. One gram of the powder was accurately weighed into a flask and 20 mL of ultrapure water was added. After soaking for 24 hours, the solution was dissolved by ultrasonication over 1 hour. The samples were filtered through a 0.45 µm micropore film. Each reference substance was accurately weighed and dissolved in methanol to prepare the following solutions: hypoxanthine 0.1132 mg/mL, allantoin 1.026 mg/mL, gallic acid 0.494 mg/mL, amygdalin 1.018 mg/mL, gentisic acid 0.506 mg/mL, paeoniflorin 2.114 mg/mL, verbascoside 1.102 mg/mL, p-methoxyphenylacetic acid 0.466 mg/mL, liquiritigenin 0.588 mg/mL, baicalin 0.312 mg/mL, naringenin 0.51 mg/mL, paeonol 0.442 mg/mL, baicalein 0.698 mg/mL, wogonin 0.340 mg/mL, aloe emodin 0.0501 mg/mL, glycyrrhizinate 1.128 mg/mL, rhein 0.0512 mg/mL, emodin 0.0501 mg/mL, chrysophanol 0.0513 mg/mL, and physcion 0.050 mg/mL. The mixed reference solutions were collected after filtration.
Methodological Validation
The methodological validation includes the analysis of specificity, precision, stability, and repeatability. RSD values of the RRT and RPA of each common peak were calculated in the same batch of samples using peak 28 as a reference. The results of RRT showed no impurity peaks. The RSD values of the RRT and RPA of each common peak were less than 3%, indicating that the instrument worked in good condition.
Standard Substances Processing
The standard substances of the 20 characteristic components were accurately weighed and dissolved with DMSO, then diluted with Dulbecco’s modified eagle medium to a final concentration of 2% DMSO to prepare the mother solution. Dilute the mother solution to different concentrations for experiment with Dulbecco’s modified eagle medium containing 2% DMSO.
Cytotoxicity Test
SMMC-7721/DOX cells were seeded into 96-well plates at 2 × 104 cells/mL, allowed to attach overnight, and treated with different concentrations of drugs for 24 hours. Cell viability was measured by using the CellTiter-Lumi™ Luminescent Cell Viability Assay Kit. The inhibition rate was calculated using the following formula: inhibition rate = [1 - (ODdrug - ODblank) / (ODcontrol - ODblank)] × 100%. The half maximal inhibitory concentration (IC50) of drugs was calculated using GraphPad prism. The experiment was repeated 3 times. 19
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (81503308, 81873287), the Key Project of Natural Science Research in Colleges and Universities in Jiangsu Province (18KJA360009), and the National Key R&D program of China (2018YFC1706905).
