Abstract
The tea of Ocimum gratissimum (OG) leaves has been commonly consumed by people living in Ishigaki Island, Okinawa prefecture, Japan, and is considered to be effective for improving diabetes mellitus. In this study, we aimed to clarify the inhibitory potential of OG leaves extract (OG-ext) on gastrointestinal glucose absorption and to provide theoretical evidence for the anti-hyperglycemic effect of OG-ext. The increase of blood glucose after oral administration of α-starch and glucose in mice was suppressed by co-administration of OG-ext. An in vitro enzymatic assay suggested that amylase and maltase were inhibited weakly by the addition of OG-ext. In Caco-2 cells, a human intestinal epithelial model, the sodium-dependent glucose transporter (SGLT) 1-mediated uptake of fluorescence glucose analog was inhibited significantly by the addition of OG-ext in a concentration-dependent manner. These results indicate that the inhibitory effect on SGLT1 is one of the mechanisms of the anti-hyperglycemic effect of the tea of OG leaves.
Ocimum gratissimum (OG, manjericon in Japanese) belongs to the Lamiaceae family and grows wild in the tropical areas of Asia, India, Brazil, and West Africa. Previous studies have suggested that OG leaves possess various beneficial effects, such as anti-oxidant, 1 anti-diarrheal, 2 anti-microbial, 3 and anti-inflammatory 4 effects. In Japan, the tea brewed from OG leaves, which is thought to be effective in improving diabetes mellitus, has traditionally been drunk by people living in the Ishigaki Island, Okinawa prefecture. In fact, extract of OG leaves exhibits anti-diabetic effect in some diabetic rat models. 5,6 However, the underlying mechanism of the anti-diabetic effect of OG leaves remains unclear. Recently, our laboratory reported that constituents of Cistanche tubulosa, a Chinese traditional medicine, suppress postprandial increase of blood glucose by inhibiting the sodium-dependent glucose transporter (SGLT) 1. 7 Dietary glucose is transported into small intestinal epithelial cells by SGLT1 in a sodium-dependent manner 8 and then, glucose is transported into enterocytes as it crosses the basolateral membrane via sodium-independent glucose transporter (GLUT) 2. 9 Previous studies suggested that the dysfunction of SGLT1 by an inactive mutation or the chemical inhibition of SGLT1 decreased blood glucose level. 10,11 In this study, we aimed to clarify the inhibitory effect of OG leaves on gastrointestinal carbohydrate digestion and glucose absorption, to validate scientific evidence regarding the anti-diabetic effect of OG tea.
To evaluate the inhibitory effect of OG leaves on the increase of postprandial blood glucose, mice were orally administered α-starch and glucose with OG leaves extract (OG-ext, 250 or 500 mg/kg). OG-ext suppressed an increase of blood glucose 15, 30, and 60 minutes after oral-loading of α-starch (Figure 1(a)). Similarly, OG-ext inhibited an increase of blood glucose 30 and 60 minutes after oral-loading of glucose in a dose-dependent manner (Figure 1(b)). These results suggested that OG leaves have an inhibitory effect on the increase of postprandial blood glucose.
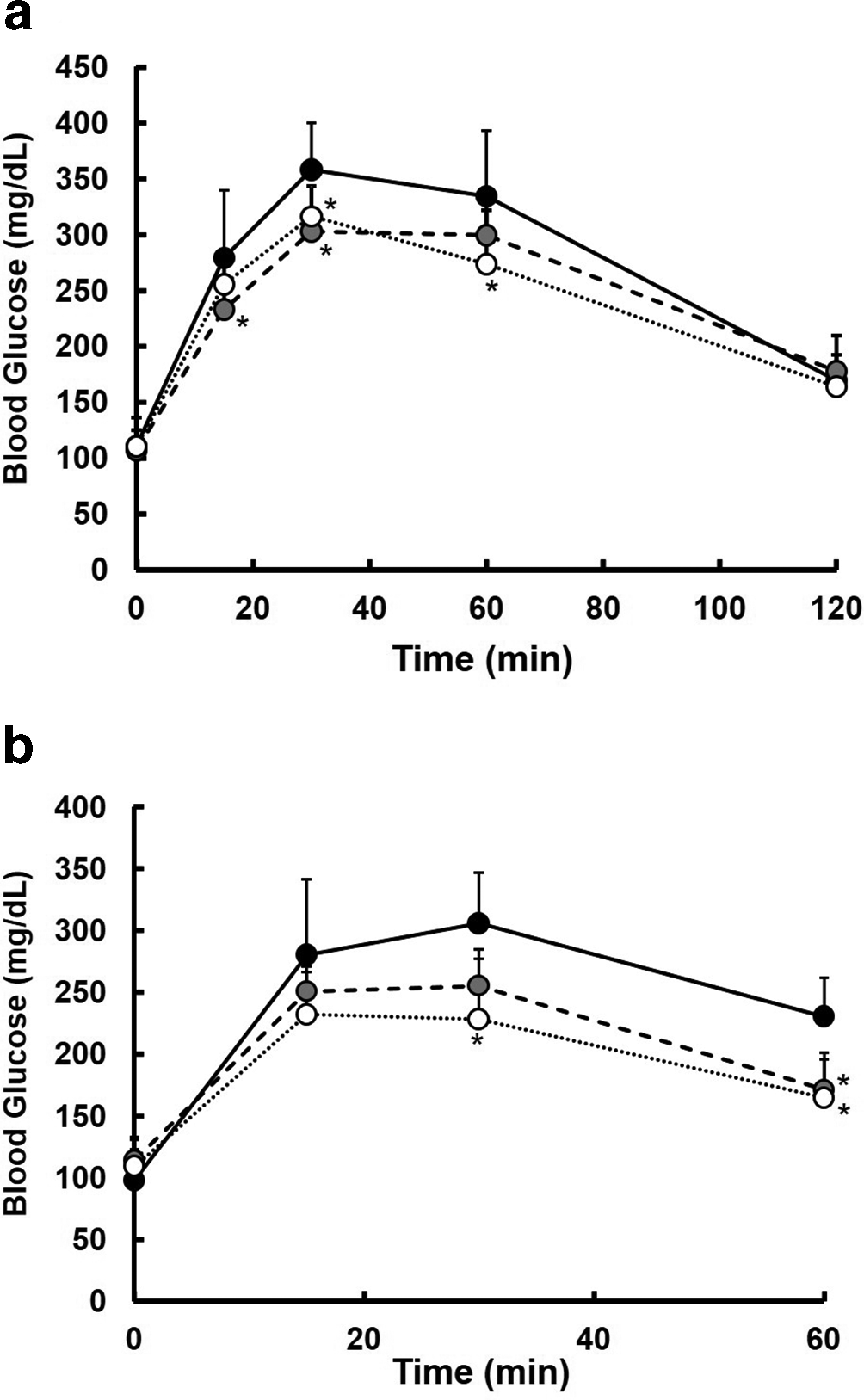
Effect of OG-ext on postprandial increase of blood glucose in mice. The time course of blood glucose after oral α-starch (a) and glucose (b) administration with 0 (Ctrl, black circle with solid line), 250 (gray circle with dashed line), or 500 (white circle with dotted line) mg/kg OG-ext. Each point represents the mean ± SD (n = 4-8). * indicates a significant difference from Ctrl at each time point by Dunnett’s test after an analysis of variance with P < 0.05.
In order to clarify the anti-hyperglycemic mechanism of OG leaves, we investigated their inhibitory potential against the digestive enzymes of carbohydrate and SGLT1-mediated glucose absorption. Dietary α-starch is digested into glucose mainly by α-amylase and maltase in the digestive tract. In this experiment, acarbose, a commercially available α-glucosidase inhibitor, was employed as a positive control. First, we examined the inhibitory effect of OG-ext on α-amylase (Figure 2). Acarbose showed a significant inhibition of α-amylase activity at a concentration greater than 0.2 mg/mL (Figure 2(a)). On the contrary, a significant decrease of α-amylase activity was observed in the group treated only with 2 mg/mL of OG-ext (Figure 2(b)). Next, the inhibitory effect of OG-ext on maltase-mediated maltose degradation was examined. Acarbose inhibits maltase activity significantly at a concentration greater than 0.25 µg/mL (Figure 3(a)). In comparison with acarbose, OG-ext has a slight inhibitory effect on maltase (Figure 3(b)). These results suggest that OG-ext exerts a weak inhibitory effect on digestive enzymes of carbohydrates.

Inhibitory effect of OG-ext on α-amylase-mediated α-starch degradation. α-Starch was digested in α-amylase containing solution for 10 minutes at 37°C in the absence or presence of acarbose (a) or OG-ext (b). Each bar represents the mean ± SEM (n = 4). * indicates a significant difference from remaining α-amylase activity in the absence of acarbose (a) or OG-ext (b) by Dunnett’s test after an analysis of variance with P < 0.05.

Inhibitory effect of OG-ext on maltase-mediated maltose degradation. Maltose was digested in maltase containing solution for 30 minutes at 37°C in the absence or presence of acarbose (a) or OG-ext (b). Each bar represents the mean ± SEM (n = 4). * indicates a significant difference from remaining maltase activity in the absence of acarbose (a) or OG-ext (b) by Dunnett’s test after an analysis of variance with P < 0.05.
To investigate the inhibitory effect of OG-ext on SGLT1-mediated intestinal absorption of glucose, we performed an uptake study using a human intestinal model cell line, Caco-2, and the fluorescence glucose analog, 2-deoxy-2-[(7-nitro-2,1,3-benzoxadiazol-4-yl)amino]-d-glucose (2-NBDG), as previously described. 7 Caco-2 cells differentiate into mature intestinal epithelial-like cells and express SGLT1 and other sodium-independent glucose transporters, such as GLUT1, 2, 3, and 5. 12,13 Moreover, previous studies have suggested that both SGLT1 and GLUTs transport 2-NBDG as a substrate. 14,15 Thus, separation of SGLT1- and GLUTs-mediated 2-NBDG uptake by Caco-2 cells is necessary to investigate the inhibitory potential on SGLT1. To prove the contribution of SGLT1 to sodium-dependent 2-NBDG uptake by Caco-2 cells, we studied the inhibitory effect of phlorizin, a known inhibitor of SGLTs, under Na+-containing and Na+-free conditions (Figure 4). Although a significant inhibition of 2-NBDG uptake by the addition of 1 mM phlorizin was observed under Na+-containing conditions, 1 mM phlorizin did not reduce 2-NBDG uptake under Na+-free conditions. Therefore, sodium-dependent 2-NBDG uptake by Caco-2 cells seems to be almost mediated by SGLT1.

Phlorizin-sensitive Na+-dependent 2-NBDG uptake by Caco-2 cells. 2-NBDG (300 µM) uptake for 120 minutes in the absence or presence of 1 mM phlorizin under Na+-containing (black bar) or Na+-free (white bar) conditions. Each bar represents the mean ± SEM (n = 4). * indicates a significant difference from 2-NBDG uptake in the absence of phlorizin under Na+-containing condition by Dunnett’s test after an analysis of variance with P < 0.05. n.s. means non-significant difference.
The inhibitory effect of OG-ext on SGLT1-mediated 2-NBDG uptake was examined (Figure 5). OG-ext inhibited 2-NBDG uptake by Caco-2 cells in the presence of Na+ in a concentration-dependent manner. Significant inhibition was observed over 0.5 mg/mL OG-ext. On the contrary, the maximum concentration of OG-ext had no effect on 2-NBDG uptake under Na+-free conditions. Moreover, cellular accumulated 2-NBDG amount in the presence of 1.5 mg/mL OG-ext under Na+-containing conditions was equivalent to that seen in the absence of OG-ext under Na+-free conditions. These results suggest that OG-ext has specific inhibitory potential against SGLT1-mediated glucose uptake.

Effect of OG-ext on Na+-dependent 2-NBDG uptake by Caco-2 cells. 2-NBDG (300 µM) uptake for 120 minutes in the absence or presence of various concentrations of OG-ext under Na+-containing (black bar) or Na+-free (white bar) conditions. Each bar represents the mean ± SEM (n = 4). * indicates a significant difference from 2-NBDG uptake in the absence of OG-ext under Na+-containing condition by Dunnett’s test after an analysis of variance with P < 0.05. n.s. means non-significant difference.
In the present study, our results indicate that OG-ext exhibits an inhibitory effect on the postprandial increase of blood glucose. Since OG-ext suppresses the increase of blood glucose after glucose-loading in mice and inhibits phlorizin-sensitive sodium-dependent glucose uptake in Caco-2 cells, the anti-hyperglycemic effect of OG-ext is mainly exerted by the inhibition of SGLT1-mediated intestinal glucose uptake. Moreover, OG-ext inhibits digestive enzymes of carbohydrates, albeit weakly. Thus, we could successfully provide theoretical evidence for using OG leaves with the aim of improving diabetes mellitus. Further investigation is necessary to identify the effective constituents of OG leaves.
Experimental
Plant Material
Leaves of OG were harvested during November and December in 2016 at a local farm on Ishigaki Island, Okinawa, Japan. A voucher specimen was deposited in the Faculty of Pharmacy, Kindai University.
Preparation of OG-Ext
An amount of 588.1 g of raw OG leaves was crushed and then extracted 2 times with ethanol/water (1:1, v/v). The extract was passed through a paper filter and finally the filtered solution was evaporated at 40°C. Resultant residues were freeze dried and kept at room temperature until use. An amount of 13.21 g of dried extract was obtained as OG-ext.
HPLC Analysis of OG-Ext
OG-ext was diluted in 50% acetonitrile at 0.5 mg/mL and centrifuged at 10 000 × g for 5 minutes at 4°C. Then, ingredients contained in 0.5 mg/mL OG-ext were analyzed by using Agilent 1200 HPLC system (Agilent Technologies, Palo Alto, CA, USA). OG-ext and blank (50% acetonitrile) were injected at 30 µL and the analytical column was a COSMOSIL Packed Column 5C18-MS-II (150 mm×4.6 mm, Nacalai Tesque, Kyoto, Japan) maintained at 40°C. Gradient elution was performed using mobile phase composed of water (A) and acetonitrile (B) at a flow rate of 1 mL/min. The gradient profile was 10% to 90% B for 0 to 30 minutes. The ingredients were detected by UV absorption at 254 nm. The obtained chromatogram is shown in Figure 6.
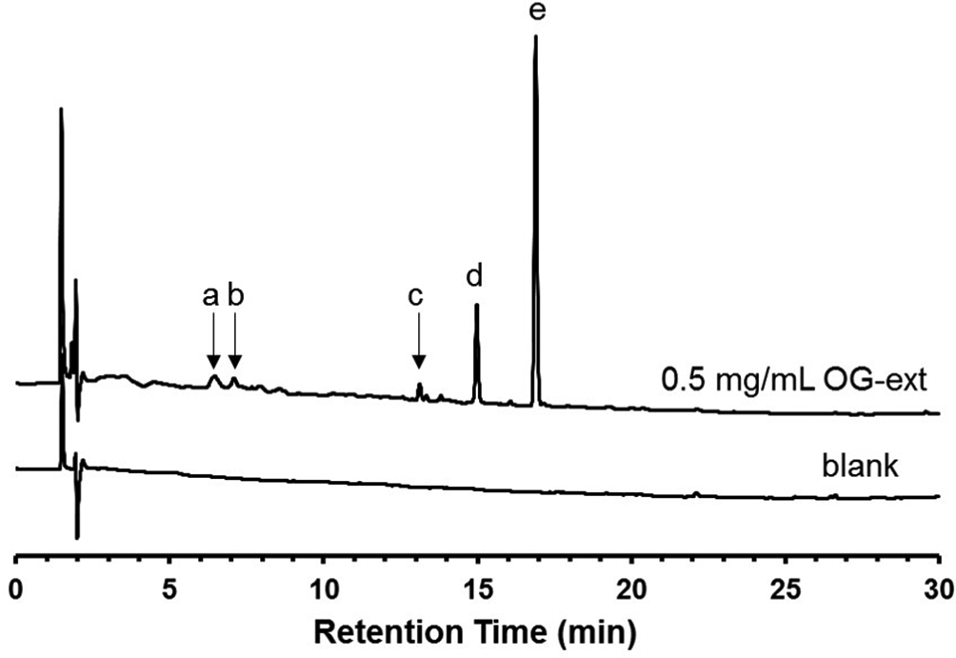
Chromatogram obtained in the HPLC-UV analysis of OG-ext. The peaks derived from 5 unknown compounds contained in 0.5 mg/mL OG-ext diluted in 50% acetonitrile were detected at 6.452 (a), 7.090 (b), 13.126 (c), 14.980 (d), and 16.892 (e) minutes.
Inhibitory Effect of OG-Ext on Blood Glucose Levels in α-Starch or Glucose-Loaded Mice
Male ICR mice (5-7 weeks) were used in this study. All mice were allowed free access to food and water under a standard 12 hours light/12 hours dark cycle in a temperature-controlled room (23°C ± 2°C). The animals were fasted for 16 to 20 hours prior to the beginning of the experiment, but were allowed free access to water. OG-ext (0, 250, or 500 mg/kg) was suspended in an α-starch (4.0 g/kg) solution or glucose (2.0 g/kg) containing 0.5% (w/v) carboxymethyl cellulose solution and administrated orally to fasted mice. Then, blood glucose was monitored using LAB Gluco (ForeCare Japan, Co., Ltd. Tokyo, Japan) at the indicated time points. All animal experimentation was carried out in accordance with the requirement of Kindai University Institutional Animal Care and Use.
α-Amylase Activity Assay
α-Amylase activity assay was performed as previously described with some modifications. 16 Briefly, α-amylase (Wako, Osaka, Japan) diluted to 50 U/mL in citrate-phosphate buffer (150 mM, pH 6.9) containing 50 mM NaCl and 5 mM CaCl2 was preincubated for 5 minutes at 37°C in the presence or absence of acarbose (0.2 or 2 mg/mL) or OG-ext (0.2 or 2 mg/mL). OG-ext was dissolved in dimethyl sulfoxide (DMSO) and assay was performed under presence of 1% DMSO as final concentration in all experiments containing non-treated control. The reaction was initiated by adding the 1% α-starch solution for 10 minutes at 37°C. The reaction was terminated by boiling for 3 minutes and stored on ice. Produced glucose was measured using LabAssay Glucose (Wako).
Maltase Activity Assay
Maltase activity assay was performed by using α-Glucosidase Inhibitory Activity Assay Kit (Wako). Briefly, maltose solution was preincubated for 3 minutes at 37°C in the presence or absence of acarbose (0.25 or 1 µg/mL) or OG-ext (0.125-2 mg/mL). OG-ext was dissolved in DMSO and assay was performed under presence of 1% DMSO as final concentration in all experiments containing non-treated control. The reaction was initiated by adding the maltase solution for 30 minutes at 37°C. The reaction was terminated by boiling for 3 minutes and stored on ice. Produced glucose was determined as described above.
Cell Culture
Caco-2 cells were obtained from RIKEN BioResource Center (Ibaraki, Japan). Caco-2 cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% (v/v) fetal bovine serum (Sigma-Aldrich, St Louis, MO, USA), 1% nonessential amino acid (Nacalai Tesque), 100 units/mL penicillin, and 100 µg/mL streptomycin, at 37°C in a humidified 5% CO2 atmosphere.
Uptake Assay in Caco-2 Cells
Glucose uptake was assayed using 2-NBDG (Cayman Chemical, Ann Arbor, MI, USA) as previously described, with slight modifications. 7 Briefly, Caco-2 cells were seeded in a 96-well plate at a density of 1.0 × 104 and cultured for 16 to 20 days before use for the experiment. The cells were washed and preincubated for 15 minutes at 37°C with Na+-containing buffer (125 mM NaCl, 4.8 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4, 1.2 mM CaCl2, and 25 mM HEPES, pH 7.4) or Na+-free buffer (125 mM choline chloride, 4.8 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4, 1.2 mM CaCl2, and 25 mM HEPES, pH 7.4) in the absence or presence of phlorizin (1 mM) as well as OG-ext (0.05-1.5 mg/mL). Then, the buffer was removed, and 2-NBDG uptake was initiated by adding the Na+-containing or Na+-free buffers containing 300 µM 2-NBDG in the absence or presence of phlorizin or OG-ext at the same concentration as that used during preincubation. OG-ext and phlorizin was dissolved in DMSO and assay was performed under presence of 1% DMSO as final concentration in all experiments containing non-treated control. The reaction was terminated by washing with ice-cold Na+-containing or Na+-free buffer, and then the cells were solubilized in 0.1% (w/v) Triton X-100. 2-NBDG-derived fluorescence was measured using a fluorescence microplate reader (FLUOROSKAN, Thermo Scientific, Rockford, IL, USA) at excitation and emission wavelengths of 485 and 535 nm, respectively. The protein concentration in cell lysates was measured with the Pierce BCA Protein Assay Kit (Thermo Scientific) according to the manufacturer’s instructions. Intracellular 2-NBDG accumulation was normalized by protein content.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the “Antiaging” Project for Private Universities, with a matching fund subsidy from the Japanese Ministry of Education, Culture, Sports, Science, and Technology (MEXT). This research was also supported in part by the MEXT-Supported Program for the Strategic Research Foundation at Private Universities, 2014–2018 [grant number S1411037].
