Abstract
Pueraria peduncularis belongs to the genus Pueraria DC., and has a wide range of medicinal and agricultural values. Previous studies have shown that methanol extracts of P. peduncularis had broad range bioactivities against different pests such as insects, phytopathogens, and snails; however, the specific studies with regard to active compounds against these pests have not been reported. In the current study, we systematically assessed the effects of P. peduncularis root extract against cancer cells, and we also isolated, purified, and analyzed the active ingredients of 8 different compounds from the root of P. peduncularis. To the best of our knowledge, coumestrol (compound
Diversity of plant resources are available in China at various agro-ecological zones in abundant quantity. Since the 1960s, Chinese medicinal experts have researched and developed a batch of natural anticancer drugs with definite efficacy and relatively small or no adverse reactions. At present, more than 2000 kinds of plants have been screened out for anticancer active ingredients. 1 These plants are mainly concentrated in the N yssaceae, A pocynaceae, C ephalotaxaceae, L eguminosae, R anunculaceae, C elastraceae, L iliaceae, R ubiaceae, A maryllidaceae, S olanaceae, R utaceae, M enispermaceae, A nnonaceae, S imarubaceae, L amiaceae, T hymelaeaceae, P odocarpaceae, E uphorbiaceae, T axodiaceae, C ucurbitaceae, U mbelliferae, and R utacerae. According to the statistics, from 1940 to 2002, about 40% of antitumor drugs came from plants themselves, while 8% came from bionic plants. 2
Thus, it is obvious that plants can produce a wide variety of secondary metabolites that are worthwhile and can be used for several purposes. There are more than 400 000 kinds of secondary metabolites found on earth. 3 Among these, approximately 200 are plant-based secondary metabolites which possess great anticancer activities that were tested in various experiments on animals. The secondary metabolites of these plants are mainly alkaloids, terpenoids, polysaccharides, phytoprotein, podophyllin lignans, harringtonine, homoharringtonine, flavonoids, saponins, taxanes, and polypeptide. 4,5 Some drugs derived from these secondary metabolites have been used to treat various types of cancer, for example, paclitaxel is mainly used for the first-line treatment of advanced ovarian cancer, 6,7 breast cancer, and other tumor cells; in addition, it is also used as the second- and third-line treatment for liver, advanced gastric, and prostate cancer. 8 Moreover, isovinblastine is used in the clinical treatment of breast cancer, lymphoma, esophageal cancer, and other tumors. Furthermore, podophyllum lignans have broad-spectrum anticancer activity and have good curative effects against small cell lung cancer, 9 leukemia, lymphatic sarcoma, glioma, Hodgkin’s disease, and other tumors. 10 Thus, at present, the derivatizations of various compounds are increasingly being performed, and these compounds are expected to be applied in the clinics as soon as possible.
Beside other plants, Pueraria peduncularis (Grah. ex Benth.) Benth. has a broad range of medicinal and agricultural values. It belongs to the genus Pueraria within the Leguminosae family, and is widely distributed across India, Pakistan, Burma, Nepal, and Southwestern China. 11 Previous studies had shown that methanol extracts of P. peduncularis have broad range bioactivities against different pests such as insects, phytopathogens, and snails, however, studies with reference to these active compounds against these pests have rarely been reported. In our previous study, we identified the active compounds such as pedunsaponin A and pedunsaponin C that were used against snails. 12 But in our current study, we isolated, purified, and analyzed the active ingredients from the root of P. peduncularis, and thereafter, we systematically assessed their effects against different phytopathogens and cancer cells. Thus, our study provides a breakthrough and scientific basis for the development and application of P. peduncularis extracts.
Materials and Methods
Isolation and Structural Analysis
Plant material and extraction of root compounds
Roots of P. peduncularis were collected from wild populations of P. peduncularis in Yaan City located in Sichuan Province, PR China (29.90°N, 102.92°E) in October 2015. Roots were initially dried in a drying oven at 60°C and then were ground using a powder grinder. 13 A total of 20.0 kg of root powder was extracted using 100 L of extraction mixture (ethanol/sterile water, 4/1, v/v) at 60°C for 6 hours, and the same procedure was repeated 3 times. The organic solvent was combined and concentrated at 60°C using a rotary evaporator (Rotavapor R-205; Büchi, Essen, Germany). Then, a total of 20 L of concentrated solution was harvested, and concentrated solution was further extracted 2 times by adding 20 L of n-butanol through a separating funnel. The n-butanol phase was evaporated at 60°C to produce 1.13 kg of crude extracts.
Isolation and purification of root compounds
The crude extract of n-butanol was suspended in 20 L of H2O. The diluents were absorbed by AB-8 resins (15 kg) for 24 hours, and then resins were eluted with 0%, 10%, 30%, 85%, and 95% (v/v) ethanol. For the antitumor assay, the solution of 95% ethanol phase, numbered KG02, was collected and concentrated at 60°C.
KG02 fraction was further separated into different fractions (Fr.Q1-Q6) via a reverse-phase medium-pressure column and eluted with 55%, 80%, and 100% (v/v) methanol. And the flow of each segment was concentrated.
The flow fraction Q2 was mixed according to the ratio of silica gel = 1:1. After the sample was dried, it was added to the top of the silica gel column and eluted with ethyl acetate-methanol gradients of 1:0, 3:1, and 0:1. The Q2-1 and Q2-2 segments were obtained.
The Q2-1 segment and Q3 segment were added to the sephadex column and then eluted with mixtures of dichloromethane and methanol (1:1, v/v) and 80% (v/v) methanol, respectively. The eluent was collected and concentrated, denoted as Q2-1-1 and Q3-1.
The Q2-2 was eluted with 35% acetonitrile-water (containing 0.02% trifluoroacetic acid) via a reverse-phase medium-pressure column , and the eluted fluid was condensed into Q2-2-1.
Sections Q4 and Q6 were dispersed with 10% tetrahydrofuran-water and extracted with ethyl triploid ethyl acetate, and the water layers were recorded as Q4-1 and Q6-1, respectively.
These fractions were further separated by high-performance liquid chromatography (HPLC) (Agilent, 1200 series, Waldbronn, Germany) equipped with a Phenomenex C18 column (250 × 10 mm, Phenomenex; Aschaffenburg, Germany) and a UV detector. Q2-1-1 and Q3-1 were eluted with 80% methanol water, Q2-2-1 was eluted with 35% acetonitrile-water, Q4-1 was eluted with 84% methanol water, and Q6-1 was eluted with 88% methanol water. The controlled flow rate was 1 mL/min. The main chromatographic peak number, peak time, and separation degree of each flow were collected by peak. Q2-1-1 was eluted to obtain compound
The purification of all these compounds was performed using HPLC by following protocol and conditions: Chromatographic column: Agilent Extend-C18, 2.5*50 mm, 1.8 µm; chromatographic conditions: 30°C; flow rate 0.4 mL/min; UV detection: maximum absorption of different compounds; mobile phase: different compounds have a different mobile phase proportion of acetonitrile and distilled water containing 0.1% formic acid.
Chemical Structural Elucidation
The chemical structures of these compounds were identified using nuclear magnetic resonance (NMR) and mass spectral data 1H- and 13C-NMR spectra on a Bruker DRX 500 spectrometer and electron ionization mass spectrometry (EI-MS) on an Agilent 1290/6460 Triple Quadrupole liquid Chromatograph Mass Spectrometer (LC/MS).
Antitumor Activity Assay
Cell culture
The lung adenocarcinoma cell line A549, hepatoma cell line QGY7703, hepatoma cell line HuH7-HCV, and breast cancer cell line MCF-7 (purchased from the Cell Bank of the Chinese Academy of Sciences) were cultured in dulbecco's modified eagle medium (DMEM), while the colon adenocarcinoma cell line SW480 (purchased from the Cell Library of the Chinese Academy of Sciences) and prostate cancer cell line DU149 (purchased from ATCC) were cultured in RPMI-1640 medium. All cells were incubated at 37°C with 5% CO2 in incubator for further study.
Cell detection
The 96-well plate densities of A549, SW480, 7703, Huh7, DU149, and MCF-7 cells were 5000, 12 000, 5000, 5000, 12 000, and 5000 per well, respectively, which were used by following the procedure of Kang et al. 14 The cells were cultured in an incubator at 37°C with 5% CO2. After the cells adhered to the wall, a final concentration of 10 μmol compound was added and then cells were further cultured for 24 hours at 37°C. After the cell culture was terminated, 5 µL CCK8 solution (Blue Skies Biological Technology Co., Ltd.) was added in each hole. After incubation for 4 hours, the absorbance rate of each well at 450 nm was measured using a microplate reader (BioTek, Synergy 4 MLFPTAD), and then cell inhibition rate was calculated.
Results
Isolation, Characterization, and Chemical Structures of Isolated Compounds
In our study, a total of 8 different compounds were isolated from the root of P. peduncularis. The characteristics, composition, and skeletal structure of these compounds are summarized here:
Compound 1
Light yellow powder, ESI-MS m/z: 269 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 7.83 (1H, d, J = 8.5 Hz, H-7), 7.68 (1H, d, J = 8.4 Hz, H-1), 7.16 (1H, d, J = 1.9 Hz, H-10), 6.94 (1H, m, H-4), 6.92 (1H, m, H-2), 6.90 (1H, J = 1.9 Hz, H-4); 13C-NMR (100 MHz, DMSO-d
6) δ: 122.7 (C-1), 113.7 (C-2), 161.2 (C-3), 103.0 (C-4), 154.6 (C-4a),157.6 (C-6), 102.0 (C-6a), 114.5 (C-6b), 120.6 (C-7), 114.0 (C-8), 155.9 (C-9), 98.7 (C-10), 157.0 (C-10a), 159.4 (C-11a), 104.1 (C-11b). The 1H-NMR and 13C-NMR data are similar to the results of previous reports.
15
On the basis of the aforementioned evidence, the structure of compound
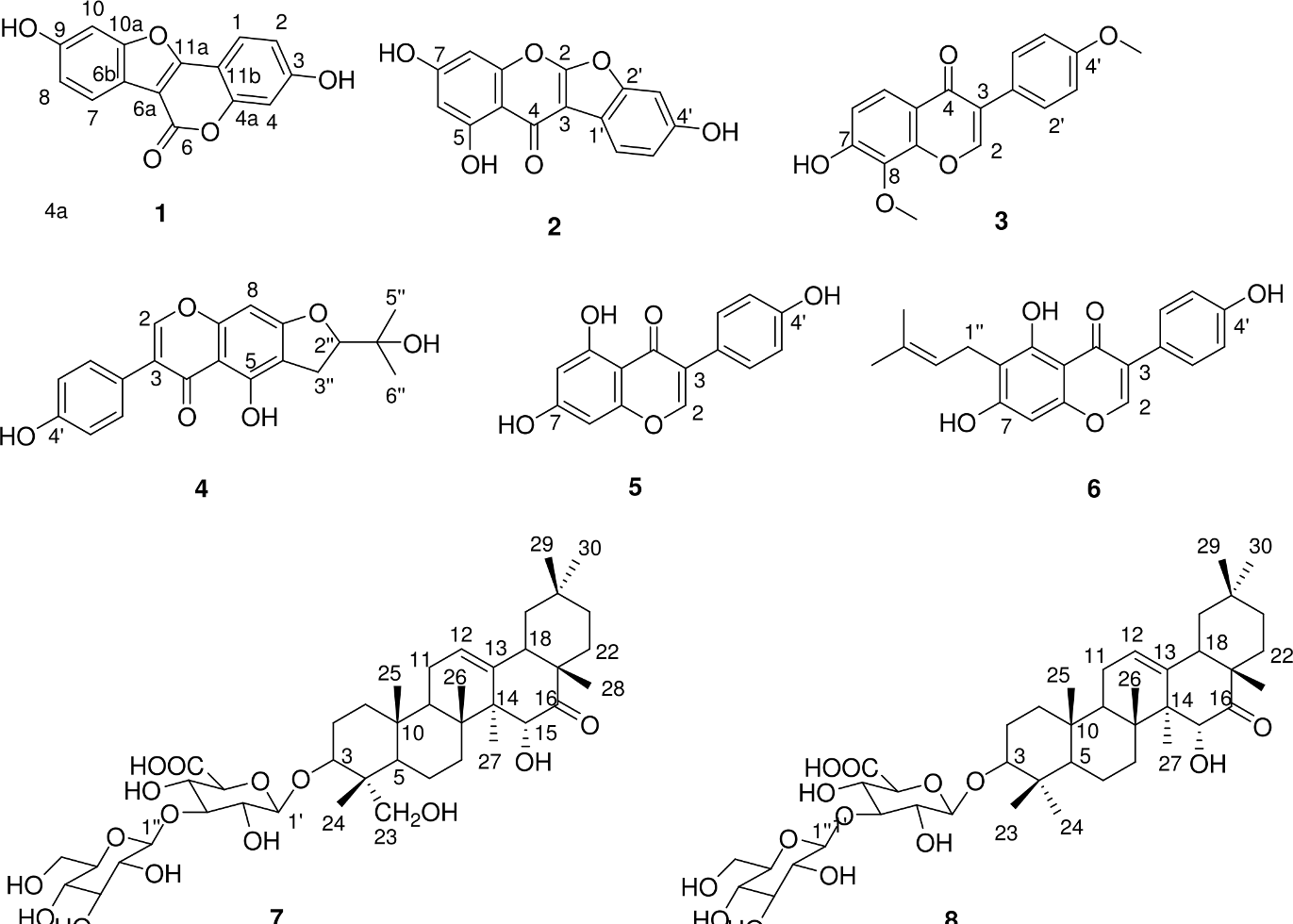
Chemical structure of compounds.
Compound 2
Light yellow powder, ESI-MS m/z: 285 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 7.71 (1H, d, J = 8.4 Hz, H-6′), 7.07 (1H, d, J = 1.8 Hz, H-3′), 6.90 (1H, dd, J = 8.4, 2.0 Hz, H-5′), 6.52 (1H, d, J = 1.9 Hz, H-8), 6.27 (1H, d, J = 1.9 Hz, H-6); 13C-NMR (100 MHz, DMSO-d
6) δ: 149.9 (C-2), 113.3 (C-3), 178.1 (C-4), 162.1 (C-5), 99.7 (C-6), 164.3 (C-7), 94.9 (C-8), 156.3 (C-9), 102.8 (C-10), 97.1 (C-1′), 154.6 (C-2′), 98.8 (C-3′), 162.1 (C-4′), 113.8 (C-5′), 121.0 (C-6′). The 1H-NMR and 13C-NMR data are similar to the results of previous reports.
16
On the basis of the aforementioned facts, compound
Compound 3
Light yellow powder, ESI-MS m/z: 299 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 8.42 (1H, s, H-2), 7.72 (1H, d, J = 8.9 Hz, H-5), 7.51 (2H, d, J = 8.7 Hz, H-2′, 6′), 7.04 (1H, d, J = 8.9 Hz, H-6), 6.99 (2H, d, J = 8.7 Hz, H-3′, 5′), 3.87 (3H, s, -OCH3), 3.78 (3H, s, -OCH3); 13C-NMR (100 MHz, DMSO-d
6) δ: 153.1 (C-2), 123.0 (C-3), 174.8 (C-4), 120.8 (C-5), 115.3 (C-6), 154.9 (C-7), 134.7 (C-8), 150.7 (C-9), 117.4 (C-10), 124.2 (C-1′), 130.1 (C-2′), 113.6 (C-3′), 159.0 (C-4′), 113.6 (C-5′), 130.1 (C-6′), 60.8 (8-OCH3), 55.2 (4′-OCH3). The 1H-NMR and 13C-NMR data are similar to the results of the previous study.
17
The aforementioned evidence revealed that compound
Compound 4
Light yellow powder, ESI-MS m/z: 355 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 13.18 (1H, s, 5-OH), 9.64 (1H, s, 4′-OH), 8.36 (1H, s, H-2), 7.39 (2H, d, J = 8.5 Hz, H-2′, 6′), 6.83 (2H, d, J = 8.6 Hz, H-3′, 5′), 6.54 (1H, s, H-8), 4.76 (2H, m, H-2″), 3.10 (2H, d, J = 8.5 Hz, H-3″), 1.16 (3H, s, H-6″), 1.14 (3H, s, H-5″); 13C-NMR (100 MHz, DMSO-d
6) δ: 154.1 (C-2), 122.3 (C-3), 180.4 (C-4), 156.1 (C-5), 109.3 (C-6), 166.3 (C-7), 88.7 (C-8), 157.5 (C-9), 105.4 (C-10), 121.2 (C-1′), 130.2 (C-2′, 6′), 115.1 (C-3′, 5′), 157.5 (C-4′), 91.6 (C-2″), 25.8 (C-3″), 70.0 (C-4″), 24.9 (C-5″), 25.8 (C-6″). The 1H-NMR and 13C-NMR data are similar to the results of previous studies.
18
These evidence demonstrated that the structure of compound
Compound 5
White crystal, ESI-MS m/z: 271 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 12.96 (1H, s, 5-OH), 10.88 (1H, s, 7-OH), 9.60 (1H, br s, 4′-OH), 8.31 (1H, s, H-2), 7.37 (2H, d, J = 8.5 Hz, H-2′, 6′), 6.82 (2H, d, J = 8.6 Hz, H-3′, 5′), 6.38 (1H, d, J = 2.0 Hz, H-6), 6.22 (1H, d, J = 2.0 Hz, H-8); 13C-NMR (100 MHz, DMSO-d
6) δ: 154.0 (C-2), 122.3 (C-3), 180.2 (C-4), 162.0 (C-5), 99.0 (C-6), 164.3 (C-7), 93.7 (C-8), 157.4 (C-9), 104.5 (C-10), 121.2 (C-1′), 130.2 (C-2′, 6′), 115.1 (C-3′, 5′), 157.6 (C-4′). The 1H-NMR and 13C-NMR data are also similar to the results of previous reports.
19
Based on the aforementioned evidence, compound
Compound 6
Light yellow powder, ESI-MS m/z: 339 [M+H]+, 1H-NMR (400 MHz, DMSO-d
6) δ: 13.22 (1H, s, 5-OH) 10.92 (1H, s, 4′-OH), 8.29 (1H, s, H-2), 7.36 (2H, d, J = 8.6 Hz, H-2′, 6′), 6.82 (2H, d, J = 8.6 Hz, H-3′, 5′), 6.45 (1H, s, H-8), 5.16 (2H, t, J = 7.0 Hz, H-2″), 3.22 (2H, d, J = 7.0 Hz, H-1″), 1.72 (3H, s, H-4″), 1.62 (3H, s, H-5″); 13C-NMR (100 MHz, DMSO-d
6) δ: 153.8 (C-2), 122.2 (C-3), 180.3 (C-4), 104.3 (C-5), 158.9 (C-6), 111.1 (C-7), 162.0 (C-8), 92.9 (C-9), 155.4 (C-10), 121.4 (C-1′), 130.2 (C-2′, 6′), 115.1 (C-3′, 5′), 157.4 (C-4′), 21.1 (C-2″), 122.2 (C-3″), 130.8 (C-4″), 17.8 (C-5″), 25.5 (C-6″). The data of 1H-NMR and 13C-NMR are also similar to the results of previous studies.
19
On the basis of the above evidence, the structure of compound
Compound 7
White powder, ESI-MS m/z: 833 [M+Na]+, 1H-NMR (400 MHz, Pyridine-d
5) δ: 4.32 (1H, m, H-3), 5.42 (1H, br s, H-12), 4.76 (1H, s, H-15), 4.32 (1H, m, H-23), 3.70 (1H, d, J = 10.8 Hz, H-23), 0.95 (3H, s, H-24), 0.96 (3H, s, H-25), 1.16 (3H, s, H-26), 1.25 (3H, s, H-27), 1.16 (3H, s, H-28), 0.82 (3H, s, H-29), 0.77 (3H, s, H-30); 13C-NMR (100 MHz, Pyridine-d
5) δ: 38.8 (C-1), 26.2 (C-2), 81.9 (C-3), 43.5 (C-4), 47.0 (C-5), 18.2 (C-6), 35.5 (C-7), 41.7 (C-8), 47.1 (C-9), 36.7 (C-10), 24.0 (C-11), 125.7 (C-12), 142.0 (C-13), 54.1 (C-14), 72.7 (C-15), 217.3 (C-16), 46.3 (C-17), 52.9 (C-18), 47.8 (C-19), 30.9 (C-20), 35.8 (C-21), 30.9 (C-22), 64.0 (C-23), 13.7 (C-24), 16.3 (C-25), 17.7 (C-26), 21.9 (C-27), 28.0 (C-28), 33.0 (C-29), 23.2 (C-30), 105.9 (C-1′), 74.3 (C-2′), 87.5 (C-3′), 71.6 (C-4′), 77.5 (C-5′), 172.2 (C-6′), 106.0 (C-1″), 75.7 (C-2″), 78.3 (C-3″), 71.7 (C-4″), 78.8 (C-5″), 62.5 (C-6″). The 1H-NMR and 13C-NMR data are similar to the results of previously conducted studies.
20
On the basis of the aforementioned facts, compound
Compound 8
White powder, ESI-MS m/z: 817 [M+Na]+, 1H-NMR (400 MHz, Pyridine-d
5) δ: 3.34 (1H, dd, 11.7, 4.2, H-3), 5.44 (1H, br s, H-12), 4.78 (1H, s, H-15), 2.55 (1H, dd, J = 13.9, 3.0 Hz, H-18), 1.26 (3H, s, H-23), 0.99 (3H, s, H-24), 0.87 (3H, s, H-25), 1.13 (3H, s, H-26), 1.32 (3H, s, H-27), 1.17 (3H, s, H-28), 0.81 (3H, s, H-29), 0.84 (3H, s, H-30), 4.98 (1H, d, J = 7.7 Hz, H-1′), 5.38 (1H, d, J = 7.8 Hz, H-1″); 13C-NMR (100 MHz, Pyridine-d
5) δ: 38.7 (C-1), 26.6 (C-2), 89.0 (C-3), 39.4 (C-4), 55.3 (C-5), 18.5 (C-6), 35.8 (C-7), 41.7 (C-8), 46.9 (C-9), 36.8 (C-10), 24.0 (C-11), 125.7 (C-12), 142.0 (C-13), 54.1 (C-14), 72.7 (C-15), 217.3 (C-16), 46.3 (C-17), 52.9 (C-18), 47.9 (C-19), 30.9 (C-20), 35.9 (C-21), 30.9 (C-22), 28.0 (C-23), 15.7 (C-24), 17.0 (C-25), 17.7 (C-26), 21.9 (C-27), 28.0 (C-28), 33.0 (C-29), 23.2 (C-30), 106.8 (C-1′), 74.3 (C-2′), 87.5 (C-3′), 71.7 (C-4′), 77.4 (C-5′), 172.2 (C-6′), 105.9 (C-1″), 75.6 (C-2″), 78.2 (C-3″), 71.5 (C-4″), 78.7 (C-5″), 62.4 (C-6″). The 1H-NMR and 13C-NMR data are similar to the results of previous reports.
21
Based on the aforementioned evidence, the structure of compound
Antitumor Activity of the Compounds
The cytotoxic activity of different compounds, derived from P. peduncularis root, against various cancer cells were determined at the concentration of 10 µg/mL, and the cytotoxic activity of these active compounds is shown in Table 1. The results showed that extract of the P. peduncularis root had a strong cytotoxic activity on the lung adenocarcinoma cell line A549 and breast cancer cell line MCF-7, with inhibition rates of 31.0% and 33.0%, respectively. However, the compounds such as lupinalbin A (compound
Cytotoxic Activity of Treatment With 10 µg/mL P ueraria peduncularis Extract on Different Cancer Cells.

Note: Values with different lowercase letters indicate statistically significant differences at 0.05 probability level (DPS 9.50, Duncan’s multiple rage test).
Discussion
The importance of herbal medicine has been realized since ancient times. Currently, the use of plant derivatives against several human, animal, and plant diseases has gained vital place due to no or sometime rare side effects. Pueraria peduncularis belongs to the genus Pueraria DC., is believed to possess a wide range of medicinal and agricultural activities, and has good therapeutic potential. To better develop and utilize the resources of P. peduncularis, different researchers have studied the chemical composition of P. peduncularis. Flavonoids, saponins, esters, amino acids, and other substances have already been identified from P. peduncularis
21
; however, there are still many components that have not been identified yet, and the relationship between the biological activities and different compounds is also not clear yet. In our study, 8 different compounds were isolated and identified from the root of P. peduncularis. These compounds are primarily coumarins, isoflavones, and saponins. To the best of our knowledge, among these compounds, coumestrol (compound
It has been reported that P. peduncularis contains more than 10 different kinds of essential trace elements, which can promote child bone development and intellectual growth, and have also therapeutic effects against hypertension, coronary heart disease, and angina pectoris.
22
Currently, no any report has been found available especially on the anticancer aspect of P. peduncularis extract. However, our study found that the P. peduncularis extract had a strong cytotoxic activity on the lung adenocarcinoma cell line A549 and breast cancer cell line MCF-7. Further studies on the anticancer compounds of P. peduncularis showed that lupinalbin A (compound
In our study, erythrinin C (compound
Supplemental Material
Supplementary material - Supplemental material for Compounds From the Root of Pueraria peduncularis (Grah. ex Benth.) Benth. and Their Antitumor Effects
Supplemental material, Supplementary material, for Compounds From the Root of Pueraria peduncularis (Grah. ex Benth.) Benth. and Their Antitumor Effects by Huabao Chen, Yong Qian, Xiaomin Zhao, Tianxing Lv, Bin Wang, GuoShu Gong, Xiaoyan Qiu, Liya Luo, Min Zhang, GuangWei Qin, Muhammad Ibrahim Khaskheli and Chunping Yang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Key R&D Program of China under Grant no. 2018YFD0200500.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
