Abstract
A new eremophilanolide sesquiterpene, 1α,6β,10β-trihydroxyeremophila-7(11),8-dien-12,8-olide (macrophyllalarin A,
There are more than 110 species of Ligularia (Compositae) in China, mainly distributed in southwest, northwest, and north China.
1,2
So far, about 40 species of Ligularia have been studied for their chemical constituents. Many research groups have isolated and identified various chemical constituents (sesquiterpenoids, monoterpenes, diterpenoids, triterpenoids, lignans, sterols, benzofurans, phenylpropanes, pyrrolizidine alkaloids [PA], phenylpropanoids, and flavonoids) from the genus Ligularia.
3
-5
Some previous study reported that the chemical constituents of Ligularia macrophylla collected from different places showed large differences, then the diversity of chemical substances in this species is present.
6
-9
In order to enrich the chemical composition diversity of the same plant species at different collection sites, the chemical constituents of L. macrophylla which grows in the Yili river valley of Xinjiang were systematically studied. This paper reports the isolation and characterization of 1 new eremophilanolide sesquiterpenes, macrophyllalarin A (
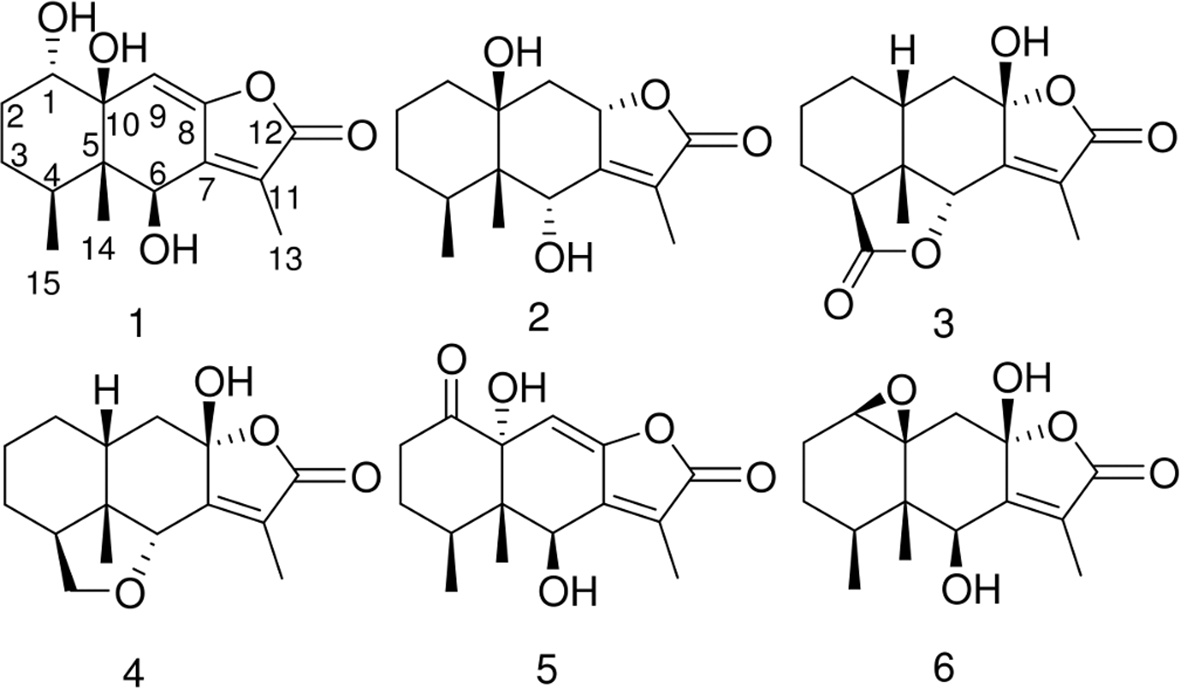
The structures of compounds 1 to
Compound
NMR Spectral Data of Compounds
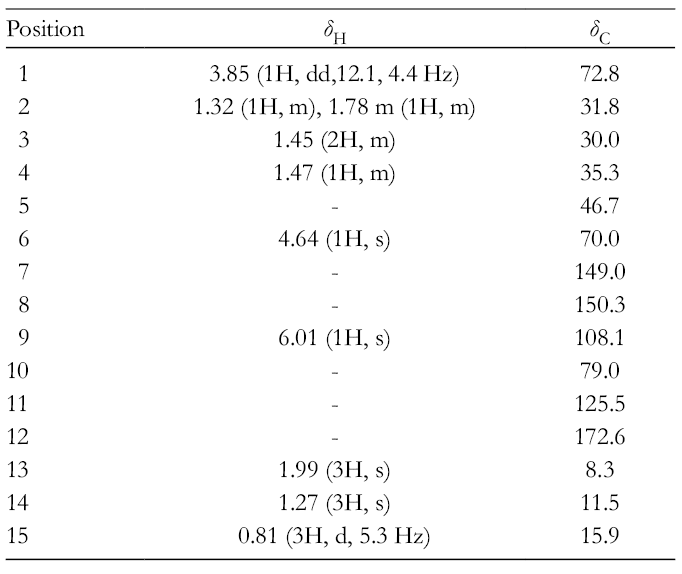

Key HMBC, 1H-1H COSY, and NOESY correlations of 1.
The stereochemistry of the compound
Compound
Ligularia macrophylla is mainly distributed in China.
1
It is reported that the characteristic chemical constituents of L. macrophylla collected from Lijiang in Yunnan province are lignans,
23
unlike this the characteristic chemical constituents of L. macrophylla collected from Xinjiang regions are eremophilane sesquiterpenes. Focusing on eremophilanes in L. macrophylla, most of them are furanoeremophilanes or eremophilan-12,8-olides with a variety of oxygen functionalities. In a sample from Kashikar region of south Xinjiang, the major characteristic chemical constituents were 9-oxygenated furanoeremophilanes.
6,12
These compounds were not obtained from a sample in Manas River of north Xinjiang, but benzofuran type of rearranged eremophilanes was isolated together with 1(10)-epoxyfuranoeremophilanes.
7
In contrast, less-oxidized eremophilanes, such as furanoeremophilan-15,6a-olide, were isolated from a sample of Kaskelen canyon of Kazakhstan.
24
Interestingly, the samples from Yili River of north Xinjiang (this study, 600 km east from Manas River), both 15,6α-olide (
Experimental
General
Melting points were determined by YRT-3 Melting Point Instrument (Henan Mingjun Instrument Co., Ltd. Henan, China) and are uncorrected. The IR spectra were recorded as KBr pellets on an Avatar 360 FT-IR ESP spectrophotometer (Nicolet, United States). The 1D and 2D NMR spectra also were measured by a Varian MR 400 NMR and a Bruker AV400 NMR (Bruker Corp, Germany). AR 2140 one part per million electronic balance (Mettler Corp, Ltd., Switzerland). HR-ESI-TOF-MS spectra were acquired using an Agilent 6210 ESI/TOF (Agilent Technologies, Santa Clara, CA, United States). UV-2401 PC UV-visible spectrophotometer (Shimadzu, Japan). FTS165 infrared spectrum analyzer (Bole company, United States). Spots of compounds were observed under UV light and visualized by spraying with vanillin and 10% H2SO4, followed by heating. Column chromatography (CC) was performed on silica gel (200-300 and 100-200 mesh; Qingdao Marine Chemical Factory, Qingdao, China).
Plant Material
The roots and rhizomes of L. macrophylla DC. were collected in July 2009 in Yili Nileke County, Xinjiang Province, P.R. China. The plant material was identified by Prof. Ping Yan, College of Life Sciences, Shihezi University, and a voucher specimen (WQ-LM-09-01) has been stored at the Herbarium, Department of Pharmacognosy, School of Pharmacy, Shihezi University, Xinjiang, China.
Extraction and Isolation
The air-dried roots and rhizomes parts (10.6 kg) of L. macrophylla were soaked and extracted with 95% EtOH at room temperature. The EtOH extract (3 × 15 L, 7 days each time) was concentrated under reduced pressure to give a semisolid (1900 g), a portion of which (1900 g) was suspended in H2O (2 L) and partitioned successively with petroleum ether (PE), AcOEt, and BuOH (each 3 × 2 L). The PE extracts (334 g) were chromatographed on silica gel column (5 kg of SiO2) by the use of a gradient of PE/acetone (from 100:1 to 0:100) to afford 11 fractions (Fr.1-Fr.11). After appropriate fractions were assembled, 3 major components were selected for further isolations. Fr.8 was subjected to repeated CC (SiO2; PE/acetone 8:1) to give subfractions Fr.8 A-D, Fr.8-A was subjected to repeated CC (SiO2; PE/AcOEt 8:1) to give
1α,6β,10β-Trihydroxy-Eremophil-7(11),8(9)-Dien-8(12)-Olide (Macrophyllalarin A, 1)
Colorless Crystal, M.p. 176°C-178°C (CH3OH);
UV (MeOH) λ max (log ε) 227 (3.256) nm;
IR (KBr): 3444, 2924, 1766, 1625, 1375, 1163, 1078, 1056 cm−1.
1H and 13C NMR (CD3OD): Table 1.
HRMS-ESI: m/z [M+Na]+calcd for C15H20O5Na+ 303.1203; found: 303.1207.
Supplemental Material
Supporting information - Supplemental material for A New Eremophilanolide Sesquiterpene From Ligularia macrophylla and Diversity of the Species
Supplemental material, Supporting information, for A New Eremophilanolide Sesquiterpene From Ligularia macrophylla and Diversity of the Species by Chuanqi Lu, Wei Dai, Wei Zhou, Lin Zhang and Qi Wang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by grants from the National Natural Science Foundation of China (30960463) and Science and Technology Planning Project (2011AB033) of the Xinjiang Production and Construction Corps, China (to Q.W.).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
