Abstract
Monascus pilosus BCRC 38093 is a mutant strain of M. pilosus BCRC 38072. The 95% ethanol extract of red yeast rice fermented by M. pilosus BCRC 38093 showed 4 major signals on high performance liquid chromatography (HPLC) examination. The extraction of metabolites and chromatography of the ethyl acetate crude extract on silica gel yielded 2 azaphilone derivatives, monascin (
The androgen receptor (AR) is a member of the steroid hormone receptor subfamily of nuclear receptors, which also includes the estrogen, progesterone, mineralocorticoid, and glucocorticoid receptors. 1 The natural steroid androgens, namely testosterone, the circulating hormone, and dihydrotestosterone (DHT), its active metabolite, are responsible for the development and maintenance of normal prostate cells.
Either the overexpression of an AR or expression of mutated AR genes has been found in several diseases, such as cancer, including prostate and breast cancers, as well as other disorders such as polyglutamate disease, androgen-dependent alopecia, hirsutism, acne, prostatic hyperplasia, spinal and muscular atrophy, and Kennedy’s disease. However, this condition can also promote the malignant growth of the prostate gland, and it is known that most prostate cancers (PCas) are androgen dependent. 2,3 The mechanism of action of the androgens involves an interaction with a specific AR (AR a ), and this receptor is postulated to play a crucial role in the development of PCa. 4
Red yeast rice (ang-kak, red koji), made by filamentous Monascus species, has traditionally been used in East Asia in the production of natural food colorant, such as for red rice wine, red soybean cheese, meat, meat products, and fish, to aromatize and conserve meat, fish, and soybean products. 5 It is also used as a folk medicine. 5 There are 4 representative species: Monascus pilosus, Monascus purpureus, Monascus rubber, and Monascus anka (class Ascomycetes and family Monascaceae). 5 Red yeast rice is employed for medicinal purposes like promotion of digestion and blood circulation, strengthening of the spleen, and removal of blood stasis. 6 The above-mentioned yeasts can produce several pigments and biologically active metabolites when grown on cooked rice. 5
In a series of studies on anti-androgen activity of natural products, we were especially interested to study the chemical composition of red yeast rice, and 1 specific pink mutant, M. pilosus BCRC 38093, which is different from other traditional red mutant Monascus species, and which has been found to be one of the active ones. Careful examination of the EtOAc-soluble fraction of a 95% EtOH extract of the red yeast rice produced by M. pilosus BCRC 38093 has resulted in the isolation of 2 azaphilone derivatives, monascin (

Chemical structures of compounds 1 to
Monascus pilosus BCRC 38093 was mutated from M. pilosus BCRC 38072. The red yeast rice produced by fermentation with M. pilosus BCRC 38093 showed high monacolin K production. The red yeast rice was extracted with 95% ethanol, and the extract was then partitioned with ethyl acetate, and water, successively. The crude ethyl acetate layer showed strong anti-androgen activity. Further bioassay-directed fractionation of the active fraction led to the isolation of 2 yellow pigments, monascin (
To study the anti-androgen activity of the azaphilone derivatives isolated from M. pilosus, luminescent secreted alkaline phosphatase (SEAP) assays were used of MDA-MB-453 cells transiently transfected with SEAP reporter construct containing prostate-specific antigen (PSA) promoter. Transfected cells were treated with 10 nM DHT and azaphilone compounds for 48 hours and then harvested and assayed for SEAP activity. Chemiluminescence readout was normalized relative to the readout from cells treated with DHT alone. Monascinol (
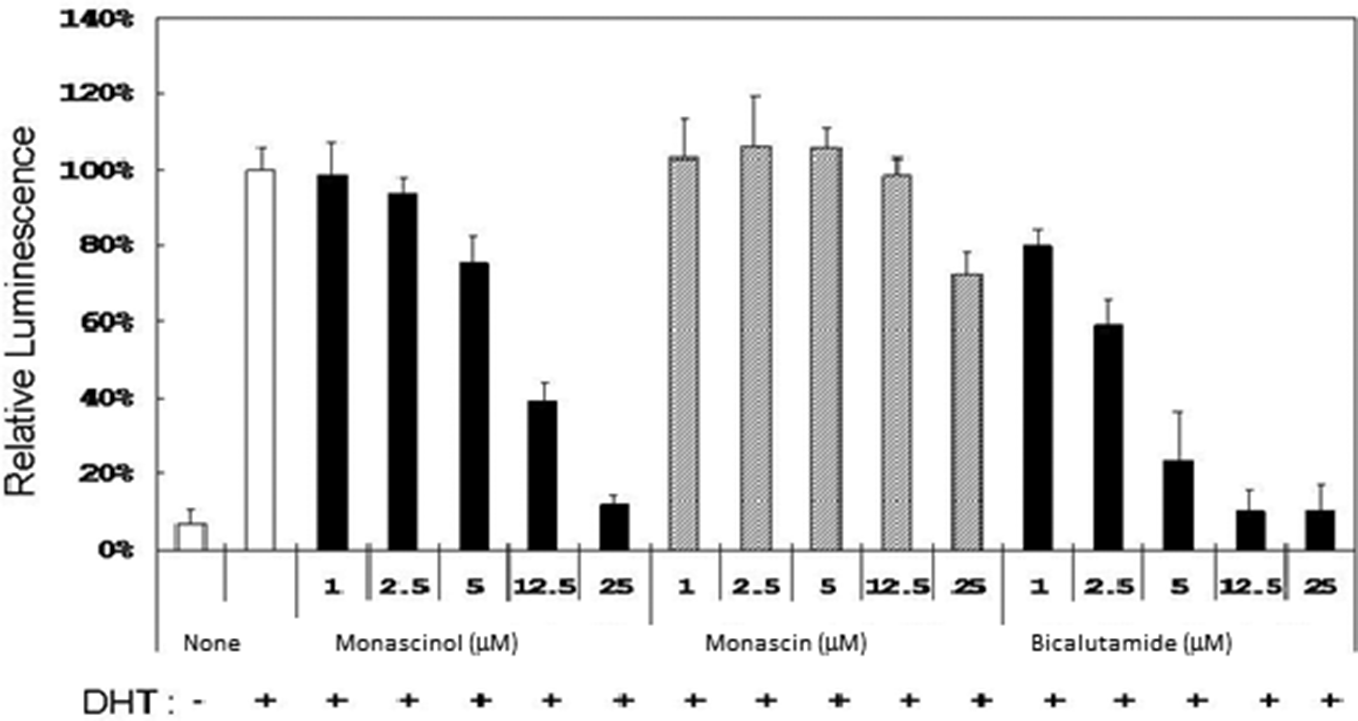
Anti-androgen activity of azaphilone analogs produced by Monascus pilosus. Luminescent secreted alkaline phosphatase assays of MDA-MB-453 cells transiently transfected with secreted alkaline phosphatase reporter construct containing PSA promoter. Transfected cells were treated with 10 nM dihydrotestosterone for 48 hours and then harvested and assayed for secreted alkaline phosphatase activity. The reported values are the means ± SD (n = 3).
In this study, we developed a HPLC technique using a gradient of phosphoric acid and acetonitrile, which allows quantitative analysis of the red yeast rice of M. pilosus and identification of 4 compounds.
From the bioactive fraction, we isolated 2 azaphilone derivatives, monascin (
The major anti-androgen active components were identified as monascin (
Experimental
General
Optical rotations were measured on a Jasco P-1020 digital polarimeter, UV spectra were obtained on a Jasco UV-240 spectrophotometer in MeOH, and IR spectra (KBr or neat) were taken on a Perkin-Elmer System 2000 FT-IR spectrometer. 1D (1H, 13C, and DEPT) and 2D (COSY, NOESY, HSQC, and HMBC) NMR spectra using CDCl3 and CD3COCD3 as solvents were recorded on Varian Unity Plus 400 (400 MHz for 1H NMR and 100 MHz for 13C NMR) and Varian INOVA-500 (500 MHz for 1H NMR and 125 MHz for 13C NMR) spectrometers. Chemical shifts were internally referenced to the solvent signals in CDCl3 (1H, δ 7.26; 13C, δ 77.0) with tetramethylsilane (TMS) as the internal standard. Low-resolution ESI-MS were obtained on an API 3000 (Applied Biosystems) and high-resolution ESI-MS on a Bruker Daltonics APEX II 30e spectrometer. Silica gel (70-230 and 230-400 mesh; Merck) was used for column chromatography, and silica gel 60 F-254 (Merck) was used for thin layer chromatography (TLC) and prep. TLC. The prep-HPLC separations were performed using a Lab Alliance series III with a Model V520 UV detector on an Inertsil ODS (Octa Decyl Silane)-3 column, 5 µm, 250 mm × 10 mm i.d. (GL Sciences, Tokyo, Japan). All organic solvents used for partition and column chromatography were of analytical grade. Acetonitrile for HPLC analysis was of HPLC grade from Merck (Darmstadt, Germany). Cell lines were obtained from BCRC (Bioresource Collection and Research Center, Food Industry Research and Development Institute, Hsin-Chu). Cell culture medium RPMI 1640, penicillin, streptomycin, and FBS were purchased from Gibco BRL (Invitrogen Corporation, Carlsbad, CA, USA).
Microorganism and Preparation of Red Yeast Rice
Monascus pilosus BCRC 38093 was used throughout in this study, and specimens were deposited at the Bioresource Collection and Research Center (BCRC) of the Food Industry Research and Development Institute. Monascus pilosus BCRC 38093 was inoculated on Potato Dextrose Agar (PDA, Difco, United States) plates and incubated at 30°C for 7 days. The spores were washed from the PDA plate using sterile water (7 mL per plate) and the concentration of the spore suspension was calculated. Sterile zailai rice (50 g) was put in a jar and mixed with 8 mL of spore suspension (2 × 106/mL) and 12 mL of sterile water. The rice was incubated at 25°C and left for 14 days to obtain red yeast rice.
Extraction and Separation
The dried red yeast rice of M. pilosus BCRC 38093 (2.986 kg) was extracted 3 times with 95% EtOH at room temperature. The ethanol syrupy extract was partitioned between EtOAc and H2O (1:1) to afford 30.1 g of the EtOAc soluble fraction. This was chromatographed over silica gel (250 g, 70-230 mesh), eluting with n-hexane/ethyl acetate: 12:1, 10:1, 8:1, 6:1, 4:1, 2:1, 1:1, acetone, and methanol to produce 36 fractions. Fraction 17 was purified by preparative TLC (n-hexane-EtOAc, 1.5:1) to give monascin (
Analysis of Red Yeast Rice Compounds by HPLC
One gram of red yeast rice was extracted with 5 mL of ethanol by sonication. The ethanol crude extract was analyzed by HPLC with UV spectroscopic detection (waters 600 pump and 996 photodiode array detector) on an Inertsil ODS-3 column, 5 µm, 250 mm × 4.6 mm i.d. (GL Sciences, Tokyo, Japan) using isocratic elution with water (0.1% H3PO4)/acetonitrile (35:65, v/v) at a flow rate of 1 mL/min. Chromatograms were recorded at a wavelength of 237 nm. In this HPLC analytical method, 4 compounds were identified.
Assay for Anti-Androgen Activity
Human breast carcinoma MDA-MB-453 cells were maintained in roswell park memorial institute (RPMI) -1640 medium containing penicillin (25 U/mL), streptomycin (25 mg/mL), and 10% FBS in a humidified 5% CO2 atmosphere. Transfections were performed using the electroporation method with a pPSA-SEAP plasmid (courtesy of Prof Chi-Huei Wang of Kaohsiung Medical University). Briefly, 1 × 106 cells were resuspended in 0.5 mL medium with 10 µg plasmid DNA and pipetted into a 0.4 cm electroporation cuvette. Cells were incubated on ice for 5 minutes, electroporated at 250 V and 960 µF using a gene pulser (Bio-Rad, Hercules, CA, United States), and incubated for 5 more minutes on ice. Transfected cells were resuspended in growth medium with 10% charcoal-stripped FBS and seeded into 96-well microplates. After an overnight incubation, cells were treated with DHT (10 nM) and samples of interest for 48 hours. Aliquots of culture media were analyzed for SEAP activity using a phospha-light reporter chemiluminescence assay kit (Applied Biosystems, United States) and the resulting luminescence was detected using a VICTOR Light 1420 luminescence counter (PerkinElmer, United States). 10,11
Monascinol
Yellowish oil.
[α]D: –42.1 (c 0.21, CHCl3).
Rf: 0.56 (CHCl3-MeOH, 15:1).
IR (Neat): 3400, 1738 cm−1.
UV/Vis λmax (MeOH) nm (log ε): 230 (4.32), 388 (4.11).
1H NMR (600 MHz, CDCl3): 0.90 (3H, t, J = 6.8 Hz, CH3-6′), 1.25-1.46 (4H, m, CH2-4′, 5′), 1.42 (3H, s, CH3-16), 1.50-1.61 (4H, m, CH2-2′, 3′), 1.86 (3H, dd, J = 6.8 Hz, CH3-15), 2.53-2.60 (1H, m, H-5), 2.70-2.76 (4H, m, H-5, 11), 2.99-3.06 (4H, m, H-6), 4.20 (1H, m, H-1′), 4.70 (1H, d, J = 12.8 Hz, H-1), 5.05 (1H, d, J = 12.8 Hz, H-1), 5.28 (1H, s, H-4), 5.89 (1H, br. d, J = 12.8 Hz, H-13), 6.49 (1H, m, H-14).
13C NMR (100 MHz CDCl3): 14.0 (6′-CH3), 17.6 (16-CH3), 18.5 (15-CH3), 22.6 (CH2), 25.9 (CH2), 30.9 (CH2), 31.5 (CH2), 34.9 (CH2), 41.2 (C-6), 49.0 (C-11), 63.8 (C-1), 69.4 (C-1′), 83.1 (C-7), 103.3 (C-4), 114.0 (C-9), 124.4 (C-13), 135.3 (C-14), 150.9 (C-10), 160.3 (C-3), 175.1 (C-12), 190.7 (C-8).
HRESI: m/z [M+Na+] calcd for C21H28O5Na: 383.1834; found: 383.1832.
Supplemental Material
Supplemental material - Supplemental material for Screening of Azaphilone Derivatives From Monascus pilosus-Fermented Rice (Red Yeast Rice) and Their Evaluation as Nonsteroidal Androgen Receptor Antagonists
Supplemental material, Supplemental material, for Screening of Azaphilone Derivatives From Monascus pilosus-Fermented Rice (Red Yeast Rice) and Their Evaluation as Nonsteroidal Androgen Receptor Antagonists by Ming-Der Wu, Ming-Jen Cheng, Yen-Lin Chen, Tai-Wei D. Liu and Kai-Ping Chen in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work was supported by a grant from the Ministry of Economic Affairs of Republic of China.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
