Abstract
Panax ginseng C.A. Meyer is recognized as one of the most important crude drugs in ancient Chinese medicine. Numerous pharmacological studies investigated P. ginseng; however, these studies were limited to ginsenosides, which are typical constituents in P. ginseng. We focused on the essential oil (EO) from P. ginseng as it has a typical aroma. Herein, we report the inhibitory activities of EO against β-secretase, acetylcholinesterase (AChE), and butyrylcholinesterase (BChE), which were investigated in order to demonstrate the potential of EO as a preventative and therapeutic agent against Alzheimer’s disease (AD). Essential oil (250 µg/mL) showed 41.4% inhibition against β-secretase, 77.4% inhibition against AChE, and 94.1% inhibition against BChE. In addition, spathulenol (8.82%, content % in EO), bicyclogermacrene (6.23%), β-elemene (3.94%), and α-humulene (3.69%) were identified as high content by Gas chromatography mass spectrometry (GC/MS) analysis. Furthermore, β-elemene and α-humulene showed high activity among 3 compounds with 50% inhibitory concentration (IC50) values of 77.2 and 137.3 µM for AChE, and 298.2 and >2000 µM for BChE, respectively. In this report, we showed the inhibitory activity of EO from P. ginseng against β-secretase, AChE, and BChE, and demonstrated that EO could be a candidate to treat AD. This is the first research to report the anti-AD effect of EO and determination of its volatile components. Especially, β-elemene and α-humulene are expected to be highly bio-available compounds due to their small molecular size and lipophilicity. From these results, EO from P. ginseng may be a promising candidate for AD treatment.
Dementia is a serious disease and the number of patients is increasing. Patients reached over 46.8 million worldwide in 2015 that are expected to reach 131.5 million in 2050. 1 Dementia can be largely classified into 4 types according to its pathogenesis, Alzheimer’s disease (AD), vascular dementia (VaD), dementia with Lewy bodies, and frontotemporal dementia. 2 Alzheimer’s disease is the major type and accounts for up to 75% of dementia. 2 The amyloid hypothesis is well known in the pathogenesis of AD. The amyloid precursor protein is hydrolyzed by 2 aspartic proteases, β-secretase and γ-secretase, and amyloid β is produced. Amyloid β is pigmented on brain tissue to form senile plaque, which expresses cytotoxicity to brain nerve cells and the subsequent appearance of AD symptoms. From these pathogenic mechanisms of AD, the inhibition of β-secretase is a suitable target for AD prevention, since the inhibition of γ-secretase leads to a suppression of the Notch signal. Our previous research on screening of plant material, especially spices and herbs, was successful and some active principles were disclosed. 3,4 On the other hand, the cholinergic hypothesis is well known as a mechanism in cognitive function. In the hypothesis, damage to brain nerve cells by senile plaque leads to a decrease in choline transferase activity 5 and a loss of cognitive function. In the clinical stage, acetylcholinesterase (AChE) and butyrylcholinesterase (BChE) inhibitors, donepezil, galantamine, and rivastigmin, are administered as therapeutic medicines. Among them, galantamine is widely found in various Amaryllidaceae plants. Thus, natural plant resources possessing cholinesterase inhibitory activity may have the potential to improve AD symptoms.
Essential oils (EOs) from natural resources and volatile compounds are expected to have therapeutic effects on AD. 6 Two effects of EO for the improvement of AD are known: aroma therapeutic and enzyme inhibitory effects. 7,8 The aroma therapeutic effect is attributed to the relaxing effect caused by inhaling an aromatic substance, while the enzyme inhibitory effect is attributed to the inhibition of enzymes related to AD and the deterioration of symptoms. In particular, the constituents of EO are often small molecules that possess lipophilicity. These molecules are expected to pass through the blood-brain barrier easily and reach the therapeutic target. 9
These facts led us to focus on the screening of volatile aromatic extracts for β-secretase and cholinesterases inhibitions. Among the plant sources available, we focused on the roots of Panax ginseng C.A. Meyer. Panax ginseng roots are recognized as one of the most important crude drugs in ancient Chinese medicine and “Kampo” prescriptions as they show a variety of pharmacological activities. Panax ginseng was also described in “Bencao Gangmu” as an “intelligent medicine” and extensive studies were performed to demonstrate its antidementia activity. Among them, protective activity on nerve cells in the brain using a cerebral ischemia model was found by Wen et al. 10 In addition, β-secretase inhibition was reported by Choi et al. 11 Active principles for these pharmacological activities were reported as ginsenosides, which are typical constituents in P. ginseng. On the other hand, studies on antidementia and the pharmacological activities of the nonginsenoside constituents of P. ginseng have not been widely performed.
Considering these circumstances, we focused on the aromatic compounds of P. ginseng since the plant has a typical aroma. The studies of aromatic compounds are limited to chemical analysis of the constituents 12,13 and the only pharmacological study was reported by Bak et al, 14,15 which found anti-inflammatory activity of EO extracts from P. ginseng. In this report, inhibitory activities against β-secretase, AChE, and BChE by EO from P. ginseng were investigated as indicators for the prevention and therapy of AD.
Results on inhibitory activities of extracts from P. ginseng during steam distillation against β-secretase are shown in Figure 1. Essetial oil showed 41.4% inhibition at 250 µg/mL against β-secretase, while water extract showed 46.5% and 56.6% inhibition at 50 and 250 µg/mL, respectively. These data suggested that water extract showed higher inhibitory activities at a lower concentration compared to EO.
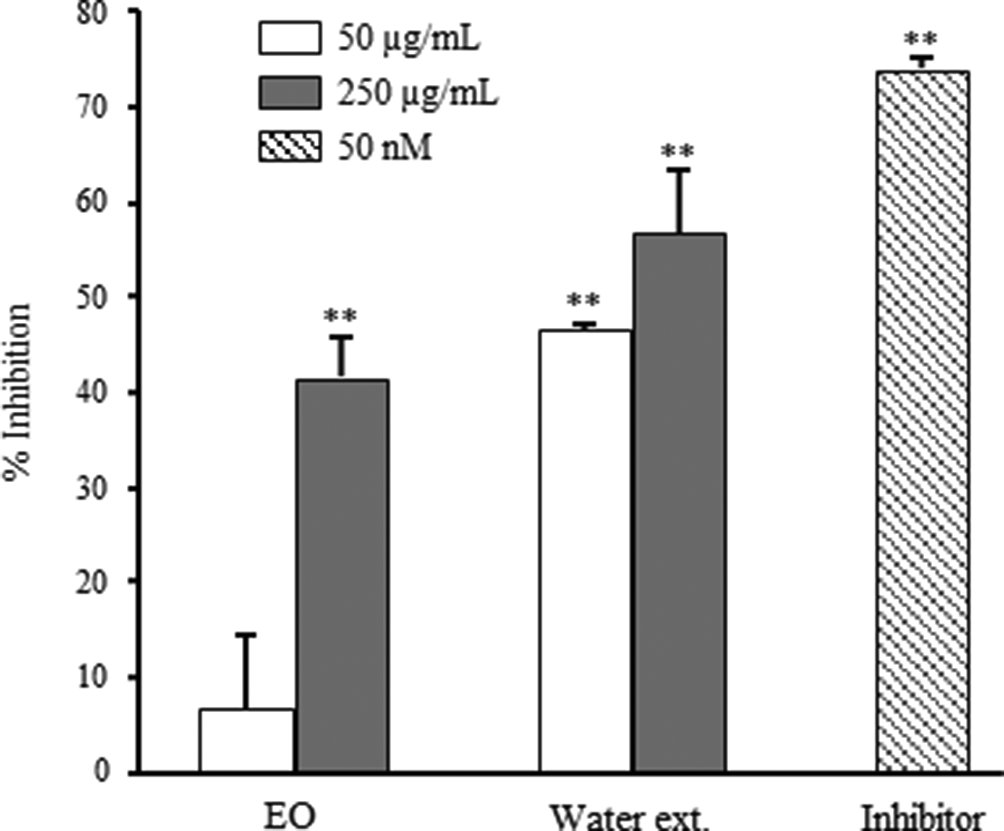
Inhibitory activities on β-secretase of essential oil and water extract prepared from Panax ginseng. Data are shown as the mean with standard deviation as error bars. Significantly different from the control group, **P < 0.01.
Results on inhibitory activities of extracts against AChE and BChE are shown in Figures 2 and 3, respectively. Essential oil showed 68.4% and 77.4% inhibition at 50 and 250 µg/mL against AChE, respectively. Moreover, EO showed 92.1% and 94.1% inhibition at same concentrations against BChE, respectively. In contrast, water extract showed no significant inhibitory activities at the same concentrations against AChE and BChE.

Inhibitory activities on acetylcholinesterase of essential oil and water extract prepared from Panax ginseng. Data are shown as the mean with standard deviation as error bars. Significantly different from the control group, **P < 0.01.
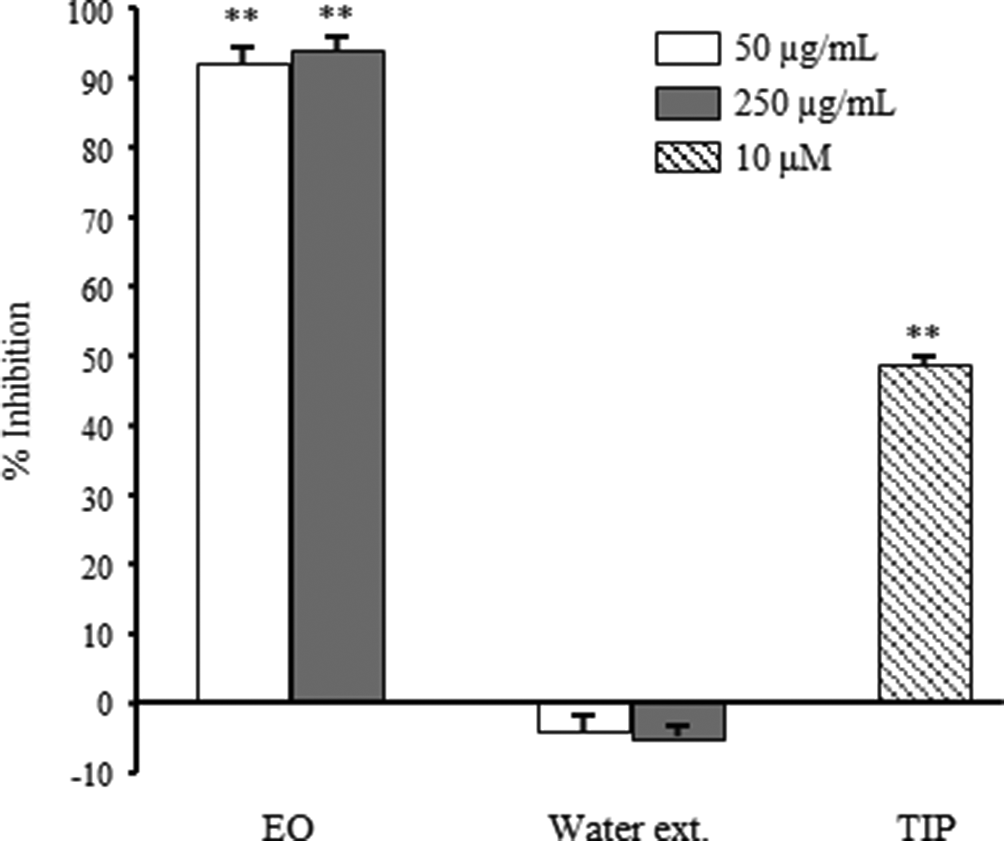
Inhibitory activities on butyrylcholinesterase of essential oil and water extract prepared from Panax ginseng. Data are shown as the mean with standard deviation as error bars. Significantly different from the control group, **P < 0.01. TIP, tetraisopropylpyrophosphoramide.
Gas chromatography mass spectrometry (GC/MS) analysis was performed to clarify the constituents of EO (Table 1). As a result, spathulenol (8.82%, % content in EO), bicyclogermacrene (6.23%), β-elemene (3.94%), and α-humulene (3.69%) were identified as high contents (Figure 4). Among these compounds, authentic β-elemene (
The 8 Major Components of Essential Oil From Panax ginseng Determined by GC/MS.
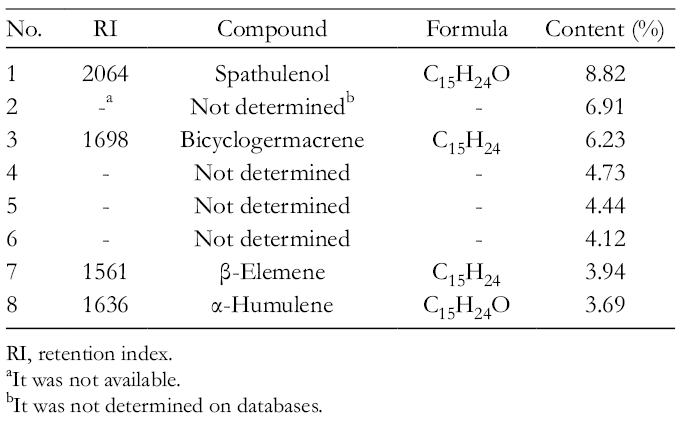
RI, retention index.
It was not available.
It was not determined on databases.

Total ion chromatogram of the essential oil from Panax ginseng: (a) β-elemene, (b) α-humulene, (c) bicyclogermacrene, and (d) spathulenol.
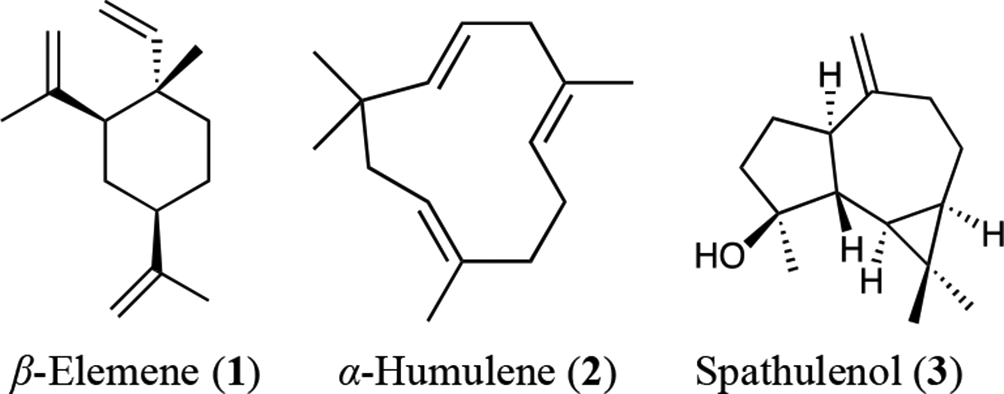
Chemical structures of β-elemene, α-humulene, and spathulenol.
Compounds
Fifty Percent Inhibitory Concentration (IC50) Values of Compounds on Inhibition of Cholinesterases.

AChE, acetylcholinesterase; BChE, butyrylcholinesterase.
Positive control of AChE.
It was not determined.
Positive control of BChE.
We successfully confirmed the inhibitory activity of EO against β-secretase, AChE, and BChE, so it is a potential candidate for the treatment of AD. Active principles were determined as
Recently, inhibitory activities of natural EO against enzymes related to the pathogenesis and the therapeutics of AD were found. 3,16 However, their active principles are yet to be determined. This may be due to the low yield of EO from raw material and to the lack of an efficient separation method for the components of EO. To overcome this obstacle, we introduced GC/MS to determine the compounds contained in EO and demonstrated activities using authentic samples instead of performing classical activity-guided purification. The contents of EO in some plants were reported to pass through the blood-brain barrier easily, 9 and translocation to the brain is expected to be high. Therefore, EO is a potential therapeutic agent for AD.
The pharmacological activities of P. ginseng and ginsenosides were reported extensively. Among them, improvement of VaD 10,18 and AD 19 by P. ginseng was reported and ginsenosides were determined as active principles. Choi et al 11 reported that ginsenosides showed inhibitory activities against cholinesterases and β-secretase. Furthermore, the bio-availabilities of ginsenosides were low due to their complex metabolism when administered orally. Essential oil components such as β-elemene and α-humulene can be recognized as highly bio-available compounds due to their small molecular size and lipophilicity.
Matsuda et al 20 reported the antiproliferative activities of P. ginseng extract and ginsenosides against human premyelocytic leukemia cells (HL-60), and the activities were enhanced by the addition of a fraction without ginsenosides. This suggests that it is important to focus on the nonginsenosides contained in P. ginseng. Study of the EO of P. ginseng may provide important insights into the complex and interesting pharmacological activities of the plant.
Experimental
Materials
Roots of P. ginseng (Raw ginseng, 6 years old) were purchased in Gyeondong Market in Korea. Fast Blue B Salt (FBB) and Triton X-100 were purchased from MP Biomedicals (Santa Ana, CA, United States). Sodium dodecyl sulfate (SDS) was purchased from GE Healthcare (Tokyo, Japan). Acetylcholinesterase (from Electrophorus electricus) and BChE (from equine serum) were purchased from Sigma-Aldrich (St Louis, MI, United States). Galantamine hydrobromide was purchased from Tokyo Chemical Industry Co., Ltd. (Tokyo, Japan). Fluorescence-quenching substrate for β-secretase [MOCAc-Ser-Glu-Val-Asn-Leu-Asp-Ala-Glu-Phe-Arg-Lys(Dnp)-Arg-Arg-NH2] and β-secretase inhibitor [Lys-Thr-Glu-Glu-Ile-Ser-Glu-Val-Asn-Sta-Val-Ala-Glu-Phe] was purchased from the Peptide Institute, Inc. (Osaka, Japan). All other reagents were purchased from FUJIFILM Wako Pure Chemical Corp. (Osaka, Japan) or Nacalai Tesque (Kyoto, Japan).
Preparation of EO From Panax ginseng Roots
Freeze dried roots of P. ginseng (rhizomes were removed from whole roots, 100 g) were pulverized and extracted with 1 L distilled water using EO quantifying apparatus (Japanese Pharmacopeia 17th edition) by a steam distillation method (5 hours, 100°C). Essential oil (pale yellow) was obtained in 0.02% yield. In addition, distilled water (used for steam distillation) was filtered and freeze dried to obtain water extract (60.0% yield).
GC/MS Analysis
Essential oil was analyzed using GC/MS under the following conditions. GC conditions: column, Inertcap Pure Wax (GL science, Tokyo, Japan, 0.25 µm, 0.25 mm i.d. × 60 m); oven temperature, 0 minute (50°C), 2 minutes (50°C), 78 minutes (240°C), 98 minutes (240°C, postrun); injection temperature, 250°C; split ratio, 1/80; injection volume, 1.0 µL; detector, flame ionization detector; MS conditions: EI source, electron energy of 70 eV; ionization temperature, 230°C; quadrupole temperature, 150°C; scanning range, 25 to 350 amu. The components were identified by matching their mass fragments and retention times with the databases (Wiley 7th and NIST08).
Assay for β-Secretase Inhibition
Assays were performed according to the method reported by Murata et al. 3 Samples in a dimethylsulfoxide (DMSO) solution (2 µL) at an appropriate concentration were diluted with 78 µL of assay buffer (20 mM acetate buffer, pH = 4.5 containing 0.1% Triton X-100) in a 96-well microtiter plate. Ten microliters of β-secretase solution in assay buffer (100 U/mL) was added to the diluted solution and incubated at 37°C for 10 minutes. After incubation, 10 µL of substrate solution with assay buffer (0.1 mM) was added and incubated at 37°C for 1 hour. After incubation, 50 µL of 2.5 M sodium acetate solution was added to the reaction solution. The reaction solution (100 µL) was moved to HPLC vial and diluted with 900 µL of water. The HPLC vial was incubated at 80°C for 10 minutes to terminate the reaction. The reaction solution was analyzed by HPLC under the conditions listed below. Column: Cadenza CD-C18 (Imtakt Co., Kyoto, Japan, 3 µm, 4.6 mm i.d. × 150 mm); mobile phase, 0.1% (v/v) formic acid/acetonitrile [0.1% (v/v) formic acid]: 0 minute (9:1), 18.0 minutes (1:1), 18.1 minutes (5:95), and 23.0 minutes (5:95); column temperature 40°C; flow rate, 1 mL/min; detection, fluorescent of excitation at 325 nm and emission at 395 nm; and injection volume, 10 µL. The peak area of the degradative fluorescent fragment (R t 15.4 minutes) was integrated. Inhibitory activity of the sample was calculated using the following equation.
The β-secretase inhibitor was used as a reference compound.

Assay for AChE Inhibition
Assays were performed according to the method reported by Marston et al, 21,22 with modifications in order to introduce a microtiter plate for higher throughput. Samples in a DMSO solution (5 µL) at an appropriate concentration were diluted with 180 µL of assay buffer (50 mM Tris-HCl buffer, pH = 7.8) in a 96-well microtiter plate. Ten microliters of enzyme solution (2.0 U/mL) and 5 µL of 1-naphthylacetate (18 mM) were added to the mixture and incubated at 37°C for 1 hour. After incubation, 25 µL of 5% (w/v) SDS solution and 25 µL of FBB (2 mM) were added. Absorbance at 600 nm was measured and the inhibition was calculated using the equation below. Moreover, enzyme nonadded group was provided in order to subtract the absorbance value of the sample itself.
Galantamine hydrobromide was used as a reference drug.

Assay for BChE Inhibition
Assays were performed according to the same method as in AChE, using BChE as an enzyme. Tetraisopropylpyrophosphoramide was used as a reference drug.
Statistical Analysis
All data were analyzed with Statcel 3 (The Publisher OMS, Tokorozawa, Japan), add-in software for Excel, using one-way analysis of variance. Significant difference was analyzed by Dunnett’s algorithm at P < 0.01 or P < 0.05.
Footnotes
Acknowledgments
We are grateful to Dr Shinichi Matsumura and Yuri Yoshioka at INABATA KORYO CO., LTD. for GC-MS analyses. We are also grateful to the late Professor Hideaki Matsuda at Kindai University for valuable discussions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
