Abstract
In the cultivation of
Obesity is recognized as a major lifestyle-related disease, especially in the developed countries. Pancreatic lipase is well known as a key enzyme for lipid absorption by hydrolysis of total dietary fats.
1
Two pancreatic lipase inhibitors, namely orlistat (Xenical)
2
in the United States and cetilistat (Oblean)
3
in Japan, have been approved so far for the treatment of obesity. To search for novel pancreatic lipase inhibitors from natural resources, the screening of plant extracts has been considered as one of the successful strategies. Hitherto, several extracts derived from plants parts, such as
In a preceding paper,
7
we reported the utility value of pruned mango leaves, which were discarded hitherto during the cultivation of mango fruits. A methanol extract of old mango dark green leaves possessed a porcine pancreatic lipase inhibitory activity. In this study, we focused on the utility of pruned
In the preliminary study, the methanol
Inhibitory Activities of
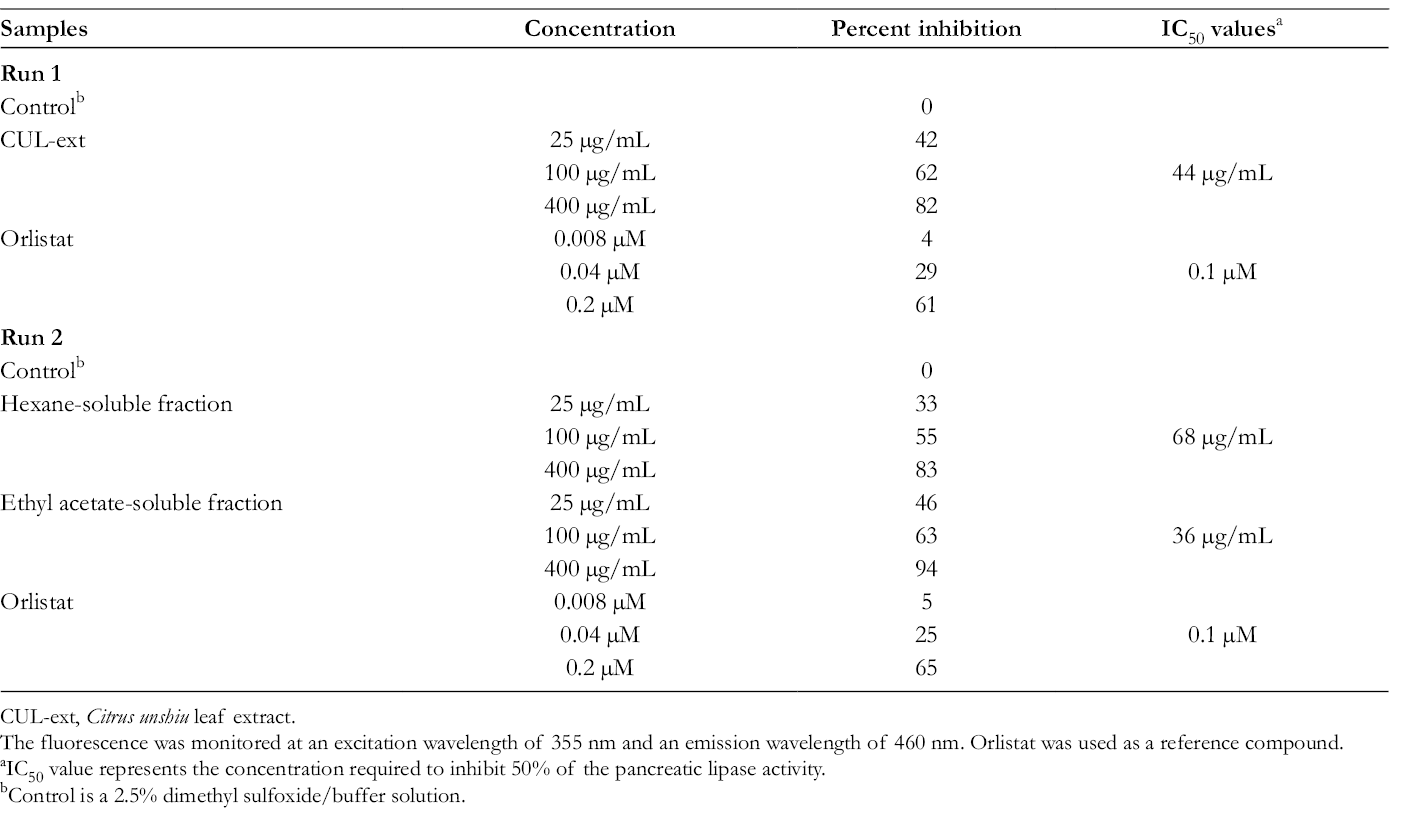
CUL-ext,
The fluorescence was monitored at an excitation wavelength of 355 nm and an emission wavelength of 460 nm. Orlistat was used as a reference compound.
IC50 value represents the concentration required to inhibit 50% of the pancreatic lipase activity.
Control is a 2.5% dimethyl sulfoxide/buffer solution.
To identify the active constituents, the CUL-ext was fractionated by solvent extraction to give a hexane-soluble fraction, ethyl acetate-soluble fraction, water-soluble fraction, and ethyl acetate-water-insoluble intermediate fraction, as described in the section “Fractionation of CUL-ext.” Among these fractions, the hexane-soluble and ethyl acetate-soluble fractions showed potent activities against pancreatic lipase with IC50 values of 68 and 36 µg/mL, respectively (Table 1). On the contrary, the water-soluble and ethyl acetate-water-insoluble intermediate fractions were almost inactive at the concentrations tested. A known pancreatic lipase inhibitor, orlistat, was used as a reference compound; the IC50 value of orlistat was 0.1 µM (corresponding to 0.0495 µg/mL) in accordance with the reported IC50 value (0.05 µg/mL)
9
as cited in Table 1. On the basis of thin-layer chromatography (TLC)
10
and liquid chromatography-electrospray ionization-mass spectrometry/mass spectrometry (LC-ESI-MS/MS) analyses
11
of these active hexane-soluble fractions and ethyl acetate-soluble fractions, it was found that nobiletin (
Inhibitory Activities of Nobiletin (1) and Rutin (2) on Pancreatic Lipase.

Orlistat was used as a reference compound.
IC50 value represents the concentration required to inhibit 50% of the pancreatic lipase activity.
In addition, the pancreatic lipase inhibitory activities of other flavonoids, such as
Considering these reported data, a part of the pancreatic lipase inhibitory activity of CUL-ext described above was attributable to
In the cultivation of
Experimental
Plant Materials
Leaves of

Photographs of (a) pruning in late winter and (b) pruned
Reagents
4-Methylumbelliferyl oleate, lipase (type II, from porcine pancreas, Lot #: SLBN3801V), authentic
Extraction
The leaf powder (10 g) was extracted with methanol (MeOH, 200 mL) for 72 hours at room temperature. The extract solution was evaporated under reduced pressure to produce MeOH extract. The yield of MeOH extract of leaves was 15% (w/w).
In Vitro Pancreatic Lipase Inhibition Assay
Porcine pancreatic lipase (type II, from porcine pancreas) activity was measured according to the method described by Nakai et al 16 with minor modifications. The test sample was dissolved with dimethyl sulfoxide (DMSO) and diluted with 13 mM Tris-HCl buffer containing 150 mM NaCl, 1.3 mM CaCl2 (pH 8.0) to a final DMSO concentration of 2.5% v/v. 4-Methylumbelliferyl oleate was used as a substrate. The substrate and the enzyme were both diluted in the above-mentioned buffer immediately before use. An aliquot of 25 µL of the test solution and 50 µL of 0.1 mM 4-MU solution was mixed in black microtiter plates, followed by the addition of 25 µL of 0.2 mg/mL enzyme solution to each well to start the reaction. After incubation for 30 minutes at 37°C, 100 µL of 0.1 M citrate buffer (pH 4.2) was added to stop the reaction. The fluorescence associated with the enzymatically released 4-methylumbelliferone product was monitored at an excitation wavelength of 355 nm and an emission wavelength of 460 nm using a multi-label counter (PerkinElmer 2030 ARVO X4; PerkinElmer Life and Analytical Sciences). Orlistat, a known inhibitor of pancreatic lipase, was used as a reference compound. The activity of negative control was also evaluated by adding stop solution before the enzymatic reaction. The inhibition activity was calculated using the following formula:
where
Each concentration of samples was confirmed in triplicate (
Fractionation of CUL-Ext
A suspension of the CUL-ext (10 g) in water (100 mL) was extracted with hexane (200 mL × 3) followed by ethyl acetate (200 mL × 3). Evaporation of the solvent resulted in a hexane-soluble fraction (1.4 g), an ethyl acetate-soluble fraction (0.9 g), a water-soluble fraction (5.7 g), and an ethyl acetate-water-insoluble intermediate fraction (1.5 g), which was obtained as an intermediate layer during the process of ethyl acetate extraction. The pancreatic lipase inhibition percentage in each fraction was evaluated.
Thin Layer Chromatography Analysis
Thin layer chromatography analysis was carried out according to the method described by Tosa et al
10
with minor modifications. Each dissolved sample (in MeOH, 5 mg/mL) was spotted on TLC (silica gel 60F254), and developed (solvent, chloroform/MeOH/water, 6:4:1 v/v; detection, UV 254 nm; and 10% H2SO4 followed by heating). Under the above condition, the
Flavonoid Content
Flavonoid content of the CUL-ext was determined (
Statistical Analysis
The experimental data were evaluated for statistical significance using Bonferroni/Dunn’s multiple-range test with GraphPad Prism for Windows, Ver. 5 (GraphPad Software Inc., 2007).
Footnotes
Acknowledgment
We are grateful to all technical staff of Yuasa Experimental Farm, Kindai University for the collection of
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
