Abstract
A novel C20-diterpenoid alkaloid with an unprecedented 19 carbon atoms, designated as tatsienensine (
Keywords
Delphinium, a large genus within the Ranunculaceae family, is estimated to contain about 350 species, mainly found in the northern hemisphere, of which 173 are found in mainland China. 1 Diterpenoid alkaloids are believed to be the major bioactive components of the genus Delphinium. 2 -5 A large number of these compounds have been isolated from various species of Delphinium and Aconitum and are classified according to their structures as C18-, C19-, and C20-diterpenoid alkaloids, 2 with a wide range of fascinating bioactivities.
Delphinium tatsienense Franch grows mainly in China in southwest Sichuan and northwest Yunnan.
2
Our previous investigation of the whole plants of this species resulted in the isolation of three new C20-diterpenoid alkaloids.
6
Gratifyingly, further chemical investigation of this plant, collected in Miyi County, Sichuan Province, P. R. China, led to the discovery of a structurally unique diterpenoid alkaloid, tatsienensine (
Tatsienensine (

Chemical structure of tatsienensine (1) and key 1H-1H COSY ( ) and HMBC (
) and HMBC ( ) correlations of tatsienensine (
) correlations of tatsienensine (
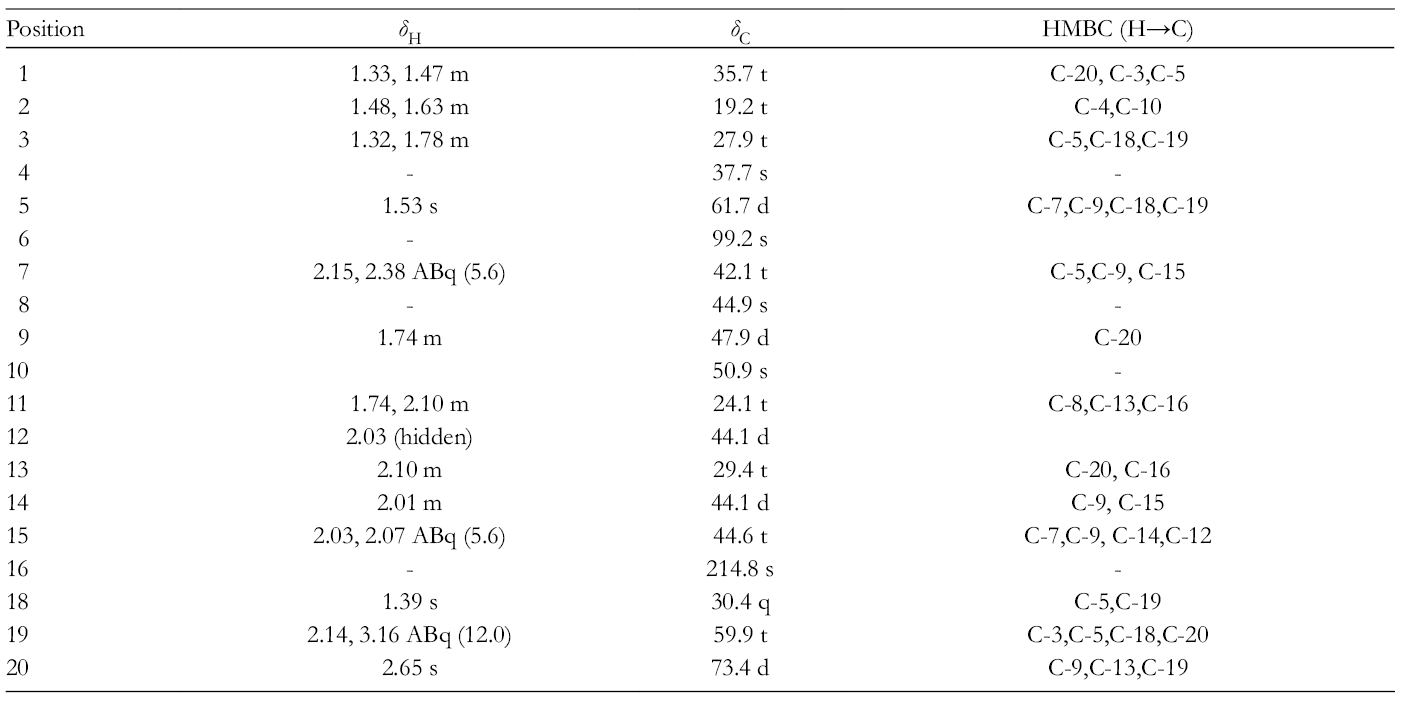
1H and 13C NMR Spectroscopic Data and 2D NMR Correlations of

ORTEP drawing of tatsienensine (1).
Experimental
General
Column chromatography (CC): silica gel H (Qindao Marine Chemical, Inc., Qingdao, P. R. China). TLC: silica gel plates; detection by spraying with Dragendorff reagent. IR spectra: Nicolet FT-IR 200S spectrometer. Optical rotations: Perkin-Elmer 341 polarimeter. 1D and 2D NMR spectra were measured on a Varian Unity-INOVA-400 instrument with TMS as internal standard. ESI-MS: Finnigan LCQ; in m/z (rel.%). HR-ESI-MS: VG Auto Spec 3000 spectrometer.
Plant Material
Whole plants of D. tatsienense Franch were collected in Miyi County, Sichuan Province, P. R. China, in September 2007. The plant was authenticated by Professor Q. E. Yang of the Beijing Institute of Botany of the Chinese Academy of Sciences, where a voucher specimen (No. 200709-1) has been deposited.
Extraction and Isolation
Air-dried and powdered whole herbs of D. tatsienense (3.0 kg) were percolated with 0.1 M HCl (40 L). The obtained acid aqueous solution was basified with 10% aqueous NH4OH to pH 9, then extracted with ethyl acetate (20 L × 3). Removal of the solvent under reduced pressure afforded the total crude alkaloids (12.1 g) as a yellowish amorphous powder, which was chromatographed over a silica gel column, eluting with a cyclohexane-acetone (8:1→1:2) gradient system, to give Frs. A (94 mg), B (399 mg), C (2.3 g), D (2.2 g), E (2.1 g), F (1.3 g) and G (2.8 g). Purification of Fr. G by silica gel CC using CHCl3-CH3OH (80:1 → 10:1) as eluent generated tatsienensine (
Tatsienensine
Colorless monoclinic crystal.
[α]
IR (KBr) ν max: 3720, 2919, 1738, 1233 cm−1.
1H and 13C NMR: Table 1.
ESI-MS m/z: 300 [M+H]+; HR-ESI-MS m/z: 300.1958 [M+H]+ (calcd for C19H25NO2, 300.1963).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Science and Technology Department of Sichuan Province (Nos. 2018JY0065, 2016JY0195) and Key Laboratory of Chemistry for Natural Products and Small Organic Molecular Catalyst of Leshan Normal University (No. TRCWYXFZCH2016003).
