Abstract
Collagen from fish scale waste is currently being studied as a promising biological material to replace collagen from animals because of advantages such as safe, fat-free, not suffering from communicable diseases, and easy absorption in human body solutions. Finding the suitable process of extracting fish scale collagen is necessary because extracting collagen from fish scales by chemical methods often requires a long time. Therefore, in this paper, some bases and acids at different concentrations were chosen to find the most suitable condition for extracting fish scale collagen from the wastage of different scale fishes belonging to the familiar Cyprinus genus. The characterizations of the extracted collagen including structure, morphology, element composition, relative molecular weight, amino acid composition, denaturation temperature, crystal structure, and thermal stability were investigated. In addition, the amino acid sequence of the extracted collagen was also determined and compared with the National Center for Biotechnology Information protein database.
Keywords
In some Asian, African, and European countries, the carp fishes are a common freshwater fish and often used as food. Fish scales are by-products in fish processing which cause pollution to the environment directly. Interestingly, fish scales contain a large content of useful materials including organic components about 40% to 55% (collagen, fat, lecithin, scleroprotein, various vitamins, etc.) and inorganic components about 7% to 25% (hydroxyapatite, calcium phosphate, etc.). Especially, collagens extracted from fish skin or fish scale waste are type I collagen, the most plentiful protein in the organisms and human body. This collagen has a triple helix structure, consisting of 2 homologous chains (α1) and an additional sequence with slight change in its composition (α1). 1,2 Fish collagens have many advantages consisting of high safety (no foot-and-mouth disease of pig or bovine spongiform encephalopathy of cattle and transmissible spongiform encephalopathy of cow), high absorbability, no religious hindrances, cheap, and biocompatible, which could give fish collagen a high level of application in various fields such as health foods, cosmetics, and biomedicine. 3 -7
Some methods were used for extracting collagen from fish skins or fish scales, eg, the chemical method using alkali and acid, the biochemical method using acid and enzyme or acid and microorganisms, and the extrusion-hydro-extraction. However, the extractions of acid-soluble collagen and pepsin-solubilized collagen are 2 widespread types. Fengxiang Zhang et al prepared and characterized type I collagen extracted from fresh carp fish scales in which fat and proteins were removed by NaCl solution before demineralization by HCl solution. The demineralized scales were immersed in acetic acid solution for extracting collagen. The residues from the acetic acid extraction were re-extracted with acetic acid solution (acid-soluble collagen) or suspended in acetic acid and digested with pepsin (pepsin-solubilized collagen). The obtained acid-soluble collagen and pepsin-solubilized collagen are type I collagen and contain 2 α chains, α1 and α2, with molecular weights ca. 117.3 and 107.4 kDa, respectively. The authors also showed that the fresh carp scale collagens contain 18 amino acids and have relatively high haft-cysteine but lower denaturation temperature than porcine dermis collagen, carp fish ordinary muscle collagen, and other seawater fish collagens. 8 The extrusion-hydro-extraction process was applied for extracting collagen from tilapia (Oreochromis sp.) fish scales. 9 The advantages of this method are shorter time for extracting process and higher protein extraction yield in comparison with original methods. The tilapia scale collagens were type I collagens and could have hopeful applications in the food, cosmetic, and medical industry. Type I collagens have also been extracted from the Egyptian Nile tilapia scales, 7 carp scales, 10 Baltic cod skins, 11 channel catfish skins, 12 red drum fish scales, 13 striped catfish skin, 14 cat fish (Tachysurus maculatus) skin, 15 teleost skin in Alaska pollack, 16 tilapia (Oreochromis niloticus) scales and skin, 17,18 and so on.
Nowadays, applications of type I collagen in medicine, exclusively in drug delivery, are developing; however, the type I collagens were often extracted from bovine skin, cell matrix, rat-tail, fish scale, etc. 19 -27 Collagens from fish with many advantages, in comparison with collagen-derived products from land-based animals, are promising biomaterials in the future. So, study on the preparation and characterization of fish collagens is essential to find new collagen products to replace collagen-derived products from land-based animals.
There are many literatures related to extracting collagens from fish; however, extracting collagens from fish still attracts study because of differences in biodiversity. The biodiversity has a strong impact on the molecular and amino acid composition of fish collagen. Muyonga et al indicated that there are significant modifications in the physical properties of protein and the quantity of amino acids of collagen extracted from different fish species, and these are rigorously correlated with the external temperature of the animal’s environment. 28 Moreover, most published papers related to collagen only report the relative molecular weight and amino acid compositions; they rarely present the exact molecular weight and amino acid sequence. Therefore, the aim of this work is to determine the physical properties of collagens and the quantity of amino acids in collagens which were extracted from fresh carp scales living in tropical area. The fresh carp fish scales (Cyprinus carpio) were chosen for study because carp fish is one of the main fish species in freshwater breeding industry in Vietnam. In addition, the amino acid sequence of these collagens, a highlight of this paper, is also investigated and reported.
Materials and Methods
Demineralization Process
Freshwater carp fish scales were collected from the markets in Hanoi, Vietnam. The scales were washed thoroughly with fresh water and then with distilled water and stored in refrigerator until use. All reagents used were of analytical grade.
To find the suitable condition for extracting collagen, a lot of experiments with different concentrations of alkali or acid were carried out as shown in Table 1. First, the scales were immersed in alkali solutions (Ca(OH)2 or NaOH or mixture of Ca(OH)2/NaOH) to remove fat and protein by stirring the solution for 24 or 8 hours, respectively (dry scales:solution = 1:20 or 1:8). Then, the protein-free scales were washed with distilled water until neutral pH. Next, the protein-free scales were demineralized by immersing in acid solution (HCl, H2SO4, H3PO4) for 20 minutes (dry scales:solution = 1:20). The demineralized scales were washed with distilled water until neutral pH.
Types and Concentrations of Acid and Alkali Used in Collagen Extraction Process.

Extracting Collagen
The demineralized scales were immersed in CH3COOH 0.5 M solution to extract collagen by stirring for 24 hours. After each 4 hours of immersing, the solution was stirred by high-speed stirrer with speed of 20 000 rpm for 30 minutes to increase the contact between scales and CH3COOH 0.5 M solution. The solution was filtered to obtain the collagen solution without any scales. Raw collagen was precipitated from the collagen solution by using NaCl 10% solution. Next, the raw collagen was redissolved in CH3COOH 0.5 M solution and reprecipitated before purifying collagen by distilled water for 48 hours using osmosis membrane. Thereafter, the purified collagen was obtained after lyophilization and stored in refrigerator. All the experiments for extracting collagen were performed at 4°C.
Characterization
Infrared spectroscopy ( IR ): IR spectrum of the freeze-dried collagen was recorded on a Nicolet iS10 spectrometer (Thermo Scientific, USA) at room temperature, in the wavenumbers ranging from 400 to 4000 cm−1, by averaging 32 scans with a resolution of 8 cm−1.
Field emission scanning electron microscopy ( FESEM ): FESEM images of fish scales and collagen were obtained using S-4800 FESEM machine (Hitachi, Japan). The samples are solid and covered by platinum to increase the electrical conductivity.
X-ray diffraction (XRD): XRD analysis of freeze-dried collagen was performed on a Siemens D5000 X-ray Diffractometer (XRD) (CuK α radiation source, λ = 0.154 nm) at a generator voltage of 40 kV with step 0.03° and a current of 30 mA at a scan speed of 0.043°/s in the 2θ scan range from 2° to 80°.
Energy-dispersive X-ray spectroscopy (EDX): EDX spectrum of freeze-dried collagen was carried out on EDX 6000E device (Japan).
Differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA): DSC and TGA diagrams of freeze-dried collagen were performed on a DSC-131 and TG analyzer (SETARAM) under argon atmosphere from room temperature to 200°C (for DSC) and 850°C (for TGA) at a heating rate of 10°C/min.
Ultraviolet-visible spectroscopy (UV-Vis): UV-Vis spectrum of collagen solution was carried out on a UV Spectrophotometer (CINTRA 40, GBC, USA) in the range of 200 to 400 nm.
Amino acid analysis: Freeze-dried collagen samples were dissolved in CH3COOH 0.5 M solution and the amino acid compositions were analyzed by a high-performance liquid chromatography (HPLC, Shimadzu [model 10A]) with test method H.HD.QT.046 (for amino acids) and H.HD.QT.112 (only for tryptophan).
Sodium dodecyl sulfate polyacrylamide gel electrophoresis (SDS-PAGE): SDS-PAGE of freeze-dried collagen was carried out according to the methods of Laemmli 29 using SDS-PAGE sample loading buffer 1× for modification at 95°C for 10 minutes.
Amino acid sequence analysis: After SDS-PAGE process, the gel was washed and the interested protein bands based on the electrophoresis results were cut. Gel pieces were washed with solution (acetonitrile 50%, NH4HCO3 100 mM) (solution A) at 30°C until Coomassie Brilliant Blue was completely removed before adding acetonitrile 100% to remove the water in the gel for 5 minutes. The gel pieces were dried and reduced by reducing solution (dithiothrietol 10 mM, NH4HCO3 100 mM) at 56°C for 45 hours and then cooled at room temperature. After reducing the disulfide bonds between cysteine radicals in the protein molecule, the gel pieces were alkylated with iodoacetamide 50 mM solution in NH4HCO3 100 mM solution. The gel pieces were incubated continuously at room temperature in the dark for 45 minutes. Then the gel pieces were cleaned with a washing solution (solution A) and water removed by adding acetonitrile 100% for 5 minutes. The gel pieces were dried and reconstituted with hydrolyzate solution (10 ng/µL trypsin in NH4HCO3 50 mM solution, acetonitrile 10%). The gel pieces were continuously incubated in ice for 30 minutes and at 30°C overnight after adding NH4HCO3 50 mM solution. Formic acid was introduced to the hydrolysis samples (the final formic acid concentration is 1%) to stop the enzymatic reaction. The supernatant was transferred to a clean eppendorf tube. The extract (acetonitrile 50%, formic acid 5%) was added into the gels, and the samples kept for 45 minutes and then ultrasonicated for 5 minutes. The supernatant was moved to the eppendorf tube as in the previous step. This step was repeated again by adding acetonitrile 90%/formic acid 5% and keeping the samples for 5 minutes before transferring the supernatant to the eppendorf tube as in the previous step (3 extraction cycles in total). The mixture of peptide extract was dried by SpeedVac (Massachusetts, USA). The peptide mixture was removed as salt by Ziptip C18 column (Millipore, MA, USA) before carrying out mass spectrometry analysis by nano liquid chromatography-mass spectrometry/mass spectrometry system. Nano LC-MS/MS system consists of QSTARXL MS/MS hybrid quadrupole system/time (Applied Biosystems/MDS Sciex, Ontario, Canada), which is equipped with nanoelectrospray (ESI) source connected to reverse phase C18 column with tip Sillica PicoTip (New Objective, USA). The MS/MS spectra of the peptide mixture were recorded and analyzed by Mascot software (Matrix Science Inc., USA). Proteins are searched on the NCBIprot database. The chemicals used in this experiment were provided by Sigma Aldrich (USA), Bio-Rad (USA), and Merck (Germany).
Determination of denaturation temperature: The denaturation temperature of purified collagen was measured according to the method of Nagai et al. 30 Freeze-dried collagen was dissolved in CH3COOH 0.5 M solution for viscosity measurement by using an Ostwald viscometer. The thermal determination curve was achieved by measuring the viscosity of the collagen solution at different temperatures from 20°C to 50°C. The temperature was increased stepwise and kept for 30 minutes at each point. This experiment was done 3 times.
Results and Discussion
Effect of Extraction Conditions on the Collagen Extraction Yields
The chemical method is usually used to extract collagen from fishes. The advantage of this method is easy to carry out with popular chemicals and simple machines. However, the limitations of this method are need for a long time (several days for a collagen extracting process) and removal of the chemicals to environment. The amino acid composition in collagen of each fish species is different, leading to the extraction condition being changed. Therefore, finding a suitable condition for extracting collagen from tropical carp fish scales with less time and lower concentration of chemicals is very important. In this study, different alkalis and acids were used as well as their concentrations changed for collagen extraction process from the carp fish scales. The concentrations of alkalis and acids used and the weight of raw collagen (collagen obtained without reprecipitation and purification) corresponding to each experiment of 5 g of original fish scales are expressed in Table 1. From Table 1, it can be seen that the increase in acid concentrations in step 2 (removing minerals) led to rising weight of the obtained raw collagen. Using the mixture of HCl 0.2 M and H2SO4 0.5 M solutions (1/2 v/v) in step 2 could yield the largest weight of raw collagen in comparison with other concentrations of acids in step 2. Utilization of NaOH solution in step 1 (removing fat and proteins) could save the time and volume of solution more than using saturated Ca(OH)2 solution. The CH3COOH 0.5 M solution used for extracting collagen from fish scales was referred to from other reports. 8,31 On the application of NaOH 0.1 M and saturated Ca(OH)2 solutions (2/1 v/v) in step 1, HCl 0.2 M and H2SO4 0.5 M (1/2 v/v) solutions in step 2, and CH3COOH 0.5 M solution in step 3 (extracting collagen), the weight of obtained raw collagen was highest. This can be explained by the hydrolysis of collagen in acidic and alkali environment, leading to decrease in the obtained weight of collagen. Therefore, the concentration of solutions as well as time for extraction of collagen had an important role in the extracting process of collagen. If using a high concentration of acid or base or extracting for a long time, collagen chains could be hydrolyzed, leading to lower collagen content. In the review of Wu Suo-Lian, the extraction yield of collagen from grass carp scale by using CH3COOH solution was 15.33% (extracting time of 25 hours) while the extraction ratios of collagen from grass carp scale by CH3COOH, citric acid, and lactic acid solutions were 45.82%, 74.34%, and 49.31%, respectively. 32 The collagen yield extracted from different types of fish scales was reported from 1.350% to 50.90%, depending on the fish species (carp scales yielded 1.35%, red seabream scales yielded 37.50%, Japanese sea bass scale yielded 41.00%, and sardine scales yielded 50.90%). 33,34 Differences in collagen extraction yield were caused by different amino acid compositions of each fish species, which directly affect the structure of the collagen. The scales of carp fish contain lower amounts of collagen compared with the other fish and marine species. The total time for extracting collagen from carp fish scales is 126 hours. 34 Samantha Cheng Fehng Pang used a 4-factor, 5-level central composite design to find optimum conditions for the maximum yield of collagen extraction from fish scales. The suitable extraction conditions were CH3COOH 0.62 M solution, pH of 2.96, temperature of 19.03°C, and time of 77.51 hours, and the yield of collagen extraction was 0.13%. 35 It can be seen that the time for extracting collagen from carp fish scales in this study was shorter than that in other researches but the extracted yield was quite high (13.6% of raw collagen).
Morphology of Fish Scales
The change in structural morphology of fish scales was observed by FESEM images displayed in Figures 1 and 2. Morphology of fish scales has a powerful variation after each step of treatment. The original scales had a compact and rigid structure with rough surface; the first part of fibers looks like the particles were connected continuously and arranged following a trend (Figure 1a). When fracturing the original scales, the fibril bundles of proteins, collagen, and other organic and inorganic substances were revealed and settled in parallel bunches. The components in scales were linked tightly (Figure 2a).

The field emission scanning electronmicroscopy images of surface fish scales before (a: original scales) and after treatment with saturated Ca(OH)2 solution (b: protein-free scales); mixture of HCl 0.2 M and H2SO4 0.5 M (c: mineral-free scales) and acetic acid 0.5 M (d: collagen-free scales) at different magnifications.
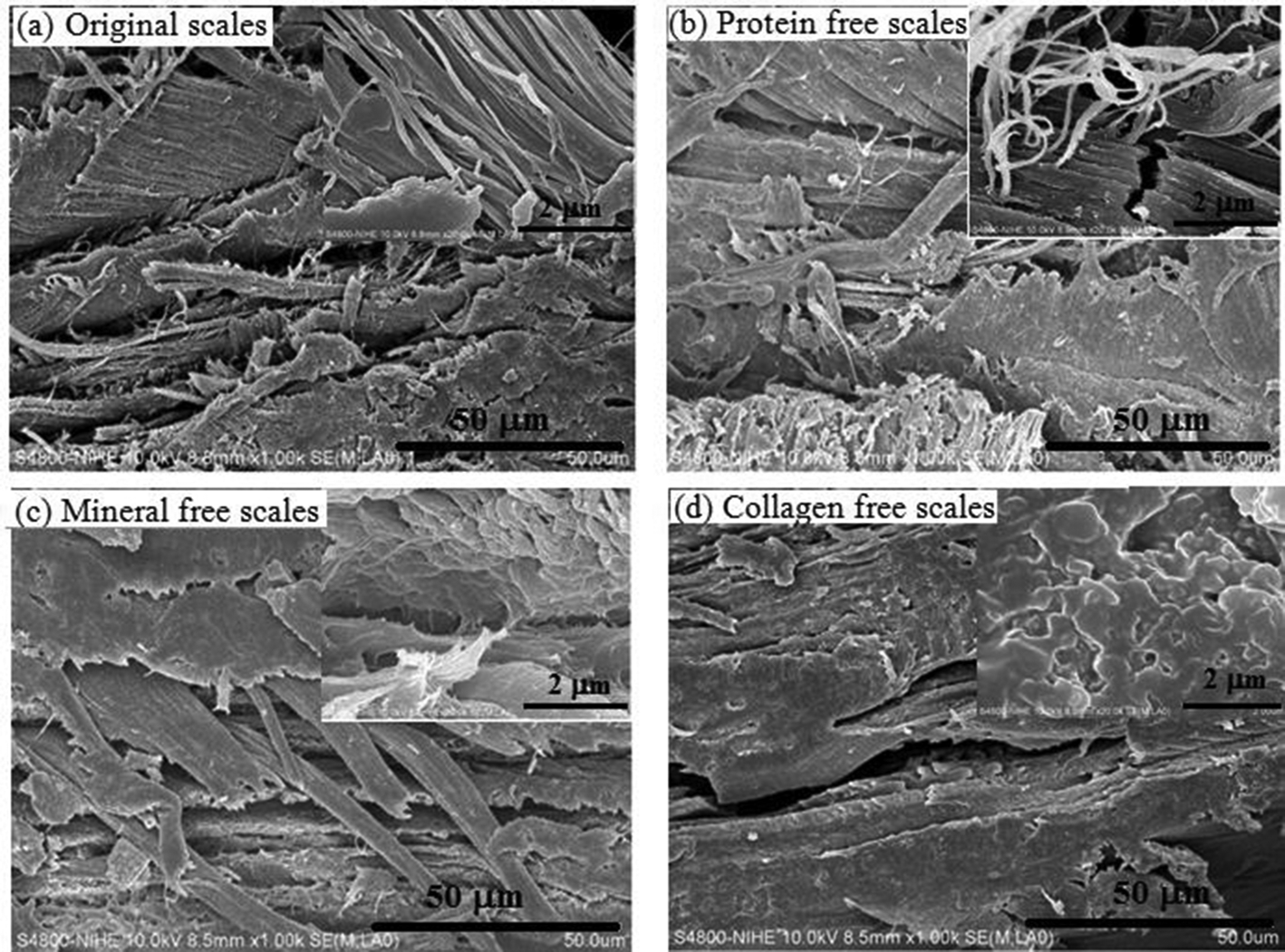
The field emission scanning electronmicroscopy images of fractured surface of fish scales before (a: original scales) and after treatment with saturated Ca(OH)2 solution (b: protein-free scales); mixture of HCl 0.2 M and H2SO4 0.5 M (c: mineral-free scales) and acetic acid 0.5 M (d: collagen-free scales) at different magnifications.
After removing fat and protein, mineral particles appeared on the surface of scales and were grouped layers (Figure 1b). The fibers in scales were broken (Figure 2b). The scales exhibited a visibly heterogonous structure. The modification of surface indicated that fat and protein were removed from scales after treating scales with alkali solution. However, the inside structure of protein-free scales was still hard.
Observation from Figures 1(c) and 2(c) showed the loss of mineral particles in the structure of scales. The structure of mineral-free scales became less compact, and collagen fibers appeared. This was an advantage for extracting collagen from these scales.
After extracting collagen, big holes were left on the surface of collagen-free scales (Figure 1d). The fibril structure of collagen was treated completely and the sheet structure of keratin was relayed. The elements in collagen-free scales are C, O, N, S, Cl as obtained from EDX spectrum (Appendix 1). The keratin structure is a stable fibrous structural protein which is difficult to be decomposed by acid and alkali solutions. This property helps to protect the fish body better.
Characterization of Purified Collagen Extracted From Carp Scales
FESEM Images of Collagen
FESEM images of raw collagen before purifying by osmosis membrane are presented in Figure 3(a). It can be seen that raw collagen contains NaCl particles which were used in precipitation of collagen.

Field emission scanning electronmicroscopy images of raw collagen (a) and purified collagen (b).
After purifying, the collagen had the typical fibril structure as other collagens obtained from fish. 33,34 The fibers were concentrated to form the fiber bundles (Figure 3b). The NaCl crystals did not appear in collagen structure.
Element Composition Analysis in Collagen
Table 2 demonstrates the element composition obtained by EDX analysis in raw collagen and purified collagen. The raw collagen consisted of C, O, N, S, Na, and Cl elements. The presence of Na and Cl elements was due to the use of NaCl in collagen precipitation. The existence of S elements was the evidence for cysteine, a sulfur-containing amino acid in raw collagen. The total composition of Na and Cl elements (84.24%) was the highest among elements in raw collagen; therefore, purification of raw collagen was essential to obtain high-quality collagen. This result was similar to the FESEM images of raw collagen in Figure 3(a).
Element Compositions in Collagen Before and After Purification.
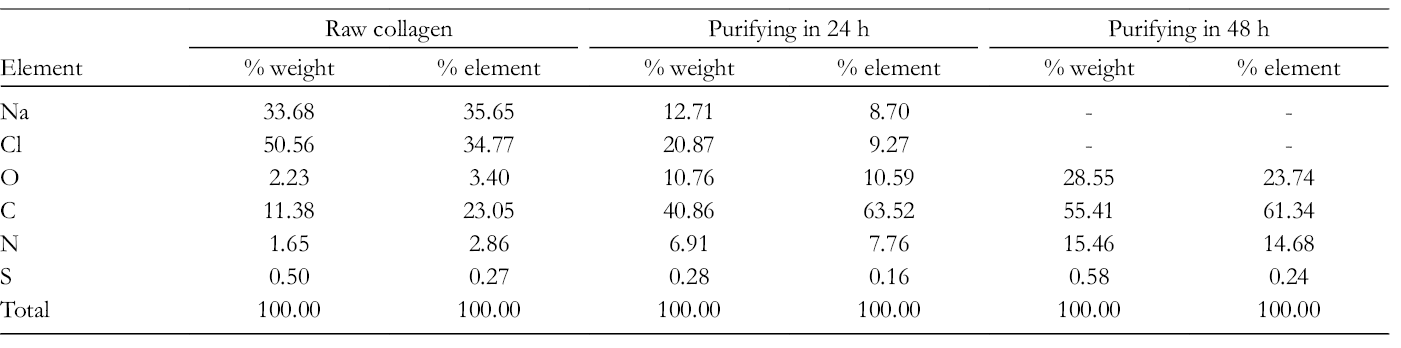
From the results in Table 2, the time for purifying collagen by distilled water using osmosis membrane had an influence on the quality of collagen. If collagen was purified for 24 hours, NaCl still existed in collagen structure (total composition of Na and Cl elements was 33.58%). As the time for purification was increased up to 48 hours, the Na and Cl elements disappeared completely. In this case, the collagen contained only C, O, N, S elements and the quantity order of these elements was C > O > N >S. This result showed the obtained collagen to have high purity.
Amino Acid Composition of Collagen
As per our knowledge, fish collagens have been recognized to vary broadly in their amino acid composition. In Table 3, the amino acid composition of collagen extracted from Vietnamese carp scales in this study was compared with that of other acid-soluble collagens extracted from Chinese carp scales. 8,34 According to Muyonga et al 28 and Foegeding et al, 36 the living environment has a strong effect on the amino acid composition in collagen extracted from fish species. By the H.HD.QT.046 (HPLC) and H.HD.QT.112 (HPLC) (only for tryptophan) test method, collagen obtained from Vietnamese carp scales contained 18 amino acids with high quantities of threonine, proline, glutamic acid, arginine, serine, alanine, and glycine as well as very low amounts of methionine, tryptophan, and tyrosine. Although glycine content in fish is the highest and present in all fish skins, bones, and scales, it exists mainly on fish skin. 35 Collagen extracted from carp scales could contain cysteine or not, due to the difference in living environment. The collagen extracted from Vietnamese carp scales in this study had a variance of amino acid composition in comparison with other carp scale collagens; in particular, it was the presence of tryptophan, the least plentiful of all 22 amino acids. 8,34 The high content of glutamic acid and arginine and the average content of glycine and alanine were similar with native fish collagen (mackerel) obtained by Sunet al. 37 There were very essential amino acids for the nutrition of young animals such as threonine (39.79%), cysteine (1.38%), lysine (1.00%), leucine and isoleucine (0.91% and 0.51%), phenylalanine (0.65%), valine (0.61%), methionine (0.47%), and tryptophan (0.28%). This result can open the application of collagen in biomedicine.
Amino Acid Composition of Some Collagens Extracted From Carp Scales of Different Countries.

Relative Molecular Weight of Collagen
Subunit composition of extracted collagen was determined by SDS-PAGE method and displayed in Figure 4. The obtained collagen was suggested as type I collagen with α1 and α2 chains corresponding to 139 and 129 kDa, respectively, and a β chain. The relative staining intensity of α1 band was higher than that of α2 band, so it can be recommended that the carp scale type I collagen has a composition of (α1)2α2 heterotrimer. The existence of a β chain in collagen is popular in the above fish. The fish scale collagen was type I collagen and composed of α1 (132.04 kDa) and α2 (120.06 kDa) chains (type (α1)2α2). 38 Collagen of Chinese fresh carp fish scales was also classified as type I collagen (type (α1)2α2) with 2 α chains (α1 117.3 kDa and α2 107.4 kDa). 8 In another literature, the collagen extracted from Chinese carp scales was type I collagen with 2 α1 and α2 chains, in which the molecular weight of α2 was exactly 116 kDa. 34 The difference in molecular weight of the 2 α chains in collagen may be due to the different resources of fish scales.

Sodium dodecyl sulfate polyacrylamide electrophoresis pattern of purified collagen.
Amino Acid Sequence of Collagen
Using a combination of SDS-PAGE, MS/MS methods, and NCBIprot database, 19 proteins belonging to the skin-scale protein group (collagen) of fish were identified. However, only 5 proteins having higher protein score, high number of significant matches, and high number of significant sequences were 5 typical collagen proteins as per the following information: Protein No.1: hypothetical protein cypCar_00045321, partial [Cyprinus carpio] (accession: KTF73577.1, protein score: 318, molecular (M r) (Da): 115 674, pI: 9.18, amino acid sequence: Appendix 2 (SEQ ID NO. 1)); Protein No.2: collagen type I alpha 2 [Carassius auratus] (accession: BAG72201.1, protein score: 294, molecular (M r) (Da): 127 652, pI: 9.41, amino acid sequence: Appendix 2 (SEQ ID NO. 2)); Protein No.3: collagen type I alpha 1 [Ctenopharyngodon idella] (accession: ADK35755.1, protein score: 231, molecular (M r) (Da): 138 104, pI: 5.44, amino acid sequence: Appendix 2 (SEQ ID NO. 3)); Protein No.4: collagen alpha-1(I) chain isoform X1 [Pygocentrus nattereri] (accession: XP_017540674.1, protein score: 149, molecular (M r) (Da): 137 928, pI: 5.46, amino acid sequence: Appendix 2 (SEQ ID NO. 4)); and Protein No.5: hypothetical protein cypCar_00006571 [Cyprinus carpio] (accession: KTG43775.1, protein score: 101, molecular (M r) (Da): 136 996, pI: 5.60, amino acid sequence: Appendix 2 (SEQ ID NO. 5)).
The proteins in the obtained collagen were compared to proteins on NCBIprot database which have clear amino acid sequence and exact molecular weight. They were hypothetical protein Cyprinus carpio or type I collagen with α1 or type I collagen with α2 chain. The MS/MS spectra of identified typical peptide fragmentation in collagen are demonstrated in Appendix 3.
The hypothetical protein Cyprinus carpio is similar to collagen α2 (I) chain and is a full-body transcriptome and proteome resource for the European common carp. From the above results, it can be seen that collagen was extracted successfully from the carp (C. carpio) scales. The amino acid sequence of fish collagen, especially, collagen extracted from fish scales, is still rarely published. Based on the highest protein score of 318, it could be confirmed that the amino acid sequence of fish collagen extracted from Vietnamese carp scales was much similar to that from European common carp. In the report of Shizuka Yamada et al, the collagen obtained from wasted fish skins and bones had an arginine-glycine-aspartic acid (RGD) motif which is a representative amino acid sequence with cell adhesion properties of arginine (Arg)-glycine (Gly)-aspartic acid (Asp). 39
IR Spectrum of Collagen
IR reflectance spectrum of the obtained collagen indicated the appearance of 5 major absorption peaks of amide groups (Figure 5). For instance, the position of vibrations of amide band A, amide band B, amide I, amide II, and amide III was at 3294.69, 3076.20, 1630.20, 1546.60, and 1238.19 cm−1, respectively. These absorption bands of the obtained collagen were similar to those of other collagens. 8,9,35,38 Moreover, C-H stretching and bending vibrations were also found at 2934.38, 1452.06, and 1337.66 cm−1, respectively. The wavenumbers of amide stretching and bending vibrations in the collagen were lower than that of free N-H vibrations. This proved these amide groups were involved in hydrogen bonds in each collagen molecule, and the IR absorption ratio between amide III and CH bending band confirmed that helical structure of the collagen was kept well. 8

IR spectrum of purified collagen.
UV-Vis Spectrum of Collagen
Proteins are large biological molecules comprised of 1 or multiple amino acids. In the UV-Vis spectrum of proteins in the investigated sample, there was an absorption peak at 220 nm due to the peptide bonds and an absorption peak at 280 nm belonging to the absorption of aromatic amino acids. Phenylalanine, tyrosine, and tryptophan had absorption bands in the range of 250 to 270, 270 to 290, and 280 to 300 nm, respectively. In the UV-Vis spectrum of purified collagen which was solubilized in acetic acid 0.5 M, there was a strong absorption peak at 192.07 nm which was the characteristic absorption of collagen type I (Figure 6). This result nearly agreed with the result of UV-Vis spectrum analysis of type I collagen from calf skin solubilized in 0.01 M NaOAc, 0.02 M NaCl (pH 4.0) buffer. 40

UV-Vis spectrum of purified collagen.
X-Ray Diffraction Analysis of Collagen
Crystal structure of collagen was examined by XRD and presented in Figure 7. There were 2 sharp diffraction peaks located at the diffraction angles about 3.53o and 7.16°, corresponding to the characteristic diffraction peaks of collagen which contains proteins having ordered structure or ordered structure snippet. The d-spacing values of the sharp peak were 12.32 and 24.84 nm, contributing to the diameter of the trihelix chain in collagen. 8
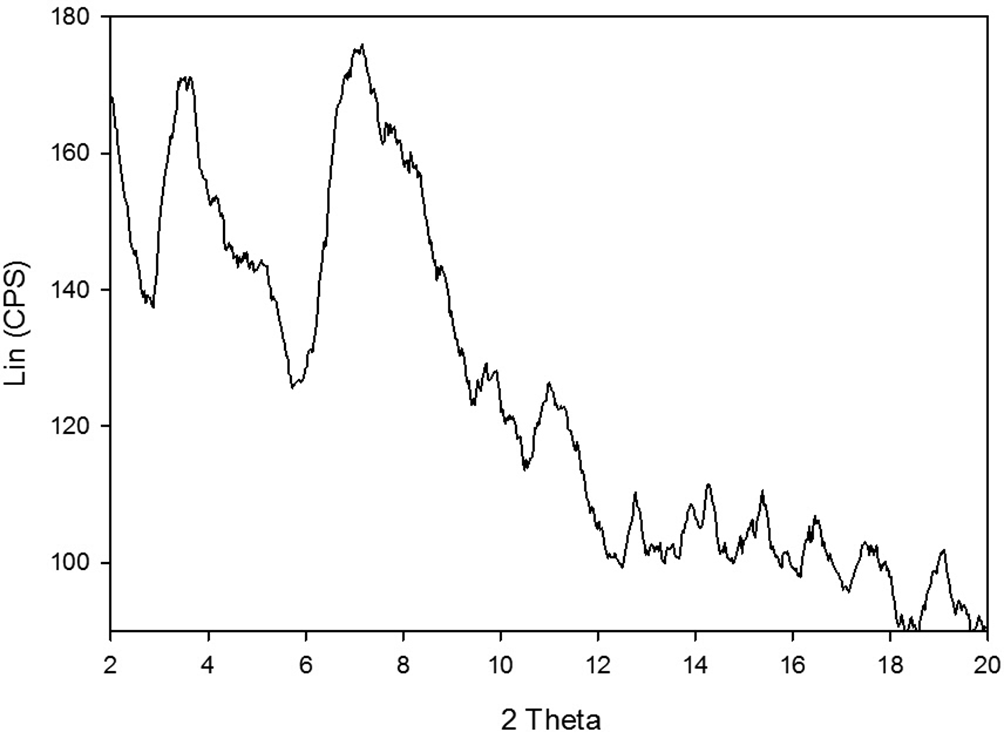
X-ray diffraction pattern of purified collagen.
Denaturation Temperature
As per our knowledge, denaturation temperature of collagen was recognized as the temperature where the fractional change in viscosity of collagen solution was 0.5. 8,34 It can be seen that the obtained collagen had denaturation temperature at 32.2°C (Figure 8). This value was similar to the denaturation temperature of acid-soluble collagen of fresh carp fish scale (tropical fish) (32.9°C) 8 but different from the acid-soluble collagen of carp scale (cold-water fish) (around 28°C). 34 The difference in denaturation temperature of tropical fish species and cold-water fish species is explained by the variance in fish’s living environmental and body temperatures. The denaturation temperature of some other tropical fish’s collagen such as bigeye snapper and brown striped snapper was found at 30.4°C and 31.5°C, respectively. 35 The collagen in this work having high denaturation temperature has promising applicability in food and pharmaceutical industry.

Thermal denaturation curve of purified collagen.
Thermal Behavior of Collagen
In Figure 9, there was an endothermic peak at 116°C (ΔH = 3.7 J/g) on the DSC diagram obtained from the first heating scan of freeze-dried collagen. This result is similar to the endothermic structural transition of dry collagen from a helical to a coiled state in the publication of Héctor Capella-Monsonís et al, with the onset temperature at 108.97°C and endothermic peak at 116.94°C. 41 Collagen molecules are closely packed in the dry state. However, in the case of our collagen, the onset temperature of endothermic process is recorded at 78°C, corresponding to both the thermal dehydration and structural transition process in collagen samples. In the second heating scan, that endothermic peak did not appear due to the disruption of the collagen structure during the first heating cycle.
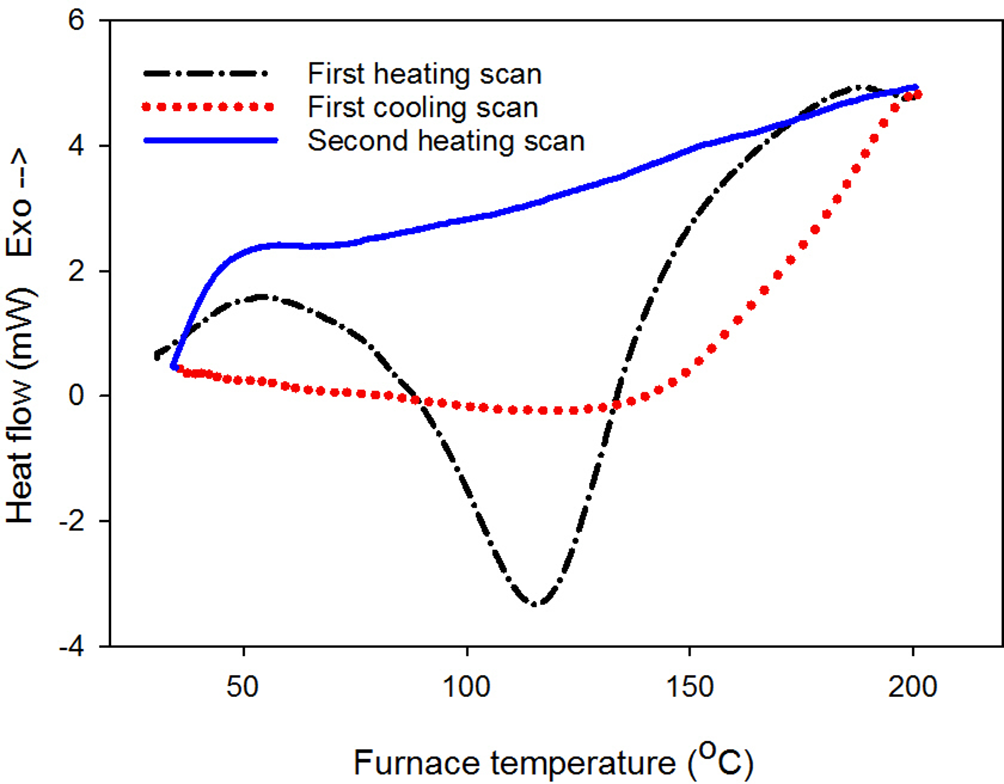
Differential scanning calorimetry diagram of purified collagen.
TGA combined with DSC analysis was used to determine the thermal properties of the collagen. From the dTG diagram of collagen, it can be seen that the collagen was degraded according to 3 steps related to 3 temperatures of maximum rate of weight loss (T max) (Figure 10). The first step was observed at T max1 88.68°C, corresponding to the loss of weight 10.32%. This decomposition step of collagen was attributed to the loss of water and residues in collagen. The next degradation of collagen occurring at T max2 334.43°C, corresponding to the loss of weight 44.32%, was mainly due to degradation of proteins that existed in collagen (methionine, tyrosine, arginine, lysine, serine, threonine, etc.,) and collagen structure forming molecules with low weight such as H2O, NH3, CO2, H2S, SO2. The last degradation step of collagen was detected at T max3 636.53°C (mass variation of 34.55%) due to the combustion of carbon or the loss of inorganic phase in collagen which remains. 42,43 The T max value of collagen in this work was higher than that of collagen sheet from fish (Lates calcarifer) scales. 42 This may be due to changing of water temperature and fish’s living environment, leading to the difference in amino acid composition, amino acid sequence, molecular weight, as well as thermal behavior of collagen.

TG (a) and dTG (b) diagrams of purified collagen.
Conclusions
In this paper, the characterizations of collagen extracted from fresh water carp fish scales (C. carpio) were determined. The suitable condition for extraction process of collagen was proposed by a combination of steps of removing fats, proteins, and minerals by dissolving in CH3COOH 0.5 M solution to receive an extraction yield for raw collagen of 13.6%. The data relating to amino acid composition of purified collagen showed that the collagen contains 18 amino acids with the presence of tryptophan, a rare amino acid in collagen extracted from carp fish scales. The collagen in the above fish scales was collagen type I, containing α1, α2, and β chains in structure with the type chain of (α1)2α2. The amino acid sequence of purified collagen was similar to collagen extracted from European common carp (Hypothetical protein cypCar_00045321, partial [Cyprinus carpio]). It had the molecular weight of 115 674 Da. The purified collagen having fibril structure and good physicochemical properties can be used as an alternative source of collagen in photography, food, pharmaceutical fields, etc.
Supplemental Material
Supplementary material - Supplemental material for Characterization of Collagen Derived From Tropical Freshwater Carp Fish Scale Wastes and Its Amino Acid Sequence
Supplemental material, Supplementary material, for Characterization of Collagen Derived From Tropical Freshwater Carp Fish Scale Wastes and Its Amino Acid Sequence by Nguyen Thuy Chinh, Vu Quoc Manh, Vu Quoc Trung, Tran Dai Lam, Mai Duc Huynh, Nguyen Quang Tung, Nguyen Duy Trinh, and Thai Hoang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 104.02-2017.326, period of 2018 to 2021.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
