Abstract
The aim of the present study was to evaluate the hypolipidemic effect of Trixis angustifolia aqueous extract (TxAE) on Triton WR-1339- and high-fat diet-induced hyperlipidemic mice. In the Triton model, treatment with TxAE at 100 and 200 mg/kg body weight produced a significant decrease in triglycerides and very low-density lipoprotein levels and a significant increase in high-density lipoprotein (HDL). Similarly, administration of TxAE along with the high-fat diet induced a significant decrease in serum total cholesterol, low-density lipoproteins, and increase in HDL. In addition, a phytochemical study of TxAE led to the isolation of 2 previously described compounds: pebrellin and xanthomicrol. This is the first time that these compounds have been identified in a plant extract with hypolipidemic effect. The results suggest the possible therapeutic potential of TxAE as a hypolipidemic agent supporting the usage of T. angustifolia as a traditional medicine.
Trixis angustifolia (Compositae), better known as “Hierba del viento,” is a plant endemic of Mexico spread in the states of Durango, San Luis Potosí, Querétaro, Zacatecas, and Aguascalientes. It is used in traditional medicine as an antipyretic agent and as a treatment for rheumatism. 1 Some members of the genus Trixis have been used to treat various complaints, including diarrhea, pain, diabetes, and venereal diseases. 2 A variety of phytochemicals with significant bioactivities have been discovered in this genus, including flavonoids, coumarins, sesquiterpene lactones, and some germacrene derivatives. 3,4
From a pharmacological point of view, the antibacterial activity against Mycobacterium tuberculosis H37Rv of organic extracts (hexane, chloroform, ethyl acetate, and methanol) of T. angustifolia aerial parts has been described. 5 In traditional Mexican medicine, the aerial parts of T. angustifolia have been used as a water-extract formulation to reduce blood glucose in alloxan-induced diabetic mice. 6 However, there is no literature about the hypolipidemic activity of this species that supports the ethnomedical usage of the plant. Therefore, the present study was conducted to evaluate the hypolipidemic effect of T. angustifolia aqueous extract (TxAE) on Triton WR-1339- and high-fat diet-induced hyperlipidemic mice. Additionally, the main components present in the aqueous extract were identified.
A single injection of Triton WR-1339 in mice (Group II) caused a marked increase of serum triglyceride (TG) and very low-density lipoprotein (VLDL) levels and a decrease in high-density lipoprotein (HDL) level compared with the control group (Table 1). When TxAE was administrated to hyperlipidemic Groups IV and V, TG serum levels were reduced by 44% and 48%, respectively, compared with Group II. Triton WR-1339 modifies TG catabolism by inhibition of lipoprotein lipase (LPL) lipolytic activity. 7 Our results suggest that TxAE is able to restore TG catabolism, whose restoration by LPL stimulation and/or beta oxidation has been related to the administration of other plant extracts. 8 Additionally, VLDL serum levels in Group V were reduced by 48.1% compared with Group II. Triglyceride serum level alterations lead to modifications of liver VLDL synthesis. 9 Our study showed that TG levels reduction was parallel with the decrease in VLDL levels by TxAE treatment. Group V also showed increased levels of HDL compared with Group II. It is known that esterification, transfer, and remodeling of HDL in blood are closely related to TG. 7 That TG and VLDL decrease in Group V, while HDL increases, indicates the hypolipidemic effect of TxAE.
Effect of Trixis angustifolia Aqueous Extract on Serum Lipid Profile in Triton WR-1339-Induced Hyperlipidemic Mice.

H, hyperlipidemic; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglyceride; TxAE 50, 100, and 200, Trixis angustifolia aqueous extract 50, 100, and 200 mg/kg, respectively; VLDL, very low-density lipoprotein.
Data expressed as a mean ± standard error; n = 8 animals/group, analyzed by analysis of variance and Student-Newman-Keuls multiple range median test.
* P < 0.05; significant difference with respect to Group I.
** P < 0.05; significant difference with respect to Group II.
On the other hand, mice fed for 6 days with the high-fat diet (Group II′) resulted in a significant increase in serum total cholesterol (TC) and low-density lipoprotein (LDL) levels, as well as a decrease in HDL, compared with the group fed with standard chow diet (Group I′). After TxAE treatment at different doses (Groups III′-V′), only at 100 mg/kg LDL level was significantly reduced (P < 0.05), while the level of HDL was elevated compared with Group II″ (Table 2). Based on this effect, a possible contribution of TxAE in the reduction of cardiovascular risk factors could be proposed. A significant body weight gain was observed in Group II′ compared with Group I′ (Figure 1a). Body weight in hyperlipidemic Groups IV′ and V′ was similar to Group I′, indicating that the administration of TxAE doses of 100 and 200 mg/kg are able to maintain body weight, even under a hypercholesterolemic diet. Increases in liver size relative to body weight in Group II′ were also observed (Figure 1b). In part, this change can be explained by hepatic lipid accumulation that could be related to hepatic steatosis. 10 When TxAE was administrated at 100 mg/kg, liver size relative to body weight was significantly lower (16%) than that in Group II′. If TxAE is able to stimulate lipid catabolism as discussed above, lipid accumulation and, therefore, body weight gain, and liver size decrease could be avoided.
Effect of Trixis angustifolia Aqueous Extract on Serum Lipid Profile in High-Fat Diet-Induced Hyperlipidemic Mice.

H, hyperlipidemic; HDL, high-density lipoprotein; LDL, low-density lipoprotein; TG, triglyceride; TxAE 10, 100, and 1000, Trixis angustifolia aqueous extract 10, 100, and 1000 mg/kg, respectively; VLDL, very low-density lipoprotein.
Data expressed as a mean ± standard error; n = 8 animals/group, analyzed by analysis of variance and Student-Newman-Keuls multiple range median test.
* P <0.05; significant difference with respect to normal group.
** P <0.05; significant difference with respect to hyperlipidemic control.

Effect of Trixis angustifolia aqueous extract administration on weight gain (a) and liver size relative to body weight (b) of high-fat diet-induced hyperlipidemic mice (data are mean ± standard error; n = 8 animals/group). *P < 0.05 as compared with normal group. **P < 0.05 as compared with hyperlipidemic group. H, hyperlipidemic; TxAE 10, 100 , and 1000, Trixis angustifolia aqueous extract 10, 100, and 1000 mg/kg, respectively.
With regard to the toxicological aspects of T. angustifolia extract, mortality and abnormal behavior were not observed in mice during the 14-day acute toxicity test. In addition, no significant pathological changes in vital organs color and texture were observed by macroscopic examination. In this sense, TxAE LD50 value could be >2000 mg/kg.
As far as we know, there is only 1 phytochemical screening regarding the use of water as solvent for the extraction of T. angustifolia constituents. 6 This is important since popular use occurs more frequently with infusions, decoctions, and macerations, and little is known about the constitution of this kind of extract. Thus, as part of the phytochemical study, we have determined the phenolic composition of TxAE. Phenolic compound levels (940.3 ± 12.2 mg gallic acid equivalents (GAE)/100 g dry plant) could be considered high compared with other medicinal plants. 11 Plants with high levels of phenolic compounds have demonstrated positive effects for prevention and treatment of hyperlipidemia. 12 Based on ESI (−) mass spectrometry, 3 main molecular ions were identified in TxAE displaying at m/z 353.0856, m/z 191.0519, and m/z 179.0461 (Supplementary Material 1). These are similar parent molecular ions to chlorogenic acid, quinic acid, and caffeic acid, respectively, 13 suggesting the presence of these phenolic acids in TxAE. Considering lipid-lowering properties previously reported for compounds found in TxAE, some of them could be related to the TxAE hypolipidemic effect acting alone or in synergism.
As different compounds isolated from Trixis species have shown biological activities,
4
we fractionated TxAE by liquid-liquid partition with ethyl acetate to improve the identification and purification of compounds probably involved in the TxAE hypolipidemic effects. Four known compounds were identified by high-performance liquid chromatography (HPLC)-atmospheric pressure chemical ionization (APCI)-mass spectrometry (MS) and nuclear magnetic resonance (NMR) analysis of the ethyl acetate fraction. Compounds
A hypolipidemic effect on streptozotocin-induced diabetes and diet-induced hyperlipidemia models has been observed after the administration of flavonoid-containing extracts.
14,15
Furthermore, in vitro and in vivo lipid catabolism modulation was observed after polymethoxylated flavones exposure by decreasing synthesis of lipids, such as TC and TG.
16
These data together with our results support the potential use of flavonoids-rich extracts, such as TxAE, on lipid-lowering treatments for different medical conditions. Additionally, the TxAE hypolipidemic ability reported in this work justifies its ethnomedical use as part of Mexican traditional medicine. To the best of our knowledge, only antibacterial activity has been demonstrated in T. angustifolia hexane extract, where flavones
In conclusion, TxAE exhibited an hypolipidemic effect on hyperlipidemic mice induced by Triton WR-1339 and a high-fat diet. Therefore, results of the present study are the first evidence supporting the use of T. angustifolia in traditional medicine and suggest its potential use as a novel natural source of new molecules for metabolic disease treatments.
Experimental
Plant Collection and Authentication
Trixis angustifolia was collected in Durango, Mexico, in April 2015. The botanical identification and authentication of the plant samples were performed by Quintos Escalante of CIIDIR-IPN. A specimen was deposited in the herbarium of the Escuela Nacional de Ciencias Biológicas, Instituto Politécnico Nacional (ENCB-IPN) with reference code: Col. M. González y S. Acevedo 2144.
Preparation of Extract
Trixis angustifolia aqueous extract was prepared following the traditional medicine used in Durango State. 17 Based on the volume of macerate administered to a young human adult (70 kg) per day as a treatment of several diseases, a certain volume of macerate with water was considered to estimate treatment doses. The aerial parts of the plant were dried and crushed into powder using a mortar. The dried powder (2 g) was mixed with 100 mL of water and the resulting macerate was filtered through Whatman filter paper No. 4.
Experimental Animals
Eighty-three male and 3 female ICR mice weighing 20 to 30 g were used for this study. Animals were housed in a room at a constant temperature of 22°C ± 1°C and a 12-hour light/dark cycle and were fed with standard rodent diet and water ad libitum. Animal distribution into groups and treatments was randomized. All experiments were approved by the Laboratory Animal Care Committee of the National School of Biological Sciences (National Polytechnic Institute) and were conducted in compliance with the Mexican Official Standard (NOM-062-200-1999) technical specifications for the production, care, and use of laboratory animals.
Acute Oral Toxicity
Acute oral toxicity was carried out according to guideline No. 423 provided by the Organization of Economic Co-operation and Development (OECD), with slight modifications. 18 This procedure was used to minimize the number of animals required for acute oral toxicity testing. Trixis angustifolia aqueous extract was intragastrically (IG) administered after a 12-hour fasting period at doses of 1000 and 2000 mg/kg body weight (bw) to 3 female and 3 male mice in each group. The control group received only water. The general behavior of mice was observed 4 hours after treatment and then every 24 hours for 14 days. At the end of the experiment, all animals were euthanized by cervical dislocation, and gross pathological changes in vital organs (liver, kidneys, heart, lung, spleen, ovaries, and testes) were identified.
Effect of TxAE on Triton WR-1339-Induced Hyperlipidemia
Triton WR-1339-induced hyperlipidemia was carried out according to the method of Mendieta et al. 19 After 1 week adaptation, male mice were randomly divided into 5 groups (8 mice per group), with Group I serving as a control (vehicle). Hyperlipidemia was induced in Groups II to V by a single intraperitoneal injection of Triton WR-1339 (400 mg/kg bw). Groups III to V were IG administrated with TxAE (50, 100, and 200 mg/kg bw, respectively) 1 hour before, and 22 and 48 hours after the Triton WR-1339 injection. At the end of each treatment (4 days), blood samples were drawn by puncturing the retro-orbital sinus and mice were euthanized by cervical dislocation.
Effect of TxAE on High-Fat Diet-Induced Hypercholesterolemic Mice
Male mice were randomly divided into 5 groups (8 mice per group), with Group I′ serving as the control (vehicle). Hypercholesterolemic diet (1% cholesterol, 0.5% sodium cholate, 5% butter, 30% sucrose, and 10% casein; food standard Rodent Lab 5001, 53.5%) was supplied to the animals ad libitum for 6 days in order to induce hypercholesterolemia. 20 Groups II′ to V′ received the hypercholesterolemic diet, and Groups III′ to V′ were administered with TxAE (10, 100, and 1000 mg/kg b.w., respectively) once a day by gavage. Doses were chosen according to the acute toxicological study divided by a security factor of 10. At the end of each treatment (6 days), blood samples were drawn by puncturing the retro-orbital sinus and mice were euthanized by cervical dislocation; adipose tissue and liver were removed and weighed. Body weight of animals was determined before and after the experiment, and weight gains were computed.
Biochemical Analysis
Serum samples were subjected to lipid analysis according to Argüelles et al. 21 Dry tubes containing collected blood were centrifuged at 13 000 × g for 15 minutes to obtain serum, which was stored at −20°C until the measurement of biochemical parameters. TC, HDL, and TG were quantified using the corresponding reaction kits for the automatic Vitalab selectra 2 instrument (Wiener Lab, Amsterdam, Netherlands).
Very low-density lipoprotein and LDL cholesterol were calculated using formulae 1 and 2 of Friedewald et al 22 :
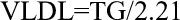
Total Phenolic Content
The total phenolic content was measured using a modified Folin-Ciocalteu method according to Stintzing et al. 23 Results were expressed as mg GAE/100 g dry plant.
Mass Spectrometry Analysis
Trixis angustifolia aqueous extract was filtered through a 0.25 µm polytetrafluoroethylene (PTFE) filter to remove any impurities. After this, the sample was analyzed in a micrOTOF-Q II™ spectrometer (Bruker Daltonics, Billerica, MA, United States) using desorption electrospray ionization-MS in positive or negative mode. A constant volumetric flow rate (8 µL/min) was achieved using a 74900-00-05 Cole Palmer syringe pump (Billerica, MA, United States) loaded with 100 µL of the sample. The capillary voltage was set to 2700 V, and nitrogen was used as the drying and nebulizing gas, with a flow rate of 4 L/min (0.4 bar) and a gas temperature of 180°C. Continuous spectra were collected in the m/z range of 50 to 1500, with a total run duration of 1 minute. Mass spectrometric data were processed using Bruker Compass Data Analysis 4.0 (Bruker Daltonics).
High-Performance Liquid Chromatography-APCI-MS Analysis
Trixis angustifolia aqueous extract was fractionated by liquid-liquid partition with ethyl acetate (EtOAc). The EtOAc extract was concentrated under reduced pressure to yield a brown viscous mass (560 mg). Ethyl acetate extract analyses were carried out on a HPLC (Agilent 1200 Series Binary SL) and a mass spectrometer with an APCI interface operating in the positive ionization mode (Bruker Esquire 6000). An Eclipse Plus C18 column (3.5 µm 100A 100 × 2.1 mm) was used. The injection volume and the flow rate applied were 5 µL and 0.2 mL/min, respectively, and formic acid 1% (v/v) in water (solvent A) and formic acid 1% (v/v) in methanol (solvent B) were used as the mobile phase. The gradient elution profile was the following: 0 to 15 minutes, 15% B; 15 to 25 minutes, 25% B; 25 to 35 minutes, 50% B; 35 to 50 minutes, 75% B; 50 to 55 minutes, 85% B. The extract was solubilized using methanol and water (1:1) and filtered in Millex filters (0.22 µm × 3.0 mm, PTFE, Millipore).
General
Melting points were determined on an electrothermal capillary melting point apparatus. Analytical thin-layer chromatography (TLC) was performed using precoated TLC plates with Silica Gel 60-F254 and visualized using combinations of UV and ceric sulfate staining. Purification of compounds was performed by column chromatography on silica gel (Merck 230-400 mesh). NMR experiments were conducted on a Varian NMR System at 500 MHz using CDCl3 or CD3OD as solvent and tetramethylsilane as the internal standard. Chemical shift values are reported in ppm (δ) (tetramethylsilane δ = 0 for 1H; chloroform-d δ = 77.0 for13C, and methanol-d 4 δ = 3.31, 4.78 for 1H, and δ = 49.2 for 13C). Spectroscopic data of each compound were compared with those previously reported. 4
Extraction of Phenolic Compounds
To purify the phenolic compounds observed in HPLC-APCI-MS analysis, the EtOAc extract was applied to silica gel flash column chromatography, eluting with an EtOAc-MeOH gradient system (99:1, 95:5, 90:10, 80:20, and 1:1), to obtain 50 fractions, which were grouped according to their chromatographic similarity. Further separation of fractions 3 to 5 (30 mg) by silica gel column chromatography, eluting with CH2Cl2-EtOAc (1:1-3:7), gave 28 fractions. Fraction 5 was further column chromatographed (2:7:1, Hex-CH2Cl2-EtOAc) and purified by crystallization from a n-hexane-CH2Cl2 system to yield pure
Statistical Analysis
All data are expressed as mean ± standard error of the mean (SEM). To perform statistical analyses, SigmaPlot 11.0 software was used. Statistical analyses were performed with one-way analysis of variance followed by Student-Newman-Keuls multiple range median test. Significant differences were set at P values less than 0.05.
Supplemental Material
Supplementary Material - Supplemental material for Hypolipidemic Effect of Trixis angustifolia Aqueous Extract on Triton WR-1339- and High-Fat Diet-Induced Hyperlipidemic Mice
Supplemental material, Supplementary Material, for Hypolipidemic Effect of Trixis angustifolia Aqueous Extract on Triton WR-1339- and High-Fat Diet-Induced Hyperlipidemic Mice by Anuar Salazar-Gómez, Anahí C. Sánchez-Chávez, Gerardo Zepeda-Vallejo, Germán Chamorro-Cevallos, Leticia Garduño-Siciliano, and Elena Vargas-Díaz in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by SIP-IPN (20181909 and 20171699). MEVD, LGZV, and LGS are COFAA, EDI, and SNI fellows. ASG is CONACyT (267721) and IPN-BEIFI fellow.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
